Abstract
Objectives
The aim of this report was to describe the clinical signs, diagnostic imaging findings, surgical management, histopathological findings, outcome and possible risk factors for cats that developed retroperitoneal fibrosis (RPF) following renal transplantation.
Methods
Medical records of cats that underwent renal transplantation and developed clinically significant RPF between 1995 and 2019 were reviewed.
Results
Eighty-one cats underwent 83 renal transplantations. Of these 81 cats, six developed clinically significant RPF. For all six cats, renal transplantation was performed using cold organ preservation solution and ureteral papilla implantation. Immunosuppression protocol included ciclosporin and prednisolone. All cats had at least one subtherapeutic trough ciclosporin level (<250 ng/ml) in the postoperative period. Cats presented with moderate-to-severe azotemia 39–210 days following renal transplantation. Abdominal ultrasonography and contrast pyelography revealed various degrees of hydroureter and hydronephrosis of the transplanted kidney. Surgical examination revealed a layer of dense fibrous tissue surrounding the transplanted kidney, ureter and bladder resulting in ureteral obstruction. Ureteral obstruction was managed by reimplantation of the proximal ureter or renal pelvis to the bladder. Histopathologic examination of the fibrous tissue and affected portion of the distal ureter revealed fibrous connective tissue with lymphoplasmacytic infiltration and perivascular inflammation suggestive of an autoimmune type reaction. Of the six cats, two died within 5 days after revision surgery, two developed signs consistent with recurrent partial ureteral obstruction (40 and 41 days after revision), one was euthanized 6 years later for an unrelated disease and one was lost to follow-up.
Conclusions and relevance
The incidence of RPF in this population of cats was relatively low (7%), but still represents a significant cause of morbidity and mortality. The cause of RPF remains unknown, although investigation into suboptimal immunosuppression as a potential cause for local rejection reaction is warranted.
Keywords
Introduction
Renal transplantation is a well-established treatment option for end-stage renal disease in cats. Overall outcome is relatively successful; however, potential intermediate and long-term complications include allograft rejection, immunosuppression-associated neoplasia or infection, and retroperitoneal fibrosis (RPF). 1 RPF is a local inflammatory process resulting in fibrous tissue proliferation in the retroperitoneal space that may result in partial or complete obstruction of the ureter. 2 RPF in cats has been described in two other reports, both detailing the condition as a complication after renal transplantation.3,4 In both reports, cats developed ureteral obstruction and surgical dissection (ureterolysis) was required to re-establish urine flow from the transplanted kidney.3,4 Clinical presentation and diagnostic findings were relatively similar for all cats, but a specific cause for RPF was not identified.
RPF in humans is classified as primary (idiopathic) or secondary. An immunologic etiology for idiopathic RPF has recently been investigated as an immunoglobulin G4-related disease (IgG4RD). 5 Causes of secondary RPF include reactions to various medications, infectious organisms, malignant disease and exposure to radiation.2,5,6 Risk factors for RPF in humans include hypertension, diabetes mellitus, neoplasia, coronary heart disease, dyslipidemia, infections, lymphoproliferative disease, cerebral vascular disease and tobacco exposure.2,6 Renal transplant-associated RPF has also been reported in humans, and is suspected to be secondary to retroperitoneal insults such as surgical trauma, infection, urine extravasation or exposure to surgical glove talcum powder.7,8 Additionally, acute and chronic transplant rejection episodes have been described as potential risk factors. 9
The aim of this paper was to describe the clinical signs, diagnostic imaging findings, surgical management, histopathologic findings, outcome and possible risk factors for cats that developed RPF after undergoing renal transplantation at one of the few institutions currently performing renal transplants in cats.
Materials and methods
Medical records review
Records were reviewed for all cats with a clinical diagnosis of RPF after renal transplantation between the years 1995 and 2019 at the University of Wisconsin School of Veterinary Medicine. Data collected included signalment, preoperative clinicopathologic findings, cause of renal failure, comorbidities, pretransplant management, anesthetic protocol, renal transplantation technique, postoperative management, complications and outcome. Additional data collected from the time of re-presentation for RPF included clinical signs, clinicopathologic findings, diagnostic imaging findings, medical and surgical management, histopathologic findings, postoperative complications and outcome.
Results
Eighty-one cats underwent 83 renal transplantations for chronic renal failure between 1995 and 2019. Of these 81 cats, six developed clinically significant RPF requiring surgical correction. Of these six cats, five were castrated males and one was a spayed female. There were five domestic shorthair cats and one domestic longhair. The median age was 11.5 years (range 3–17). The cause of renal failure was chronic kidney disease (CKD) of unknown etiology for five cats and obstructive urolithiasis for one (cat 2).
Preoperative screening and clinicopathologic findings
Prior to renal transplantation, each cat underwent a physical examination and bloodwork, including serum chemistry panel, complete blood count, urinalysis with aerobic culture and sensitivity, urine protein:creatinine (UPC) ratio, blood typing with cross-match, total thyroxine, feline leukemia virus (FeLV)/feline immunodeficiency virus (FIV) serology and toxoplasma titers. Abdominal ultrasound was performed to evaluate the kidneys, ureters and bladder. Cardiac function was evaluated including thoracic radiographs, blood pressure, electrocardiogram and echocardiography.
At the time of transplantation, median creatinine and blood urea nitrogen (BUN) were 3.8 mg/dl (range 3.3–4.5; reference interval [RI] 0.6–2.0) and 74.5 mg/dl (range 66–90; RI 18–36), respectively, for all cats. All cats had moderate-to-severe anemia (packed cell volume [PCV] 14–25%; median PCV 17%), which was suspected to be secondary to CKD. Five of six had comorbidities at the time of transplantation. Cat 1 had a Coombs’ positive status and hypertension, which was treated with amlodipine (0.25 mg/kg PO q24h). Blood pressure was normal at the time of transplantation. Cat 2 was proteinuric with a UPC of 1.99 and was treated with benazepril (2 mg/kg PO q24h). Additionally, cat 2 had a history of obstructive urolithiasis but did not have any evidence of urolithiasis at the time of renal transplantation. Cat 3 had a grade II/VI left parasternal heart murmur and was diagnosed with dynamic right ventricular outflow tract obstruction, which did not require any treatment. Cat 5 had a grade IV/VI left parasternal systolic heart murmur and was diagnosed with trivial mitral valve regurgitation and did not require any treatment. Cat 6 was diagnosed with dynamic left ventricular outflow tract obstruction and hypertension, and was treated with atenolol (1.1 mg/kg PO q12h). Urine culture and infectious disease testing (FeLV/FIV, toxoplasmosis) were negative in all cats. All cats were blood type A and were compatible (major and minor cross-match) with the kidney donor cat.
Preoperative transplant management
All cats were treated with intravenous crystalloid fluid therapy (lactated Ringer’s solution or Plasmalyte 40–60 ml/kg/day IV based on the individual needs of the patient) and maintained on an oral phosphate binder (aluminum hydroxide 13–100 mg/kg PO q8–24h) prior to transplantation. Blood products (30 ml packed red blood cells [n = 2] or 24–60 ml whole blood [n = 4]) were administered to all cats for the management of anemia. The immunosuppression protocol included ciclosporin A (CsA; 3.48–4.14 mg/kg PO q12h) and prednisolone (0.5 mg/kg PO q12h) starting 48 h before surgery.
Recipient anesthetic protocol
The anesthetic protocol was formulated at the discretion of the attending board-certified anesthesiologists. Protocols included premedication with fentanyl (2.85–5.35 µg/kg IV) and midazolam (0.1–0.28 mg/kg IV), induction with propofol (2.6–4.75 mg/kg IV; n = 5) or etomidate (0.75 mg/kg IV; n = 1), and maintenance with isoflurane and 100% oxygen. Cefazolin (22 mg/kg IV) was administered at the time of surgical induction and every 90 mins thereafter. In five cats, mannitol (0.5 g/kg IV) was administered prior to completion of the vascular anastomosis. In three cats, nitroprusside was administered for severe intraoperative hypertension (systolic blood pressure >180 mmHg; n = 1) or at the discretion of the surgeon, based on subjective evaluation of the anastomosis site and turgidity of the kidney (n = 2) at the time the transplanted kidney was reperfused.
Surgical procedure
The renal transplant procedure was performed as previously described by Sutherland et al. 10 The left kidney was removed from the donor cat and immediately flushed with (~20 ml) of cold, buffered sucrose phosphate preservation solution. The kidney was stored in preservation solution (~4°C) for approximately 1–4 h prior to implantation. The kidney was implanted via end-to-side anastomosis of the renal vein and artery to the vena cava and aorta, respectively. The ureter was implanted using the extravesicular ureteral papilla implantation technique. 10
Immediate postoperative care and monitoring
Postoperative management was based on individual requirements and included monitoring of vital signs, indirect blood pressure, PCV/total solids, serum electrolytes, BUN, creatinine and urine output. Medications included cefazolin (22 mg/kg IV q8h, 1–28 days while intravenous catheters were in place [n = 6]), enrofloxacin (5–10 mg/kg IV or PO q12–24h, 5 days [n = 2]), cephalexin (20 mg/kg PO q8h, 1 day [n = 1]) and heparin therapy (5–10 mg/kg SC q8h, 5–7 days [n = 6]). Hydralazine (1–2.5 mg SC q12–24h, 2–6 days) was administered for control of hypertension (systolic blood pressure >160 mmHg) in all six cats. Four cats were administered ketoconazole (10 mg/kg PO q24h) as a means of prolonging CsA metabolism and allowing for once-daily administration. Additional blood transfusions (20–60 ml whole blood) were administered to five cats. Color Doppler ultrasound of the transplanted kidney was performed on days 1–7 to evaluate renal size, perfusion, resistive index and urine outflow. Serum creatinine and trough (12–24 h) whole-blood CsA levels (high-performance liquid chromatography) were monitored every 1–3 days until CsA levels stabilized on a consistent dose. Target trough level of CsA was 400–600 ng/ml for the first 12 months after transplantation.
Immediate postoperative outcome
All cats had normal creatinine (0.6–2.0 mg/dl) within 1 day of renal transplantation. There were no major complications in the immediate postoperative period, with the exception of one cat (cat 1) that was febrile with an inflammatory leukogram 10 days postoperatively. Cat 1 responded to reinstitution of crystalloid intravenous fluids and antibiotic therapy (enrofloxacin, amoxicillin–clavulanic acid) and was discharged without evidence of infection. Mean hospitalization time for all cats was 18 days (range 9–32).
Post-hospitalization care
The CsA dose was adjusted as necessary to maintain trough levels between 400 and 600 ng/ml. Serum creatinine and CsA levels were monitored at a minimum of every 3–4 weeks for the first 3 months and then every 4–12 weeks depending upon the variability of the previous levels. All cats had a transient subtherapeutic CsA blood trough level (<250 ng/ml) in the postoperative time period despite consistent monitoring.
Re-presentation
All six cats re-presented between 39 and 210 days after renal transplantation. Clinical signs included anorexia (n = 3), lethargy (n = 2), vomiting (n = 3), urinating outside of the litter box (n = 2), grinding teeth (n = 1), abdominal distention (n = 1), polyuria/polydipsia (n = 1) and fever (n = 1). One cat had no overt clinical signs but was azotemic on routine bloodwork. All cats were azotemic, with a median BUN of 153.5 mg/dl (range 110–194) and a median creatinine of 7.8 mg/dl (range 2.3–10.4). Two cats were anemic, with a PCV of 16% and 17%, respectively. Median urine specific gravity was 1.013 (range 1.007–1.015). All cats had a negative aerobic urine culture at the time of re-presentation. Cat 1 was febrile and had retroperitoneal fluid on abdominal ultrasound. Cytological examination of the fluid revealed a transudate with poor cellularity consisting of occasional erythrocytes and superficial squamous cells. This cat was diagnosed with Mycoplasma haemofelis after transplantation and was receiving enrofloxacin (2.5 mg/kg PO q12h).
Further diagnostic testing included abdominal ultrasound (n = 6), abdominal CT (n = 1) and ultrasound-guided direct contrast pyelography (n = 6). Ultrasound findings were relatively consistent, including hypertrophy of transplanted kidney (n = 3), hydronephrosis (n = 6) and lack of distinguishable urine jet from the transplanted ureter (n = 4) (Figure 1). Contrast CT was performed in one cat which revealed enlargement of transplanted kidney with decreased contrast uptake and scant amount of contrast in the urinary bladder suggesting severe but incomplete ureteral obstruction. Ultrasound-guided contrast pyelography was performed in all six cats and revealed obstruction of the proximal to mid-ureter with decreased or no contrast passing into the bladder (Figure 2).
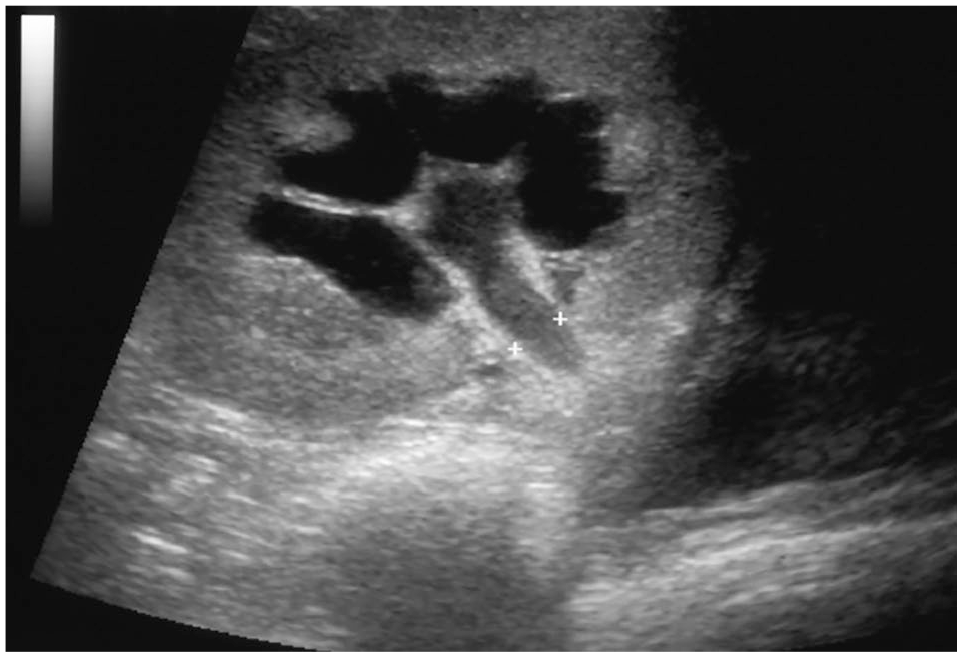
Ultrasound image of the renal pelvis and proximal ureter (

Direct contrast pyelogram of the transplanted kidney demonstrating dilation of the renal pelvis and obstruction of the proximal ureter
Surgical management of ureteral obstruction
Surgical correction of the ureteral obstruction was performed in all cats. Intraoperative examination of the abdominal cavity revealed a dense layer of fibrous tissue surrounding the transplanted kidney, ureter and bladder in all six cats, as well as adhesions between the body wall and mesentery or colon in two cats (Figure 3). Examination of the ureter confirmed either stricture (n = 4) or kinking (n = 2) of the ureter due to the surrounding fibrous tissue. In addition, one cat (cat 1) was found to have necrosis in the area of stricture with purulent material noted on transection of the affected ureter. Owing to the severity of the fibrous adhesions and degree of ureteral obstruction, the ureter was transected immediately proximal to the obstruction and re-implanted into the bladder. The proximal ureter was re-implanted into the apex of the bladder using an extravesicular ureteroneocystotomy technique (n = 5) and direct renal pelvis-to-bladder anastomosis (n = 1).

Intraoperative image of the transplanted kidney demonstrating the thick, white, fibrous tissue encapsulating the kidney (K) and proximal ureter (*), with the distal aspect of the ureter entering the bladder (B) apex
Samples of the fibrous tissue and the resected ureter (n = 5) were collected for histopathologic evaluation. Standard histologic processing was performed for hematoxylin and eosin staining, and samples were reviewed by a board-certified veterinary pathologist.
Five of six cats recovered from surgery without immediate complications. Cat 1 developed cardiac and respiratory arrest in the immediate postoperative period and did not regain consciousness. Cat 3 remained persistently azotemic and hypotensive postoperatively, developed mentation changes and died of respiratory arrest 5 days later. In the remaining four cats, BUN and creatinine normalized 1–8 days after revision. Cats 5 and 6 experienced recurrence of partial ureteral obstruction 40 and 41 days after revision, respectively. Additional revision surgery was declined, and both cats were managed medically until they were lost to follow-up 30 and 550 days later, respectively. Cat 2 was euthanized 6 years later due to metastatic carcinoma of the colon and cat 4 was lost to follow-up 60 days after revision.
Histopathologic evaluation revealed dense, moderately cellular fibroblastic to fibrous connective tissue surrounding the ureter in all samples. Ureteral serosa in the area of obstruction showed edema and hemorrhage with occasional moderate neutrophilic infiltration. Also noted was a diffuse lymphoplasmacytic infiltration into the fibrous tissue, with occasional perivascular aggregates of lymphoid inflammatory cells (largely lymphocytes and plasma cells) (Figure 4). Cat 4 had evidence of a more severe inflammatory infiltrate with degenerate neutrophils. Cat 1 had evidence of microabscessation in the walls of the ureter, consistent with intraoperative findings of pyuria.

Histopathologic images of the cross section of the ureter demonstrating the fibrous reaction within the seromuscular layer. Tissues stained routinely with hematoxylin and eosin at (a) × 4 and (b) × 20 magnification
Discussion
Six of 83 (7%) cats undergoing renal transplantation at the University of Wisconsin School of Veterinary Medicine between 1995 and 2019 developed clinically significant RPF. All six cats developed ureteral obstruction of the transplanted kidney and underwent ureteral reimplantation.
In this study, 5/6 cats were male, ranging in age from 3 to 17 years. Owing to the small sample size, no assessment was made as to whether gender or age could be a predisposing factor for RPF, although males appear to be overrepresented in both the human and veterinary literature,3,4,5,11,12 and the two previous veterinary reports3,4 describe a similar range in age, from 4 to 13 years.
In humans, the idiopathic form of RPF is considered a systemic inflammatory or IgG4RD. 5 IgG4RD is a fibroinflammatory autoimmune condition characterized by infiltration of IgG4-positive plasma cells affecting tissues of the salivary glands, pancreas, biliary tract, thyroid, prostate, kidney and retroperitoneal space. IgG4RD in humans often responds to corticosteroids and immunosuppressive therapy. 5 Additionally, the antiproliferative agent mycophenolate mofetil has shown some benefit in humans with RPF,13,14 and potentially should be investigated as part of the management of feline patients.
Histopathologic examination of the tissue samples from the cats in this study revealed chronic perivascular inflammation suggestive of an autoimmune type reaction, characterized primarily by lymphoplasmacytic infiltration. These findings may suggest an exaggerated immune response despite the use of the established immunosuppressive protocol, or perhaps a complication due to transient subtherapeutic CsA levels in the early postoperative period.
Currently, CsA combined with prednisolone is the most widely accepted immunosuppressive regimen for cats undergoing renal transplantation. Individual variation in absorption and metabolism of CsA makes maintaining blood concentrations relatively challenging and requires frequent monitoring to guide dose adjustments. All cats in this study had at least one trough sample below accepted immunosuppressive levels (250 ng/ml) within 30 days of transplantation. Owing to the fact that levels were checked every 1–3 days, it is impossible to know precisely how long blood levels were subtherapeutic, but the estimated duration was 3, 3, 7, 14, 7 and 10 days for cats 1–6, respectively. Similarly, 4/6 cats had additional subtherapeutic levels after the 30-day postoperative period when levels were checked less frequently. Levels of CsA in this time period remained <250 ng/ml for a range of 3–23 consecutive days. Although it is impossible to know exactly how long blood levels were subtherapeutic in each of these cats, the histologic features suggest an immune reaction may have played a role in RPF formation.
RPF has also been reported secondarily to neoplasia, diabetes mellitus, infection, medications (including antihypertensives such as hydralazine) and surgical trauma.2–5,15,16 In the current study, none of the cats had evidence of diabetes or cancer at the time of diagnosis of RPF. Only one (16%) had evidence of infectious disease (M haemofilis) vs 48% in a previous study. 3 Two of six cats (33.3%) received amlodipine or benazepril for treatment of hypertension, and all cats received at least one dose of hydralazine (0.5–2.5 mg/kg SC) post-transplantation for the management of hypertension. The mechanism of RPF following the administration of hydralazine in humans is unknown, but it has been proposed that repeated arteriolar vasoconstriction and vasodilation causes perivascular edema and transudation of plasma contents leading to RPF.16,17 In the previously mentioned studies,3,4 only 50% and 13.7% of cats, respectively, were administered any antihypertensive medications and the fact that hydralazine was not used in any of the 29 cats reported by Wormser et al 3 makes it difficult to implicate that drug as a potential cause for RPF in cats.
The incidence of clinically significant RPF following renal transplantation in this study (n = 6/83 [7%]) was less than previously described (n = 29/138 [21%]); however, the reason for the difference is unknown and direct comparison is challenging owing to the relatively small number of cases and inherent variation in patient population and comorbidities. Donor and recipient screening protocols, perioperative management and immunosuppressive protocols appear similar at both institutions. The only notable differences in surgical technique include the use of cold storage preservation solution and the technique for ureteral re-implantation. Cold storage has been shown to protect the kidney from adverse effects induced by warm ischemia (acute tubular necrosis), which may ultimately reduce the inflammatory response of the kidney and thus a stimulus for RPF. 18 Use of this technique obviously did not eliminate the risk for RPF, but may have contributed to the reduced incidence.
All of the cats in this report underwent resection of the obstructed ureter and re-implantation using an extravesicular approach. The severity of the fibrous adhesions surrounding the ureter and the concern for recurrent stricture, prompted the decision to resect and re-implant the ureter. This is in contrast to the two reports where ureterolysis was used to establish patency without the need to re-implant the ureter.3,4
Revision procedures were associated with a high mortality (n = 2/6 [33%]) and of the four cats that survived revision surgery, two (50%) experienced recurrence of ureteral obstruction. It is unknown whether surgical revision technique has an impact on the recurrence of ureteral obstruction, but the rate of recurrence suggests that persistent or recurrent fibrosis after revision surgery is likely and monitoring for re-obstruction is warranted.
Although not evaluated in these cats, C-reactive protein (CRP) may be useful for earlier detection of RPF. CRP is an acute-phase protein that increases with inflammation and can be used as a sensitive biomarker to detect the presence of underlying inflammation in the absence of other clinical or laboratory indicators. Studies have shown CRP blood levels are elevated in humans with RPF, independent of the cause,6,13 and it has diagnostic utility in monitoring progression and improvement in various inflammatory diseases including pancreatitis, cholangiohepatitis, immune-mediated diseases, pneumonia and neoplasms such as lymphoma and hemangiosarcoma.19,20
The limitations of this study include the small number of cats on which to base any conclusions regarding causes or risk factors for RPF. Additionally, it is possible that other cats from this institution could have developed RPF that went undetected owing to the lack of obvious clinical signs or involvement of the transplanted ureter. Finally, it is also possible that, despite careful evaluation of the medial records, these six cats experienced some unique causative factor that was not detected during retrospective evaluation.
Conclusions
RPF should be considered as a cause for azotemia in the intermediate postoperative period in cats following renal transplantation. Although the incidence of RPF in this study was relatively low (7%), the high morbidity and mortality associated with this complication warrants careful monitoring and early diagnosis to potentially minimize the effect of ureteral obstruction on the transplanted kidney. The potential association between RPF and subtherapeutic CsA levels in the immediate postoperative period warrants further investigation.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognized high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee was therefore not necessarily required.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or nonexperimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
