Abstract
Objectives
The aim of the present study was to report the postoperative short- and long-term outcomes in cats with benign ureteral obstruction treated surgically with ureteral stent, subcutaneous ureteral bypass (SUB) or ureterotomy.
Methods
Signalment, surgery time, hospitalization duration, pre- and postoperative biological and biochemical parameters, postoperative revision surgery rate, and date of death or last follow-up date were recorded. Pre- and postoperative biological and biochemical parameters, risk of major complications requiring revision surgery and survival time were statistically analyzed.
Results
A total of 105 cats (128 ureters) met the inclusion criteria, consisting of 34 ureters in the stent group, 43 in the SUB group and 51 in the ureterotomy group. There was no significant difference in sex, body weight (BW), serum creatinine, potassium, phosphorus, pH, base excess or packed cell volume (PCV) preoperatively between the three surgical groups. Surgery time and hospitalization duration were significantly longer in the stent group compared with the other two groups. Major complications requiring revision surgeries were 41%, 23%, 15% in the stent, SUB and ureterotomy groups, respectively. The median survival time (MST) was 1426 days in the stent group; MST was not reached in the SUB or ureterotomy groups during the study. Five cats (4.4%; one stent, three SUBs, one ureterotomy) died within 10 days postoperatively. The ureterotomy group had higher long-term PCV and BW.
Conclusions and relevance
Ureterotomy demonstrated favorable outcomes for the management of feline benign ureteral obstruction, including the lowest rate of revision surgery. Improvements in PCV and BW were most pronounced in this group. In addition, surgical and hospitalization durations were comparable to those of SUB. In this single-center, single-surgeon cohort, these findings support ureterotomy as a viable and durable surgical option in appropriately selected cases, while recognizing that the results may not be generalizable to all clinical settings.
Introduction
Feline ureteral obstruction can be caused by ureteroliths, strictures, soft tissue (blood clots, debris, purulent material and solidified dried blood), trauma, neoplasia and iatrogenic causes.1 –6 Medical intervention has been used with various success rates in the range of 8–30%. 4 Owing to the low chance of clinical resolution by medical management, surgical intervention is often necessary. Surgical intervention includes ureteral stenting, subcutaneous ureteral bypass (SUB), ureterotomy and ureteroneocystostomy, with mixed success rates and known complications.3,4,6,7 –16 There are no standardized guidelines for type of surgical treatment pursued, and the choice of procedure is often decided based on the surgeon’s experience, preference and available equipment, as well as the cause and location of the obstruction.
Reported complications of ureteral stents include stent migration, severe pollakiuria, dysuria, hematuria or urinary tract infections.7,8,14,15,17 Reported stent replacement or reobstruction rates are in the range of 15.4–27%,12,14,15 and non-infectious cystitis and dysuria have been observed and reported in 25–37.7% of cases.7,13 –15 Short-term postoperative mortality rate in cats receiving a ureteral stent was in the range of 7.6–15%,7,8,12 –14 with a reported median survival time (MST) of 419 days (range 44–994) in 26 cats. 14
Reported complications of SUB placement include luminal obstruction or catheter kink, implant migration and infection.18,19 The obstructive complication rate has been reported to be in the range of 7–29%.11,12,18 –21 Non-infectious cystitis and dysuria have also been observed in 38.5% of cats receiving a SUB. 9 The reported mortality rate to discharge or at 9 days is in the range of 5.7–15.4%.9,12,18 In feline patients, the survival rate post-SUB placement at 1, 3 and 6 months was 84.6%, 83.3% and 72.7%, respectively. 9
Ureterotomy has been described for benign ureteral obstructions in cats, with reported complications of uroabdomen, stricture formation and reobstruction.3,4,6,10 Reobstruction was reported in 40% of cats, with 10% of survivors needing additional revision surgeries, of which there was a 30% mortality rate. 4 Stricture was seen in over 30% of cats in this same study, 4 and additional surgery was needed in 12.8% of cases. 10 Lower urinary tract signs occurred in only 2% of ureterotomy patients. 7 The mortality rate associated with ureterotomy in cats is reported to be in the range of 8–30%.4,7 –10 Reported survival rates at 1, 3 and 6 months were 70%, 50% and 37.5%, respectively. 9
These higher complication and mortality rates suggest ureterotomy may not be the ideal surgical option for feline benign ureteral obstruction.
The authors are unaware of any current literature that compares the short- and long-term outcomes of ureteral stent placement, SUB placement and ureterotomy for benign ureteral obstruction (ureterolith, soft tissue, stricture) in cats from one institution in the same study. The objective of this study was to report the major postoperative complication rate defined as events that required an additional surgical or interventional procedure to correct the complication (level 3 adverse events), 22 outcomes in biochemical and biological parameters, short-term mortality rates (defined as up to 10 days postoperatively) and long-term survival times in these three surgical procedures.
We hypothesized that the 10-day postoperative mortality rate after ureterotomy would be lower than the 8–30% previously reported in the literature4,7 –10 and would not differ significantly among the three surgical interventions, the major postoperative complication rate after ureterotomy would not be as high as the 10–12.8% previously reported,4,10 and postoperative survival times would not differ significantly among the three procedures.
Materials and methods
A retrospective review of medical records from the veterinary teaching hospital between January 2012 and December 2020 was conducted to identify cats who underwent surgical intervention for benign ureteral obstruction (ureterolith, soft tissue, stricture), including ureteral stent, SUB or ureterotomy. Informed consent for use of clinical information for research purposes is routinely obtained at admission. Cats were included if they had clinical, biochemical and imaging follow-up for at least 1 month postoperatively, up to 24 months, either at our institution or with their primary veterinarian.
Preoperative data obtained from medical records included signalment and body weight (BW [in kg]). Biochemical data included serum blood urea nitrogen (BUN), creatinine (Cre), potassium (K), phosphorus (P), packed cell volume (PCV), pH and base excess (BE). Surgery records were assessed and surgery time, drain placement for the ureterotomy group and type of procedure were recorded. Surgery time was defined as the time from skin incision to completion of skin closure, whereas anesthesia time (not analyzed statistically) was defined as the time from induction to extubation.
Postoperative data collected included hospitalization time, biochemical data, presence of uroabdomen for the ureterotomy group and other major complications. Major postoperative complications were defined as the need for revision surgery: a level 3 adverse event. 22 During postoperative (1 month, 3 months, 6 months, 12 months and 24 months) follow-up examinations, cats were monitored for the following data: serum BUN, Cre, K, P, PCV, BW and urinary tract-focused ultrasonography to detect early obstruction. Follow-up was primarily performed at our institution for all three surgical groups; however, in cats who were clinically stable, lived far from the hospital or whose primary veterinarians were able to perform follow-up care, monitoring was carried out by the referring veterinarians. Owners were advised to return every 1–3 months. Owners were also instructed to seek immediate re-evaluation at our institution if any clinical concern arose.
Time of death or latest follow-up information was obtained from either our hospital records, referring veterinarians or owners.
The exclusion criteria included procedures that did not represent a single stand-alone intervention for a benign ureteral obstruction (eg, pyelotomy, ureteroneocystostomy, nephrostomy, ureteronephrectomy or concurrent procedures, such as SUB on one ureter and ureterotomy on the contralateral side), as well as cases with malignant obstruction.
Cats that required stent or SUB revision – such as stent replacement, conversion from stent to SUB or removal of a SUB because of obstruction, malfunction or other complications – were included, and all revision procedures were recorded as major postoperative complications. Group assignment was based solely on the initial procedure; however, cats were only retained in that group as long as the original treatment modality remained in place.
Accordingly, for survival and longitudinal analyses, cats were censored at the time of conversion to a different procedure (eg, stent to SUB), device removal, nephrectomy or any event resulting in loss of the original intervention, as they no longer represented the initial treatment group.
In contrast, cats who underwent stent replacement but continued with a stent remained in the stent group and contributed follow-up data until their final evaluation or censoring point.
Choice of procedure
The choice of surgical technique (stent, SUB or ureterotomy) was not randomized. The decision was based on the cause and location of obstruction, ureteral anatomy, device availability and surgeon preference. Earlier in the study period, ureteral stenting was more commonly performed (2010–2015), as only ureteral stenting, ureterotomy combined with stent placement and ureteroneocystostomy combined with stent placement were performed at that time; however, concurrent procedures were excluded from this study. After 2016, SUB devices were implemented at our institution and were generally selected for ureteral strictures, multiple small nephroliths or ureteroliths, or in cats who had already developed stent-related complications. Beginning in 2017, ureterotomy was increasingly favored for cats with one or two discrete ureteroliths measuring 1 mm or more and causing focal obstruction, as some SUB-associated complications (eg, obstruction, persistent pollakiuria) were observed, and the operative duration for ureterotomy was perceived to be comparable to SUB. All surgeries (stent, SUB and ureterotomy) were performed by a single experienced small-animal soft tissue surgeon.
Statistical analysis
Pre- and postoperative biochemical and hematological parameters (serum BUN, Cre, K, P, PCV, preoperative BE and pH), BW and age were evaluated among three groups. For descriptive statistics, data were assessed for normality using the Shapiro–Wilk test and reported as mean ± SD for normally distributed data or median (range) for non-normally distributed data. Variables that were not normally distributed were log-transformed before further analysis. The overall survival rate was estimated using Kaplan–Meier curves and Cox models. The preoperative parameters, 1, 3, 6, 12 and 24 months postoperative parameters, and comparison among surgeries were evaluated using mixed-effects models with the pre-values, covariates (potentially influencing the response variables), duration of surgery and time by surgery interaction as fixed effects, with patients as random effects. Follow-up duration was analyzed using a Kruskal–Wallis test. A linear mixed-effects model was used to analyze surgery time and length of hospitalization.
All analyses were conducted using commercially available software (SAS version 9.4). P <0.05 was considered significant.
Results
A total of 105 cats (128 ureters) met the inclusion criteria, consisting of 34 ureters in the stent group, 43 in the SUB group and 51 in the ureterotomy group. Eight cats were excluded for undergoing a combination of surgical procedures.
The mean age was 7.1 ± 3.3 years and mean BW was 3.95 ± 1.1 kg. There were three female cats, 48 spayed female cats, six male cats and 48 castrated male cats. The breeds were domestic shorthair (n = 30), American Shorthair (n = 29), Scottish Fold (n = 14), Maine Coon (n = 8), Munchkin (n = 5), British Shorthair (n = 4), Singapura (n = 3), Egyptian Mau (n = 2) and Bengal Cat, Chinchilla, Cornish Rex, Himalayan, Minuet, Norwegian Forest Cat, Ragdoll, Russian Blue, Selkirk Rex and Tonkinese (n = 1 each). Age, sex and BW in each group are summarized (Table 1). There was no significant difference in sex and weight between the three groups. The cats were significantly older in the SUB group compared with the stent group (P = 0.046).
Age (in years), sex and body weight (BW) in each group

Data are mean ± SD. There was no significant difference in sex distribution or BW among the three groups. Age was significantly higher in the subcutaneous ureteral bypass (SUB) group compared with the stent group (P = 0.046).
F = female; FS = female spayed, M = male; MN = male castrated
Clinical pathology preoperative findings included normal to severely elevated BUN, Cre, K or P, and normal to severely decreased PCV, pH or BE (Table 2). Baseline BUN in the ureterotomy group was statistically lower than that in the SUB group (P = 0.01). However, Cre, K, P, PCV, pH and BE were not statistically different across the three groups preoperatively.
Preoperative clinical pathology parameters

Data are mean ± SD. Blood urea nitrogen (BUN), creatinine (Cre), potassium (K) and phosphorus (P) ranged from normal to severely elevated values. Packed cell volume (PCV), pH and base excess (BE) ranged from normal to severely decreased values. Baseline BUN was significantly lower in the ureterotomy group compared with the subcutaneous ureteral bypass (SUB) group (P = 0.01). No significant differences were observed among the groups for Cre, K, P, PCV, pH or BE
RI = reference interval
Surgical findings
Before surgery, cats were medically managed for 1–10 days with fluid therapy and transferred to our institution. Preoperative biologic data were obtained on the day of surgery. Cats were premedicated and induced with protocols at the discretion of the attending clinician. Cefazolin 22 mg/kg IV was administered every 90 mins during the procedure as perioperative antimicrobial prophylaxis in accordance with commonly recommended practices for clean-contaminated soft tissue surgical procedures.
Ureteral stents (Vet Stent-Ureter, 2+ Ft or 2.5 Ft; Infiniti Medical) were placed under ultrasonographic guidance 23 and placement confirmed with radiographs during or after the procedure. Briefly, normograde placement was attempted first, followed by retrograde placement if normograde placement was not possible. Because guidewire access to the renal pelvis is required for stent placement, renal pelvis decompression was inherently achieved during this procedure.
A commercial SUB kit for cats and small dogs (SUB 1001K or SUB 2001K kit; Norfolk Vet Products) was used in this study and placed under the ultrasound guidance as described by Butty et al. 24 Placement was confirmed by ultrasound during the procedure with concurrent saline flushing and radiographs after surgery. As the SUB procedure requires catheterization of the renal pelvis, renal pelvis decompression occurred routinely during device placement.
All ureterotomies were performed under magnification (Leica Microsystems, used at × 3–3.5 magnification). In cats with severe hydronephrosis or azotemia, renal pelvis decompression (pyelocentesis) was performed at the discretion of the surgeon. The obstructed ureter lesion was undermined from the retroperitoneum and gently held between the surgeon’s fingers to reduce bleeding. The ureter was incised adjacent to the obstruction, and after the obstruction was removed, it was closed with 8-0 or 9-0 nylon (MANI) in a simple interrupted pattern. Two to five sutures were placed depending on incision length. After ureteral closure, a leak test was routinely performed using warm saline, administered via a temporary nephrostomy catheter. Active closed-suction drains were placed in the peritoneal cavity adjacent to the ureterotomy site before abdominal closure in 39/51 (76%) cats undergoing ureterotomy.
All surgeries were performed by a single surgeon. Mean surgery time in the stent, SUB and ureterotomy groups was 181.8 ± 71.3 mins, 104.5 ± 47.3 mins and 107.7 ± 37.6 mins, respectively. The surgery time was shorter in the ureterotomy (P <0.0001) and SUB groups (P <0.0001) compared with the stent group. There was no significant difference in surgery time between the SUB and ureterotomy groups (P = 0.778).
After surgery
Cats were hospitalized in the intensive care unit for recovery. They were given a fentanyl constant rate infusion (1–5 µg/kg/h IV) postoperatively and intravenous fluid therapy. Blood transfusion, nasoesophageal tubes and antibiotics were administrated at the discretion of the surgeon. Cats were monitored with renal focused ultrasonography and blood biochemical values before discharge. The presence of a uroabdomen was monitored via an active closed abdominal drain, as well as biochemical changes in abdominal effusion (paired Cre and K concentrations) in the ureterotomy group.
Hospitalization duration
The mean hospitalization time was 7.5 ± 3.0 days in the stent group, which was statistically longer than that in the SUB group (5.1 ± 2.6 days; P = 0.004) and ureterotomy group (5.3 ± 2.7 days; P = 0.005). There was no significant difference in hospitalization time between the SUB and ureterotomy groups (P = 0.852).
Follow-up
The follow-up duration was in the range of 1–72 months for the stent group (median 9 months), 1–50 months for the SUB group (median 21.5 months) and 1–53 months for the ureterotomy group (median 18 months). Although the medians differed among groups, wide variability and overlap in follow-up duration resulted in no statistically significant group difference (P = 0.40).
Biochemical parameters
The mean BUN concentration decreased postoperatively in all groups. Although not statistically significant, the postoperative BUN trend from 1 month to 24 months showed an increase in the stent group, a plateau in the SUB group and a decrease in the ureterotomy group (Figure 1).
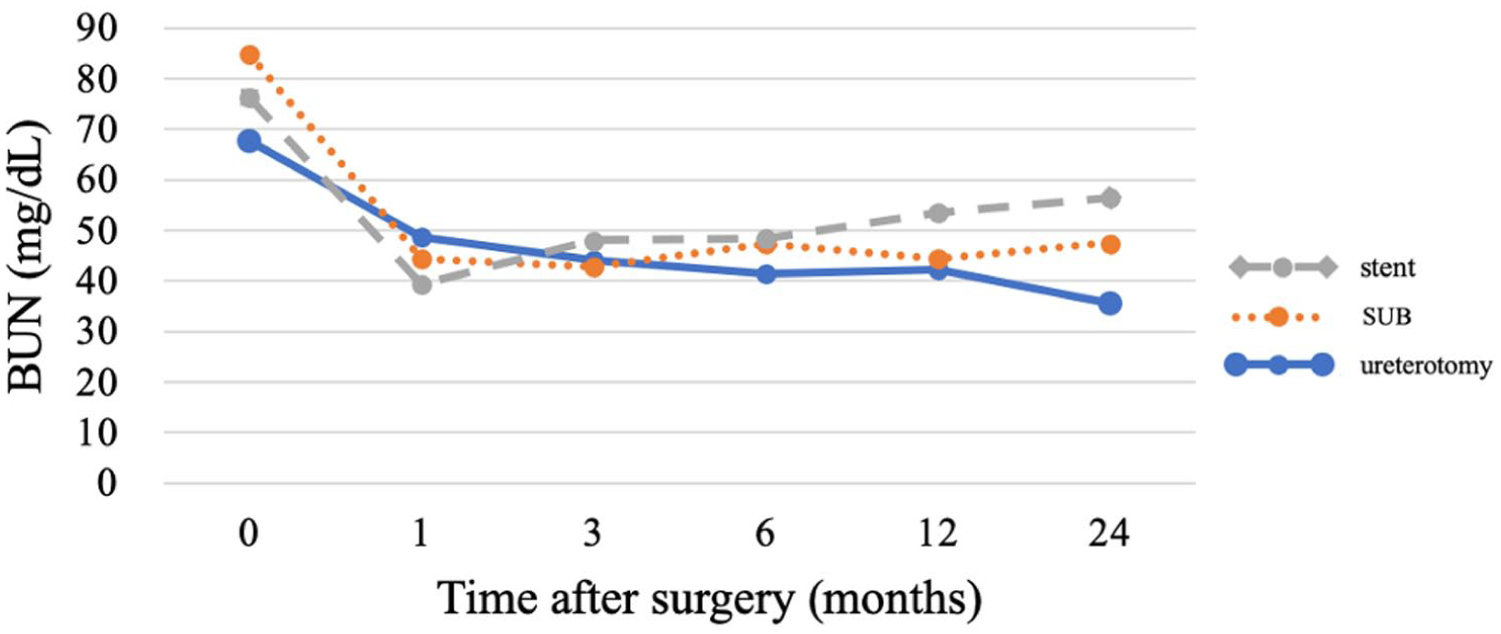
Serum blood urea nitrogen (BUN) trends after surgery. The x-axis shows time after surgery (months) and the y-axis shows serum BUN concentration (mg/dl). The stent group (gray dashed line) showed a trend toward increasing BUN, the subcutaneous ureteral bypass (SUB) group (orange dotted line) remained stable and the ureterotomy group (solid blue line) showed a decreasing trend, although these differences were not statistically significant
One month postoperative Cre was lower than the preoperative concentration and had been static from 3 months through 24 months postoperatively in all groups (mean creatinine 2.0–3.0 mg/dl) (Figure 2).
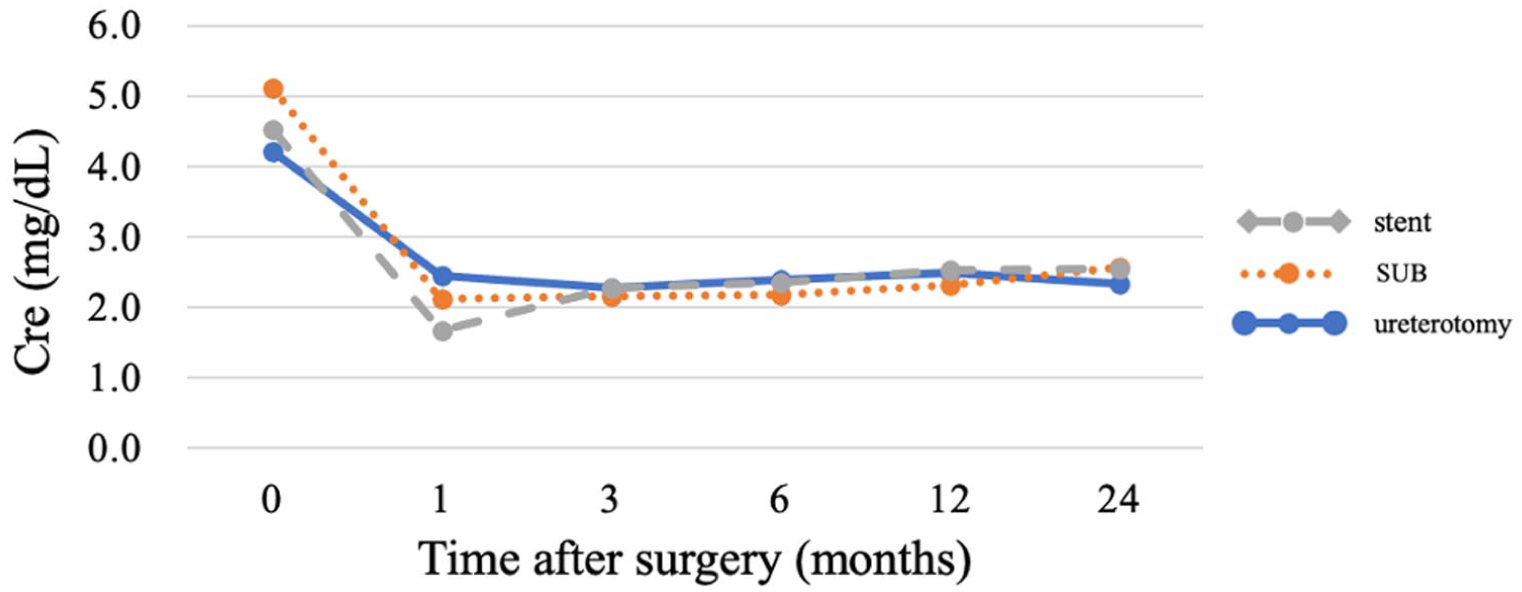
Serum creatinine (Cre) trends after surgery. The x-axis shows time after surgery (months) and the y-axis shows Cre concentration (mg/dl). The stent group (gray dashed line), subcutaneous ureteral bypass (SUB) group (orange dotted line) and ureterotomy group (solid blue line) all showed decreased creatinine concentrations at 1 month postoperatively, which remained stable at 3–24 months
Mean PCV was similar in all groups at 1 month postoperatively, and then increased gradually in every group, with PCV significantly higher in the ureterotomy group than in the SUB group at 12 and 24 months postoperatively (12 months: 4.23, 95% confidence interval [CI] 1.10–7.37; 24 months: 4.47, 95% CI 0.20–8.75) (Figure 3).

Packed cell volume (PCV) trends after surgery. The x-axis shows time after surgery (months) and the y-axis shows PCV (%). The stent group (gray dashed line), subcutaneous ureteral bypass (SUB) group (orange dotted line) and ureterotomy group (solid blue line) had similar mean PCV at 1 month postoperatively, which then increased gradually in all groups. PCV was significantly higher in the ureterotomy group compared with the SUB group at 12 and 24 months (mean difference 4.23, 95% confidence interval [CI] 1.10–7.37; 24 months: 4.47, 95% CI 0.20–8.75)
In the stent group, BW progressively decreased over time, with an absolute mean loss of 0.36 kg at 1 month, 0.40 kg at 3 months and up to 0.57 kg by 24 months. It was significantly lower than any other groups at 6, 12 and 24 months postoperatively: the effect coefficient was 3.75 (95% CI 3.50–4.00), 3.56 (95% CI 3.27–3.86) and 3.45 (95%CI; 2.99-3.92), respectively. In contrast, BW remained relatively stable in the SUB group, with maximal changes in the range of −0.17 to +0.06 kg. In the ureterotomy group, consistent weight gain, with increases of 0.20 kg at 3 months, 0.41 kg at 6 months and 0.54 kg by 24 months, indicating clinically meaningful improvement in nutritional status. BW was significantly higher than any other groups at 3, 6, 12 and 24 months postoperatively: the effect coefficient was 4.22 (95% CI 4.08–4.36), 4.43 (95% CI 4.28–4.58), 4.49 (95% CI 4.33–4.64) and 4.56 (95% CI 4.34–4.78), respectively (Figure 4).
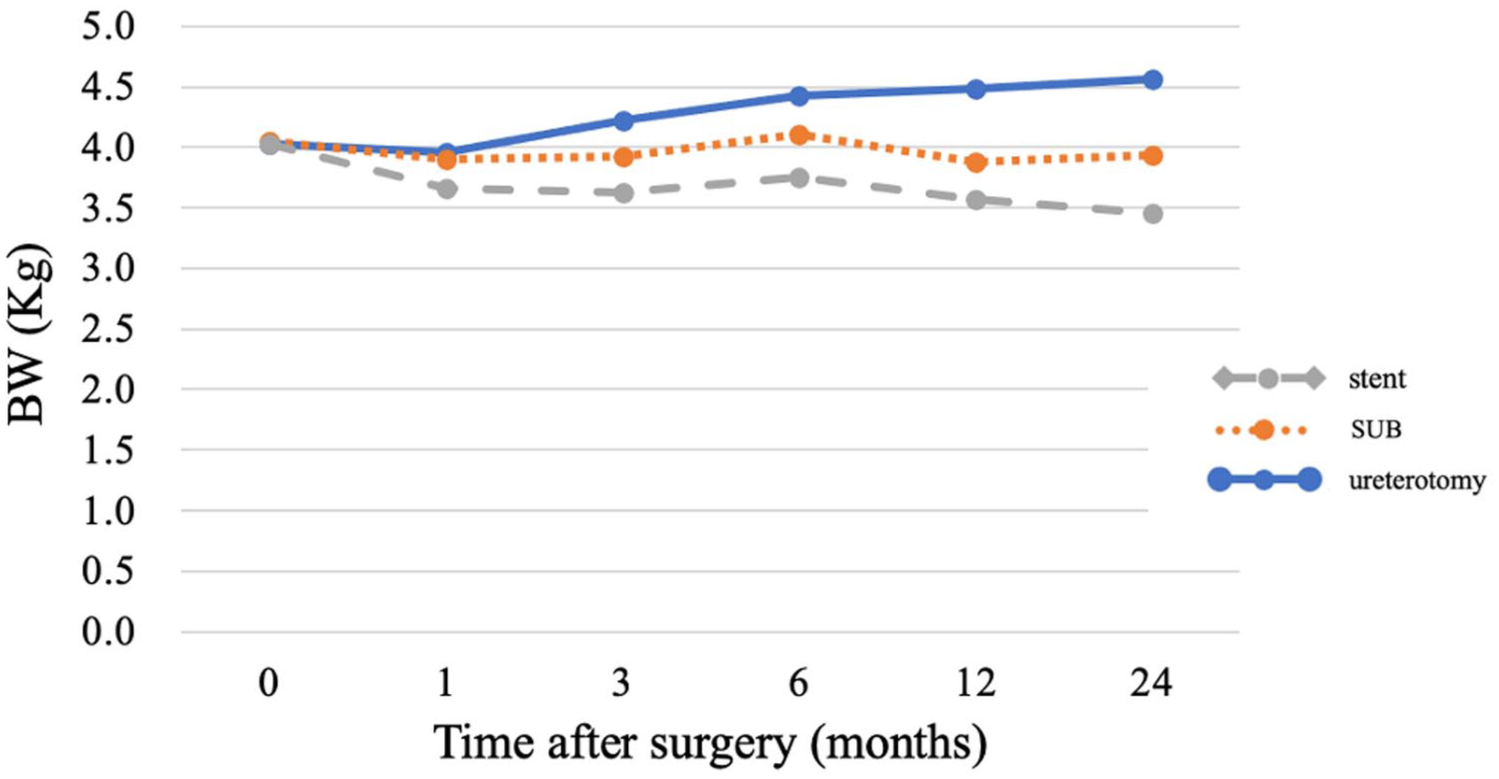
Body weight (BW) trends after surgery. The x-axis shows time after surgery (months) and the y-axis shows BW (kg). The stent group (gray dashed line) showed a gradual decrease in mean BW during follow-up and was significantly lower than the other groups at 6, 12 and 24 months postoperatively (effect coefficient 3.75, 95% confidence interval [CI] 3.50–4.00; 3.56, 95% CI 3.27–3.86; and 3.45, 95% CI 2.99–3.9), respectively). The ureterotomy group (solid blue line) had significantly higher body weight than the other groups at 3, 6, 12 and 24 months (4.22, 95% CI 4.08–4.36; 4.43, 95% CI 4.28–4.58; 4.49, 95% CI 4.33–4.64; and 4.56, 95% CI = 4.34–4.78, respectively)
Complications
Overall major complications requiring additional revision surgery (level 3) 22 were observed in 25% in all cats, with 41%, 23% and 15% in the stent, SUB and ureterotomy groups, respectively. The stent group had a higher hazard ratio (2.80, 95% CI 1.23–6.38; P = 0.01) than those in the SUB and ureterotomy groups. Level 3 postoperative complications in the stent group included obstruction (n = 6), stent migration (n = 4), severe pollakiuria and stranguria (n = 4), followed by stent replacement (n = 6), switching from a stent to a SUB placement (n = 4) or stent removal without any surgical treatments afterward (n = 1). In the SUB group, revision surgery was required for intraluminal blood clot formation or mineralization in or around the tube (n = 6), which needed to be replaced, or shunting port skin erosion (n = 4). The SUB group showed a tendency for late level 3 complications at approximately 2.5 years postoperatively: 4/43 (9.3%) SUBs experienced a skin erosion around the shunting port and required revision surgery during this study period. Skin erosion was observed at the medial aspect of the shunting port (Figure 5). Shunting port skin erosions were observed 12–36 months after SUB 2.0 placement (SUB 2001K Kit; Norfolk Vet Products), and no patient encountered this complication in the first generation of SUB (SUB 1001K Kit; Norfolk Vet Products).

Shunting port skin erosion in cats after subcutaneous ureteral bypass (SUB) placement. Skin erosion was observed at the medial aspect of the shunting port at 12–36 months postoperatively. Of 43 cats in the SUB group, four (9.3%) developed this late complication and required revision surgery. All cases occurred in the second-generation SUB system (SUB 2.0 Kit; Norfolk Vet Products), and no skin erosion was recorded in the first-generation SUB devices
In the ureterotomy group, uroabdomen occurred in 2/44 (4.5%) cats; however, none of these cases required revision surgery, none were fatal and all were self-limiting with resolution within 48 h. Reobstruction in the ureterotomy group occurred in eight cats, and all cats had recurrent ureterolithiasis, which was caused by a previously diagnosed nephrolith, and resolved with additional ureterotomy or SUB. None of these eight ureters had strictures from the first ureterotomy or stone formation at the previous site, with the new ureterolith obstructed distally to the initial site in all cats.
Ten-day postoperative mortality rate
Five cats (4.8%) died within 10 days postoperatively: one (4.2%) in the stent group, three (8.1%) in the SUB group and one (2.3%) in the ureterotomy group. Cause of death included progressive renal failure (SUB n = 2), neural deficits (eg, seizure, deteriorating consciousness) (SUB n = 1) or shock (ureterotomy n = 1, stent n = 1), most likely secondary to severe acute kidney injury and associated hemodynamic or electrolyte derangements. The cat that developed seizures had exhibited twitching before surgery and showed severe preoperative metabolic derangements consistent with uremic encephalopathy. Although the biochemical abnormalities improved postoperatively and the cat was discharged in a stable condition, it developed acute seizures at home on postoperative day 3 and died.
Survival time
MST was 1426 days in the stent group, but was not reached in the SUB or ureterotomy groups during our study.
The overall mortality rates during our study period in the stent, SUB and ureterotomy groups were 33%, 30% and 23%, respectively. No statistical difference in mortality among surgical procedures was observed during our follow up period (P = 0.87) (Figure 6).
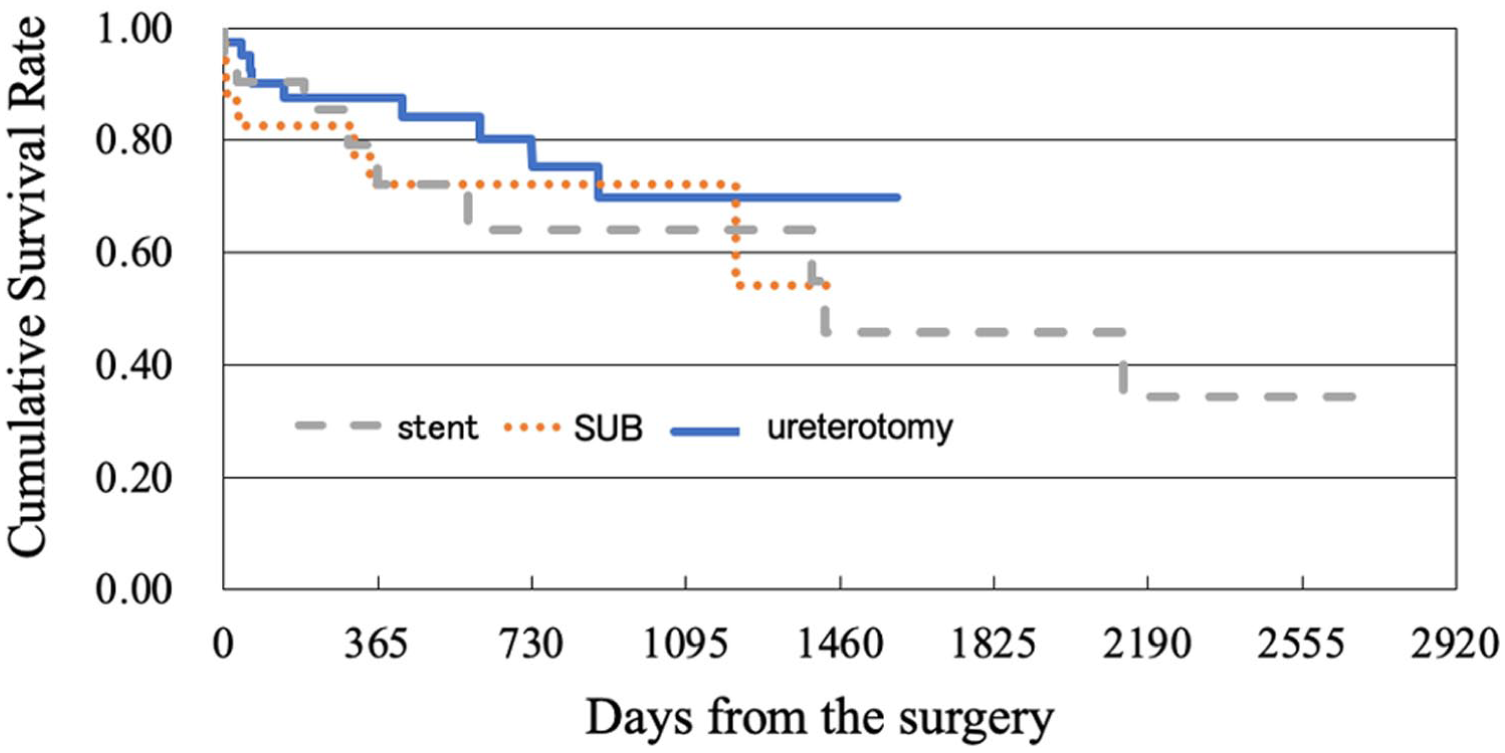
Kaplan–Meier survival curve after surgery. The x-axis shows days from surgery and the y -axis shows cumulative survival rate. The stent group (gray dashed line), subcutaneous ureteral bypass (SUB) group (orange dotted line) and ureterotomy group (solid blue line) had overall mortality rates of 33%, 30% and 23%, respectively, during the follow-up period. No statistically significant difference in mortality was observed among groups (P = 0.87)
Discussion
This study demonstrated notably improved outcomes in the ureterotomy group compared with previously published reports.3,4,6,7 –10 The ureterotomy group showed comparable duration of surgery and hospitalization, the lowest level 3 postoperative complication rate during long-term follow up, a markedly reduced short-term mortality rate (2.3% vs previously reported 8–30%)4,7 –10 and an 80% survival rate over a median follow-up of 18 months. These findings support our hypotheses and suggest reconsideration of ureterotomy as a viable and favorable surgical option for feline ureterolithiasis.
Several factors likely contributed to the improved outcomes in our ureterotomy group: the high case volume, consistent surgical technique and perioperative management by a single experienced surgeon, and the procedural learning curve – with our team evolving from early stent use, transitioning to SUBs and more recently favoring ureterotomy for cats with ureteroliths. This continuity and experience may have influenced short- and long-term outcomes and represents an important source of selection bias.
The incidence of uroabdomen in ureterotomy cats was low (4.5%) in our study population, contrasting with previous reports ranging from 6% to over 30%.2,4,10 This reduction may reflect improved surgical skill or a strategy of conservative postoperative fluid management.
In contrast, stent and SUB procedures were associated with higher rates of complications, especially long-term issues. Shunting port-associated skin erosion was identified as a delayed complication in 4/43 (9.3%) ureters in the SUB group, typically developing 12–36 months postoperatively. Affected cats often had poor body condition score (1–2/5) and lesions included superficial skin ulceration and port exposure, requiring revision surgery ranging from debridement to device removal or ureteronephrectomy. Access port skin erosion, which is rarely reported in cats with a SUB, 25 is recognized as a long-term complication in the human literature, with a 0.6–9% occurrence.26 –29 This is also noted in manufacturer documentation (Norfolk Medical Products, Bard). All four cases in our study were initially aseptic, with either negative cultures or skin flora differing from urine isolates, suggesting a non-infectious, multifactorial etiology. Contributing factors likely included subcutaneous fat reduction, dermal thinning, as reported in human medicine,26 –29 and possibly mechanical pressure from the larger SUB 2.0 port. The delayed onset and recurrence, despite medical or surgical management, highlight the need for long-term monitoring, especially in underconditioned patients.
Across all groups, serum Cre concentrations decreased at 1 month and remained relatively stable at 3–24 months postoperatively (approximately 2.0–3.0 mg/dl), suggesting initial improvement in renal function after relief of obstruction followed by stabilization at a chronic kidney disease stage determined by pre-existing nephron loss. This pattern may explain the lack of significant differences in long-term survival among groups. Cre stabilization by approximately 3 months postoperatively across all groups aligns with known renal recovery physiology after relief of ureteral obstruction. Experimental studies in dogs have demonstrated that the glomerular filtration rate improves rapidly after decompression and typically plateaus within 4–12 weeks, depending on the duration of the obstruction. 29 Similar patterns have been reported in clinical feline studies. Cats treated with SUB show the greatest reduction in serum Cre during the first 1–3 months postoperatively, with minimal change thereafter. 11 Ureteral stent-treated cats likewise demonstrate early improvement followed by stabilization over long-term follow-up.14,15 The consistent plateau observed in our population therefore likely reflects expected renal physiological recovery and suggests that long-term Cre values primarily reflect the degree of irreversible nephron loss present at the time of obstruction relief.
Ureterotomy cats exhibited the most significant and consistent improvements in PCV over 12 and 24 months, and BW over 3, 6, 12 and 24 months postoperatively, in contrast to the weight loss observed in stent cats and static trends in SUB cats. It is possible that improved appetite and nutritional status were facilitated by fewer persistent lower urinary tract signs (eg, pollakiuria, dysuria)9,13 –15 compared with cats in which a ureteral stent or SUB device had been placed. PCV and BW are easily measurable parameters, and improvements may therefore represent markers of recovery and potentially predictors of long-term clinical success. Although PCV values in the stent group appeared slightly higher than those in the SUB group at certain time points, these differences were not statistically significant, and only the ureterotomy group demonstrated a significant long-term increase in PCV. The mild elevation observed in some stent cats should be interpreted cautiously, as PCV is a multifactorial parameter influenced by hydration status, nutritional state, erythropoietin responsiveness and chronic inflammation – including potential inflammatory reactions associated with indwelling implants such as stents or SUB devices. Because serum Cre concentrations did not differ among groups over time, a renal-mediated explanation for PCV differences is unlikely.
In contrast, the sustained PCV improvement in the ureterotomy group, accompanied by parallel gains in BW, likely represents a true clinical improvement and may reflect reduced lower urinary tract signs, improved appetite and overall better systemic recovery in these cats.
This study has several limitations. Patients were inconsistently treated before transfer to our facility, and because of the retrospective nature of this study, potential biases such as inconsistent medical record documentation or missing data may have affected the results. Preoperative intravenous fluid therapy may also have influenced biochemical and hematologic values. Although all cats received fluid therapy before surgery, variation in the duration and intensity of treatment may have contributed to hemodilution and lower preoperative PCV values in some cats, as well as variable effects on renal parameters. Because documentation regarding perioperative blood transfusion was incomplete, the exact number of cats receiving transfusions could not be reliably determined. However, transfusions, when given, occurred only in the immediate perioperative period, and no cats received transfusions after discharge. In addition, there were no significant differences in PCV between groups at baseline or at 1 month postoperatively, suggesting that perioperative transfusion was unlikely to have influenced long-term PCV assessment. Urinalysis, stone composition analysis, urine culture results and the cause of obstruction (eg, stone, soft tissue, stricture) were not incorporated into the analysis, which limits interpretation regarding renal function, potential infectious contribution and risk of recurrence. All cats were treated at a single institution by the same surgeon, but at different time points over several years, with the potential for skills and protocol changes to influence outcomes. Procedure allocation was not randomized; device availability and evolving surgeon preference also influenced the choice of technique. Although cats in the SUB group were significantly older than those in the stent group, age did not significantly affect survival or the hazard of major complications in our multivariable analyses, suggesting that age was unlikely to have influenced the comparative outcomes among procedures. These factors limit the generalizability of our findings and should be considered when comparing groups. Although long-term follow up was included, the study period was not sufficient to assess the overall survival time or capture delayed complications such as postoperative level 3 events. In addition, MST was not reached for either the SUB or ureterotomy groups. A prospective, randomized clinical trial would be needed to meet these goals.
Conclusions
Ureterotomy demonstrated favorable outcomes for feline benign ureteral obstruction, with the lowest rate of major revision surgery, a low 10-day mortality rate comparable to other procedures and no significant differences in long-term survival among techniques. Cats undergoing ureterotomy showed the most consistent improvements in PCV and BW, suggesting enhanced clinical recovery and long-term stability. These findings challenge historical concerns regarding the morbidity of ureterotomy and indicate that its risks may have been previously overestimated. Although results reflect a single-center, single-surgeon experience and should be interpreted accordingly, ureterotomy appears to be a practical, effective and durable option when applied to appropriately selected cases.
Footnotes
Acknowledgements
We would like to express our sincere gratitude to Toshihito Furukawa (Biostatistical Research) and Dr Kimi Togari for their valuable contributions to the statistical analysis in this study.
Author note
This study was presented in part at the 2025 Society of Veterinary Soft Tissue Surgery Annual Meeting.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
