Abstract
Objectives
Anaplasma phagocytophilum is considered the most significant rickettsial pathogen affecting cats. The organism is transmitted by ticks of the species Ixodes ricinus in Central Europe and can cause granulocytic anaplasmosis in pets, wildlife and humans. The aims of this study were to assess the frequency of positive test results for A phagocytophilum in cats in Germany, Austria and Switzerland, as well as to evaluate clinical and laboratory findings in cats with positive PCR results.
Methods
This study included the results of direct (PCR) and indirect detection methods (immunofluorescence antibody tests [IFAT]) requested by veterinarians in Germany, Austria and Switzerland between 2008 and 2020 from the LABOKLIN laboratory (Bad Kissingen, Germany). The veterinarians treating the PCR-positive cats were contacted by telephone to enquire about their clinical signs, laboratory findings, management and outcomes.
Results
In total, 244/1636 cats (15%) tested positive by direct (PCR: n = 27/725 [4%]) and/or indirect detection methods (IFAT: n = 221/956 [23%]). In 18/26 cats with PCR results positive for A phagocytophilum, additional information about clinical signs, laboratory findings, treatment and outcome was obtained. Of these 18 cats, five had comorbidities independent of their infection with A phagocytophilum. The most common clinical signs in PCR-positive cats (total/without comorbidities) were lethargy (83%/92%), fever (83%/85%) and thrombocytopenia (61%/62%). Overall, more than half (57%) of the cats with and without comorbidities recovered clinically.
Conclusions and relevance
Infections with A phagocytophilum should be considered as differential diagnoses in cats with tick infestation, lethargy, fever and thrombocytopenia. The clinical signs and laboratory findings are consistent with published case reports in cats. Ectoparasite prophylaxis in cats is recommended throughout the entire year.
Introduction
Anaplasma phagocytophilum is a Gram-negative, obligate intracellular bacterium. 1 It is known to cause granulocytic anaplasmosis in humans, as well as domestic and wild animals. 2 The distribution of the pathogen is largely dependent on the prevalence of its primary vector, ticks of the Ixodes species. 3 Diagnostic methods include PCR testing and/or testing for antibodies to the pathogen in paired serum samples over 4 weeks, with a four-fold increase in antibody titres. 4 The microscopic detection of morulae is highly suggestive but has low sensitivity. Dye residue, nuclear fragments or basophilic precipitants in blood smears may be mistaken for morulae. 5 The diagnosis of acute infection should thus be based primarily on PCR results. 4
A phagocytophilum has been detected by PCR and/or demonstration of morulae in blood smears in 0–23% of cats tested in Europe, while antibodies were detected in 0–33%. 6 Among cats tested by PCR in Germany, 0.3% (Bavaria/Lower Saxony) 7 and 0.4% (Southern Germany, Berlin/Brandenburg)8,9 tested positive. Immunofluorescence antibody tests (IFATs) revealed serum antibodies in 16% of cats from Bavaria/Lower Saxony, 7 and 9% of cats from Berlin/Brandenburg. 9 A literature review reported clinical signs in 33/34 cats (97%), primarily lethargy (94%), fever (88%) and anorexia (76%). The main laboratory abnormalities were thrombocytopenia (59%) and anaemia (26%). 6
The aim of the study was to evaluate the number of cats with positive IFAT and/or PCR results in Germany, Austria and Switzerland, as well as to evaluate cats with positive PCR results in terms of clinical signs, additional laboratory findings, management and outcome.
Materials and methods
This study included direct and indirect detection assays for A phagocytophilum in cats for which samples were provided by veterinarians working in Germany, Austria or Switzerland to the LABOKLIN laboratory (Bad Kissingen, Germany) between January 2008 and December 2020. Direct detection methods included a TaqMan real-time PCR (Applied Biosystems/Life Technologies) using EDTA blood. Sequencing of PCR-positive tested samples was performed to confirm A phagocytophilum infection. MegaFLUO ANAPLASMA (MegaCor Diagnostik; ⩾1:40 positive) was used to detect antibodies in serum. Initial samples sent in for each cat were included. Further test results, such as platelet-bound antibody testing (LABOKLIN; Immunology Study Group, University of Veterinary Medicine Hannover, Germany), Coombs tests (LABOKLIN), testing for feline leukaemia virus (FeLV) antigen and feline immunodeficiency virus (FIV) antibodies (on-site testing in clinics; NovaTec VetLine Feline Immunodeficiency Virus ELISA and NovaTec VetLine Feline Leukemia Virus Antigen ELISA [NovaTec Immundiagnostica]) and PCR testing for additional pathogens (haemotropic Mycoplasma species; real-time PCR, based on Peters et al, 10 [LABOKLIN]), were additionally evaluated, if available. The veterinarians who provided samples from cats with positive PCR results were contacted by telephone to obtain information regarding history, clinical findings, laboratory results, management and outcome. A descriptive statistical analysis was carried out in SPSS for Windows (version 27.0; IBM).
Results
A total of 1423 cats from Germany, 133 cats from Austria and 80 cats from Switzerland were included. Of the 1423 cats form Germany, 206 (15%) tested positive for A phagocytophilum by direct or indirect detection methods (18/619 cats [3%] by PCR, 191/847 cats by IFAT [23%]). The detection rate of antibodies to A phagocytophilum in cats in Germany increased from 8–12% of cats tested between 2008 and 2012 to 35–45% between 2018 and 2020 (Table 1). A similar trend was noted when analysing the PCR results as 0–2% of cats tested positive between 2009 and 2013 and 2–10% tested positive between 2017 and 2020. The highest incidence of cats that tested positive by either method occurred in Berlin/Brandenburg (total: 26/145 [18%]; PCR: 7/101 cats [7%]; IFAT: 21/49 cats [43%]). In other German federal states, 0–7% of cats tested positive by PCR, and antibodies were detected in 14–59% of cats examined; however, some regions were represented by only a small number of samples (Figure 1).
Annual prevalence of Anaplasma phagocytophilum in cats between January 2008 and December 2020 as provided by veterinarians working in Germany to the LABOKLIN laboratory

Data are n tested positive / n total (%)
IFAT = immunofluorescence antibody test

Prevalence of Anaplasma phagocytophilum in cats in Germany between January 2008 and December 2020 provided by veterinarians working in Germany to the LABOKLIN laboratory. Data are n tested positive / n total (%).
In Austria, 25/133 (19%) cats tested positive for A phagocytophilum (5/64 [8%] by PCR and 21/71 [30%] by IFAT). Of the 80 cats from Switzerland, 13 (16%) tested positive (4/42 [10%] by PCR, 9/38 [24%] by IFAT). Owing to the low total numbers of tested cats, a further analysis of separate regions within these countries was not included.
Of the cats with positive PCR results, further information was collected from the treating veterinarians for 11/18 cats in Germany, 3/5 cats in Austria and 4/4 cats in Switzerland. Twelve of 18 cats were European Shorthair, 2/18 were Maine Coon or mixed breed, respectively, and 1/18 cat each was LaPerm Longhair or British Shorthair. The median age of these 18 cats was 8 years (mean 7.9; range 9 months to 17 years). Sex was known in 16/18 cats (9/16 castrated males; 1/16 entire male; 4/16 spayed females; 2/16 entire females). Of the 18 cats, 15 had outdoor access, while living conditions were unknown for the remainder.Comorbidities were known in 5/18 cats.
In 13/18 cats, no comorbidities were identified. All cats without comorbidities showed clinical signs, and most commonly presented with lethargy (12/13 cats) and fever (11/13 cats, 39.2–41.4°C), followed by anorexia (6/13 cats) and lymphadenomegaly (5/13 cats) (Table 2). Haematology results were available for all 13 cats (Table 3), as well as biochemistry results in 10 cats (Table 4). The most common abnormality in the cats without comorbidities was thrombocytopenia (8/13 cats), which was confirmed by manual counting or blood smear evaluation in 6/8 cats. In 3/8 cats with thrombocytopenia, testing for platelet-bound antibodies was negative. One cat had a haemolytic anaemia (haematocrit 0.19 l/l, hyperbilirubinaemia, negative Coombs test). IFAT for A phagocytophilum (LABOKLIN) was performed in 2/13 cats; both tested positive. FeLV antigen and FIV antibody testing was performed in 3/13 cats; all tested negative. One cat was tested solely for FeLV antigen with a negative result. PCR testing for Haemoplasma species was performed in 4/13 cats and was negative in all. Imaging results were available for 3/13 cats, all of which had splenomegaly. In one of these three cats, a cytological investigation of splenic aspirates showed reactive hyperplasia of the white pulp, pyogranulomatous inflammation and morulae in neutrophilic granulocytes.Information about the therapy and outcome was available for 7/13 cats without comorbidities. All seven cats were treated with doxycycline 5 mg/kg q12h or 10 mg/kg q24h PO for 2–3 weeks. Owing to deterioration of disease, one owner decided to euthanase their cat after 2 days. After 3 weeks of treatment, four cats had recovered clinically. In two cats, which had been treated for 2 and 3 weeks, respectively, a clinical relapse or reinfection confirmed by positive A phagocytophilum PCR occurred 5 weeks and 2 years, respectively, after finishing the therapy. Both cats were tested again because of relapsing fever and lethargy; testing for A phagocytophilum immediately after finishing treatment was not performed for either cat.
Clinical abnormalities in 18 cats tested positive positive for Anaplasma phagocytophilum by PCR at first presentation

Data are n (%)
Comorbidities: Mycoplasma haemofelis (n = 2); hyperthyroidism (n = 1); lymphoma (n = 1); paralytic ileus (n = 1)
Range 39.2–41.4°C (median 40.2°C, mean ± SD 40.1 ± 0.64ºC)
Ascites probably linked to the paralytic ileus diagnosed during exploratory laparotomy
Complete blood count results of 18 cats that tested positive for Anaplasma phagocytophilum by PCR at the time of first presentation (different laboratories)
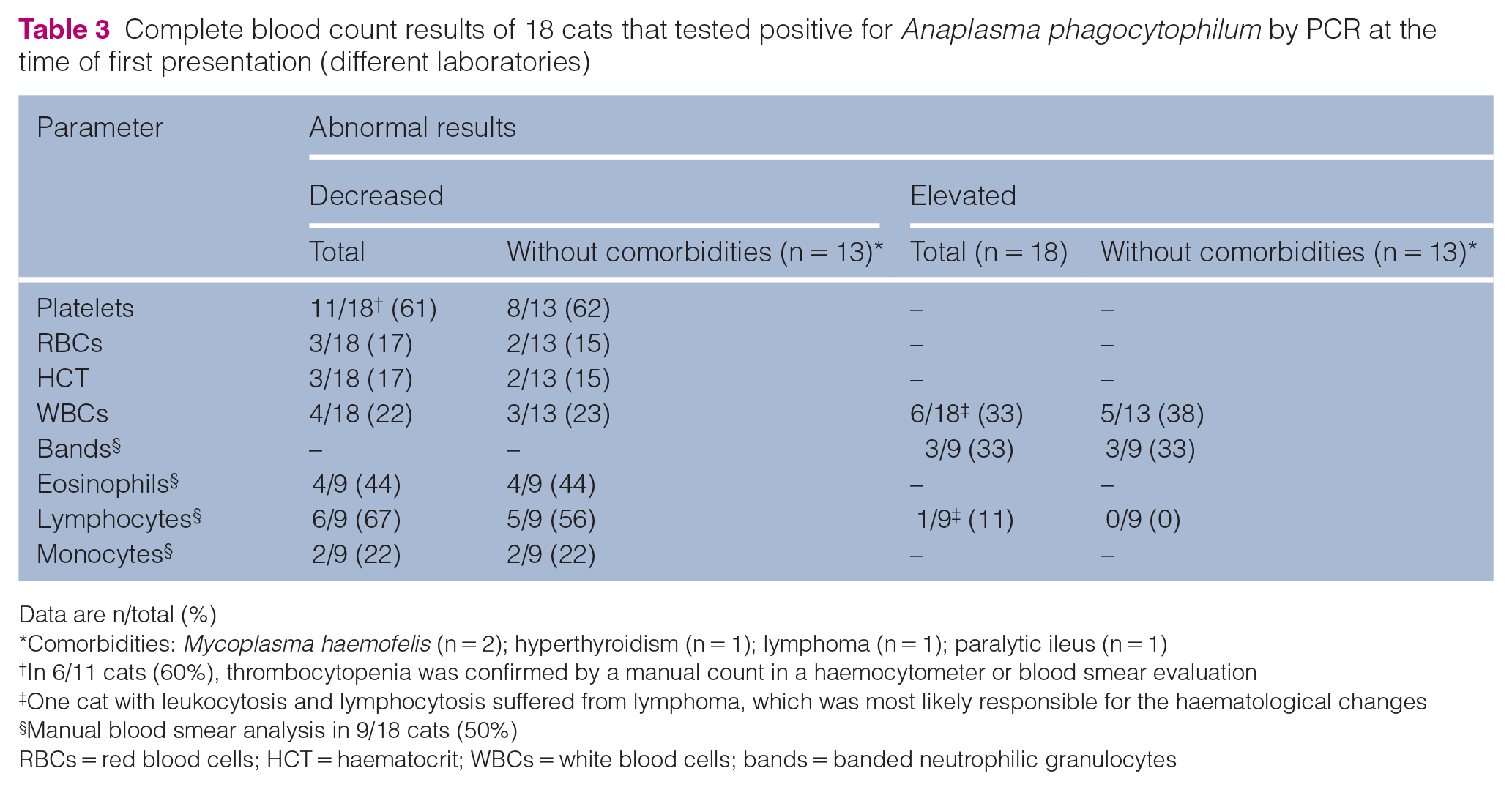
Data are n/total (%)
Comorbidities: Mycoplasma haemofelis (n = 2); hyperthyroidism (n = 1); lymphoma (n = 1); paralytic ileus (n = 1)
In 6/11 cats (60%), thrombocytopenia was confirmed by a manual count in a haemocytometer or blood smear evaluation
One cat with leukocytosis and lymphocytosis suffered from lymphoma, which was most likely responsible for the haematological changes
Manual blood smear analysis in 9/18 cats (50%)
RBCs = red blood cells; HCT = haematocrit; WBCs = white blood cells; bands = banded neutrophilic granulocytes
Biochemistry results of 15/18 cats that tested positive for Anaplasma phagocytophilum by PCR at the time of first presentation (different laboratories)

Comorbidities: Mycoplasma haemofelis (n = 2); hyperthyroidism (n = 1); lymphoma (n = 1); paralytic ileus (n = 1)
In the cat with azotaemia and increased alanine aminotransferase (ALT) and alkaline phosphatase (AP), chronic renal failure and hyperthyroidism were present
AST = aspartate aminotransferase
Comorbidities were present in 5/18 cats. One cat had been diagnosed with hyperthyroidism and chronic kidney disease (CKD). In four cats, comorbidities were detected at the index visit: 2/4 cats had positive PCR results for Mycoplasma haemofelis (none was anaemic), one cat was diagnosed with lymphoma and one cat suffered from a paralytic ileus with ascites of unknown origin. Fever and anorexia (4/5 cats), as well as lethargy (3/5 cats) were the main presenting complaints (Table 2). The cat with lymphoma had a haemolytic anaemia (haematocrit 0.12 l/l, hyperbilirubinaemia, negative Coombs test). FeLV antigen/FIV-antibody testing was performed in 3/5 cats (two cats coinfected with M haemofelis, one cat with ileus); all tested negative. One cat with hyperthyroidism tested negative for FIV antibodies.Management and outcomes were known in 4/5 cats with comorbidities. One cat was euthanased owing to a lymphoma. One cat with a paralytic ileus was initially treated with doxycycline (10 mg/kg q24h PO for 2 days), which was subsequently changed to enrofloxacin (5 mg/kg q24h PO) owing to vomiting. This cat recovered and did not have any clinical signs after completing treatment. The remaining two cats had additional infections with M haemofelis and were both treated with doxycycline (10 mg/kg q24h PO for 2 and 3 weeks, respectively). One cat recovered, while the other cat re-presented with similar signs and tested positive for A phagocytophilum by PCR 4 weeks after completing treatment.
Discussion
A total of 244/1636 cats (15%) from Germany, Austria and Switzerland tested positive for A phagocytophilum between 2008 and 2020. There seems to be a general trend of increasing rates of anaplasmosis in dogs and humans.11,12 The findings of this study suggest a similar trend in cats. This may be due to increased awareness and consideration of such infections as differential diagnoses in both human and veterinary medicine, or the change in climate and vector habitats, as well as the prevalence of pathogen reservoirs. 13
Twenty-seven of 725 cats (4%) tested positive by PCR. PCR testing has a high sensitivity in acute infection. Nevertheless, false-positive results may occur as a result of the detection of other pathogens, such as Anaplasma platys and Pseudomonas species, 14 but this was excluded by sequencing. Previously, individual cats in Germany that tested positive by PCR have been reported. In a study from Berlin/Brandenburg, 1/265 cats (0.4%) tested positive (49/265 [18%] clinically healthy, 216/265 [81%] with different clinical signs). 9 Further cases of cats with positive test results included 1/306 cats (0.3%) from Bavaria/Lower Saxony (10/306 cats [3%] suspicious for A phagocytophilum infection, 58/306 clinically healthy stray cats [19%], remainder were random diagnostic samples) 7 and 2/497 cats (0.4%) from southern Germany (random diagnostic samples). 8 In the present study, 18/619 cats (3%) from different regions of Germany had positive PCR results. It should be noted that PCR testing for A phagocytophilum may be requested by treating veterinarians owing to clinical suspicion for an infectious disease such as anaplasmosis. This may explain the higher prevalence compared with the abovementioned studies.
In total, 221/956 cats (23%) were found to be antibody positive by IFAT, being well within the range reported in the literature. 6 The assay used in this study detected A phagocytophilum IgG antibodies, and positive serology may be interpreted as the consequence of past pathogen contact. Interpreting serology results may be complicated by a lack of seroconversion in acute infections, persistently high antibody titres and possible cross-reactivity, particularly with A platys or Ehrlichia species.2,4,13,15 However, these two pathogens are transmitted by Rhipicephalus sanguineus in southern parts of Europe and are not endemic in Germany, which means that the risk of vector contact and transmission of these two pathogens is small. In addition, although this study did not consider the possibility of some cats being imported from other countries or travelling with their owners abroad from Germany, this is less of a factor for cats as for dogs. In the light of these considerations, the impact of the abovementioned cross-reactions appears to be minimal. The interpretation of IFAT can be subjective and sensitivity may decrease, especially in cats with low or borderline titres. This may especially be considered in the 119 cats with a low titre of 1:40.
With regard to regions, the highest proportion of cats that tested positive either by PCR or for antibodies was, besides Saxony-Anhalt, in Berlin/Brandenburg, where a comparatively high percentage of ticks infected with A phagocytophilum (7%) has been reported. 16 Other areas with a similarly high prevalence of Ixodes ticks infected with the pathogen were the Saarland states, with a prevalence of 17% 17 and Bavaria with 9%.17,18 Too few samples were provided for cats from the Saarland states (n = 16) to draw any conclusion (Figure 1). In Bavaria, 2% of cats tested positive by PCR and 18% tested positive by IFAT, which is below the overall mean for cats in Germany (PCR 4%, IFAT 23%) (Figure 1). However, this should not be interpreted as more than a tendency, and warrants further studies with higher numbers of tested cats.
Overall, the clinical and laboratory findings of the cats in this study are consistent with A phagocytophilum infections as described in the literature. 6 Thrombocytopenia was seen in 8/13 cats without known comorbidities and represents the most common laboratory abnormality. In cats, falsely low values may occur in mechanical platelet counting during impedance measurement due to platelet aggregates, as feline platelets are more prone to aggregate, and a relative lack of size difference to erythrocytes compared with other species. 19 In 6/8 cats in this study, thrombocytopenia was confirmed by manual chamber counting or blood smear evaluation. Leukocytosis and leukopenia were seen almost equally, which is consistent with the previous literature. 6 Anaemia is the second most common haematological abnormality reported. 6 Anaemic cats were additionally screened for the presence of haemotropic Mycoplasma species by means of PCR, as this may be a relevant differential diagnosis (all tested negative). A common blood chemistry finding was hyperglycaemia, which was present in 4/5 cats tested but was most probably a physiological response to stress. 20 Hyperproteinaemia was present in 3/10 cats tested, two of which had hyperglobulinaemia. This was most likely caused by antibody stimulation and/or inflammatory response to the A phagocytophilum infection.
Comorbidities were known in 5/18 cats. Two cats had coinfections with M haemofelis. This pathogen may cause haemolytic anaemia of varying severity, 21 but neither cat was anaemic. Both cats tested negative for FeLV antigen and FIV antibodies. This suggests that their clinical presentation and laboratory findings (inappetence, fever, lethargy and thrombocytopenia, as well as hyperglobulinaemia in one cat) can be attributed to their infection with A phagocytophilum. One cat was diagnosed with lymphoma. The leukocytosis with lymphocytosis found in this cat is most likely due to its underlying pathology. The immunosuppression associated with both lymphoma and haemoplasma infection may increase susceptibility to anaplasmosis. One cat had hyperthyroidism as well as CKD. Its azotaemia and elevated levels of alanine aminotransferase and alkaline phosphatase were most likely due to these underlying diseases, while lethargy, fever and leukocytosis might be due to A phagocytophilum infection. The clinical and laboratory abnormalities in the cat with paralytic ileus and ascites might have been caused by both conditions (ileus as well as ascites).
A phagocytophilum is an obligate intracellular pathogen, and, as such, antibiotics must penetrate the cell itself in order to be effective.22 –25 In accordance with case studies, doxycycline is currently regarded as the best treatment option in dogs and cats,4,6 and should be given at a dosage of 10 mg/kg q24h PO over 4 weeks 4 . However, as in dogs, there have been cases of cats in which infections resolved spontaneously without antibiotic treatment. 26 Eleven of 12 cats in this study were treated with doxycycline at 5 mg/kg q12h or 10 mg/kg q24h PO for 2–3 weeks. The duration of treatment was therefore shorter than recommended. 4 This was largely due to a lack of compliance in pet owners once clinical signs regressed, which has also been described in the literature. 6 In three cats treated with doxycycline for either 2 (n = 1) or 3 (n = 2) weeks, there was a second episode of clinical signs (fever and lethargy) and A phagocytophilum PCR testing within 4 weeks, 5 weeks and 2 years of treatment, respectively, was again positive. No PCR testing had been performed in order to confirm resolution of the infection after discontinuation of doxycycline in any of these three cats. Thus, persisting infection or reinfection after a separate contact with an infected vector may have occurred. Persisting infections have been described in dogs, horses and sheep that were experimentally infected with A phagocytophilum,27 –29 and their significance in natural infections is controversial. One study of 16 cats in the USA reported that the pathogen was not detectable by PCR after treatment with doxycycline for 21–45 days (most commonly 3 weeks). 20 One study detected the pathogen by PCR for up to 120 days after a 4-week course of doxycycline in cats. 20
Limitations
The limitations of this study include its retrospective design and small case numbers from Austria and Switzerland. Information on any time spent abroad was not available for any of the cats included. Furthermore, there were no data on ectoparasite prophylaxis or the living conditions of the cats, both of which could impact on pathogen prevalence. Information about presentation, clinical signs or laboratory results was not available for all cats. The 18 PCR-positive cats in Germany, Austria and Switzerland were all treated by different veterinarians. Laboratory testing was performed not only in their clinics, but also in separate external laboratories, which affects the comparison of different haematological and blood chemistry parameters. The extent to which the cats with PCR-confirmed infection were further investigated for coinfections or underlying pathology also varied substantially.
Conclusions
This is the first study to report the rate of positive detection assays for A phagocytophilum in a diagnostic laboratory for cats in Germany, Austria and Switzerland. This was also the first study to monitor changes in the incidence of A phagocytophilum in cats detected by direct, as well as indirect, detection methods over a time frame of 13 years. During this period, there was a notable increase in positive antibody assays, which may indicate increasing contact with the pathogen and/or increasing awareness. An infection with A phagocytophilum should be considered in any case where clinical signs such as lethargy, loss of appetite and fever are present in outdoor cats from Ixodes-endemic areas. Thrombocytopenia was the most prevalent haematology finding, but anaemia, leukocytosis and leukopenia were also seen. Currently, it is recommended that infections are managed with a 4-week course of doxycycline, which should be continued regardless of a possibly faster resolution of clinical signs in order to reduce the risk of recurrence. There is generally a good prognosis, and patients recover clinically shortly after initiating antibiotic therapy. Owing to the risk of vector-transmitted infections in cats, ectoparasite prophylaxis should be recommended throughout the year.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
