Abstract
Objectives
The study aimed to evaluate the efficacy and safety of insulin glargine 300 U/ml (IGla-U300) in cats with variable duration of diabetes mellitus (DM).
Methods
Thirteen client-owned cats with DM completed a prospective clinical trial. Four cats were highly suspected of hypersomatotropism and excluded from the insulin efficacy evaluation. All cats were treated with IGla-U300 SC at a starting dosage of 0.5 U/kg q12h and fed with a low carbohydrate diet. Cats were monitored for 8 weeks with a once-weekly at-home 16 h blood glucose curve (BGC) and a questionnaire evaluating the presence of DM-related clinical signs. In-clinic evaluations, including serum fructosamine measurement, were scheduled within 3 days of the first, third, sixth and eighth BGC. Glycemic variability was assessed by calculating the SD of each BGC.
Results
Excluding four cats suspected of hypersomatotropism, at the time of the eighth BGC, improved or absent polyuria, polydipsia, polyphagia, weight loss, lethargy and improved or normal general demeanor were reported in 8/9 (88%), 8/9 (88%), 7/9 (77%), 7/9 (77%), 7/9 (77%) and 8/9 (88%) cats, respectively. Two cats achieved remission after 29 and 53 days. Another two cats went into remission after the end of the study (days 82 and 96). All cats that achieved remission were newly diagnosed diabetics. Median (range) serum fructosamine concentration significantly decreased when comparing the time of enrollment (604 [457–683] µmol/l) with the eighth week of treatment (366 [220–738] µmol/l) (P = 0.02). In all 13 cats, biochemical hypoglycemia (blood glucose <60 mg/dl; <3.3 mmol/l) was detected in 13/104 (12.5%) BGCs, while clinical signs suggesting hypoglycemic episodes were not reported. Glycemic variability was significantly lower at the fifth BGC when comparing cats that achieved remission with cats that did not achieve remission (P = 0.02).
Conclusions and relevance
IGla-U300 seems effective and safe for the treatment of feline diabetes, but more long- term and comparative clinical trials are needed.
Introduction
Long-acting insulin products are currently considered the first choice for feline diabetes mellitus (DM) treatment.1,2 Glargine insulin 100 U/ml (IGla-U100) has been evaluated previously in diabetic cats, resulting in effective glycemic control.3,4 Recently, a new insulin glargine product with an increased concentration of 300 U/ml (IGla-U300) was approved for treatment of human type 1 diabetes mellitus (T1DM) and type 2 DM (T2DM). 5 IGla-U300 formulation comprises the same molecule as IGla-U100 that is delivered in a third of the original volume, which results in a smaller subcutaneous depot and reduced surface area, leading to a slower re-dissolution rate, lower bioavailability and increased daily dose in human patients. 6 These features probably explain the flatter and more prolonged insulin profiles that were obtained with IGla-U300 when compared with IGla-U100 in human patients.7,8 Clinical trials comparing IGla-U300 with IGla-U100 resulted in similar achievements of HbA1c targets, but a reduced risk of hypoglycemia in T1DM and T2DM human patients treated with IGla-U300.6,9–16 Comparison of the pharmacodynamics of IGla-U300 and IGla-U100 in healthy cats using the isoglycemic clamp resulted in similar duration of action, but importantly, IGla-U300 had a flatter time-action profile than IGla-U100. 17 Similarly, IGla-U300 was shown to have a flatter time-action profile than insulin degludec using the isoglycemic clamp method in healthy cats, and also had an average 4 h longer duration of action than degludec; generally longer than 16 h for IGla-U300. 18 Currently, clinical trials evaluating IGla-U300 in diabetic feline patients have not been performed yet. The present study aimed to assess the efficacy and safety of IGla-U300 in privately owned cats diagnosed with DM.
Materials and methods
This prospective clinical trial was conducted at the Veterinary Teaching Hospital of the University of Bologna, Italy, from December 2018 to July 2020. The trial was approved by the Scientific Ethics Committee of the University of Bologna (number 1084/2019).
DM diagnosis and exclusion criteria
DM diagnosis was performed according to the Agreeing Language In Veterinary Endocrinology (ALIVE) criteria (Table 1) established by the European Society of Veterinary Endocrinology (ESVE). 19 Client-owned cats, both newly diagnosed or previously treated with insulin, were enrolled. Cats having already received insulin were enrolled only if their DM was poorly controlled or if they were experiencing a relapse of diabetes following a period of remission as defined by the ALIVE criteria, 19 while newly diagnosed cats were defined as having never received insulin before enrollment. Cats treated with corticosteroids or progestagens during the previous 60 days from enrollment and cats with relevant concurrent chronic disorders potentially affecting overall health and life expectancy (eg, neoplasia) were excluded. Patients with diabetic ketoacidosis (DKA) or acute pancreatitis were included after clinical signs of the concurrent disorder resolved and their general condition had improved. Cats with a high suspicion of hypersomatotropism (HS) (serum insulin-like growth factor 1 [IGF-1] concentration > 1000 ng/ml; > 131 nmol/l) were enrolled but excluded from the insulin efficacy evaluation. Diagnostic investigations for acute pancreatitis or other concurrent diseases were performed if justified by a clinical suspicion.
Definition of feline diabetes mellitus (DM) diagnosis according to the Agreeing Language In Veterinary Endocrinology (ALIVE) project of the European Society of Veterinary Endocrinology (ESVE)

Applying the ALIVE criteria permits the possible existence of a subpopulation of cats where DM or stress hyperglycemia cannot be confidently confirmed or excluded; if concerns over the presence of DM persist, periodic re-evaluation is warranted
The potential for stress hyperglycemia warrants caution in interpretation of hyperglycemia of any magnitude in cats
Study design
At time of enrollment, a physical examination was performed, then a complete blood count (CBC), serum biochemistry, comprehensive of fasting serum glucose and 1,2-o-dilauryl-rac-glycero glutaric acid-(6′-methylresorufin) ester (DGGR) lipase activity, fructosamine, total thyroxine (T4) and IGF-1 concentrations were measured. Body condition score (BCS) was evaluated at enrollment and at the end of the study. A BCS of 5/9 was considered normal. In newly diagnosed diabetic cats, IGF-1 concentrations were measured after 6 weeks of insulin treatment. All cats enrolled were treated with IGla-300 (Toujeo SoloSTAR pen, Sanofi), 5 starting at a dose of 0.5 U/kg (rounded down to the nearest whole unit) of ideal body weight (estimated evaluating BCS), administered subcutaneously q12h using the manufacturer’s pre-filled insulin dosing pen and were fed low carbohydrate (< 15% ME; < 5 g/100 kcal) diets. In cats already on treatment for DM, the starting insulin dose was the same as the last dose of the previous insulin administered. In addition, cats were allowed to eat the same low carbohydrate diet received before enrollment.Owners were instructed how to administer insulin according to the manufacturer’s guidelines using the insulin dosing pen, and how to measure blood glucose with a veterinary portable blood glucose meter (PBGM). Owners were asked to perform a 16 h blood glucose curve (BGC), measuring blood glucose every 2 h, at home, once weekly for 8 weeks. Owners were given the option of performing the first at the Veterinary Teaching Hospital, to get used to the management of DM slowly. The same day the BGC was performed, the owner had to answer a questionnaire that was provided at the time of enrollment to evaluate the presence of clinical signs related to DM.In-clinic evaluations were scheduled within 3 days after the first, third, sixth and eighth BGCs, in which a full physical examination (including body weight measurement) and biochemical evaluation with serum fructosamine concentrations were performed. Every biochemistry evaluation was performed after a minimum fasting period of 8 h. Owners were instructed to withhold the insulin administration on the morning of the evaluation. Insulin dose adjustments (Table 2) were determined by a single clinician (GL) after evaluation of the owner’s response to the questionnaire, results of BGCs, 24 h water intake measurement and serum fructosamine concentration. The last dose prescribed before the cessation of insulin therapy because of remission was 1 unit administered once daily in the morning. In accordance with ALIVE criteria, diabetic remission was defined as the absence of clinical or biochemical evidence of DM for at least 4 weeks after cessation of insulin therapy. 19 Presence of biochemical hypoglycemia was defined when blood glucose concentration was <60 mg/dl (<3.3 mmol/l).
List of subsequent insulin 300 U/ml dose reductions used in diabetic cats entering remission

Clinical evaluation questionnaire
The questionnaire provided was composed of three sections; in the first, the owner had to assign a score ranging from 1–10 to evaluate the following: polyuria (1 = does not urinate, 5 = normal, 10 = extremely increased); polydipsia (1 = does not drink, 5 = normal, 10 = extremely increased); polyphagia (1 = does not eat, 5 = normal, 10 = extremely increased); weight loss (1 = severe weight loss, 5 = stable body weight, 10 = heavily increased); general demeanor (1 = strongly depressed, 5 = normal, 10 = extremely excited); and lethargy (1 = not sleeping, 5 = normal, 10 = extremely increased). In addition, in the same section, reporting the occurrence of gastrointestinal signs or signs related to hypoglycemic events was encouraged. The second section was designed to record the amount of water consumed in 24 h, measured within 2 days before or after the day the BGC was performed. The last section was dedicated to reporting blood glucose concentration results of the BGC.
Analytical methods
Owners were instructed to measure blood glucose concentration from capillary blood obtained from the inner or outer surface of the pinna, using a veterinary PBGM (AlphaTrak, Zoetis), that was previously validated for its use in cats. 20 Serum fructosamine and fasting glucose concentrations were measured using an automated chemistry analyser (AU480 Olympus/Beckman Coulter). Serum IGF-1 concentrations were measured using an automated solid-phase, enzyme-labelled chemiluminescent immunometric assay (Immulite 2000 IGF-1 assay, Diagnostic Products). 21
Statistical analysis
Statistical analysis was performed using a commercially available software (Prism V.5.01, GraphPad Software). Numeric variables were analyzed for normality with D’Agostino and Pearson tests and results were reported as mean (± SD) or as median and range (minimum-maximum value) for normally or non-normally distributed data, respectively. Fasting serum glucose concentration, serum fructosamine concentrations, water volume consumed and insulin doses were compared at different time points using the Wilcoxon paired test, while mean blood glucose concentration of BGCs was compared with the paired t-test. Within-day glycemic variability (GV) was evaluated by calculating the standard deviation of blood glucose concentrations for each BGC. GV of BGCs was compared between cats achieving remission and those that did not using the Mann–Whitney U test, and within groups at weekly time points using the Wilcoxon paired test. Evaluation of GV and time to nadir (time elapsed from insulin administration to the detection of the nadir) was performed with exclusion of BGCs in which insulin administration was interrupted or skipped because of diabetic remission or detection of hypoglycemia. Furthermore, BGCs in which insulin administration timing was not specified were excluded from the time to nadir evaluation as well as those in which insulin was administered once daily. Cats with HS were excluded from the efficacy evaluation. Therefore, these patients were only included in the evaluation of biochemical hypoglycemia, time to nadir, GV and insulin dose, and excluded from the evaluation of clinical signs, body weight, water consumption, fasting serum glucose, mean blood glucose of BGCs, serum fructosamine and remission. Cats not receiving insulin owing to remission were not considered for dose evaluation at the eighth week time point. All tests were two-tailed and significance was set at P ⩽0.05.
Results
Animal population
Thirteen privately-owned cats completed the study. Eight (61%) cats were newly diagnosed, four (31%) cats were enrolled because of poor glycemic control (two received porcine lente insulin for 172 and 125 days; two received IGla-U100 for 369 and 177 days) and one (8%) was enrolled consequent to DM relapse. Ten patients were domestic shorthair cats and three were domestic longhair cats; eight were neutered males and six spayed females. The mean age ± SD was 9.8 ± 2.6 years and the mean ± SD body weight was 5.1 ± 1.5 kg. The median (range) BCS was 4/9 (4–8/9) and 5/9 (3–8/9) at the time of enrollment and after 8 weeks of treatment, respectively. At the time of enrollment, 3/13 (23%), 10/13 (77%) and 0/13 (0%) cats had a BCS ⩾7/9 4–6/9 or ⩽3/9, respectively. After 8 weeks of treatment 4/13 (31%), 8/13 (61%) and 1/13 (8%) cats had a BCS ⩾7/9, 4–6/9 or ⩽3/9, respectively. Five cats were enrolled after DKA was resolved and one after concurrent acute pancreatitis. At the time of enrollment, the median (range) DGGR lipase activity was 36 (20–191; reference interval 6–120 U/l) U/l. In 3/13 (23%) cats, stage 2 non-proteinuric, non-hypertensive chronic kidney disease was diagnosed after the trial ended. Eleven cats were fed with Purina Pro Plan Diabetes Management (Nestlé Purina Commerciale) (four exclusively dry, two exclusively canned and five both formulations) and two with Royal Canin Diabetic dry diet (Royal Canin). In 4/13 (31%) cats, HS was highly suspected (IGF-1 >1000 ng/ml; >131 nmol/l). Of these, 3/4 (75%) were newly diagnosed diabetic patients.
Home blood glucose monitoring
A total of 104 BGCs were performed, eight for every cat enrolled. In 9/13 (69%) cats the first BGC was performed at the Veterinary Teaching Hospital. The median (range) duration of all BGCs performed was 14 (8–17) h and 14 (10.5–17) h if excluding BGCs due to an unknown precise time of insulin administration, once-daily insulin administration or skipped administration owing to the risk of hypoglycemia. In 25/74 (33.8%) of BGCs performed in patients receiving insulin twice daily the dose administered in the morning was 1 unit higher than the dose administered in the evening.
Efficacy and safety evaluation
At the time of the eighth BGC, improved or absent polyuria, polydipsia, polyphagia, weight loss, lethargy and improved or normal general demeanor were reported in 8/9 (88%), 8/9 (88%), 7/9 (77%), 7/9 (77%), 7/9 (77%) and 8/9 (88%) cats, respectively (Figure 1). Body weight at the end of the trial was stable or increased in 8/9 (88%) cats. The mean ± SD body weight at enrollment was 5.1 ± 1.4 kg and after 8 weeks of treatment was 5.5 ± 1.3 kg (P = 0.01). Water consumption measurement was performed in 5/9 (55%) cats enrolled; in 4/5 (80%) cats, a decrease was reported when comparing the first and the last measurement. The median (range) water volume consumed at the first measurement was 36.4 (10–98) ml/kg/24h and at the last measurement was 29.3 (9.8–50) ml/kg/24h (P = 0.12). The median (range) fasting serum glucose concentration at the enrollment was 424 (92–568) mg/dl (23.6 [5.1–31.6] mmol/l) and after 8 weeks of treatment was 197 (76–520) mg/dl (10.9 [4.2–28.9] mmol/l) (P = 0.08). The mean ± SD concentration of mean blood glucose concentrations of BGCs at the first week was 262 ± 155 mg/dl (13.3 ± 8.6 mmol/l) and at the eighth week was 187 ± 121 mg/dl (7.2 ± 6.7 mmol/l) (P = 0.3). The median (range) serum fructosamine concentration at the time of enrollment was 604 (457–683) µmol/l and at the time of the eighth week was 366 (220–738) µmol/l (P = 0.02). Four (44%) cats achieved remission, two after 29 and 53 days from enrollment and the other two after the end of the study when still receiving IGla-300 (day 82 and 96, respectively). All cats that achieved remission were newly diagnosed diabetics (80% of all newly diagnosed cats enrolled not suspected to have HS). Biochemical hypoglycemia was detected in 13/104 (12.5%) of all BGCs performed and in 2/32 (6%) when considering only BGCs performed in cats with HS, while clinical signs consistent with hypoglycaemia were not reported in any cat.
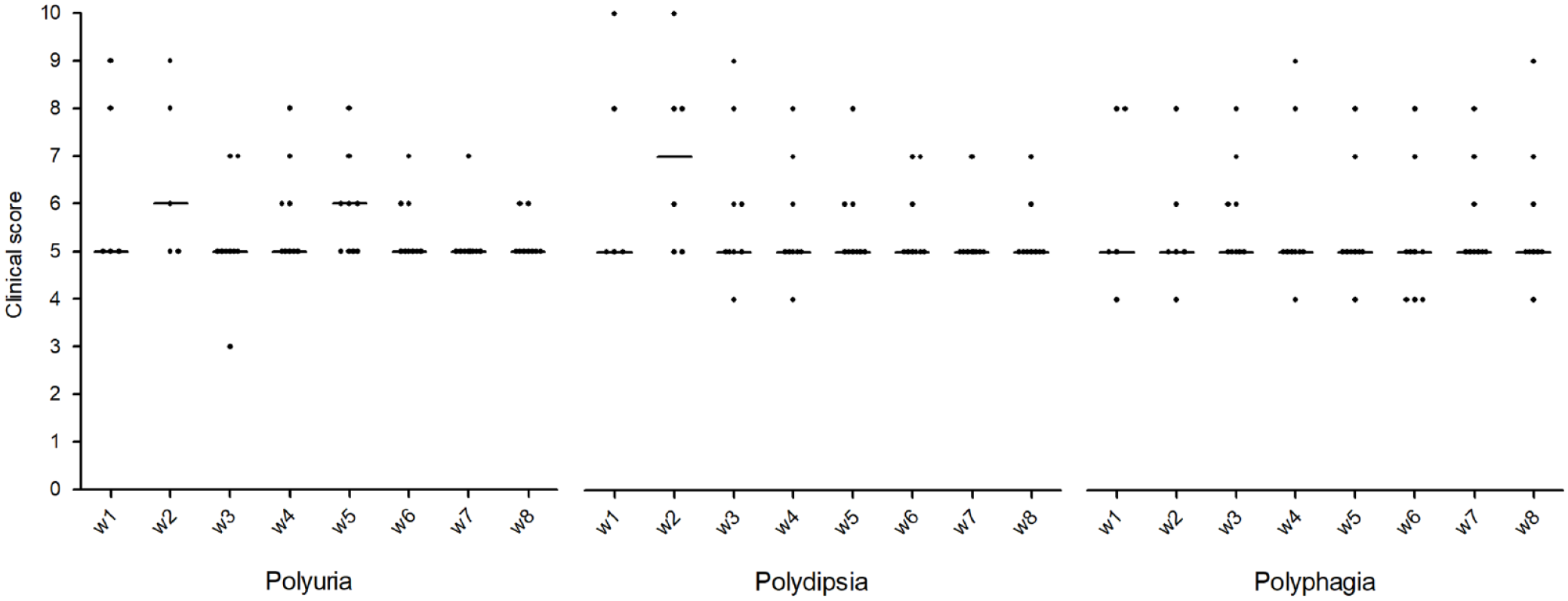
Scores describing clinical signs reported by owners of feline patients treated with insulin glargine 300 U/ml and low carbohydrate diet for 8 consequent weeks (w1–w8). Clinical signs reported are: polyuria (1 = does not urinate, 5 = normal, 10 = extremely increased); polydipsia (1 = does not drink, 5 = normal, 10 = extremely increased); and polyphagia (1 = does not eat, 5 = normal, 10 = extremely increased)
Glycemic variability
The median SD of BGCs performed in the first week was not significantly different to any other time point when considering cats achieving remission, not achieving remission or all cats enrolled (Figure 2). When comparing cats achieving remission or not, GV was significantly different only at the fifth week of treatment (P = 0.02), with the median (range) SD being 25.8 (15.2–27.5) mg/dl (1.4 [0.8–1.5] mmol/l) in cats that achieved remission and 54.3 (24.9–154.2) mg/dl (3.0 [1.4–8.6] mmol/l) in cats that did not (Figure 2).

Standard deviation, as a metric of glycemic variability, of blood glucose curves (BGCs) performed for 8 consequent weeks in feline diabetic patients treated with insulin glargine 300 U/ml and low carbohydrate diet. Dots (cats that achieved remission) and squares (cats that did not achieve remission) represent median SD and bars represent ranges. Glycemic variability was significantly different (*) when comparing cats achieving remission with cats not achieving remission at the fifth week (P = 0.02)
Insulin dose
The median (range) IGla-U300 starting dose was 0.91 (0.57–2.50) U/kg/day and, after 8 weeks of treatment, in cats still receiving insulin was 0.98 (0.17–4.00) U/kg/day (P = 0.62).
Time to nadir
Thirty out of 104 (28.8%) BGCs performed were excluded from the evaluation of the time to nadir due to an unknown precise time of insulin administration, once-daily insulin administration or skipped administration due to the risk of hypoglycemia. In 27/74 (36.5%) of BGCs evaluated, nadirs were detected within 2 h, in 43/74 (58.1%) within 4 h and 54/74 (73%) within 6 h from insulin administration (Figure 3).

Distribution of nadirs in feline patients treated with insulin glargine 300 U/ml for 8 weeks
Cats with HS
In 4/13 (31%) cats, HS was highly suspected. None of the cats enrolled had CT or MRI performed to confirm HS diagnosis. The median concentration of mean blood glucose concentrations of BGCs at the first week was 400 (110–470) mg/dl (22.2 [6.1–26.1] mmol/l) and at the eighth week was 302 (204–369) mg/dl (16.8 [11.3–20.5] mmol/l) (P = 0.34). The median (range) SD of blood glucose concentrations of BGCs performed in cats with HS was 70 (29–160) mg/dl (3.9 [1.6–8.9] mmol/l) and in cats with only DM was 31 (6–147) mg/dl (1.7 [0.3–8.2] mmol/l) (P < 0.01) (Figure 4). The median (range) insulin dose at the time of enrollment for cats with HS was 1.1 (0.8–2.1) U/kg/24h and after 8 weeks of treatment was 2.4 [2.1–3.1] U/kg/24h (P = 0.06). When comparing cats with HS and cats with only DM after 8 weeks, the median insulin dose was not significantly different (P = 0.41).

Standard deviation, as a metric of glycemic variability, of blood glucose curves (BGCs) performed for 8 consequent weeks in cats with highly suspected hypersomatotropism (HS) and cats with only diabetes mellitus (No HS) treated with insulin glargine 300 U/ml and low carbohydrate diet. Glycemic variability was significantly different (*) when comparing BGCs performed in HS and No HS cats (P <0.01)
Discussion
Management of feline DM aims at resolving or improving clinical signs as well as minimizing possible complications, while assuring a good quality of life for both the pet and the owner. 1 Remission of clinical signs is considered one of the main goals for insulin treatment and generally is obtained by achieving appropriate glycemic control. 1 In the present study, most of the cats treated with IGla-300 had their owners reporting an improvement of clinical signs related to DM, and more objectively, serum fructosamine concentrations were significantly reduced. Four (44%) out of the nine cats without HS that were enrolled achieved remission. All of them were newly diagnosed upon enrollment. Of the five cats that did not achieve remission, four were previously diagnosed and treated with insulin prior to enrollment. Variable remission rates have been reported in previous trials,3,4,22–26 and direct comparison with the present study was not possible due to lack of standardization of treatment and monitoring protocols applied. 27 A higher chance of remission in cats exposed to shorter periods of uncontrolled DM has been reported in previous studies,4,28 supporting the hypothesis that an early reversal of hyperglycemia could limit glucose-induced toxicity directed to beta-cells and preserve their functionality. This hypothesis was supported by previously published studies performed in human patients with T2DM in which a rapid achievement of euglycemia significantly increased remission rates.29,30 Of the five cats without HS that did not achieve remission, one presented with acute pancreatitis before enrollment and had another episode of acute pancreatitis after the end of the study. In this case, the chance of diabetic remission might have been decreased because pancreatitis reduced insulin sensitivity or had been the cause of DM.31,32 In two of the five cats that did not achieve remission, clinical signs resolved and blood glucose measurements of BGCs were persistently < 200 mg/dl (<11 mmol/l); these cats received the lowest insulin dose (1 unit once daily), but remission criteria were not met. The other two cats that did not achieve remission were lost to follow-up immediately after the study finished. Considering that 50% of the cats that achieved remission did so after the end of the study, it is possible that the short length of the trial was responsible for an underestimation of the remission rate. The four cats with HS did not achieve remission.
In this clinical trial, treatment with IGla-U300 resulted in no reported clinical signs suggesting hypoglycemic episodes at any time point of the study. The low risk of clinical hypoglycemia was probably not only the result of the pharmacological characteristics of IGla-U300 as reported in previous studies,6,9–18 but also the result of the close monitoring protocol which guaranteed that weekly insulin dose adjustments could be made if necessary. GV is defined as the fluctuation of glycemic measurements over a given interval of time and classified as short-term (within-day or between-day) and long-term GV. 33 In human medicine, excess short-term GV was associated with increased risk for hypoglycemia.34–39 Within-day GV has been previously evaluated also in feline diabetic patients using the SD,40,41 which is considered one of the simplest and most used metrics available. 42 In a recent study in newly diagnosed diabetic cats treated with exenatide extended release and IGla-U100 or IGla-U100 alone, cats that achieved remission had significantly lower within-day GV after 6 weeks compared with cats that did not achieve remission. 41 In the present study, a similar result was obtained evaluating the association between GV and remission after 5 weeks from enrollment. A precise role for GV in veterinary medicine has not been defined yet, and more studies are needed to assess its relationship with glycemic control. However, the results of this study and that of Krämer and co-authors (2020) support that GV could be considered a potentially useful and simple index to predict diabetic remission in feline patients.41 Cats with HS had significantly higher GV compared with cats not having HS. To the author’s knowledge, this is the first time that GV has been evaluated in a population of cats with HS. Previously, a recent study did not find a difference in GV, assessed by SD, when comparing cats with or without concurrent diseases potentially influencing insulin sensitivity, but in that case cats with HS were not evaluated. 43 Considering the limited knowledge available in veterinary medicine about GV, it is impossible to give a precise explanation for this result. However, we hypothesize the existence of a connection between elevated GV and complicated glycemic control, which is common in cats with HS treated only with insulin and low carbohydrate diet. 44 If this result is confirmed in other studies, then an elevated GV could influence the index of suspicion for HS diagnosis in poorly controlled diabetic cats.
In a recent study in healthy cats, the range of duration of action of IGla-U300 was between 16 and 18 h in 3/6 cats when assessed by the isoglycemic clamp method and between 18 and 22 h in 6/7 cats when assessed by suppression of endogenous insulin. 18 Consequently, in the present study, on a theoretical basis, the administration of IGla-U300 every 12 h likely resulted in an overlap of blood glucose lowering effect in the first few hours after each injection. In 75% of BGCs, the nadir was detected within 6 h after morning insulin administration (Figure 3) and this would fit with the hypothesis of an overlapping effect. However, if overlap of effect was the sole explanation of the post-injection nadirs, one would expect that the frequency of nadirs would be the same after the morning and evening insulin injections, if the dose is the same in both. The finding that the frequency of nadirs after the evening insulin administration was much lower suggests that other causes reducing blood glucose concentration were at play. In addition, a recent but as yet unpublished study reported that diabetic cats tend to have lower minimum blood glucose concentration during the day in comparison with night-time. 45 Our study was not designed with the purpose of performing pharmacodynamic evaluations, and the use of the time to nadir to investigate the presence of doses overlap had major limitations. In a clinical setting, the nadir does not necessarily correspond to the moment of maximum insulin action as multiple variables could affect blood glucose concentrations including feeding and stress.
In the present study, the small number of feline patients enrolled represented the main limitation, which is common when referring to clinical trials performed with privately-owned cats with naturally occurring DM. The use of a control group composed of diabetic cats treated with a traditional therapy would have allowed comparing IGla-U300 with more commonly used insulin products (eg, IGla-U100). A direct comparison between IGla-U300 and IGla-U100 would clarify if advantages in terms of reduction of hypoglycemic episodes and lower potency reported in human diabetic patients could also be confirmed in feline patients,6,9–16 justifying the use of IGla-U300 in clinical practice. The remission rate was probably underestimated due to the decision to enroll long-term diabetic cats as well as newly diagnosed cats. Another limitation was the enrollment of cats previously treated with insulin, since their diabetes might be more difficult to manage than newly diagnosed cats because of reduced insulin sensitivity or an undiagnosed concurrent disease. However, the decision to also include cats already on treatment was made to evaluate IGla-U300 in an heterogenic population that was as similar as possible to that found in a clinical setting. In this study, a clinical scoring system that has not been previously validated was used to provide categorical data for the purpose of comparison, and might not have accurately captured the clinical responses of individual patients. HS diagnosis was not confirmed with CT or MRI in any of the cats enrolled. However, a recent study reported a positive predictive value of 95% when using a serum IGF-1 cut-off of 1000 ng/ml. 44 Therefore, the diagnosis of HS in the present study was very likely.
Conclusions
IGla-U300 is an effective and safe option for treatment of feline DM. More studies are needed to evaluate if this insulin is associated with reduced risk of hypoglycemia in cats than traditional insulin treatments, as is the case in human patients. Further investigation is also warranted about the possibility of an overlapping effect of insulin action when IGla-U300 is administered twice daily, and if a once daily administration might be equally effective.
Footnotes
Author note
The study was presented as an oral abstract at the 30th ECVIM-CA Annual Congress, online, September 2020.
Conflicts of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the diet Purina Pro Plan Diabetes Management (Nestlé Purina Commerciale) was provided for free to the owners by the manufacturer.
Ethical approval
This work involved the use of non-experimental animals (owned or unowned) and procedures that differed from established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). For any animals or humans individually identifiable within this publication, informed consent (either verbal or written) for their use in the publication was obtained from the people involved.
