Abstract
Objectives
The aim of the present study was to retrospectively assess remission rates and survival in diabetic cats managed using a moderate-intensity, low-cost protocol of home blood glucose measurements and insulin adjustment by clients of a cat-only practice, and to determine if predictors of remission, relapse or survival could be identified.
Methods
The records of a cat-only practice were used to identify 174 cats with newly diagnosed diabetes managed using only pre-insulin home blood glucose measurements for insulin dose adjustments based on a protocol provided to clients aimed at maintaining pre-insulin blood glucose in the range of 6.5–11.9 mmol/l (117–214 mg/dl). Cats were excluded for the following reasons: insufficient follow-up in the records; a lack of owner compliance was recorded; they were receiving ongoing corticosteroids for the management of other conditions; they were euthanased at the time of diagnosis; or they were diagnosed with acromegaly or hyperadrenocorticism.
Results
Using only pre-insulin blood glucose measurements at home to adjust the insulin dose to maintain glucose in the range of 6.5–11.9 mmol/l, 47% of cats achieved remission, but 40% of those cats relapsed. A minority (16%) of cats were hospitalised for hypoglycaemia. The survival time was significantly longer in cats in remission and Burmese cats.
Conclusions and relevance
The cost and time burden of treating diabetic cats may cause some clients to choose euthanasia over treatment. While the highest rates of diabetic remission have been reported in studies of newly diagnosed cats treated with intensive long-acting insulin protocols and low carbohydrate diets, these protocols may not be suitable for all clients. Nearly 50% of cats with newly diagnosed diabetes achieved remission with this low-cost, moderate-intensity, insulin dosing protocol. As remission was significantly associated with survival time, discussing factors in treatment to optimise remission is important, but it is also important to offer clients a spectrum of options. No cats that started treatment in this study were euthanased because the owner did not wish to continue the diabetes treatment.
Keywords
Introduction
Diabetes mellitus is the second most common endocrine disease in domestic cats, and some cats achieve remission of their diabetes with appropriate treatment.1 –3 Diabetes can be a challenging diagnosis for owners, because treatment and management typically require twice-daily insulin injections and monitoring, which can include frequent trips to the veterinary clinic. Costs, quality of life for the cat and an impact on the owner’s schedule may make clients reluctant to pursue treatment.4,5 It has been reported that up to 10% of owners choose euthanasia on diagnosis of diabetes, rather than pursue treatment, particularly in older cats. 4
Individual clients of cats with newly diagnosed diabetes will have differing considerations when it comes to costs, treatment protocols, perceptions of quality of life and priority that their cat achieves diabetic remission. A full discussion should be had with all clients, offering them a spectrum of care options, including monitoring, diagnostic testing and potential outcomes, to allow them to make the best choice for themselves and their cat.
Diabetic remission has been associated with longer survival times and owner perception of better quality of life for their cat.5,6 However, remission rates can vary widely between protocols used, and owners must be willing to treat their cat on diagnosis and accept that this may be a lifelong treatment if remission is not achieved.
In cats with type 2-like diabetes, early institution of insulin therapy aimed at restoring glycaemic control can result in the remission of diabetes and resolve the need for ongoing exogenous insulin administration. 7 Remission is associated with the reversal of suppression of insulin secretion. This suppression is believed to result from increased glucose (described as glucose toxicity) and lipid concentrations, characteristic of untreated diabetes. 8 Diabetic remission has been previously described as maintaining euglycaemia for at least 2–4 weeks after insulin therapy has ceased.9 –12 Cats in diabetic remission have increased survival and likely have a better quality of life compared with those that remain diabetic.6,10,12,13 Studies with early intervention, frequent glucose monitoring, twice-daily glargine or detemir insulin and dose adjustments aimed at achieving normal or near-normal blood glucose concentrations, combined with low carbohydrate diets (⩽1.6 g/100 kcal), are reported to achieve the highest remission rates of up to 100%.10,13 These studies relied on either intensive home monitoring or hospitalisation of cats for blood glucose curves. However, other studies using twice-daily glargine injections have reported much lower remission rates (17–41%).14 –16 The majority of studies are also limited due to small sample size, lack of randomisation and blinding, and the absence of rigid criteria for diagnosis and inclusion between studies. 11 In addition, approximately 25–30% of cats in remission are reported to relapse and require further insulin therapy, with 30% of cats in one study relapsing within 9 months.9,10,12
Home blood glucose monitoring has been associated with better quality-of-life perceptions for both cat and owner, and also comes with lower costs. 17 In practice, it may be more realistic to expect lower remission rates where the rigorous requirements of high-intensity protocols are not always able to be implemented, as these are not always appropriate for all clients.
A limitation in prospective studies is often recruiting a sufficient number of cats. The Cat Clinic in Brisbane, Australia, has used a moderate-intensity, home blood glucose monitoring protocol with twice-daily glargine injections for the last 15 years, allowing a large number of cats to be identified retrospectively. This protocol offers cost benefits to owners, removing the requirement for frequent veterinary visits and utilising a readily available glucometer for human use that can be purchased from most pharmacies.
The aims of this study were to retrospectively identify cats with newly diagnosed diabetes managed with this protocol, evaluate remission and relapse rates, and assess survival. It is hoped that by offering clients lower-cost, less-intensive, home-management protocols, owners will be relieved of some of the burdens of diabetic treatment, and be more receptive to the management of their diabetic cat, resulting in fewer cats being euthanased.
Materials and methods
Study design
This was a retrospective study that identified client-owned diabetic cats treated with glargine and managed with a standardised moderate-intensity blood glucose management protocol from a cat-only veterinary hospital (The Cat Clinic, Brisbane, Australia). Practice records between January 2005 and September 2019 were reviewed to retrospectively identify diabetic cats whose owners were provided with the insulin dosing protocol. Cats were excluded if there was insufficient follow-up in records, lack of owner compliance, they were receiving ongoing corticosteroids for the management of other conditions, they were euthanased at the time of diagnosis, or were diagnosed with acromegaly or hyperadrenocorticism. The cat data recorded included breed, sex and date of birth. The date of diagnosis was recorded, as well as the date of remission and relapse, if applicable. The diagnosis was based on the presentation of clinical signs and a blood glucose concentration of >13 mmol/l (234 mg/dl); fructosamine concentrations were not routinely performed. Data collected from the medical records included initial blood glucose concentration, body condition score (BCS; if recorded) at the time of diagnosis, if diabetic ketoacidosis (DKA) or peripheral neuropathy was present, and if there was a recent history of steroid administration. DKA was diagnosed based on a blood glucose concentration ⩾16 mmol/l (⩾288 mg/dl), glucosuria, clinical signs, ketonuria and/or elevated betahydroxybutyrate, and decreased serum bicarbonate where available. 18 Dietary data were collected where available. In addition, any episodes of hypoglycaemia requiring hospitalisation were recorded. Diabetic remission was considered achieved if cats recorded a non-fasted casual glucose concentration of ⩽6.5 mmol/l (⩽117 mg/dl) a minimum of 2 weeks after ceasing insulin therapy and had no clinical signs of diabetes. The remission date in this study was defined as 14 days after insulin cessation, and only cats in their first remission were included.
Protocol
The protocol was developed by Dr Rhett Marshall at The Cat Clinic, Brisbane based on research conducted for his PhD (supervised by author JR), and is outlined in Table 1. 19 The key feature of the protocol was to allow insulin adjustments based on pre-insulin blood glucose concentration measurements made by the owners at home. Initial doses of insulin were 0.25 IU/kg ideal body weight for a starting blood glucose concentration of <20 mmol/l (360 mg/dl) or 0.5 IU/kg ideal body weight if higher. 13 Clients were initially encouraged to take measurements twice daily, although the time period was able to be lengthened depending on the stability of the cat. The original cutpoints for insulin dose adjustments were aimed at maintaining pre-insulin blood glucose in the range of 6–11.9 mmol/l (108–214 mg/dl). Higher cutpoints were used (6–13.9 mmol/l; 108–250 mg/dl) once the cat had been diabetic for more than 2 months (Table 1). It was recommended that cats be fed low carbohydrate veterinary prescription diets designed for diabetic cats that were available in Australia at the time (Hills m/d, Hills; Royal Canin diabetic, Royal Canin) and/or meat, although client compliance varied. Dietary data were collected through either notes in the history or sales records. For most cats, it could not be determined with confidence from the records how long they were fed the diet. Twice-daily feeding was not enforced, with cats allowed to eat in their preferred pre-diagnosis pattern. Clients were also taught to recognise signs of clinical hypoglycaemia, including lethargy, confusion, weakness and ataxia. If these clinical signs were noted, blood glucose measurements were obtained, and the clinic contacted for admission to hospital. For cats that achieved remission, clients were encouraged to continue blood glucose measurements at least once a week and monitor for the recurrence of any clinical sign of diabetes, such as polydipsia, polyphagia or polyuria. This protocol was developed using Accu-Chek glucometers (Roche), which are readily available from regular pharmacists. As these are calibrated for use in humans, measured blood glucose concentrations were approximately 1–2 mmol/l (36 mg/dl) lower than the actual glucose concentration when in the euglycaemic range. 20 The AlphaTRAK (Zoetis) is a glucometer specifically calibrated for use in cats; however, this had only recently become available in Australia, and can be difficult and expensive for clients to source. No cats in this cohort used AlphaTRAK glucometers.
Insulin dosing protocol provided to clients for adjustments of glargine dose based on pre-insulin blood glucose measurements in cats with newly diagnosed diabetes (if remission was not achieved within 2 months, dosing guidelines were altered)

Statistical analyses
A multivariate Cox proportional hazard model was initially used to examine survival time, defined as the time from diagnosis to death. The following variables were used to determine their association with overall survival time: sex (male or female); breed (domestic, Burmese, other); blood glucose concentration at diagnosis; peripheral neuropathy; DKA; and corticosteroid administration before diagnosis. Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated. Cats had censored survival time if they were alive by the end of the study or were lost to follow-up. To examine the association between diabetic remission and survival, the Kaplan–Meier product limit method was used. Within this analysis, the log-rank (Mantel–Cox) test was used to compare median survival times between cats that achieved remission and cats that did not.
Differences in remission rates between cats on various diets were examined using a χ2 analysis and the results are expressed as median ± SD. Differences in blood glucose concentrations at diagnosis between cats that subsequently went into remission vs those that did not were examined using an unpaired t-test, and the results are expressed as mean ± SD. For BCS, a Mann–Whitney non-parametric test was used to compare median BCS. All statistical analyses were conducted using GraphPad Prism Software (https://www.graphpad.com/). P <0.05 was considered statistically significant.
Results
Signalment
A total of 334 newly diagnosed cats that were managed using the protocol were identified, and 174 eligible cats met the selection criteria and were included in the study (103 castrated male cats, 71 spayed female cats) (Table 2). Cats were excluded if there was insufficient follow-up (41/334, 12.3%), poor owner compliance (63/334, 18.9%), the cats were receiving ongoing corticosteroids (17/334, 5.1%), the cats were euthanased at diagnosis (19/334, 5.7%) or had underlying acromegaly or hyperadrenocorticism (20/334, 6%). Of the cats euthanased at diagnosis, only one was euthanased due to the owner not wanting to start treatment. Other reasons for euthanasia included chronic renal failure (6/18), neoplasia (6/18), congestive heart failure (3/18), mobility issues (2/18) and a cryptococcus infection (1/18).
The median age at diagnosis of diabetes was 12 years (range 5–21). The most common breed was domestic shorthair (n = 80), followed by Burmese (n = 37). Multiple other breeds were represented (Table 3). BCS (on a nine-point scale) was recorded for 115 cats (median 5; range 2–9).
Signalment, biochemical parameters and comorbidities of all cats (n = 174) at the time diabetes was diagnosed, and compared between cats that went into remission (n = 82) and those that did not (n = 92)

Data are n (%), mean ± SD or median (range)
Estimated dietary carbohydrate content (g/100 kcal) after diagnosis, during treatment
BCS = body condition score; DKA = diabetic ketoacidosis; fPLI = feline pancreatic lipase immunoreactivity
Breeds of 174 cats with newly diagnosed diabetes retrospectively identified as using the protocol for home blood glucose monitoring

Diagnosis
All cats were newly diagnosed and had not received any insulin treatment before starting the protocol. At diagnosis, the median recorded blood glucose was 23 mmol/l (414 mg/dl) (range 13.2–38.6 mmol/, 237–694 mg/dl; n = 171). The median cholesterol concentration was 6.5 mmol/l (range 3.1–14.7 mmol/l; n = 122; reference interval [RI] 2.2–5.5). Only 22 cats had feline pancreatic lipase immunoreactivity (fPLI) recorded at the time of diagnosis, with a median of 22 µg/l (range 2.1–50 µg/l; RI 0.1–3.5). Of these cats, 18 (82%) had an fPLI above the RI. DKA was diagnosed in 23/174 (13.2%) cats at presentation and peripheral neuropathy was recorded in 15/173 (8.7%) cats. A history of recent corticosteroid administration (within the last month) was reported in 25/169 (14.8%) cats, and one cat had received megestrol acetate (Table 2).
Remission and relapse
Overall, a remission rate of 47% (82/174) was achieved in cats treated with the current protocol, with a median time to remission of 93 days (range 18–1364) (Table 2). Of the 82 cats achieving remission, 22 (27%) went into remission within the first 2 months of starting the protocol (12.6% of all cats). By 6 months, a further 37 cats had achieved remission (34% of all cats in the study, 59/174). Of those that went into remission, the median age at diagnosis was 12 years (range 5–19) and the median blood glucose concentration at the time of diagnosis was 24.1 mmol/l (433 mg/dl) (range 13.2–38.6 mmol/l; 237–694 mg/dl). By comparison, cats that did not go into remission had a median age at diagnosis of 13 years (range 6–21) and a median blood glucose concentration at the time of diagnosis of 22.0 mmol/l (369 mg/dl) (range 13.6–33.9 mmol/l; 244–610 mg/dl). Statistical analyses revealed there was no major difference in median age at diagnosis between the groups (P = 0.25), although there was a small but significant (P < 0.01) difference in mean blood glucose concentration at diagnosis (Table 2). Cats that went into remission had marginally higher mean blood glucose (24.3 ± 5.3 mmol/l, 437 ± 95 mg/dl) than those that did not go into remission (22.4 ± 4.4 mmol/l, 403 ± 79 mg/dl), with four cats that went into remission exhibiting very high blood glucose concentrations (>35 mmol/l) at diagnosis (Figure 1). The median BCS was significantly different in cats that went into remission compared with those that did not (5 vs 4).
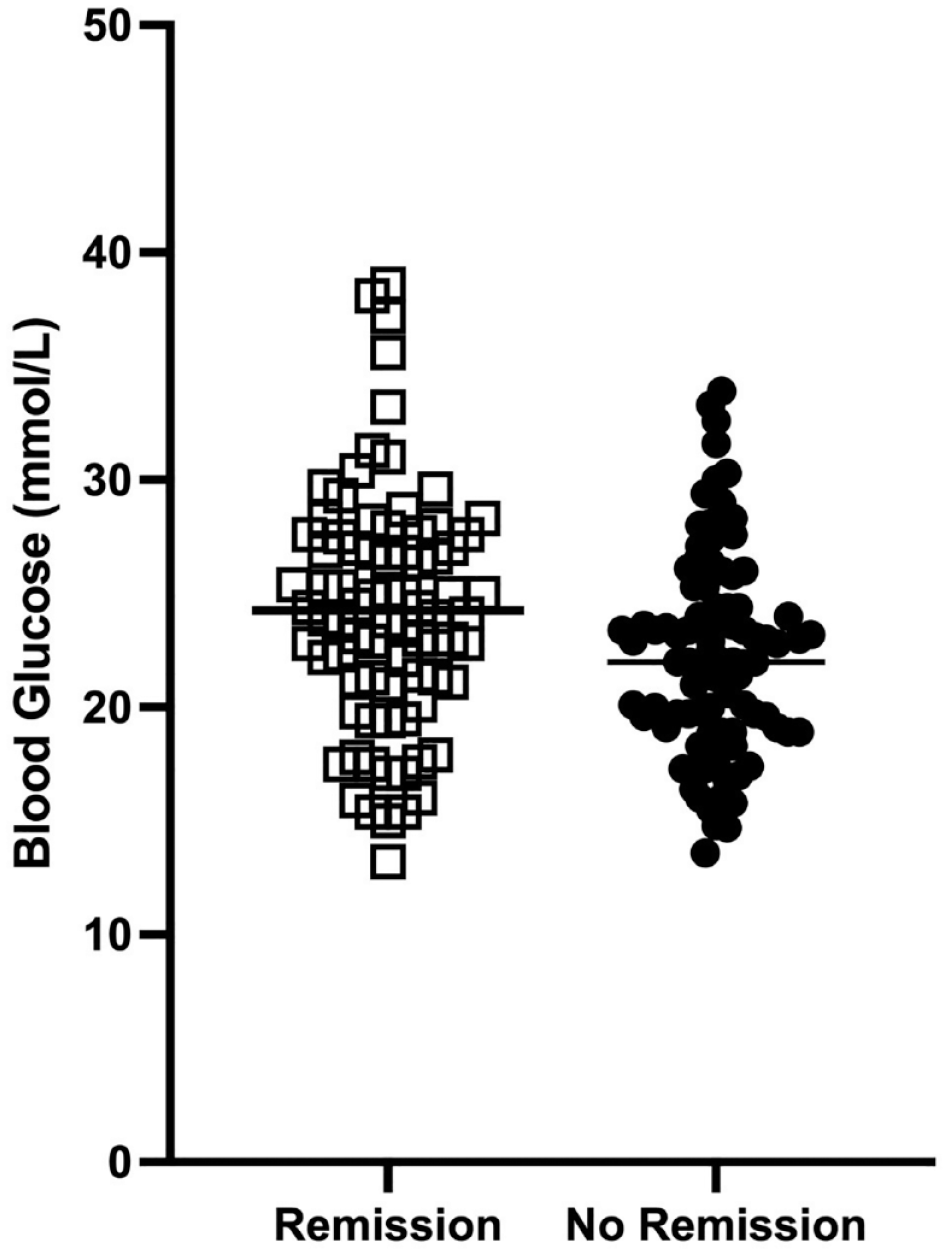
Blood glucose concentration at the time of diagnosis of cats that subsequently went into diabetic remission in response to the protocol (open squares, n = 82) or did not (closed circles, n = 92). The horizontal line is mean glucose concentration for each group. The difference in mean blood glucose is significant (P <0.05)
Of the 82 cats that went into diabetic remission, eight (9.8%) had DKA at the time of diagnosis, seven (8.5%) were recorded as having peripheral neuropathy at the time of diagnosis and 20/80 (25%) had recorded corticosteroid use within 1 month before the time of diagnosis, with one cat receiving megestrol acetate.
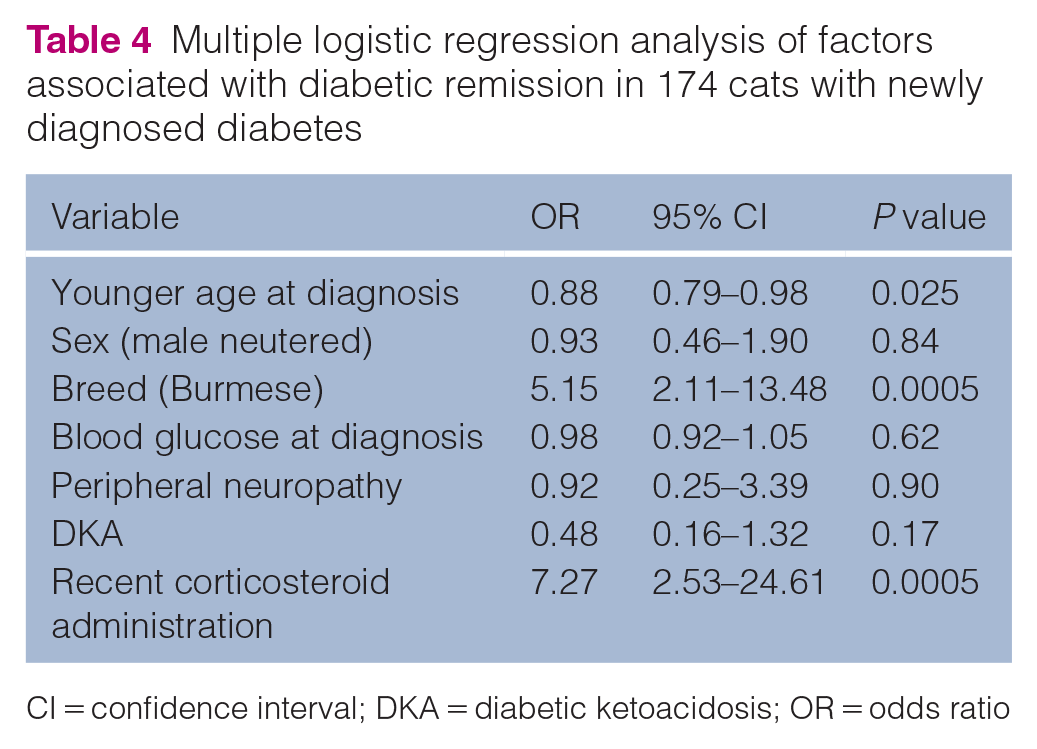
Multiple logistical regression revealed factors associated with remission (Table 4). Significant (P <0.05) factors included age at diagnosis, breed and recent corticosteroid administration. Older cats had a slightly lower chance of remission, while Burmese cats were five times more likely to achieve remission than domestic (short- and medium hair) breeds. Corticosteroids in the month before diagnosis were associated with a sevenfold greater chance of remission. Notably, blood glucose at the time of diagnosis was not associated with diabetic remission, despite a small significant difference noted in mean values between the groups.
Multiple logistic regression analysis of factors associated with diabetic remission in 174 cats with newly diagnosed diabetes

CI = confidence interval; DKA = diabetic ketoacidosis; OR = odds ratio
Of the cats that went into remission, 33/82 (40.2%) relapsed, with a median time to relapse of 206 days (range 17–1992). Of these cats, 23 (70%) relapsed within the first year, resulting in a relapse rate of 28% within 12 months after achieving remission, despite a small significant difference noted in mean values between the groups.
Hypoglycaemia
Of the 174 cats managed using this protocol, 27 (15.5%) were hospitalised for the management of clinical hypoglycaemia; of these, five (18%) were due to an accidental overdose of insulin by the owner. Some cats had multiple episodes of clinical hypoglycaemia (seven cats had two episodes, two cats had three episodes).
Diet
Limited dietary information was available for 117/174 (67.2%) cats. Of those cats, 72 (61.5%) were reported as eating diabetic wet and dry foods (either Hills m/d or Royal Canin Diabetic) and/or meat. The remission rate in cats eating diabetic and meat diets was not significantly different (P = 0.25) from the remission rate in cats eating other foods. The carbohydrate content in the diet was estimated from the available dietary information and was found to be not significantly different (P = 0.14) between those cats that went into remission (5.7 ± 2.9 g/100 kcal, range 0.0–11.4; n = 33) and those that did not go into remission (4.6 ± 2.3 g/100 kcal, range 0.0–10.1; n = 44). Mean carbohydrate intake was 5.1 g/100 kcal and only four cats were fed diets with a carbohydrate intake of less than 3.5 g/100 kcal.
Follow-up
Cats were followed up until euthanasia, death, the owner moved away or the time of the last communication with the client. On follow-up, of the 174 cats, 100 (57.5%) were euthanased, 13 (6.9%) died at home, six (3.5%) had owners that moved away and 56 (32.2%) had no further follow-up recorded as of 30 September 2018. The most common reasons for euthanasia were cancer (24/100) and end-stage renal disease (18/100). The median time to euthanasia after diagnosis was 1015 days (range 34–3193), with 21 (21%) cats euthanased within the first year of being diagnosed. No cats were euthanased due to clients not wanting to continue diabetic treatment. Causes of death or euthanasia are outlined in Table 5.
Causes of death in 113 cats that were euthanased or died during the follow-up of the 174 cats with newly diagnosed diabetes treated with the moderate-intensity protocol (range of survival 34–3193 days)
FATE = feline aortic thromboembolism; IBD = inflammatory bowel disease; UTI = urinary tract infection
Survival
Of the 113 cats that had follow-up until death, the majority (93/113, 82%) survived for more than 1 year after diagnosis, 40 (35%) survived for more than 3 years and 15 (13%) survived for more than 5 years. Overall, regardless of diabetic remission, the median survival time for cats after diagnosis was 773 days (range 472–1458). Two factors had a significant association with survival time. Multivariate Cox proportional hazard modelling indicated that the expected hazard of dying was approximately 30% greater for each year of age at diagnosis (HR 1.30, 95% CI 1.20–1.40, P <0.0001), while Burmese cats were more likely to have increased survival (HR 0.56, 95% CI 0.32–0.94, P = 0.035). Factors such as sex, BCS, cholesterol, fPLI, blood glucose at diagnosis, cholesterol, DKA, peripheral neuropathy and recent corticosteroid administration were not significantly associated with survival time.
A Kaplan–Meier product limit analysis revealed that diabetic remission was associated with increased survival time (Figure 2). The survival time in cats that achieved remission was significantly increased during the 4 years after diagnosis (median survival 959 vs 746 days for remission and non-remission cats, respectively; P = 0.03 log-rank test). Despite fewer records, significant increases in median survival times were also noted in remission cats in later years (7 years: median survival 1364 vs 933 days; P = 0.03; 8 years: median survival 1377 vs 943 days; P = 0.05 log-rank tests).

Kaplan–Meier survival curve of cats that either went into diabetic remission in response to the protocol (dashed line, n = 82) or did not (solid line, n = 92). The survival time in years is expressed on the X axis. The difference in survival between the groups is significant (P <0.05)
Discussion
In this retrospective study of 174 cats with newly diagnosed diabetes, 47% achieved remission when managed by owners using pre-insulin blood glucose concentrations measured at home. The insulin dose was adjusted by owners using a protocol aimed at maintaining pre-insulin blood glucose in the range of 6–11.9 mmol/l (108–214 mg/dl).
Remission rates
The remission rate of 47% was lower than in two previous studies using glargine, which reported 100% remission in eight cats with newly diagnosed diabetes, and 84% remission in 32 cats that had been diabetic for less than 6 months.10,13 However, it is comparable with or higher than in three other studies using glargine, where remission rates were in the range of 17–40% (Table 6).14 –16 There were considerable differences between the studies, including whether only newly diagnosed or previously diagnosed cats were studied, variations in diet and insulin dosing protocols. Therefore, it is not clear what factors are most influential in achieving high remission rates. In our study, only newly diagnosed cats were included for evaluation. Only two previous studies (Marshall et al, 13 with 100% remission, and Riederer et al, 14 with 20% remission) enrolled cats with newly diagnosed diabetes exclusively, with the other studies having a mix of newly diagnosed and previously treated cats. Of the two studies with remission rates over 50% using glargine, one was only in newly diagnosed cats and the other predominantly involved cats previously treated with protamine zinc insulin or lente.10,13 Sample sizes were also limited, with 4/5 studies having only 5–15 cats.
Published studies of remission rates in diabetic cats treated with twice-daily glargine*

Only cats in the control group treated with glargine are included in this table; the group treated with glargine and exenatide extended-release are not included
NPH = neutral protamine Hagedorn; PZI = protamine zinc insulin
Frequency of blood glucose testing
In our study, blood glucose was measured by the client at home twice a day, just before each insulin injection. This reduced the cost burden for the client and was less stressful for most cats and clients than repeated clinic visits. Home blood glucose monitoring has been linked to improved quality of life for the client and the cat.17,21 In contrast, 4/5 studies reporting remission rates in glargine-treated cats admitted the cats to the clinic for blood glucose curves to be performed at various frequencies (Table 6). Only one other study in glargine-treated cats was based exclusively on home blood glucose monitoring, but the owners were required to measure glucose concentration a minimum of three times a day, and a mean of five measurements was made each day. These cats were recruited through an online feline diabetes forum, and thus the owners were likely to be more willing to follow this intensive monitoring protocol. 10 A requirement for this frequency of glucose monitoring may not be practical or desirable for many clients.
By creating a moderate-intensity, home-based protocol, clients may be more willing to undertake treatment of their cat. The current availability of continuous glucose monitoring systems, such as the Freestyle Libre (Abbott Laboratories), has made glucose monitoring more feasible for some clients. These devices provide continuous interstitial glucose measurements without the need for blood glucose measurements via ear prick for calibration and are sufficiently accurate for use in cats. 22 Enabling more frequent measurements in the initial few weeks after diagnosis facilitates earlier implementation of tighter glycaemic control, which may increase remission rates. However, these devices only last up to 2 weeks (and may stop working sooner), are quickly removed by some cats and come with significant cost to the client, so may not be suitable for long-term use for many clients. In our study, clients were able to be taught blood glucose monitoring using an ear prick and the readily available human-use Accu-Chek glucometer was relatively inexpensive for the client (Accu-Chek guide prices: USD$29.99 or AUD$41.99 for meter, and USD$55.99 or AUD$47.99 for 100 strips [prices from wallgreens.com and chemistwarehouse.com.au, accurate on 29 June 2023]). It remains to be determined whether increased remission rates using continuous glucose monitoring justify the higher initial costs for the client.
Timing of blood glucose testing
To simplify this protocol for clients, only pre-insulin blood glucose concentrations were measured, and therefore blood glucose nadirs were not known. While this protocol was largely considered safe, 27 (15.5%) cats developed clinical hypoglycaemia using the protocol, and five of these were due to an accidental overdose of insulin by the client. An important part of this protocol is training clients to recognise, monitor and act upon any hypoglycaemic episodes.
Cutpoints
Only 2/5 studies reporting remission rates in glargine-treated cats described cutpoints for insulin dose adjustments (Table 6). One study that utilised multiple measurements throughout the day aimed to keep blood glucose concentrations in the range of 2.8–11.1 mmol/l (50–199 mg/dl). 10 The lower frequency of glucose measurements in our study did not allow for this level of tight glycaemic control. Cutpoints for pre-insulin blood glucose and dose adjustments in our study were lower than those reported in another study, which adjusted the dose based on cats coming in to the hospital every 2 weeks for blood glucose curves, with no in-between home monitoring. 13 Insulin was adjusted to maintain nadir glucose above 7 mmol/l (126 mg/dl) and pre-insulin glucose above 12 mmol/l (216 mg/dl). However, the cats in that study may have been receiving a higher insulin dose relative to their non-stress-affected blood glucose concentrations at home, because blood glucose in individual cats can be increased by up to 10 mmol/l (180 mg/dl) and is, on average, 4 mmol/l (72 mg/dl) higher due to the stress of travel and admission to a veterinary clinic. 23
For newly diagnosed cats, the cutpoints in our protocol were lower for the first 2 months to allow for tighter initial control. After this, the cutpoints were increased to maintain pre-insulin blood glucose levels in the range of 6–13.9 mmol/l (108–250 mg/dl) providing a greater safety margin because the frequency of blood glucose measurements decreased the longer the cat was diabetic (Rhett Marshall, personal communication). After diagnosis, clients were initially encouraged to perform twice-daily pre-insulin blood glucose measurements. As the cats stabilised, the time between glucose measurements increased, depending on the cat’s response to insulin. In many cats, once they were on a stable dose of insulin, the target range of blood glucose concentrations (6–13 mmol/l, 108–234 mg/dl) was still above the normal range (<6.5 mmol/l, 115 mg/dl). The lack of tight glycaemic control may have contributed to decreased remission rates. 13 The change in the insulin dosing guidelines after 2 months was based on the PhD study by Marshall et al, 13 where 6/8 (75%) cats were in remission by this time, with the remaining 2/8 (25%) in remission within 16 weeks. However, the protocol used for those cats had key differences: blood glucose curves were generated in hospital; different glucose cutpoints were used; and the cats were fed a lower carbohydrate diet (1.6 g/100 kcal, Purina DM wet food; Nestlé Purina Pet Care), which was not available in Australia at the time of our study. Within 2 months of using our protocol, only 12.6% of cats were in remission. Continuing the use of lower blood glucose cutpoints for a longer period may result in higher remission rates.
Blood glucose concentrations at diagnosis
While cats that went into remission had significantly higher blood glucose concentrations at diagnosis, this difference is unlikely to be clinically relevant. Four cats that went into remission had blood glucose concentrations at diagnosis of >35 mmol/l, while the highest concentration at diagnosis of the cats that did not go into remission was 33.9 mmol/l (Figure 1). The four cats with the highest blood glucose concentrations did not have other underlying complications, such as peripheral neuropathy, DKA or recent corticosteroid administration. This suggests that blood glucose concentration at the time of diagnosis does not impact the probability of remission.
Diet
Low carbohydrate diets are an important component of diabetic management unless other dietary requirements take precedence. For example, diabetic cats treated with a variety of insulins and fed a low carbohydrate diet of 3.5 g/100 kcal had significantly increased remission rates (68%) compared with those fed a 7.6 g/100 kcal diet (41% remission) after 16 weeks. 24 Recent diagnosis, twice-daily glargine adjusted to achieve tight glycaemic control, combined with a low (⩽3.5 g/100 kcal) or ultra-low (⩽1.6 g/100 kcal) carbohydrate diet have been associated with the highest remission rates (80–100%).10,13 In contrast, our cohort had a remission rate of 47%, but the average dietary carbohydrate content was 5.1 g/100 kcal, and only four cats consumed diets estimated at lower than 3.5 g/100 kcal. Mean carbohydrate content did not differ significantly between remission and non-remission cats. However, limited dietary data were available based on written histories or sales records. Proportions of different diets were estimated as an even split between the two, and duration was uncertain in most cats. There was also no significant difference found in remission rates in an earlier study between diabetes-specific food and over-the-counter adult maintenance foods. However, the carbohydrate content of those diets did not differ significantly, similar to our study. 15 The only two commercially available dry diabetic diets available to cats in our study had carbohydrate content above 3.5 g/100 kcal (3.9 g/100 kcal, Hills m/d and 4.9 g/100 kcal, Royal Canin Diabetic). Wet varieties had a carbohydrate content of 3.9 g/100 kcal (Hills m/d) and 3.2 g/100 kcal (Royal Canin Diabetic). Advising clients on balanced diets with carbohydrate content ⩽3.5 g/100 kcal may increase the probability of remission. Several cats were also fed meat (0 g/100 kcal), which can effectively lower overall carbohydrate content combined with a balanced diet.
History of corticosteroid use
The highest remission rates were in cats previously treated with corticosteroids (80%), consistent with previous findings that recent corticosteroid use is a predictor for remission.9,12 One cat that received megestrol acetate before diagnosis also went into remission. Cats that required ongoing administration of corticosteroids for other underlying issues after a diagnosis of diabetes were excluded from our analysis. As corticosteroids may exacerbate pre-existing pancreatic β-cell dysfunction by causing insulin resistance that compounds underlying reduced β-cell capacity, cats continuing to receive corticosteroids should have their blood glucose concentrations monitored on an ongoing basis.
Peripheral neuropathy and DKA
Almost half (47.7%) the cats with peripheral neuropathy on diagnosis went into remission, which was not different from the overall remission rate in our study. The presence of peripheral neuropathy has been reported to be a negative indicator for remission and was postulated to result from a longer disease process before clinical signs of diabetes occurred.10,12 DKA was concurrently diagnosed in 13.2% of cats at diagnosis, and 38.4% of these cats achieved remission. Consistent with other reports, the presence of DKA at the time of diagnosis of diabetes in our study did not impact the probability of remission.12,25
Relapse
Our study had a higher proportion of cats that relapsed (40%) than previously reported (25–30%).9,10,12 This is likely because of the longer follow-up period, as only 28% of our cats relapsed within 12 months. However, the majority (70%) of cats that relapsed did so within the first year. While clients were strongly advised to continue weekly blood glucose monitoring of cats in remission, there were not sufficient data available to know how many adhered to that. Cats in diabetic remission still have altered states of glucose tolerance, glucose metabolism and insulin sensitivity, so it is important that clients are encouraged to maintain glucose monitoring to facilitate early intervention if blood glucose increases. Exposure to potential triggers of relapse should be minimised, such as avoiding corticosteroids where possible, maintaining an ideal body condition and continuing to feed a low carbohydrate diet (⩽3.5 g/100 kcal).9,26,27
Survival
Consistent with one previous report, diabetic remission in our study was significantly associated with longer survival times over 4 and 8 years. 28 Median survival time over 4 years was increased by approximately 200 days (remission 959 days vs no-remission 746 days). Another study found that of 59 cats followed from the diagnosis of diabetes to death, the median survival time of 15 cats that went into remission was 913 days (range 35–3468) compared with 25 days (range 1–1470) of 44 cats that did not go into remission, though the significance was not stated. 6 The number of observations in later years in our study was limited because the average age at diagnosis of diabetes was 12 years, 1 year older than cats in other studies, and relatively few cats lived for another six or more years.6,28,29
Diabetes is almost four times more common in Burmese cats in Australia, the UK and Europe, and Burmese was the second most common breed (21%) in our study.2,3,30 Burmese cats managed with this protocol were five times more likely to achieve remission than other breeds and were more likely to have increased survival. A study of 93 diabetic cats reported that age at diagnosis was significantly higher in Burmese cats compared with other breeds (13.6 vs 10.9 years), and it has also been reported that remission is more likely in cats with a higher age at diagnosis.12,30 However, in our study, there was no significant difference in age at diagnosis between breeds. Burmese cats are known to be genetically predisposed to diabetes, so earlier monitoring and detection may result in higher remission rates. This information was not available in the data; however, Burmese cats in our study had marginally higher mean blood glucose concentrations at diagnosis (22.8 vs 25.1 mmol/l), so early diagnosis does not appear to be a factor in our findings.
Other studies have compared crossbred and purebred cats, not individual breeds, and did not report significant differences in remission or survival between those groups.6,12 Increased age at diagnosis was significantly associated with decreased survival. Euthanasia or death was not associated with diabetes-related complications, and a shorter survival time in general is expected as age increases.
Limitations
While a retrospective study allowed for a large number of cats to be included, the data collected in our study were limited by what was recorded in the history. As such, not all data were available. While a main aim of this protocol was to enable clients to monitor and manage their cat’s diabetes independently, this resulted in some variation in monitoring frequency, which may have impacted remission rates and outcomes. Dietary information was extremely limited, and this made it difficult to fully evaluate the effect it may have had on remission rates.
Only one cat was excluded from the study because the owner decided not to start treatment after they were provided with the insulin dosing protocol, and the cat was euthanased. However, the records identified for potential inclusion were generated on a search of the medical records for cats that had the handout for the insulin dosing protocol printed for the client. Therefore, all clients included were initially willing to consider the management and treatment of diabetes. It is unknown what proportion of cats with newly diagnosed diabetes overall may have been euthanased, because based on the search of medical records, cats that did not have the protocol printed for the client were not flagged for inclusion in this study.
Conclusions
Reported remission rates for diabetic cats vary widely, depending on factors of the study design used, and this can make it challenging for veterinarians to give clients a realistic indication of the likelihood of remission or prediction of survival. In addition, the ongoing management of a diabetic cat at home may feel challenging to some clients, and the costs and time associated with intensive protocols have been reported as reasons why some clients opt to euthanase their cat at the time of diagnosis. The protocol outlined in our study has been used within a cat practice for over 15 years and allows clients to monitor and manage their diabetic cat at home, with veterinary guidance always available when needed. This removes the costs and logistics associated with frequent veterinary visits. Additionally, the protocol used a glucometer readily available at pharmacies, which further helps to reduce costs. It is important to have a spectrum of care options to discuss with clients on diagnosis, to enable them to choose the best option for them and their cat. However, there are some limitations in advising on the probability of an expected outcome based on the small number of studies and the small number of cats in most studies.
Our protocol had a remission rate of 47% for diabetic cats fed a range of diets and with some variability in how often clients monitored their cats. Remission was significantly associated with increased survival time, particularly over the first 4 years after diagnosis. Recommending a lower carbohydrate diet (⩽3.5 g/100 kcal) and using lower glucose concentration cutpoints for 6 months instead of 2 months may result in a higher probability of remission.
Further research is needed to provide a clearer understanding of which treatment factors are most strongly associated with remission, such as frequency of monitoring, glucose cutpoints for increasing or decreasing insulin dose, carbohydrate content of diet and identification of other factors that may impact survival time. This would facilitate clients to make a more informed choice on the most appropriate treatment option for them and their cat.
Footnotes
Accepted: 26 January 2024
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study received funding from Abbot Animal Health and Nestlé Purina.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
