Abstract
Objectives
Assessment of heart size in kittens is important, and there is a need for reference intervals (RIs) to prevent misinterpretation of cardiomegaly in this patient population. The purpose of this study was to generate RIs for echocardiographic and radiographic quantification of cardiac size in healthy kittens.
Methods
In total, 88 kittens aged 6–16 weeks were enrolled in this study. Physical examination, and radiographic and echocardiographic evaluations were performed without sedation. Thoracic radiographs and echocardiographic images were measured to establish RIs for vertebral heart score (VHS), cardiac thoracic ratio (CTR) and multiple echocardiographic variables. N-terminal pro B-type natriuretic peptide (NT-proBNP) was measured. Statistical correlations between echocardiographic parameters and age, body weight and sex were all evaluated and RIs were generated.
Results
Low-grade heart murmurs were appreciated in 26/88 kittens (29.5%). Kittens had a median VHS of 9.5 vertebrae (95% RI 8.0–10.9) and a median CTR of 67.2% (95% RI 54.4–79.8%). Measured NT-proBNP levels were comparable to healthy adult cats with a median of 31 pmol/l (upper reference limit 75 pmol/l). Multiple moderate-to-strong correlations between body weight and age with various echocardiographic parameters were observed and allometric scaling was performed for body weight. RIs for echocardiographic parameters were generated based on patient weight using allometric scaling formulas. Tricuspid valve regurgitation was a common finding and was present in 37.5% (n = 33) of the kittens.
Conclusions and relevance
This study establishes RIs for thoracic radiograph assessment, echocardiography and cardiac biomarkers in kittens, which fills a critical gap in the veterinary literature. The VHS reported in this study is higher than previously reported for adult cats.
Keywords
Introduction
Interpretation of normal cardiac size in kittens is important given the prevalence of heart murmurs, need for preanesthetic evaluation and review in cases of respiratory distress in kittens. This is especially relevant in shelter and rescue situations where limited financial resources could result in dire consequences such as euthanasia if congenital cardiac disease was suspected. Physiologic heart murmurs are common in young animals, 1 and subjective radiographic assessment of kittens and puppies can lead to misdiagnosis of cardiomegaly in some cases. 2 Radiographs, biomarkers such as N-terminal pro B-type natriuretic peptide (NT-proBNP) and echocardiogram are tools used to identify occult cardiac disease in adult cats,3–5 and are likely useful tools in kittens. However, no reference intervals (RIs) exist for the aforementioned diagnostic tests in kittens, making it impossible to objectively assess kittens for normal cardiac size.
In adult cats, radiographic evaluation of cardiac size using the vertebral heart score (VHS) 6 and the cardiac thoracic ratio (CTR) 7 can provide an objective assessment of cardiac enlargement. VHS is a method of assessing cardiac size that compares the sum of the long- and short-axis cardiac dimensions with the length of the vertebral bodies as an internal control. 6 In one study, healthy young cats (aged 3–6 months) had a higher VHS than reported normal values for adult cats. 8 The use of CTR compares the cardiac width with the width of the thoracic cavity on ventrodorsal (VD) projections to assess cardiac size. 7 A study in cats of varying ages demonstrated no significant difference in the CTR measured on VD projections. 9 However, the youngest cats were 6–15 months old in this study.
Additional imaging modalities, such as echocardiography, may be employed to evaluate heart size in kittens. Identification of congenital cardiac defects in kittens has been established,2,10 but normal chamber dimensions have not been reported.
Cardiac biomarkers, such as NT-proBNP, are inexpensive, potentially useful tests to increase suspicion for occult cardiomyopathies4,11 and congenital heart disease3,12 in cats. B-type natriuretic peptides are enzymes that are released in response to ventricular stretch. 13 Studies in human patients have demonstrated the need for age-based RIs for this cardiac biomarker, 14 but NT-proBNP values have not been investigated in kittens.
The objective of this study was to define RIs for radiographic, echocardiographic and NT-proBNP measurements in kittens with the hypothesis that kittens have a larger cardiac size-to-body weight ratio than previously determined values in adult cats.
Materials and methods
This study was approved and conducted in accordance with University of California, Davis Institutional Animal Care and Use Committee protocol #20582.
Animals and study design
Ninety-three apparently healthy kittens (6–16 weeks of age) that were presented to the Community Surgery Service at the University of California, Davis, School of Veterinary Medicine, for spay or castration, were prospectively enrolled between June and September 2018. A physical examination, a packed cell volume (PCV) and total solids (TS), feline leukemia virus (FeLV) and feline immunodeficiency virus (FIV) test, plasma NT-proBNP concentration, thoracic radiographs and echocardiogram were performed for all kittens within a 24 h period and prior to anesthesia. Body weight, sex and murmur grade (if present) were recorded. Kittens exhibiting signs of systemic disease, including significant pulmonary disease, anemia (PCV <25%), 15 congenital cardiac defects, vertebral malformations or an inability to be safely re-strained without sedation, were excluded. All examinations were performed without sedatives or chemical restraint.
Blood sampling and handling
Venous blood samples were collected and placed into EDTA tubes to measure plasma NT-proBNP concentrations, SNAP Combo FeLV Ag/FIV Ab Test (IDEXX Laboratories) and placed in microhematocrit tubes for PCV and TS assessment. For PCV/TS, the microhematocrit tubes were filled, sealed with clay and centrifuged for 3 mins. The tubes were placed on a hematocrit reader chart, and the percentage that intersected the top layer of red blood cells was recorded (PCV). To measure TS, 1–2 drops of plasma from the tube were placed on the glass plate of a refractometer and the observed value was recorded in g/dl. FeLV and FIV testing was performed using the SNAP Combo FeLV Ag/FIV Ab Test (IDEXX Laboratories) based on the manufacturer’s instructions. The EDTA tubes were centrifuged and plasma separated within 60 mins of collection. The plasma samples were stored at −80°C until they were shipped overnight on dry ice to IDEXX Laboratories. Plasma NT-proBNP concentrations were tested using the commercially available, second-generation feline assay (Cardiopet proBNP; IDEXX Laboratories).
Thoracic radiography
Three-view thoracic radiographs were obtained. Thoracic radiographs were reviewed by one observer (CGH) for the presence of vertebral anomalies, severe pulmonary pathology or poor positioning. If identified, these cases were excluded. Cardiac measurements were performed by a trained observer (NF) and reviewed by a board-certified cardiologist (CGH).
VHS was measured from the right lateral projection in all kittens (Figure 1a). The cardiac long axis was measured from the ventral border of the tracheal bifurcation (carina) to the left ventricular apex, and the cardiac short axis was measured perpendicular to the long-axis measurement at the point of maximal cardiac width. Using an adjustable caliper, the long- and short-axis measurements were then repositioned beginning at the cranial aspect of the fourth thoracic vertebrae as previously described. 6 The number of vertebrae within the long-axis measurement and the short-axis measurement were recorded to the nearest 0.1 vertebrae. The sum of both these values was recorded as the VHS.

(a) Right lateral and (b) ventrodorsal thoracic radiographs in a kitten demonstrating the objective measurement of heart size. (a) The vertebral heart score is calculated by the sum of the long-axis (L) and short-axis (S) dimensions of the cardiac silhouette transposed onto the vertebrae and measured as the number of vertebrae beginning from the cranial aspect of the fourth thoracic vertebrae (T4). (b) The ratio between the maximum transverse width of the cardiac silhouette (W) and the transverse width of thorax (T) at the same level are used to calculate cardiac thoracic ratio
The CTR was measured from the VD projection (Figure 1b). The maximal transverse width of the cardiac silhouette was measured. This measurement was divided by the transverse width of the thorax at the same level to obtain the CTR. 8
Echocardiography
All echocardiograms were performed by one of two board-certified veterinary cardiologists (JAS or CGH) using a 12-4 MHz sector array transducer (Philips iE33 Ultrasound and Phillips EPIQ 7c Ultrasound; Philips Healthcare). Each kitten was manually restrained in right and then left lateral recumbency. Standard long- and short-axis views were obtained. 16 Two-dimensional echocardiography (2D), M-mode, color Doppler and spectral Doppler echocardiographic images were recorded. All measurements were performed by one observer (ANS) using an offline work station (Syngo Dynamics; Siemens Medical Solutions). The average value for three consecutive cardiac cycles was obtained for each measurement whenever possible. Images that could not be adequately interpreted were excluded from evaluation.
Heart rate (HR) was obtained using M-mode. Two-dimensional left atrial size was quantified in short axis and long axis. For short-axis assessment, the right parasternal basilar view was used. The left atrial diameter in short axis (LA) at early diastole, defined as the first visible frame after aortic valve closure, was measured and indexed to the aortic root diameter. The diameter of the aorta (Ao) was measured parallel to the commissure of the non-coronary and right coronary aortic valve cusps. The LA was measured parallel to the commissure of the left coronary and non-coronary aortic valve cusps as previously described (Figure 2a). 17 The left atrial diameter in long axis (LAD) was quantified from the right-parasternal four-chamber view by measurement of the maximal left atrial dimension at end systole, defined as the frame prior to the mitral valve opening, as previously described (Figure 2b). 18
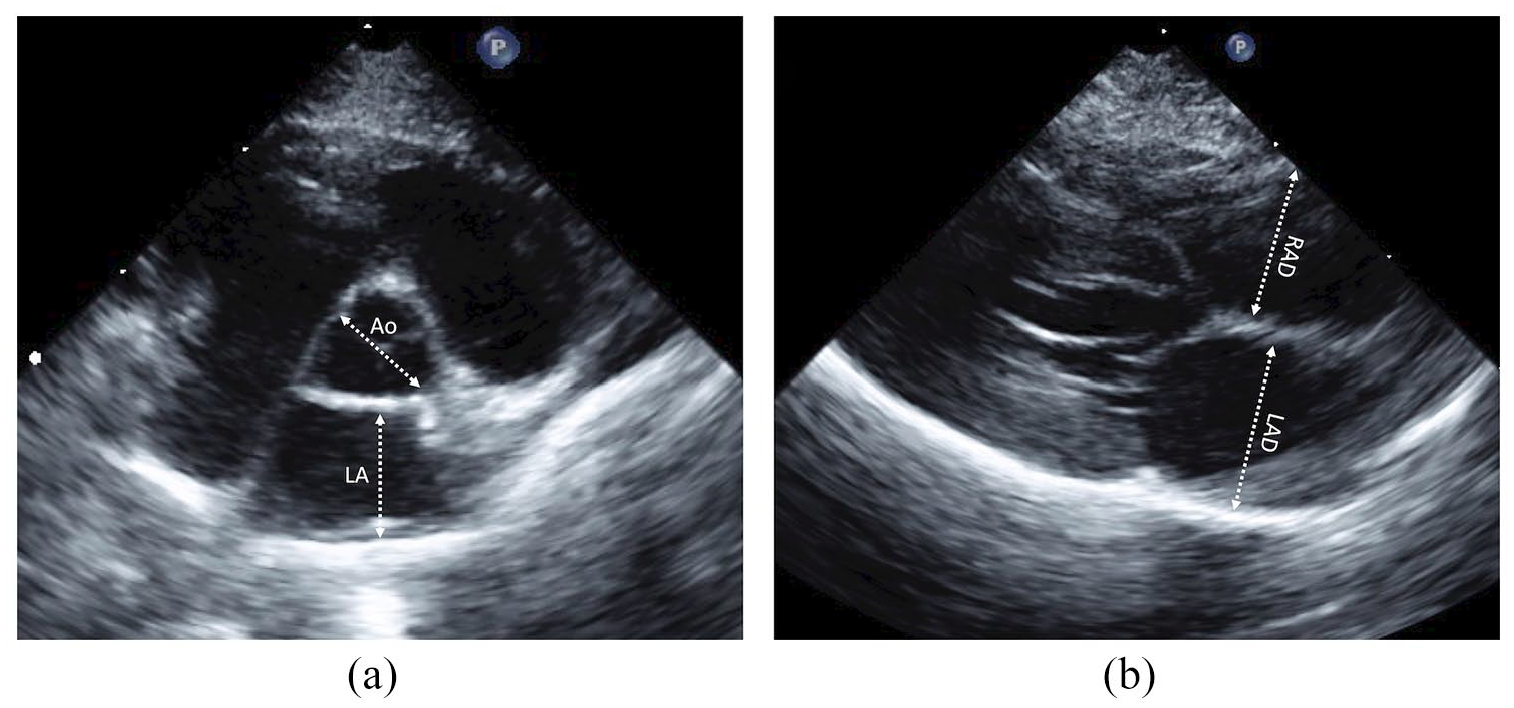
Echocardiographic images from a kitten. (a) The right parasternal short-axis basilar view depicting the measurement of the left atrium (LA) and aorta (Ao); and (b) the right parasternal long-axis view demonstrating the measurements of the left atrial diameter (LAD) and right atrial diameter (RAD)
Maximal left ventricular wall measurements (interventricular septum [IVS] and left ventricular free wall [LVFW]) were obtained using 2D and M-mode at the level of the chordae tendineae and measured using the leading edge-to-leading edge method during systole (s) and diastole (d). Two-dimensional maximal diastolic dimensions of IVS and LVFW also were obtained in right parasternal long-axis imaging planes for each kitten. The maximal aortic velocity (AoV Vmax) was obtained from the left parasternal apical view using continuous wave Doppler aligned parallel with aortic flow, and maximal pulmonary velocity (PV Vmax) was obtained from the right parasternal short-axis view using pulsed wave Doppler with the gate placed just past the level of the valve.
Right atrial and right ventricular size were quantified from the right parasternal long-axis four-chamber view (Figure 2b). Maximal right atrial diameter (RAD) was measured at end systole from the mid-point of the intra-atrial septum to the right atrial free wall, parallel to the tricuspid valve. 19 Right ventricular internal dimension in diastole (RVIDd) and systole (RVIDs) were measured parallel to the tricuspid valve annulus where the tips of this valve contact the endomyocardium when open. 19
Valvular regurgitation was evaluated as present or absent by subjective assessment of color flow Doppler images. Dynamic right ventricular outflow tract obstruction was defined by aliasing of color flow Doppler in the right ventricular outflow tract with dynamic systolic narrowing of the proximal outflow region and corroborating evidence of a dynamic flow profile on continuous wave Doppler with a peak instantaneous systolic velocity ⩾1.7 m/s. 20
Statistical methods
All analyses were performed using commercially available statistical analysis software (GraphPad Prism v7 and STATA v15.1). A D’Agostino and Pearson Omnibus normality test was performed to assess for data distribution. Mean ± SD is reported for all normally distributed data and median and interquartile range (IQR) reported for non-parametric data. RIs were computed using MedCalc (version 15.0; MedCalc Software) using the Robust method with 90% confidence intervals (CIs) for radiographic and echocardiographic data in accordance with the American Society for Veterinary Clinical Pathology Guidelines. 21 For NT-proBNP, a right-sided RI was generated using the non-parametric percentile method. Outliers were detected via Tukey’s test but were not removed. Pairwise Pearson correlation was performed to assess the correlations between patient characteristics and various radiographic and echocardiographic variables. Absolute values of r between 0.1 and <0.4 were considered as weak, 0.4 and <0.7 as moderate, and 0.7 and 1 as strong correlation.
The patient characteristics included weight (kg), age (weeks), sex (male vs female) and HR (beats per min [bpm]). Multiple regression analysis was performed between all patient characteristics and the following echocardiographic parameters: LAD, LA, Ao, IVSd, IVSs, LVFWd, LVFWs, left ventricular internal dimension in diastole (LVIDd), left ventricular internal dimension in systole (LVIDs), left ventricular fractional shortening (FS; %), RAD, RVIDd, RVIDs, right ventricular FS (%) and right ventricular free wall in diastole (RVFWd). A forward selection technique was performed by including the variables with P values <0.1. In order to test the adequacy of the multivariable regression model, a Gronnesby and Boran goodness-of-fit test was performed.
Linear echocardiographic parameters were normalized to body weight (kg) using the constants obtained from allometric scaling or power equation, Y = axb. In this equation, a represents the proportionality constant, b represents the scaling exponent, Y represents the linear echocardiographic parameter and x represents the body weight. Linear regression analysis was then performed on log10(body weight) vs each log10(echocardiographic parameter). This produces the log10 form of the allometric scaling equation, log(Y) = log (a) + b × log(x). In this equation, b represents the slope and a represents the antilog Y-intercept. Prediction intervals were determined from the constant (ac) using the formula: ac = log10−1[log(a) ± t × Sx,y]. In this equation, a is the proportionality constant obtained from the linear re-gression equation, t is the desired Student’s t-statistic for n – 2 degrees of freedom and Sx,y is the standard error of the Y estimate obtained from linear regression.
A P value <0.05 was assigned as significant for all analysis, including multivariable analysis.
Results
Ninety-three healthy kittens (6–16 weeks old) were evaluated. Five kittens were excluded; three had cardiac abnormalities (peri-membranous ventricular septal defect [n = 1], left ventricular outflow tract obstruction [n = 1] and aortic insufficiency [n = 1]), one had a vertebral abnormality and one kitten had a temperament that precluded evaluation. Radiographs and complete echocardiograms were available for the remaining 88 kittens. Plasma from 11 kittens was hemolyzed or lacking sufficient volume for testing; therefore, NT-proBNP was submitted for 77/88 kittens. All 88 kittens were domestic shorthair cats with a mean age of 10.7 ± 2.3 weeks and median weight of 1.3 kg (IQR 0.98–1.5 kg). There were 59 male and 29 female kittens. Quiet (I–II/VI) heart murmurs were appreciated in 26/88 kittens (29.5%), with the remaining kittens noted to have no murmur.
Bloodwork demonstrated a median PCV of 33% (IQR 30–35%) and TS of 6.5 g/dl (IQR 6.0–7.2 g/dl). 22 The kittens had a median NT-proBNP concentration of 31 pmol/l (IQR 24–46 pmol/l, minimum 24 pmol/l, maximum 129 pmol/l) and an upper reference limit of 75 pmol/l with 90% CI (66–129 pmol/l). Three outliers were detected with values of 78, 212 and 129 pmol/l, respectively. The lower limit of detection for the NT-proBNP assay was reported as 24 pmol/l. The median VHS was 9.5 vertebrae (IQR 8.9–9.9 vertebrae) and the median CTR was 62.7% (IQR 49.2–67.3%). Proposed RIs for VHS and CTR are given in Table 1. Analysis of body weight and VHS, weight and CTR were not significantly correlated (P = 0.22 and P = 0.81, respectively). Age and VHS, but not CTR, were weakly but significantly correlated (P = 0.011, r = −0.27 and P = 0.82, respectively).
Reference intervals (RIs) for radiographic parameters in kittens (6–16 weeks of age)

CI = confidence interval
The average HR was 216 ± 27 bpm. Echocardiographic measurements with proposed RIs for the patient population are listed in Table 1 in the supplementary material. Tricuspid valve regurgitation (TR) was observed in 33/88 kittens (37.5%) and mitral valve regurgitation was observed in 5/88 kittens (5.7%). Dynamic right ventricular outflow tract obstruction was observed in one kitten.
There was a constant effect of body weight on echocardiographic parameters; therefore, weight-based means and 95% prediction intervals of various echocardiographic variables derived from allometric scaling parameters are reported in Table 2. In the correlation matrix, HR, weight and age were significantly correlated with various echocardiographic parameters (Table 3). However, sex was not significantly correlated to any of the echocardiographic parameters tested. Multiple regression analysis was performed to predict various echocardiographic parameters from HR, weight, age and sex. HR and weight predicted various echocardiographic parameters with statistical significance (see Table 2 in the supplementary material). Body weight was the best predictor of echocardiographic variable, with significance found for 13/16 echocardiographic variables. Linear regression analysis of logarithmically transformed echocardiographic variables was performed and the results, including constants and allometric scaling exponents, are listed in Table 3 in the supplementary material.
Body weight-based means and 95% prediction intervals of echocardiographic variables in kittens aged 6–16 weeks
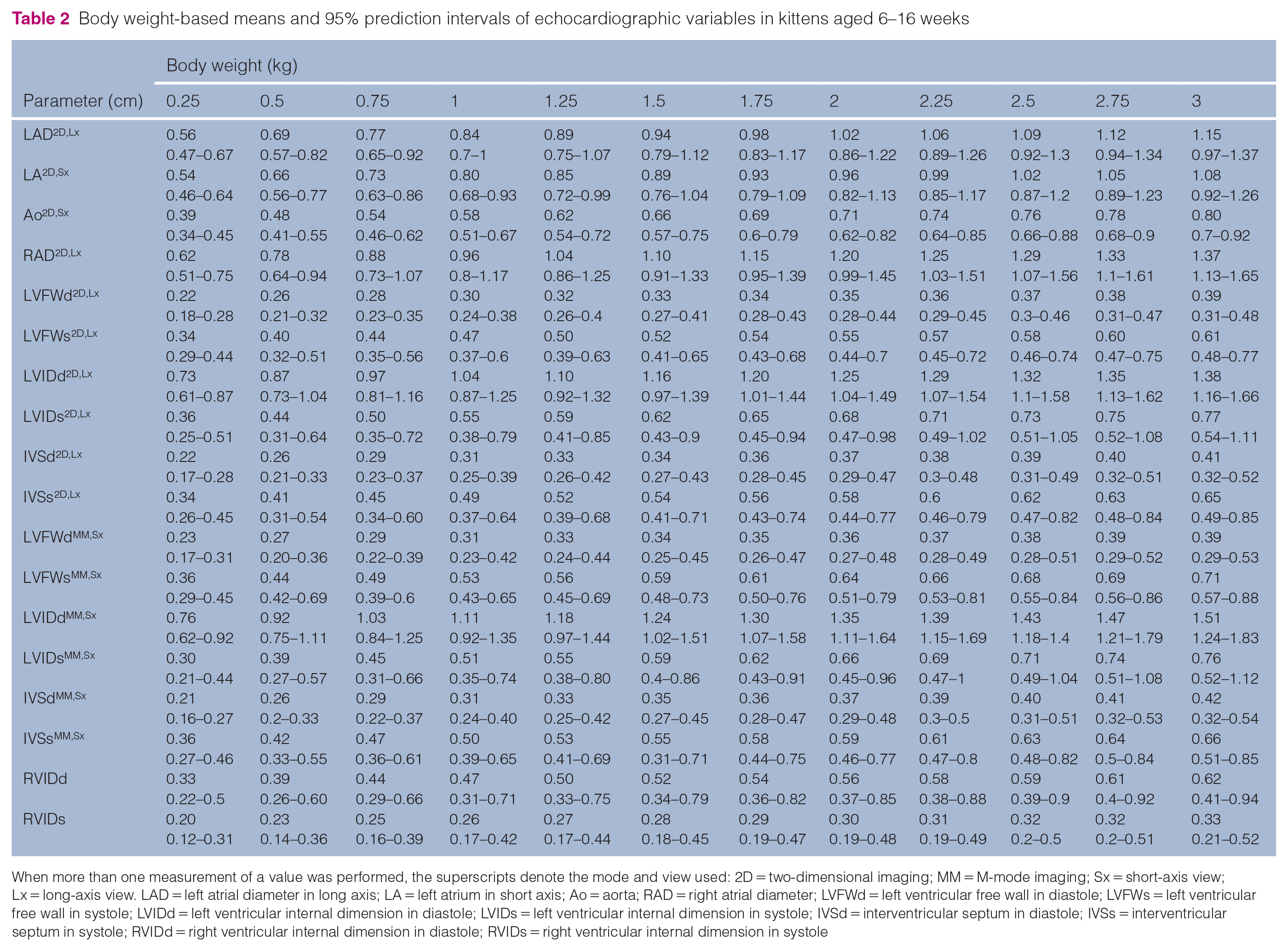
When more than one measurement of a value was performed, the superscripts denote the mode and view used: 2D = two-dimensional imaging; MM = M-mode imaging; Sx = short-axis view; Lx = long-axis view. LAD = left atrial diameter in long axis; LA = left atrium in short axis; Ao = aorta; RAD = right atrial diameter; LVFWd = left ventricular free wall in diastole; LVFWs = left ventricular free wall in systole; LVIDd = left ventricular internal dimension in diastole; LVIDs = left ventricular internal dimension in systole; IVSd = interventricular septum in diastole; IVSs = interventricular septum in systole; RVIDd = right ventricular internal dimension in diastole; RVIDs = right ventricular internal dimension in systole
Correlation of selected echocardiographic parameters to heart rate, weight and age in kittens

When more than one measurement of a value was performed, the superscripts denote the mode and view used: 2D = two-dimensional imaging; MM = M-mode imaging; Sx = short-axis view; Lx = long-axis view. bpm = beats per min; LAD = left atrial diameter in long axis; LA = left atrium in short axis; Ao = aorta; RAD = right atrial diameter; RVIDd = right ventricular internal dimension in diastole; RVIDs = right ventricular internal dimension in systole; RVFW = right ventricular free wall; IVSd = interventricular septum in diastole; IVSs = interventricular septum in systole; LVFWd = left ventricular free wall in diastole; LVFWs = left ventricular free wall in systole; LVIDd = left ventricular internal dimension in diastole; LVIDs = left ventricular internal dimension in systole; LV = left ventricular; FS = fractional shortening
Discussion
This study establishes RIs for radiographic measures of cardiac size (VHS and CTR), plasma NT-proBNP values and echocardiographic assessment in kittens. Owing to the significant effect of weight, RIs based on patient weight for echocardiographic variables were also determined. Effect of weight on echocardiographic variables has also been shown in adult cats.23,24
Cardiac disease in cats is difficult to diagnose when echocardiography is not available given the variable presence of murmurs in normal and diseased cats. Thoracic radiographs and NT-proBNP may be used by shelters or rescue groups when echocardiography is not available. The suspicion for cardiac disease may lead to potentially unnecessary euthanasia, or could affect adoption, especially when financial resources are limited. The radiographic measurement of VHS in kittens in this study was larger than previously reported RIs for adult cats, 25 with most kittens (n = 86/88 [97.7%]) exceeding the previously proposed cut-off of 7.9 vertebrae. 25 This is an essential finding for the interpretation of radiographic cardiac size in kittens.
Plasma NT-proBNP concentration was also evaluated and results are largely consistent with the median of 24 pmol/l (IQR 24–34 pmol/l) reported in normal adult cats. 4 Only 2/88 (2.3%) kittens had NT-proBNP values that exceeded the previously established cut-off for normal cats (<100 pmol/l).4,11 Variation from adult values in NT-proBNP levels have been reported in infants; 14 however, this was not appreciated in our study.
On echocardiographic evaluation, the right-sided heart measurements (RAD and RVIDd) in kittens were comparable to adult cat RIs, 19 but LAD was smaller in kittens than reported values in adult cats. 24 These findings, coupled with the smaller relative body size of kittens, support the impression of right-heart prominence in young kittens and represents the physiologic changes that occur in transition from fetal to adult circulation. 2 Physiologic TR has been previously reported in normal adult cats, 26 and was a common finding in the kittens evaluated. The finding of TR with an apparently structurally normal valve likely represents a physiologic variation and may be a result of higher right ventricular pressures as a kitten transitions from fetal to adult circulation.
There are important limitations of the current study. A single echocardiogram was performed on each kitten at varying time points throughout the day, precluding evaluation of day-to-day variation. Additionally, a single observer performed all measurements on thoracic radiographs and echocardiographic images at a single time point precluding evaluation of inter- and intra-observer differences. All examinations and measures were performed at a single institution by cardiologists or trainees from that institution; therefore, the measures reported may be biased by the training and experience of these observers. Furthermore, standard echocardiographic imaging planes typically optimize the left heart, making right-heart measurements difficult in some cases. While values for normal kitten plasma NT-proBNP levels are reported, further evaluation of NT-proBNP in kittens with cardiac disease compared with kittens with no echocardiographic abnormalities is warranted to establish the utility of this diagnostic in predicting the presence of cardiac disease in kittens.
The RIs proposed in this study are relevant for kittens aged 6–16 weeks of age; further longitudinal studies are warranted to evaluate the changes in heart size as kittens age to adults. Examination of kittens at this age is useful in evaluating for congenital heart defects, but does not rule out the potential to develop acquired cardiac diseases, including hypertrophic cardiomyopathy, in the future.
Conclusions
This study determined radiographic, echocardiographic and NT-proBNP RIs for kittens that are 6–16 weeks of age. Radiographic cardiac size as assessed by VHS is increased in comparison with published RIs in adult cats and this should be considered when relying on this diagnostic for evaluation of cardiac size in kittens. Further studies are warranted to evaluate the utility of VHS and biomarkers in detecting cardiac disease in kittens and longitudinal studies can be considered to evaluate the change in heart size as kittens age.
Supplemental Material
Supplementary Table 1
RIs for echocardiographic parameters in kittens (6–16 weeks of age)
Supplemental Material
Supplementary Table 2
Results of multiple regression analysis of echocardiographic parameters with age, body weight, heart rate and sex (only significant parameters are reported)
Supplemental Material
Supplementary Table 3
Results of linear regression analysis including proportionality constant (a), standard error of the Y estimate, allometric exponents (b), standard error of b, and R2 for logarithmically transformed echocardiographic variables.
Footnotes
Acknowledgements
The authors would like to thank the veterinary technicians from the University of California Davis Cardiology, Neurology/Neurosurgery and Community Surgery Services for their assistance with the kittens, as well as the kittens and rescue groups who participated in this study.
Supplementary material
The following files are available online:
Supplementary Table 1: RIs for echocardiographic parameters in kittens (6–16 weeks of age).
Supplementary Table 2: Results of multiple regression analysis of echocardiographic parameters with age, body weight, heart rate and sex (only significant parameters are reported).
Supplementary Table 3: Results of linear regression analysis including proportionality constant (a), standard error of the Y estimate, allometric exponents (b), standard error of b and R2 for logarithmically transformed echocardiographic variables.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by Maddie’s Fund (grant A19-1871), and the Center for Companion Animal Health, School of Veterinary Medicine, University of California, Davis (grant 2018-45 F/M).
Ethical approval
This work involved the use of non-experimental animals (owned or unowned) and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
