Abstract
Objectives
Feline herpesvirus-1 (FHV-1) is a prevalent cause of ocular disease in cats and limited topical options for treatment currently exist. The first objective of this study was to confirm the efficacy of ganciclovir against FHV-1 in vitro. The second objective was to assess the safety and ocular tolerability of topically applied ganciclovir eye gel (GEG) in healthy cats.
Methods
FHV-1 was used to infect tissue culture wells covered in maximally confluent Crandall–Rees feline kidney cells prior to the addition of three molarities of ganciclovir (8.9 µM, 17.8 µM and 89 µM) before being incubated for 48 h. Ganciclovir efficacy in vitro was then assessed using standard plaque reduction assay. Commercially available GEG (0.15%) was applied q8h to one randomly chosen eye of four healthy cats for 7 days. Commercially available lubricating eye gel (LEG) was applied to the opposite eye q8h. Complete blood counts (CBCs), blood chemistry panels (CHEM) and urinalysis (UA) were performed on all cats before and after the study period. Ocular lesions were assessed daily using a standardized scheme.
Results
Ganciclovir led to a significant reduction in FHV-1 plaque number, area and diameter at all tested molarities in vitro. The highest molarity assessed (89 µM) caused a 100% reduction in viral plaque number. There was no significant difference in ocular lesion scores between eyes receiving GEG and LEG. Animals remained healthy throughout the study period with CBC, CHEM and UA showing no clinically significant alterations.
Conclusions and relevance
Based on the in vitro results, ganciclovir appears to be effective against FHV-1 in vitro. When applied q8h as a commercial 0.15% gel to a small group of cats with normal eyes, this medication was well tolerated. Taken together, these data suggest this medication warrants further investigation in cats with ocular disease caused by FHV-1.
Introduction
Feline herpesvirus-1 (FHV-1) is the leading cause of conjunctival and corneal ulceration in cats, 1 and has been implicated in a variety of painful ocular conditions. 2 Although there have been recent advances in treatment of FHV-1 with oral antivirals,3–5 there are still limited options for topical ophthalmic use. Idoxuridine, 6 trifluridine 6 and cidofovir 7 have all been previously assessed as topical ophthalmic treatments for FHV-1 infection in cats. In vitro data have suggested that ganciclovir has superior efficacy against FHV-1 than these other agents,8,9 and has not been associated with notable ill effects when used in humans or dogs for the treatment of viral ocular surface disease.10–12 Ganciclovir (as a 0.15% topical ophthalmic preparation) has been anecdotally used by some veterinary ophthalmologists for FHV-1 ocular infection in cats. However, the safety and ocular tolerability of this medication has never been assessed, to our knowledge.
The objectives of this study were to assess the safety and ocular tolerability of topically applied 0.15% ganciclovir eye gel (GEG) in healthy cats and to confirm the efficacy of ganciclovir against FHV-1 in vitro. We hypothesized that the GEG would be safe and well tolerated, and that ganciclovir would demonstrate efficacy against FHV-1 in vitro over a range of molarities.
Materials and methods
In vitro efficacy
Crandall–Rees feline kidney cell (CRFK; ATCC) confluent monolayers (passage 7–10) were grown on a 24-well plate in Dulbecco’s Modified Eagle Medium (DMEM) containing 10% fetal bovine serum (FBS) and 1% penicillin–streptomycin (Gibco). Fifty plaque-forming units of plaque-purified wild-type FHV-1 (obtained from a PCR-confirmed archived sample) were adsorbed by the cells over the course of 1 h (at 37ºC and 5% CO2 with plates rocked every 15 mins). The supernatant from each well was then replaced with DMEM containing 1% methylcellulose, 2% FBS and 1% penicillin–streptomycin ± ganciclovir (8.9 µM/17.8 µM/89 µM). Positive (no ganciclovir) and negative controls (no virus) were included and all wells were repeated four times each. The plates were then incubated at 37°C with 5% CO2 for 48 h with visual assessment (by one investigator, ACL) of cellular viability using light microscopy prior to fixation. All wells were fixed with methanol then stained with 0.5% crystal violet solution (Fisher Scientific) before the assessment of plaque number, area and diameter using a light microscope (Olympus MVX10 Research Macro Zoom with Olympus DP72 digital camera system) and measurement software (Olympus CellSens). For plaque area and diameter, 10 plaques (or as many as were available in the case of the higher molarities of ganciclovir) were chosen from each well for measurement. Each well was divided into four quadrants. Plaques chosen for measurement were closest to the central intersection of the quadrants. Three plaques were chosen from diagonally opposite quadrants with two plaques chosen from the remaining quadrants (10 plaques total).
A one-way ANOVA was used to detect differences in plaque numbers among groups. A mixed ANOVA with each plate well as the random effect and treatment group as the fixed effect was used to detect differences in plaque diameter and area among groups. Where differences were found to exist, post-hoc multiple comparisons were performed using Tukey tests. Significance was set at P <0.05.
Clinical trial in healthy cats
All procedures were performed in accordance with an approved Louisiana State University Institutional Animal Care and Use Committee protocol (19-022). Four colony-housed, healthy adult neutered male cats aged between 6 and 7 years old, were used for this study. All animals received physical examinations by a veterinarian immediately before and after the trial. All animals received non-masked baseline eye examinations performed by a veterinary ophthalmologist (ACL), which were repeated daily throughout the 7-day study period. This included slit-lamp biomicroscopy (Kowa SL-17), indirect fundoscopy (28D [Keeler; Volk]), tonometry (TonoVet [iCare]; ‘d’ setting), Schirmer tear testing type 1 (STT; Schering-Plough Animal Health) and fluorescein staining (BioGlo; HUB). Ocular clinical scores were assigned according to a previously described scheme (SPOTS ocular scoring scheme), 13 with animals assigned a daily score of 0–58 per eye (total of 0–406 per week), with 0 representing normal and 58 representing severe disease. All animals were sedated with a combination of dexmedetomidine (3 µg/kg [Dexdomitor; Zoetis]), ketamine (5 mg/kg; Dechra) and butorphanol (0.2 mg/kg [Torbugesic-SA; Zoetis]) administered by intramuscular injection to facilitate collection of blood and urine samples.
Complete blood counts (CBCs), serum biochemistry panels (CHEM) and urinalysis (UA) were performed on samples obtained from all animals 24 h prior to the start of the study period and immediately afterwards (by Louisiana State University School of Veterinary Medicine Clinical Pathology Laboratory).
Topical ocular 0.15% GEG (Zirgan; Bausch and Lomb) was applied to one randomly chosen (by coin toss) eye q8h in each cat. In the opposite eye, a carbomer-based lubricating eye gel (LEG [Optixcare Eye Lube; CLC Medica]) was applied q8h. The volume dispensed at each dosing was sufficient to coat the ocular surface. The listed GEG inactive ingredients (in order) were carbomer homopolymer, water for injection, sodium hydroxide, mannitol and benzalkonium chloride. The listed ingredients (in order) in the LEG were sterile water, sorbitol, carbomer, sodium hydroxide, disodium EDTA and cetrimide.
Data analyses were performed using commercially available software (JMP Pro 14.1.0). Values for clinical score data, intraocular pressure (IOP) and STT were assessed for differences between eyes receiving LEG and GEG with a paired t-test and equivalence testing. The residuals from all models were checked for normality with the Shapiro–Wilk test and it was determined that with the exception of some outliers at both ends of the distribution, the residuals were normally distributed. Significance was set at P <0.05.
Results
In vitro efficacy
The results for the in vitro testing of ganciclovir against FHV-1 are summarized in Table 1. All negative control wells demonstrated an absence of viral plaque formation. All wells demonstrated evidence of healthy (normal cell morphology and cell size) CRFK cells (at approximately 100% confluence) prior to and following fixation. CRFK cell numbers were estimated to be similar in all wells, including control wells. Relative to positive controls, the use of 89 µM ganciclovir led to 100% inhibition of plaque formation. Differences in plaque number, area and diameter were significantly different among molarities shown in Table 1 (P <0.0001). Based on the results in Table 1, the calculated IC50 (number) was 11.4 µM, the IC50 (diameter) was 9.9 µM and the IC50 (area) was 7.4 µM.
Results of in vitro assessment of three molarities of ganciclovir (GCV) against feline herpesvirus-1
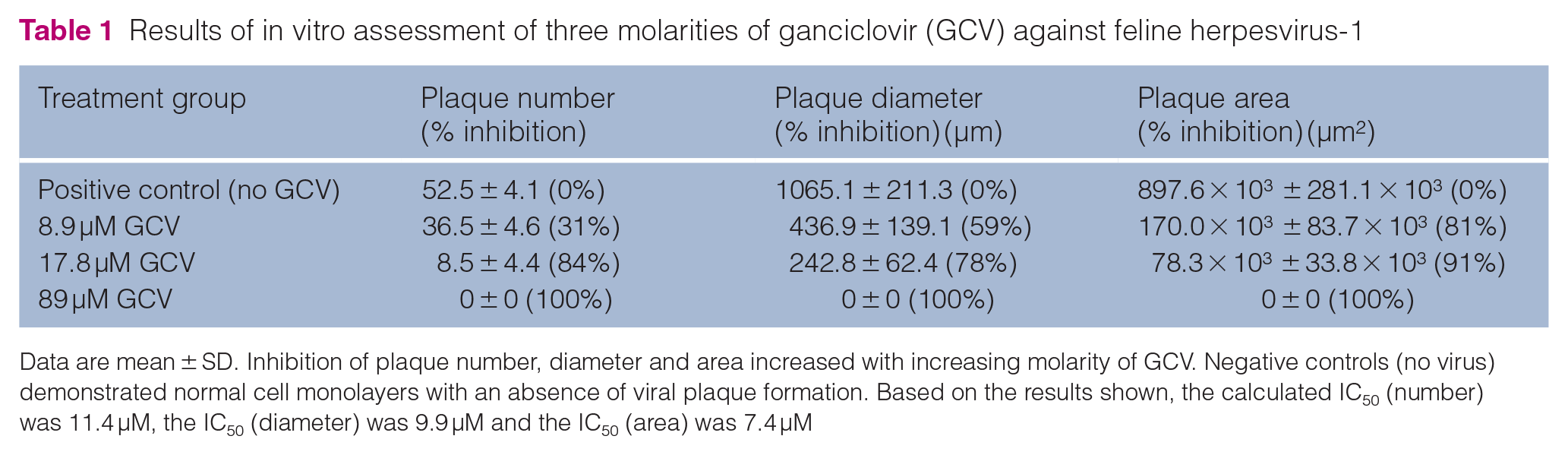
Data are mean ± SD. Inhibition of plaque number, diameter and area increased with increasing molarity of GCV. Negative controls (no virus) demonstrated normal cell monolayers with an absence of viral plaque formation. Based on the results shown, the calculated IC50 (number) was 11.4 µM, the IC50 (diameter) was 9.9 µM and the IC50 (area) was 7.4 µM
Clinical trial in healthy cats
Both GEG and LEG were well tolerated by all cats. The ocular examination findings are summarized in Figure 1. All four animals were assigned an ocular clinical score of 0/58 in both eyes at study initiation. The overall ocular lesion scores assigned over 7 days from the ocular grading scheme were 5/406 GEG and 6/406 LEG (cat 1), 5/406 GEG and 2/406 LEG (cat 2), 4/406 GEG and 3/406 LEG (cat 3) and 5/406 GEG and 7/406 LEG (cat 4). No significant difference was detected for overall ocular lesion score over 7 days (P = 0.836) between eyes receiving GEG or LEG. IOP (mean ± SD) was 22.3 ± 4.7 mmHg in eyes receiving GEG and 22.7 ± 5.7 mmHg in eyes receiving LEG (Figure 2a). No significant difference was detected for IOP (P = 0.634) between eyes receiving GEG or LEG. STT readings (mean ± SD) were 17.1 ± 3.1 mm/min in eyes receiving GEG and 17.2 ± 4.1 mm/min in eyes receiving LEG (Figure 2b). No significant difference was detected for STT (P = 0.944) between eyes receiving GEG or LEG. With a difference of ⩽3 units for IOP (P = 0.0007), STT (P = 0.0007) and ocular lesion score (P = 0.0446) it was determined that the GEG and LEG were equivalent (by equivalence testing).

Summary of ocular findings during the study period with an assigned score for eyes receiving ganciclovir eye gel (GEG) and lubricating eye gel (LEG). Each bar represents the number of eyes for each treatment, which demonstrated the specified clinical sign at some point during the study period. The maximum score for any eye at one time was 1. Note that the scoring scheme had a range of 0 to 3 for conjunctival hyperemia and conjunctival discharge and 0 to 4 for chemosis. All other parameters assessed were scored as 0 (normal)

(a) Intraocular pressure (IOP) and (b) Schirmer tear test (STT) results for eyes receiving either ganciclovir eye gel (GEG) or lubricating eye gel (LEG). No significant differences were found for either IOP or STT readings between eyes receiving GEG and LEG
Physical examinations before and after the trial period determined that the animals were overweight (all body condition scores were 7/9) but otherwise normal. No clinically significant alterations in CBC, CHEM or UA were detected after the use of GEG or LEG (see Table S1 in the supplementary material).
Discussion
This small study demonstrates that 0.15% GEG is well tolerated by cats without local or systemic toxicity over a period of 7 days when applied q8h. All animals in the study experienced some mild ocular irritation from both the GEG and the LEG. The LEG chosen as the control medication is of a similar composition to the base of the GEG used. Topical ophthalmic ointments have been previously noted to be locally irritating to cats, with varying degrees of irritation depending on the formulation. 14
The GEG was applied to one eye of each cat q8h for 7 days. In a clinical situation for the treatment of FHV-1, therapy may be more prolonged or applied more frequently owing to the typical nature of viral ocular disease in cats.6,15 Application of topical ocular gel medications in cats can be challenging and may exacerbate stress-related diseases such as FHV-1. For this reason we chose to assess a q8h frequency of application as this is the authors’ (ACL, RTC) clinical recommendation for other topical antiviral medications for cats with ocular FHV-1. However, it is recognized that higher frequencies of topical antiviral medications may be recommended clinically, given that available agents are virostatic. Four animals were assessed as it was expected that the GEG would be well tolerated in cats and using a cohort of this size allowed for detailed assessment of ocular and systemic effects of the drug. However, without using a substantially larger cohort of animals in a follow-up to this small study, it is not possible to determine if rare local/systemic toxicity in some animals may occur.
The animals used for the clinical trial were overweight but otherwise healthy. All subjects were found to have some bloodwork and/or UA values outside the reference intervals both before and after the trial period that could potentially be attributed to the use of sedation, unrelated/undetected age-related conditions or obesity. Ultimately, no clinically relevant alterations in CBC, CHEM or UA were detected following the use of GEG q8h for 7 days in this group of cats.
One potential limitation of the present study is the absence of a quantitative method of in vitro cytotoxicity assessment. If ganciclovir were to cause cytotoxic effects on the CRFK cells, this may potentially affect IC50 values obtained. However, an experienced observer (ACL) performed and observed all in vitro experiments, and no detrimental effect of the drug on the CRFK cells was visually detected at any of the concentrations assessed. Another potential limitation is the use of a single 24-well plate to perform all in vitro experiments (with replicates in individual wells on the same plate). As a result, unidentified variables could conceivably have affected the outcome of the experiment (such as incubator variables and variations in CRFK cell growth).
Ganciclovir has been assessed in vitro against FHV-1 in the past and IC50 values have been calculated.8,16 To avoid experimental induction of FHV-1 disease in host animals, we confirmed efficacy in vitro using three molarities of the drug (which are one times, two times and ten times the previously reported average IC50 value), 16 all of which were notably lower than the equivalent contained in the 0.15% commercial eye gel. The values we obtained for reduction in plaque number, area and diameter were mostly in agreement with previous assessments.8,9 The highest molarity of ganciclovir we used in vitro (89 µM) caused 100% inhibition of viral plaque formation. This concentration is much lower than that contained in the 0.15% GEG assessed (approximately 5.8 × 103 µM), suggesting that GEG may be effective when used in cats infected with FHV-1. However, differences between this in vitro result and efficacy results in vivo would be expected for many reasons, including different cell type and exposure duration. Because ganciclovir is virostatic (in common with other available topical ophthalmic antivirals), frequent daily use is likely to be most effective when treating animals with FHV-1. Future prospective controlled studies should assess the use of GEG in animals infected with FHV-1 to assess efficacy through reduction of viral load and improvement of clinical signs.
Conclusions
Based on the in vitro results, ganciclovir appears to be effective against FHV-1 in vitro. When applied q8h as a commercial 0.15% gel to a small group of cats with normal eyes, this medication was well tolerated. Taken together, these data suggest this medication warrants further investigation in cats with ocular disease caused by FHV-1.
Supplemental Material
Table S1
Summary of clinical pathology values noted to be beyond reference intervals for CBC, CHEM and UA
Footnotes
Acknowledgements
The authors would like to thank Dr Jon Fletcher for facilitating access to the animals used for this study.
Supplementary material
The following file is available online:
Table S1: Summary of clinical pathology values noted to be beyond reference intervals for CBC, CHEM and UA.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding for this study was provided by Louisiana State University School of Veterinary Medicine start-up funds (Lewin).
Ethical approval
This work involved the use of experimental animals and the study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). For any animals or humans individually identifiable within this publication, informed consent (either verbal or written) for their use in the publication was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
