Abstract
Objectives
An affordable and effective treatment is needed to manage feline hypersomatotropism. The aim of this study was to assess whether treatment with oral cabergoline for 90 days in cats with hypersomatotropism and diabetes mellitus improved diabetic and insulin-like growth factor 1 (IGF-1) control.
Methods
This was a prospective cohort non-blinded pilot study enrolling client-owned cats with spontaneously occurring diabetes mellitus and hypersomatotropism. Cats received oral cabergoline (5–10 µg/kg q24h) for 90 consecutive days. Serum IGF-1 and fructosamine concentrations were measured on days 1, 30 and 90. Quality of life was determined using the DIAQoL-pet questionnaire on days 1 and 90.
Results
Nine cats were enrolled and eight completed the study. There was no significant change in the following: IGF-1 (day 1 median 2001 ng/ml [range 890–2001 ng/ml]; day 30 median 2001 ng/ml [range 929–2001 ng/ml]; day 90 median 1828 ng/ml [range 1035–2001 ng/ml]; χ2(2) = 0.667, P = 0.805); fructosamine (day 1 median 499 µmol/l [range 330–887 µmol/l], day 30 median 551 µmol/l [range 288–722 µmol/l], day 90 median 503 [range 315–851 µmol/l]; χ2(2) = 0.581, P = 0.764); or DIAQoL-pet score (median on day 1 –2.79 [range –4.62 to –0.28], median on day 90 –3.24 [range –4.41 to –0.28]; P = 0.715). There was a significant change of insulin dose (χ2(2) = 8.667, P = 0.008) with cats receiving higher insulin doses at day 90 compared with day 1 (median on day 1 was 0.98 [range 0.63–1.49] and median on day 90 was 1.56 [range 0.49–2.55] units/kg q12h; P = 0.026).
Conclusions and relevance
Cabergoline did not improve diabetic control or normalise insulin-like growth factor concentration, or improve patient quality of life.
Introduction
Hypersomatotropism (HST) in cats is a condition caused by chronic excessive growth hormone (GH). Most cats with HST have concurrent diabetes mellitus (DM), which can be difficult to control using treatments that only target glycaemic control.
Medical management using pasireotide or surgical management via hypophysectomy has improved GH and diabetic control in cats with HST.1–3 However, these treatments are often too costly for owners and effective alternative modalities are needed.
In human medicine, HST is known as acromegaly owing to the phenotypic changes induced by the condition. There are three main medical management options for acromegaly in humans, which are: somatostatin receptor agonists (SRAs) such as octreotide or pasireotide; dopamine receptor agonists (DRAs) such as bromocriptine or cabergoline; and GH receptor antagonists, namely pegvisomant. Recommendations suggest the primary medical treatment of patients who have moderate-to-severe disease should be SRAs and patients who have mild disease (serum insulin-like growth factor 1 [IGF-1] <2 times the upper limit of the age-adjusted range) can be treated using a DRA.4,5 The direct mechanism of action of GH-secretion inhibition by DRAs is thought to be via somatotrope dopamine 2 receptors (D2Rs) within the pituitary. 6 As cats with HST have pituitary expression of D2Rs, therapy with a DRA might result in improved GH control and therefore diabetic control. 7
Cabergoline and bromocriptine are DRAs with high affinity for D2Rs in rats and monkeys.8,9 Cabergoline is the D2R-specific DRA with more favourable properties, having a longer action of duration and thus requires less frequent dosing; it is better tolerated; and it exhibits increased insulin-sensitising effects independent of GH reduction vs bromocriptine.10,11 An oral preparation of cabergoline was licensed and available for use in cats at the time of study (Kelactin; Kela NV) Cabergoline has also been proven to be well tolerated when administered for several months in both dogs and humans.12,13
The aim of the pilot study was to determine if cats with HST and DM experienced decreased serum IGF-1 and improved diabetic control determined by serum fructosamine concentration and insulin dose requirement when receiving once-daily treatment with oral cabergoline. A secondary aim of the study was to determine whether this treatment resulted in an improved quality of life (QoL) for these cats.
Materials and methods
The study was approved by the Ethics and Welfare Committee of the Royal Veterinary College, UK (URN 2016 1604). Informed written consent was obtained from all owners before enrolment. Cats with HST were prospectively enrolled between 1 October 2016 and 31 May 2017. Inclusion criteria were DM that had been treated for at least 4 weeks prior to enrolment, serum IGF-1 concentration >700 ng/ml with pituitary enlargement (>4 mm dorsoventral height) or serum IGF-1 >1000 ng/ml without pituitary imaging. As IGF-1 >1000 ng/ml has been shown to have a positive predictive value for HST of 95% in the UK diabetic cat population where the prevalence of HST was 25%, the necessity of additional pituitary imaging in this group was not deemed essential. 14 If contrast-enhanced pituitary imaging had not already been performed and the owner consented to the procedure, then this was undertaken on day 1, as previously described. 15 Exclusion criteria were poor patient tolerance of veterinary procedures, uncontrolled hyperthyroidism, insulin antagonist therapy within the preceding 4 weeks prior to enrolment or if they had a disease that was more critical to the cat’s welfare than HST, as judged by the attending clinician.
Owners of eligible cats were offered a reduced fee for contrast-enhanced CT of their cat, a free supply of PZI insulin (Prozinc; Boehringer Ingelheim) and diabetic cat food (Purina DM; Nestle Purina) for the length of the study. Owners paid for the initial period of hospitalisation and the cost of cabergoline during the study. Owners were encouraged to perform home blood glucose monitoring. As a result of the death of one cat owing to the development of congestive heart failure (CHF), all subsequently enrolled patients underwent echocardiogram examination at enrolment and risk of progressive cardiac disease was discussed with owners. Owners were instructed to intermittently monitor their cat’s resting respiratory rate at home because this is reported to be a sensitive indicator of CHF in cats. 16 Owners were instructed to contact the investigator if their cat’s average resting respiratory rate was greater than 36 breaths per minute. No owner declined to enrol on the study after receiving this information.
On day 1, a blood sample was collected for pretreatment complete blood count, serum biochemistry, serum IGF-1 and fructosamine concentration determination. Cats had a subcutaneous interstitial glucose monitor (Guardian REAL Time Continuous Glucose Monitoring System; Medtronic) placed to measure glycaemic control for an initial period of hospitalisation of 4 days. 17 Cats received the same insulin dose and frequency as prescribed by their referring veterinarian prior to enrolment on day 1. Cats were prescribed oral cabergoline once daily starting on day 2 and were discharged on day 5. Cats were monitored for possible adverse drug effects, whether there was increased sensitivity to insulin therapy as determined by glycaemic control during hospitalisation, clinical sign monitoring by their owners and repeat fructosamine measurement on days 30 and 90, and IGF-1 measurements were repeated on days 30 and 90.
Patient QoL was assessed by requesting owners to complete the psychometric DIAQoL-pet questionnaire at days 1 and 90. The DIAQoL-pet has previously been validated to quantify owner-perceived QoL of the owner and diabetic pet, and can quantify the effect of treatment upon their diabetic cat’s QoL, as well as their own. 18
Statistical analysis
A P value <0.05 was considered significant. Data were analysed for normal distribution visually using histograms and by performing Shapiro–Wilk tests. Any IGF-1 concentration >2000 ng/ml was analysed as being 2001 ng/ml. Non-normally distributed data are presented as median and range, and data with normally distributed data presented as mean ± SD. Friedman tests and post-hoc related samples Wilcoxon signed rank tests with Bonferroni adjustment where appropriate were performed to compare repeated measures IGF-1, fructosamine and insulin dose data on days 1, 30 and 90. The related samples Wilcoxon signed rank test was used to compare QoL data on days 1 and 90. The Spearman rank test was used to compare the strength of correlation between data. Statistical analyses were performed using statistical software (GraphPad Prism version 8.4.0 for macOS [GraphPad Software] and IBM SPSS Statistics Version 26.0.0.0 for macOS [IBM]).
Results
Nine cats were enrolled, eight cats completed the study and one cat (cat 1) died during the study (see the table in the supplementary material for the raw data of all nine cats). The data from the cat that did not complete the study was excluded from these analyses of insulin dose, fructosamine, IGF-1 and DIAQoL-pet scores. All nine cats were domestic shorthairs, six were male and three were female, mean age was 10.8 ± 2.8 years, mean weight was 4.8 ± 0.8 kg, mean pituitary dorsoventral height was 6.3 ± 1.6 mm and median pituitary volume was 0.088 cm3 (range 0.048–0.327 cm3). Cat 6 did not undergo intracranial CT imaging because of the concern that this patient had a high risk for CHF, as determined by echocardiographic measurements. Home blood glucose monitoring was performed by 4/9 owners. All owners reported that they were successful when giving cabergoline to their cats and that the medication had been handled as per the manufacturer’s instructions.
The first three cats enrolled on the study received 5 µg/kg cabergoline q24h but had a dose increase to 10 µg/kg q24h at day 30–35, and the remaining cats had a cabergoline dose of 10 μg/kg q24h from enrolment. Cat 7 did not have IGF-1 and fructosamine measurements at day 30.
Serum IGF-1 results
There was no significant change in serum IGF-1 concentration over the 3 months of the study (day 1 median 2001 ng/ml [range 890–2001 ng/ml]; day 30 median was 2001 ng/ml [range 929–2001 ng/ml]; day 90 median was 1828 ng/ml [range 1035–2001 ng/ml]; χ2(2) = 0.667, P = 0.805) (Figure 1). Four cats experienced a decrease and four an increase in IGF-1 from day 1 to day 90. The median pituitary volume of cats that experienced an IGF-1 reduction was not significantly different compared with those that did not experience a reduction in IGF-1 (0.086 vs and 0.133 cm3, P = 0.94).

Patient serum insulin-like growth factor 1 (IGF-1) on days 1, 30 and 90. There was no significant change of IGF-1 during the study (χ2(2) = 0.667; P = 0.805)
Serum fructosamine and insulin dose
There was no statistical difference of fructosamine concentration at any time point (day 1 median 499 µmol/l [range 330–887 µmol/l], day 30 median 551 µmol/l [range 288–722 µmol/l], day 90 median 503 µmol/l [range 315–851 µmol/l]; χ2(2) = 0.581, P = 0.764) (Figure 2). An insulin dose increase was prescribed for 6/8 cats. There was a significant change in insulin dose prescribed during the study (χ2(2) = 8.667; P = 0.008), with cats receiving higher insulin doses on day 90 vs day 1 (median on day 1 was 0.98 units/kg q12h [range 0.63–1.49 units/kg q12h] and median on day 90 was 1.56 units/kg q12h [range 0.49–2.55 units/kg q12h]; P = 0.026) (Figure 3).

Patient serum fructosamine on days 1, 30 and 90. There was no significant change of fructosamine during the study (χ2(2) = 0.581; P = 0.764)
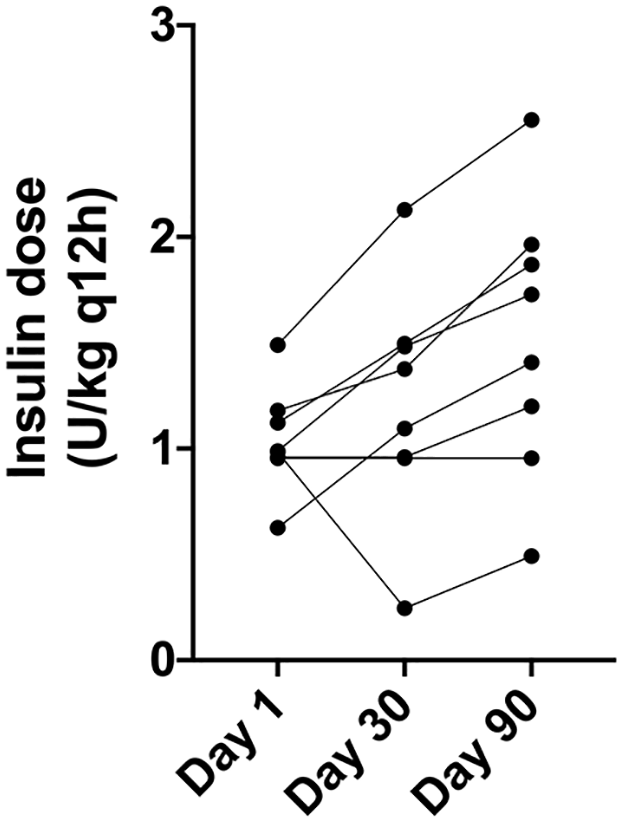
Patient insulin dose on days 1, 30 and 90. There was a significant change in insulin dose prescribed during the study (χ2(2) = 8.667; P = 0.008), with cats receiving higher insulin doses on day 90 vs day 1 (median on day 1 was 0.98 units/kg q12h [range 0.63–1.49 units/kg q12h] and median on day 90 was 1.56 units/kg q12h [range 0.49–2.55 units/kg q12h]; P = 0.026)
DIAQoL-pet scores
The DIAQoL-pet was completed by 6/8 cat owners. There was no statistically signficant change in DIAQoL-pet scores between day 1 and day 90 (median on day 1 was –2.79 [range –4.62 to –0.28], median on day 90 was –3.24 [range –4.41 to –0.28]; P = 0.715) (Figure 4). DIAQoL-pet scores negatively correlated with insulin dose on day 1 but not on day 90 (Spearman’s rank –0.871; P = 0.034 and –0.257, P = 0.623, respectively).

Patient DIAQoL-pet scores on days 1 and 90. There was no significant change of DIAQoL-pet score (P = 0.715)
Potential adverse drug effects
One cat died during the study period. General physical examination at enrolment of this cat was unremarkable apart from a grade 2/6 systolic cardiac murmur. An echocardiogram was not performed at enrolment. On day 82, the cat developed tachypnoea and was diagnosed with supraventricular tachycardia and CHF. The cat’s owners elected for it to be euthanased and a post-mortem examination was declined.
Cat 7 did not have fructosamine or IGF-1 data for day 30 because it was hospitalised at its local veterinary practice for an episode of presumed pancreatitis on day 28. Cat 5 experienced reduced appetite and small intestinal diarrhoea, which resolved within 1 week without specific treatment, and the same cat experienced asymptomatic hypoglycaemia on day 60. Two other cats experienced self-limiting inappetence of unknown cause lasting <1 week. No owner requested withdrawal from the study owing to concerns over possible adverse drug effects.
Discussion
This is the largest case series to date to describe cats with HST and DM treated with cabergoline. Although a direct measurement of insulin sensitivity was not performed, the trend for increasing requirement for exogenous insulin with similar serum fructosamine concentrations infers that the cats experienced increasing insulin resistance. This is likely due to ongoing uncontrolled HST because cabergoline did not reliably control IGF-1 concentration by decreasing it to within the reference interval (RI).
There are no published studies describing the pharmacokinetics of cabergoline in cats. The plasma elimination half-life is between 63 to 109 h in humans. 8 The initial dose of 5 μg/kg q24h by mouth was chosen because this was the licensed dose for the treatment of inappropriate lactation in cats, and this dose was effective in terminating pregnancy in queens, which suggests effective suppression of prolactin secretion. 19 This dose is equivalent to a 0.5 mg q24h dose for an average human using mg/kg dosing, which is reported to result in GH suppression in humans with acromegaly. 20 However, other studies have reported using higher doses of cabergoline to treat cats and the medication was well tolerated, and some humans with acromegaly require higher doses to achieve a biochemical response.21,22 This was part of the rationale for increasing the initial cabergoline dose from 5 to 10 μg/kg q24h for cats 4–9.
The findings of this study differ from the results of a case series of three cats with HST and DM treated with 10 μg/kg q48h cabergoline for 3 months. 23 All the cats in that study experienced decreased IGF-1 and improved insulin sensitivity after treatment. It is possible that the cause of HST in cats was different to the cause of HST in this study as different pituitary adenoma subtypes are known to respond differently to medical management.24,25 Response to cabergoline therapy can also vary depending on prior treatments, alternative splicing of DRD2 mRNA, magnitude of dopamine receptor expression at the protein level or defective signalling pathways downstream of D2R stimulation.26–29 Also, cabergoline is typically recommended for the treatment of acromegaly in humans who have mild clinical signs and IGF-1 concentrations <1.5–2 times above the RI.5,30 There were 6/9 cats that had serum IGF-1 concentrations >2000 ng/ml at the start of the study and only two cats had IGF-1 concentrations less than twice the laboratory RI. It is possible that the severity of HST in these cats was inappropriate for cabergoline treatment. Apart from decreasing IGF-1, cabergoline might exert antidiabetic effects by increasing insulin sensitivity without affecting GH levels. 31 This mechanism could explain the response of cat 5, which was receiving a lower dose of insulin and had lower serum fructosamine despite slightly increased IGF-1 at day 90 vs day 1.
Consideration and measurement of QoL is increasingly important when undertaking clinical studies and particularly important in veterinary medicine since a common reason for euthanasia of a cat with DM is an owner-perceived poor pet QoL.32–34 Acromegaly is associated with reduced QoL in humans and improves, but does not normalise, with disease control.35–38 Diabetes in cats is associated with an owner-perceived reduced QoL of their cat and improved DM control has been associated with improved QoL.18,39 QoL scores did not improve during the study, which could be explained by poor biochemical control of either HST or DM.
Clinical signs that might have been compatible with drug-induced adverse effects include an episode of presumed acute pancreatitis in one cat; 2/9 cats experienced inappetence, which was presumed to not be associated with pancreatitis; and one cat, which had experienced inappetence, also experienced self-limiting small intestinal diarrhoea lasting <1 week. Gastrointestinal adverse effects of cabergoline have previously been reported in cats receiving 15 μg/kg q24h. 21 Nausea, vomiting and vertigo are the most commonly reported side effects in cabergoline-treated humans with hyperprolactinaemia, affecting up to one-third of those treated. 40 Cabergoline does not appear to induce pancreatitis in humans, and it is possible the cat that experienced pancreatitis did so independently of cabergoline treatment.
In 2008, the Medicine and Healthcare products Regulatory Agency published a statement that cabergoline therapy might be associated with an increased risk of cardiac fibrosis, and cardiac valvulopathy should be excluded prior to starting cabergoline therapy. 41 A recent systematic review concluded that the risk of cabergoline-associated valvulopathy in patients with prolactinoma is low, but the authors recommend an initial echocardiogram prior to starting cabergoline therapy. 42 Patients affected by Parkinson’s disease often receive cabergoline doses >3 mg daily (around 40 μg/kg q24h for the average human in the UK) vs 0.25–3 mg/week (equating to 3.125–39 μg/kg per week) in patients affected by prolactinoma and appear to have an increased risk of cabergoline-induced cardiomyopathy.43–45 The doses of cabergoline used in the present study are more comparable with those used to treat prolactinoma than Parkinson’s disease. Nonetheless, as 30–50% of apparently healthy cats without a heart murmur have echocardiographic evidence of heart disease, it is possible that the use of cabergoline might have contributed to progressive cardiac disease in patient 1, which died on day 82.46,47 It is also possible that this patient experienced progressive heart disease regardless of cabergoline therapy, because HST in cats is associated with a hypertrophic cardiomyopathy phenotype and increased risk of CHF. 48
The low patient number will have affected the power of the study. However, this was a pilot study and, as a previous report described a good response of three cats with diabetes and hypersomatotropism treated with cabergoline, this study provides evidence that not all cats experience a good response. 23 Another limitation is that IGF-1 concentrations >2000 ng/ml were not diluted to obtain the exact IGF-1 concentration. The lack of exact IGF-1 enumeration will have limited our ability to determine a difference during the study. Nonetheless, despite this limitation, we can be confident reporting that no cat achieved normalisation of serum IGF-1.
Conclusions
Although the study was underpowered, cabergoline does not appear to reliably control HST as the cats in this study did not achieve IGF-1 control nor improved diabetic control.
Supplemental Material
Supplementary Table 1
Raw data for the nine cats enrolled on the study
Footnotes
Author note
Preliminary data from the study were presented as an oral research communication at 27th ECVIM CA Congress, 2018.
Supplementary material
The following file is available online:
Supplementary Table 1: Raw data for the nine cats enrolled in the study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and publication of this article. The Royal Veterinary College Diabetic Remission Clinic receives support from Boehringer Ingelheim, Nestlé Purina PetCare and Zoetis.
Ethical approval
This work involved the use of non-experimental animals (owned or unowned) and procedures that differed from established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
