Abstract
Objectives
Bisphenol A (BPA) has been mentioned as a possible factor contributing to feline hyperthyroidism. Nevertheless, there are no previous studies reporting on the concentration of BPA in feline serum and its association with thyroid function. The objectives of this study were to measure serum BPA concentration in cats aged ⩾7 years, considered as healthy by their owners, and to compare the results with clinicopathological findings.
Methods
Sixty-nine cats aged ⩾7 years considered as healthy by their owners were enrolled in the study. The concentration of BPA in feline serum was measured using liquid chromatography–tandem mass spectrometry. In all cats, signalment, living environment, diet history, and the results of haematological and biochemical analysis, including thyroxine levels, were available.
Results
The mean serum BPA concentration in feline serum was 1.06 ± 0.908 ng/ml. Significant correlation was found between BPA concentration and haemoglobin (r = 0.3397; P = 0.0043), haematocrit (r = 0.3245; P = 0.0065) and the number of red blood cells (r = 0.2916; P = 0.0151), concentration of total protein (r = 0.2383; P = 0.0486), concentration of calcium (r = 0.3915; P = 0.0009) and level of bilirubin (r = 0.3848; P = 0.0011). No other significant correlations were found. Significant differences (P <0.01) were found between mature (1.28 ± 0.994 ng/ml) and geriatric cats (0.420 ± 0.240 ng/ml), between strictly indoor cats (1.27 ± 0.992 ng/ml) and cats with outdoor access (0.660 ± 0.529 ng/ml), and between cats fed canned food (1.23 ± 0.935 ng/ml) and cats fed non-canned food (0.774 ± 0.795 ng/ml).
Conclusions and relevance
Measurable serum BPA levels were found in all examined samples. The age of the cats was revealed as a significant factor affecting BPA concentration and mature cats had the highest levels. A significantly higher concentration of BPA was found in cats living strictly indoors and in cats fed canned food. No association was found between BPA and thyroid function. Further studies are needed that focus on hyperthyroid cats for better evaluation of this relationship.
Introduction
Bisphenol A (BPA) is one of the most widely used synthetic compounds on the planet. It is used in the synthesis of polycarbonate plastics, epoxy resins and other polymer materials. Owing to their excellent chemical and physical properties, they are used to produce food and beverage containers or the linings for metal products. 1 Epoxy resins are mainly used as an internal coating in canned foods to prevent contact between the food and the metal wall, and to protect the deterioration of the cans. 2
It is generally accepted that BPA has endocrine-disrupting effects on humans by interacting with various biological receptors, such as oestrogen receptors, 3 androgen receptors 4 and thyroid hormone receptors. 5 In companion animals, BPA has been mentioned as a potential factor in the development of feline hyperthyroidism. 6
In a large controlled study of hyperthyroid cats, a greater risk of developing hyperthyroidism was found in cats that were fed food from ‘pop-top’ cans at various times throughout their lives. 7 Canned food as a risk factor for hyperthyroidism was also identified in other studies.8–11 BPA used for lining the interiors of metal cans migrates into the can’s contents during processing and storage, and contamination of pet food has been reported.12–15
The deleterious effect of BPA on thyroid function may derive from its structural similarity to thyroid hormones. 16 The results of an in vitro study suggested that BPA could impair thyroid hormone action by inhibiting triiodothyronine (T3) binding to the thyroid receptor and by suppressing its transcriptional activity. 17 BPA was able to inhibit deiodinase activity both in vivo (in adult male Wistar rats) and in vitro with increasing thyroxine (T4) concentration in serum and reduced T3/T4 ratio, thus suggesting impairment in the peripheral metabolism of thyroid hormones. 18 Study of female Wistar rats and rat thyroid cell line showed that BPA increases the production of reactive oxygen species in thyrocytes, which could lead to oxidative damage, thus possibly predisposing to thyroid disease. 19
To our knowledge, there is no previous report concerning the serum concentration of BPA in cats. The purpose of this study was to evaluate BPA concentrations in feline serum samples obtained from a study assessing clinicopathological variables in cats considered by their owners as healthy and to compare serum BPA concentrations with these findings.
Materials and methods
In total, 69 samples of feline serum taken from a study wherein the health status of cats aged ⩾7 years were evaluated. All samples were obtained between July 2016 and February 2017. All cats were considered healthy by their owners. Signalment and detailed history, including living environment and type of food, were investigated. The owners denied the presence of any apparent clinical disease and the cats were free of medication for at least 6 months. Thorough physical examination, with assessment of body condition score (BCS) and muscle condition score, was performed.20,21 Blood samples were taken from the vena jugularis or vena cephalica antebrachii.All blood samples were taken with the owners’ consent. The study was performed after obtaining permission from the Ministry of Education, Youth and Sports (no. 14648/2016-6).Routine laboratory analysis for 69 cats included complete blood count (Sysmex XT 2000iV; Sysmex) with manual evaluation of blood smear, and serum biochemistry profile, including albumin, total protein, glucose, urea, creatinine, alkaline phosphatase (ALP) activity, alanine aminotransferase (ALT) activity, gamma-glutamyl transferase activity, aspartate aminotransferase (AST) activity, total bilirubin, calcium, phosphorus, potassium and sodium (Abbott Architect c4000; Abbott Diagnostics), total T4 concentration (Immulite 1000; Siemens Healthineers) and symmetric dimethylarginine (SDMA; IDEXX Laboratories).
For the measurement of BPA, samples of blood serum were stored at –20°C. Based on published studies, BPA is stable in samples stored at –20°C.22,23 The determination of BPA was conducted using liquid chromatography–atmospheric pressure chemical ionisation– tandem mass spectrometry (LC/MS) and was based on a previously described method with some modifications. 24 A sample of blood serum (0.5 ml) was spiked with internal standard solution (5.0 μl) at a target level of 1.0 ng/ml and shaken with toluene (1.5 ml) for 100 s. The organic layer was transferred into the glass test tube and liquid–liquid extraction with toluene was repeated twice. Organic portions were combined and evaporated until dry under a gentle nitrogen stream at 55°C. In order to perform derivatisation, 250 μl of sodium bicarbonate buffer (100 mM; pH 10.5) and 250 μl of dansyl chloride solution in acetone (1 g/l) were added to the dry residues in glass test tubes, gently vortexed and incubated at 60°C for 5 mins. Subsequently, the mixture was evaporated until dry under a gentle nitrogen stream at 55°C, and dry residues were reconstituted in 500 μl of methanol and used for LC/MS analysis.
A Thermo Scientific UHPLC Accela 1250 system was connected to a Thermo Scientific TSQ Quantum Access MAX Triple Quadrupole Instrument equipped with an atmospheric pressure chemical ionisation probe. A Thermo Scientific Hypersil C18 (2.1 mm × 100 mm, 1.9 μm) column was used at a constant flow rate of 300 μl/min. The mobile phase consisted of water containing 0.1% formic acid (solvent A) and acetonitrile (solvent B). The gradient used was 0–2.0 min linear gradient from 50% to 95% B; 2.0–8.0 mins held at 95% B; 8.0–8.4 mins from 95% to 50% B; and 8.4–9.0 mins held at 50% B in order for the column to re-equilibrate before the next injection. The full loop injection volume of the sample was set at 10 μl. The atmospheric pressure chemical ionisation was operated in positive mode under the following conditions: capillary temperature 325.0°C; vaporiser temperature 300.0°C; sheath gas pressure 35.0 PSI; auxiliary (drying) gas 10 AU; discharge current 4.0 μA.
For our QA/QC program, the instrument was calibrated daily with multilevel calibration curves. Procedural blank and solvent blank were analysed for every set of five samples. The method process efficiency was 86%; extraction recovery was 97%; and the matrix effect was 9.2%. The inter-day precision expressed as a relative standard deviation (SD) was 11.5% and the accuracy was 9.2%. The limit of detection (0.063 ng/ml) was determined as 3:1 signal vs noise value. In order to avoid contamination of samples and solvents, all operations were performed without plastics using glassware.
The standards of BPA and isotopically labelled BPA-D16, sodium bicarbonate, sodium hydroxide, dansyl chloride and formic acid were purchased from Sigma-Aldrich. All solvents were residual analysis purity (Chromservis).
Statistical analysis was performed using Statistica 8.0 for Windows (StatSoft). Concentrations of BPA in blood serum were log10 transformed to approach a normal distribution. ANOVA was used to calculate the effect of age and BCS on the serum concentration of BPA. When a significant effect was found, a conservative Tukey’s test was conducted as a post hoc test to determine BPA differences between individual groups. An unpaired Student’s t-test was used to evaluate effects of sex, type of food and living environment, where two datasets for each factor were available (males/females; canned food/non-canned food; only indoor/outdoor access). The relationship between BPA content and haematological and biochemical variables was assessed by Spearman correlation. Significance was set at P <0.05. BPA serum concentrations in are presented as mean ± SD.
Results
There were samples from 43 females (all spayed) and 26 males (one intact; the other 25 were castrated). The mean age of the cats was 10.3 ± 3.1 years (range 7–18 years). The most common breeds were domestic shorthair (n = 52), followed by British Shorthair (n = 7), Maine Coon (n = 5), Sphynx (n = 2), Norwegian Forest Cat (n = 1), Siberian (n = 1) and Persian (n = 1).
Cats were divided according to their age into mature (7–10 years; n = 45), senior (11–14 years; n = 14) and geriatric (⩾15 years; n = 10). Cats were categorised according to their living environment, as living only indoors (n = 46) and cats with outdoor access (n = 23). Cats were also compared according to the food they were fed: there was a group of cats fed commercial canned food and a group of cats fed non-canned food. Different types and brands of canned food were mentioned by the owners; nevertheless, no further subgroups were created.
Based on the results of BCS assessment, cats were categorised into three groups as cats with under ideal BCS (BCS 2–3/9; n = 7), cats with an ideal BCS (BCS 4–5/9; n = 32) and cats that were over ideal (BCS 6–8/9; n = 30). There were no cats with a BCS 1/9 or 9/9. The most common clinical finding was odontolithiasis and gingivitis observed in 35 cats. In five cats, a systolic heart murmur was found.
The results of haematology and biochemistry examinations are presented in Tables 1 and 2. Mild hyperglycaemia (probably stress induced) was found in 11 cats. In three cats, mild azotaemia (maximum values of creatinine and urea were 230 µmol/l and 22.2 mmol/l, respectively) and increased SDMA concentration were observed. Increased ALT activity was found in seven cats (maximum value 4.8 µkat/l). In this group, there were three cats with serum T4 levels consistent with the diagnosis of hyperthyroidism (all three cats had a T4 concentration >136 nmol/l).
Haematology results in all cats and mature, senior and geriatric cats
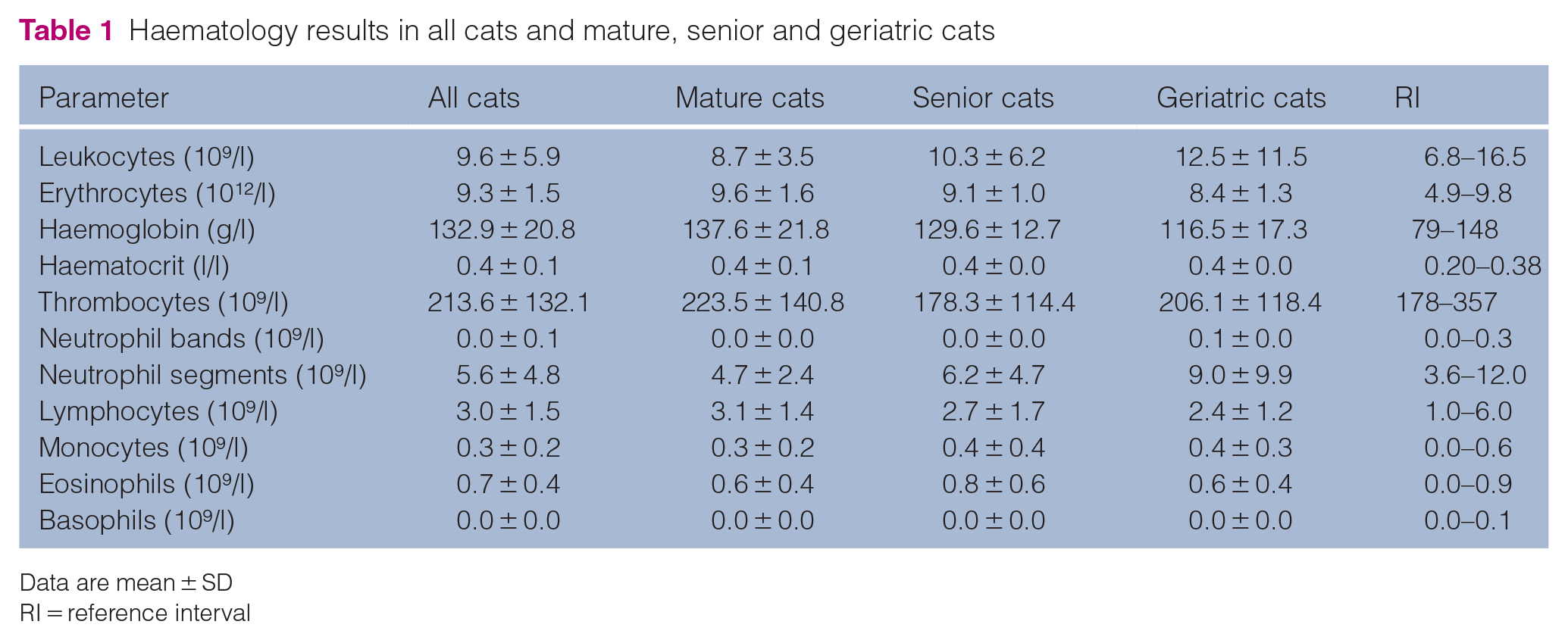
Data are mean ± SD
RI = reference interval
Results of biochemistry evaluation in all cats and in mature, senior and geriatric cats
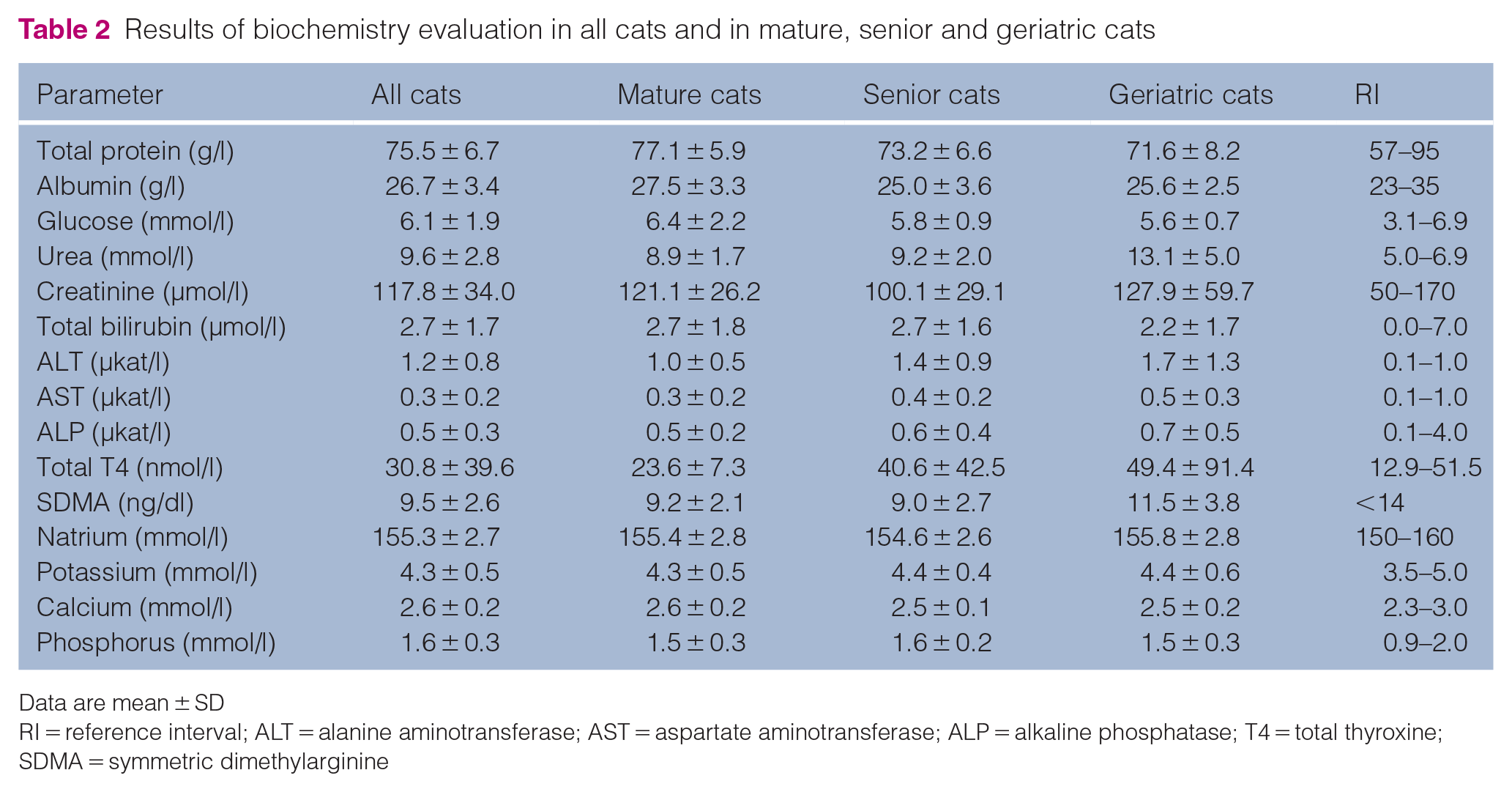
Data are mean ± SD
RI = reference interval; ALT = alanine aminotransferase; AST = aspartate aminotransferase; ALP = alkaline phosphatase; T4 = total thyroxine; SDMA = symmetric dimethylarginine
The mean serum BPA concentration in feline serum was 1.06 ± 0.908 ng/ml. ANOVA revealed an effect of age (P <0.01). When using the post-hoc Tukey’s test, concentrations of BPA were significantly higher (P <0.01) in the serum of mature cats (1.28 ± 0.994 ng/ml) compared with geriatric cats (0.420 ± 0.240 ng/ml) (Figure 1). Spearman correlation was significant between serum BPA concentration and age of the cats (r = –0.2416; P = 0.0455). ANOVA did not reveal an effect of BCS (Figure 2).

Box plot of serum bisphenol A (BPA) in 69 cats according to age: mature (7–10 years; n = 45); senior (11–14 years; n = 14) and geriatric (⩾15 years; n = 10). Boxes show the median and the upper and lower quartiles. Whiskers show the range without outliers. Circles show the outliers. Significant differences (P <0.01) in serum BPA are indicated by different alphabetic superscripts

Box plot of serum bisphenol A (BPA) in 69 cats according to body condition score (BCS): under ideal (BCS 2–3/9; n = 7), ideal (BCS 4–5/9; n = 32) and over ideal BCS 6–8/9; n = 30). Boxes show the median and the upper and lower quartiles. Whiskers show the range without outliers. Circles show the outliers
When using the unpaired Student’s t-test, cats that were living indoors only had a significantly higher (P <0.01) serum BPA concentration (1.27 ± 0.992 ng/ml) compared with cats with outdoor access (0.660 ± 0.529 ng/ml) (Figure 3). A significantly higher (P <0.01) BPA concentration (Figure 4) was found in cats fed canned food (1.23 ± 0.935 ng/ml) compared with cats fed non-canned food (0.774 ± 0.795 ng/ml). There were no statistically significant differences between males (1.14 ± 1.15 ng/ml) and females (0.992 ± 0.728 ng/ml).

Box plot of serum bisphenol A (BPA) in 69 cats according to living environment: indoors only (n = 46) and outdoor access (n = 23). Boxes show the median and the upper and lower quartiles. Whiskers show the range without outliers. Circles show the outliers. Significant differences (P <0.01) in serum BPA are indicated by different alphabetic superscripts

Box plot of serum bisphenol A (BPA) in 69 cats according to food: canned food (n = 44) and non-canned food (n = 25). Boxes show the median and the upper and lower quartiles. Whiskers show the range without outliers. Circles show the outliers. Significant differences (P <0.01) in serum BPA are indicated by different alphabetic superscripts
A significant correlation was found between BPA concentration and haemoglobin (r = 0.3397; P = 0.0043), haematocrit (r = 0.3245; P = 0.0065) and the number of red blood cells (r = 0.2916; P = 0.0151), concentration of total protein (r = 0.2383; P = 0.0486), concentration of calcium (r = 0.3915; P = 0.0009) and level of bilirubin (r = 0.3848; P = 0.0011). No other significant correlations were found.
Discussion
Despite the fact that BPA is mentioned as a possible factor contributing to feline hyperthyroidism, there are no reports evaluating serum BPA concentration in cats. Only two very recent experimental studies of the effect of BPA on cats are available. In one recent study, 25 BPA was administered to three cats intravenously and locally via intracortical injection, and visual function was assessed. The result was impairment of visual perception in cats after acute BPA exposure. 25 An inhibitory effect of BPA in vitro on spontaneous contractions of the feline uterus was found in another experimental study. 26
In all cats in this study, measurable serum concentrations of BPA were found. The values are comparable with BPA concentrations detected in the serum of people or dogs.14,27,28
The relationship between BPA and thyroid gland function is a subject of extensive research both in epidemiological studies and animal models. In a large Chinese population, a high urinary BPA level was associated with increased thyroid function, as measured by the concentration of free T3 and thyroid-stimulating hormone (TSH). 29 In Chinese women with positive thyroid autoantibodies, a higher urinary BPA concentration was associated with an increased risk of thyroid nodules. 30 In school children, a high urinary BPA concentration was negatively associated with a risk of multiple thyroid nodules and thyroid volume. 31 In patients with thyroid nodules or thyroid cancer, urinary BPA concentration was significantly higher compared with a control group. 32 Exposure to bisphenol AF, an analogue of BPA, increases susceptibility of developing differentiated thyroid cancer in patients with thyroid nodules. 33 Direct oral administration of BPA to adult rats increased T4 levels, but, in mice, 18 BPA exposure during puberty decreased T4 levels. 34 All these results and experiments indicate that BPA could affect thyroid function, but the effects may vary according to the route, dose, duration or age at exposure.
BPA has been mentioned as one of possible causes of feline hyperthyroidism owing to its endocrine-disrupting character and reports from human medicine. Our study does not support this hypothesis, where no correlation was found between serum BPA concentration and total T4 levels (Figure 5). However, only three cats had increased T4 levels, suggesting hyperthyroidism in this study. Thus, further research in hyperthyroid cats is needed to evaluate the relationship between BPA and hyperthyroidism.

Scatter plot of bisphenol A (BPA) concentrations vs thyroxine from all cats in the study (n = 69). The correlation (r = 0.0301) was not significant (P = 0.8061)
Humans and animals can be exposed to BPA through various routes, including diet, transdermally and possibly through inhalation. Diet is considered the primary route of exposure.35,36 In dogs, chewing of toys and training devices was identified as another route of exposure to BPA. 37 As BPA enters the blood through the gastrointestinal tract it can be effectively detoxified in the liver. Until the detoxification process begins, the free form of BPA can exist in the blood. 35 In mammals, BPA can be metabolised via two pathways: glucuronidation and sulfation. 38 In the rat liver, glucuronidation could be catalysed by UGT2B1, an isoform of uridine 5-diphospho-glucuronyltransferase (UGT). BPA has a short half-life; it leaves the body rapidly and does not accumulate. 39 Nevertheless, little is known about the metabolism of BPA in cats. Cats are known for their low capacity for glucuronide formation. They lack several UGTs, including UGT1A6, UGT1A9 and UGT2B7.40,41 There are no reports on feline UGT2B1 activity. The kidneys are the main route of elimination. In our study, no correlation was found between serum BPA and SDMA concentrations. It suggests that excretion of BPA is not affected by mild changes of glomerular filtration rate.
A statistically significant difference was found between concentrations of BPA in cats fed with canned food and cats fed non-canned food. As diet is considered the primary route of exposure, even short-term consumption of diet containing BPA increases its circulating concentrations. 14
A possible impact of BPA on thyroid function was considered owing to the presence of BPA in canned food and studies reporting feeding this type of diet as a risk factor for feline hyperthyroidism.8–11 However, other food-related factors may play a role in the pathogenesis of thyroid disease in cats. Increased risk of hyperthyroidism was found in cats preferring certain flavours (fish, liver or giblet). In this context, variable dietary iodine levels may cause the disease or its progression.42,43 Selenium status could also modify thyroid function. 44 Nevertheless, there are many other goitrogenic compounds that can contribute to the development of lesions in exposed cats. Genistein and daidzein as soy isoflavones are common constituents of commercially available cat food and may be present in concentrations sufficient to result in some biological effect. 45
A significant difference in BPA concentration was found between indoor cats and cats with outdoor access. A higher concentration of BPA in indoor cats may be associated with house dust, where the presence of BPA has been proved in previous reports.46,47 This contaminated dust may be ingested during grooming, which is an important part of feline comfort behaviour. House dust as a source of contaminants has been reported in the case of flame retardants, when a correlation was found between their concentration in house dust and cat hair or serum.48–50
The concentration of BPA in feline serum samples decreased with increasing age. A similar tendency was found in a human population, 51 where the urinary BPA concentration in children was significantly higher than in adolescents and adults; adolescents also had higher BPA levels than adults. The higher concentration in children was explained by possible higher food consumption and air inhalation in relation to their weight than those of adolescents and adults. Nevertheless, in our study, we compared adult cats where these differences should be minimal. The differences could also be related to variations in absorption, distribution, metabolism and excretion of BPA. In senior and geriatric cats, changes in digestive efficiency occur: 22% of cats >14 years of age had protein digestibility of <77%, and 33% of cats aged >12 years had fat digestibility of <80%. 52 Whether changes in digestive efficiency in older cats affect absorption of BPA in gastrointestinal tract remains unclear. Our results suggest that BPA is not accumulated in the body.
In humans, BPA is frequently mentioned as an obesogen. 53 Adipose tissue is now considered as an endocrine organ that actively secrets a number of adipokines. Recent studies suggest that elevated BPA exposure is associated with changes of adipokine levels that are involved in the regulation of appetite and satiety, and are generally associated with the pathophysiology of obesity.54,55 Nevertheless, we did not find any significant relationship between BPA concentration and BCS. According to a large review, there are several studies reporting a significant relationship between higher urinary or blood BPA concentrations and higher body mass index or body weight. 56 Owing to the cross-sectional nature of these studies and controversial data, the author concluded that the causal link may be complex and difficult to interpret.
A strong correlation was found between BPA concentration and parameters of an erythroid cell line (number of erythrocytes, concentration of haemoglobin and haematocrit) (Figure 6). In human medicine, the effect of BPA has been described in several reports showing its significant and haemolytic potential. Incubation of human erythrocytes with different bisphenol concentrations caused haemolysis, a high methaemoglobin concentration and echinocytosis. 57 Another in vitro study revealed an eryptotic potential of bisphenols together with increased cytosolic calcium ions, increased phosphatidylserine translocation in red blood cells and a significant increase in calapin and caspase-3 activities. 58 In carp, exposure to a sublethal concentration of BPA caused a significant decrease in haematological parameters (haemoglobin, haematocrit, mean cell volume, mean cell haemoglobin). 59 A harmful effect of BPA on haematopoietic processes was proved on an erythroblastic cell line of porcine bone marrow. 60 All these reports show a negative effect of BPA on an erythroid cell line that is in contrast with the results of our study, where a positive correlation of BPA concentration and erythroid cell line indices was found. The haemolytic effect of bisphenols has been reported in rats, together with an increase of bilirubin concentration. 61 In our study, a positive correlation between serum BPA concentrations and bilirubin levels was found. The main two causes of increased bilirubin are haemolytic and hepatobiliary diseases. According to the results of haematology, haemolysis caused by BPA can be excluded for the cats in our study. Hepatotoxicity induced by BPA with increased serum AST, ALT and ALP activities, and bilirubin concentration, has been reported in rats.61,62 In our study, the presence of liver disease associated with BPA exposure is questionable because no other significant correlation between BPA serum concentration and parameters of liver function and markers of hepatic injury were found.

Scatter plot of bisphenol A (BPA) concentrations vs haemoglobin from all cats in the study (n = 69). The correlation (r = 0.3397) is significant (P = 0.0043)
A significant correlation was found between serum BPA concentration and serum total protein levels. When there was no correlation between BPA and albumin levels, the correlation between BPA and globulins can be considered. Increased gamma globulin protein levels have been reported as a result of low-dose administration of BPA in adult rats; 63 however, our statistical analysis did not reveal any relationship between BPA and globulins. Thus, it remains unclear and further studies are needed.
A significant correlation between serum BPA and calcium levels was found. A similar finding was reported in a very recent study in women. 64 The authors suggested that increased calcium may be the result of higher intestinal absorption and/or reabsorption in the kidneys. Nevertheless, studies in laboratory rodents did not confirm this hypothesis and this association is not fully understood.65,66
A limitation of this study is its retrospective nature. Samples of feline sera originated from previous work evaluating clinicopathological variables in cats older than 7 years that were considered as healthy by their owners. Thus, only three cats with findings consistent with hyperthyroidism were enrolled in this study. Measurement of TSH might be helpful to diagnose other potential cases of subclinical hyperthyroidism; 67 nevertheless, it was not the part of the former study. In addition, there were no kittens or young adult cats to assess properly the effect of age on serum BPA concentration. Furthermore, no evaluation of house dust or feline food and subsequent comparison with serum BPA levels could be made.
Conclusions
Measurable concentrations of BPA were found in all cats with higher levels in indoor cats and cats fed with canned food. Nevertheless, no relationship was found between serum BPA and T4 levels. According to the low number of hyperthyroid cats in this study, further studies are needed for a better description of this association. It is not clear whether BPA plays a role in the pathogenesis of feline hyperthyroidism, although diet and environment appear to be important sources of exposure for cats.
Footnotes
Acknowledgements
The authors would like to thank Mr Callum McKinney for the English proofreading.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by the internal grant IGA 230/2018/FVHE of the University of Veterinary and Pharmaceutical Sciences Brno.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee was therefore not necessarily required.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). For any animals or humans individually identifiable within this publication, informed consent (either verbal or written) for their use in the publication was obtained from the people involved.
