Abstract
Objectives
The purpose of this study was to determine the impact of ageing on the cardiovascular system of healthy adult cats.
Methods
Six experimental cats were used. Echocardiography and measurements of systolic blood pressure (SBP) were performed every year for 8 years (from 1 year of age to 8 years of age) in all cats. Age-related changes to left ventricular (LV) structures, LV systolic and diastolic function, and SBP were assessed.
Results
There were no significant changes in LV structures and SBP. Peak longitudinal strain rate during systole was decreased at 8 years of age, and peak longitudinal strain rate during diastole was decreased from 6 years of age.
Conclusions and relevance
This study revealed that some measures of cardiac function recorded in six healthy cats from 1 to 8 years of age were affected as the cats got older; however, there were no structural changes or changes in measurements that are routinely assessed in clinical practice.
Keywords
Introduction
Ageing is a multifaceted process influenced by genetics and myriad internal and external factors. Age-related changes are known to lead to decreased vitality, increased vulnerability to disease and eventual death. Disorders of the cardiovascular system are among the most commonly encountered diseases in ageing. 1
Several structural and functional changes occur in the human heart as ageing progresses. Structurally, there is a significant increase in myocardial thickness as a result of increased left ventricular (LV) mass. 2 Functionally, both systolic and diastolic functions decrease with age. 3 It has been reported that ageing is a significant risk factor associated with cardiac dysfunction. 4 While ageing is not a direct cause of heart failure, the threshold for the manifestation of cardiac disease progressively lowers with increasing age.
Blood pressure (BP) is also influenced by ageing as a result of age-associated arterial stiffness and peripheral vascular resistance. 4 Hypertension is also one of the risk factors associated with heart failure in human medicine. 5 Thus, changes to the cardiovascular system as a result of ageing are important factors in the manifestation and progression of cardiac disease.
Although several reports have described the influence of ageing on cardiac function, specifically BP and LV mass, in cats,6–11 these studies did not include a longitudinal follow-up. The aim of this study was to determine the impact of ageing on the cardiovascular system using longitudinal data from healthy adult cats.
Materials and methods
Animals
This study was performed as part of a medical examination. All procedures were consistent with the guidelines of the Azabu University Animal Care and Use Committee.
Six 1-year-old experimental cats (three females and three males) were included in this study. To exclude systemic disease, a complete clinical examination comprising physical assessments, a complete blood count, blood chemistry, radiographic assessments and abdominal ultrasonography was performed every year until the cats were 8 years old, and all cats were confirmed as remaining healthy throughout the 8-year period.
Although the cats were kept in individual cages that limited their movement, the cages were large enough for them to move freely. In addition, they were removed from the cages for moderate exercise for several minutes twice daily. They were fed commercial cat food twice daily and water was available ad libitum. The room was well ventilated, with an appropriate temperature and humidity, and controlled light/dark cycles. Body weight was measured prior to each echocardiographic examination.
The cats were used for several experiments, many of which included medication or anaesthesia.
Conventional echocardiography
For all cats, echocardiography was performed every year for 8 years (from 1 year of age to 8 years) in all cats. All echocardiographic images were acquired using an ultrasound unit equipped with a 7 MHz transducer (Vivid 7 System; GE Medical Systems). All echocardiographic examinations and measurements were performed by one examiner, and performed directly on screen with freeze-frame images. The mean values of variables in five consecutive cardiac cycles were used for statistical analysis.
Cats were gently restrained in lateral recumbency without sedation. Using a two-dimensional method, the aortic and left atrial (LA) diameters at the end-ventricular systole were measured from the right parasternal short-axis view at the level of the aortic valve, and the LA-to-aortic root was measured from these values. Uniformity of the wall thickness in each cat was visually confirmed on the right parasternal short-axis view at the level of the chordae tendineae. The LV internal diameters, interventricular septum (IVS) and LV free-wall thickness at end diastole were measured from the right parasternal short-axis view at the level of the chordae tendineae on M-mode, and the LV shortening fraction was subsequently calculated. The subaortic IVS thickness was measured at end diastole from the right parasternal five-chamber view. Heart rate was calculated from M-mode images.
Structural changes in the mitral valve, tricuspid valve, aortic valve and pulmonary valve were assessed along with regurgitation by examining the right and left parasternal four- and five-chamber view.
Doppler and speckle tracking echocardiography
Cardiac function was assessed using pulsed-wave (PW) Doppler, colour tissue Doppler imaging (TDI) and speckle tracking echocardiography (STE).
Colour flow Doppler imaging was applied to facilitate the positioning of a cursor in line with mitral inflow. In addition, a sampling gate was positioned in line with this flow at the level of the open mitral valve tips. The peak early (E) and late (A) diastolic filling velocities were measured and used to calculate the E:A ratio.
All colour TDI examinations were performed as previously described. 12 The lateral and septal aspects of the mitral annulus were sampled using the left four-chamber apical view. A 2 × 2 mm sample volume without angle correction was used. The lateral peak systolic and early diastolic myocardial velocities (S′ and E′, respectively) were measured.
Longitudinal strain rate was measured using STE as previously described. 13 Peak longitudinal strain (SL) and strain rate during systole (SrLs) and early diastole (SrLe) were determined using the left apical four-chamber view and measured in six segments in the basal, middle and apical regions of the septal and lateral walls. The measurements used for statistical analysis were the average of the measurements taken from these six ventricular segments.
LV mass
LV mass was calculated using the truncated ellipse formula and the right parasternal long- and short-axis views at end diastole, as described previously. 14 LV papillary muscles were included in the measurement of LV mass.
Systolic BP
An indirect measurement of BP was carefully obtained in conscious cats using a standardised method of the general technical protocol recommended for a BP measurement session by the American College of Veterinary Internal Medicine. 15 Systolic BP (SBP) was obtained in a non-invasive manner using Doppler sphygmomanometry (Hadeco). An inflatable cuff of an appropriate size was placed on the tail and hair was clipped before placing the probe. The cuff was manually inflated until the pulse signal was no longer audible and then gradually deflated. SBP was determined when the Doppler signal was re-audible. Several consecutive BP measurements were undertaken during each session to obtain a stable set of five values; the mean value was used for statistical analyses.
Statistical analysis
All measurements were expressed as mean ± SD. Statistical analyses were performed using commercial computer software (Ekuseru-Toukei 2012; Social Survey Research Information). Data regarding body weight, echocardiographic indices and SBP at each age were visually inspected and tested for normality using the Kolmogorov–Smirnov test. All data showed normal distribution and were compared using one-way ANOVA. When a significant difference was detected, multiple comparisons were evaluated using Bonferroni correction.
Intra-observer variability for PW, TDI, STE parameters and LV mass was assessed by calculating the coefficients of variation (CV) using the formula: 16
CVs were considered clinically acceptable if <10%. 12
Results
Animals
No abnormalities were detected in any of the cats during the complete clinical examination. Body weights did not change significantly throughout the 8-year study period.
Conventional echocardiography
The results of the conventional echocardiography investigations are shown in Table 1. There were no significant differences in any indices. Valvular morphological changes and regurgitation were not detected in any cats.
The results of conventional echocardiography

Data are mean ± SD
IVSd = interventricular septum thickness at end diastole; LVIDd = left ventricular internal diameters at end diastole; LVFWd = left ventricular free wall thickness at end diastole; FS = left ventricular shortening fraction; HR = heart rate; bpm = beats per min; Ao = aortic diameter at end systole; LA = left atrial diameter at end systole; LA/Ao = left atrium-to-aortic root; IVSao = subaortic interventricular septum thickness at end diastole
Cardiac function
The results of the cardiac function investigations are shown in Table 2 and Figures 1 and 2. SrLs at 8 years of age was significantly decreased relative to the values at 1 and 5 years. With regard to SrLe, there were significant differences at 6 years of age compared with at 3 years of age, at 7 years of age compared with at 2 and 3 years of age, and at 8 years of age compared with at 1, 2 and 3 years of age. There were no significant differences in E, A, S′, E′ or SL throughout the study.
The results of pulsed wave Doppler
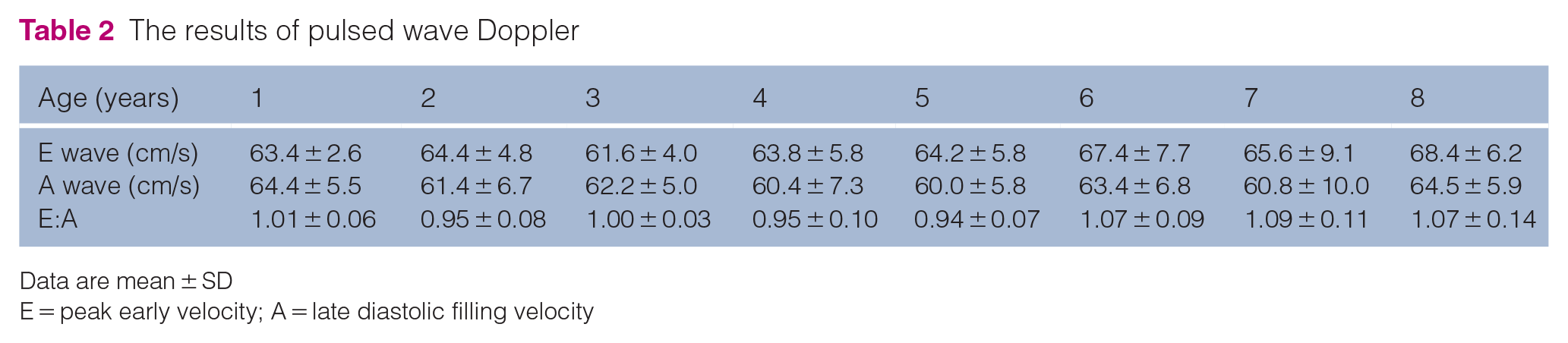
Data are mean ± SD
E = peak early velocity; A = late diastolic filling velocity

Results of tissue Doppler imaging. There were no significant differences in lateral peak systolic and early diastolic (S′ and E′, respectively)

Results of speckle tracking echocardiography. There were significant differences in strain rate during systole (SrLs) and strain rate during early diastole (SrLe). SrLs at 8 years was significantly decreased compared with that at 1 and 5 years. There were significant differences between SrLe at 6 years compared with at 3 years, at 7 years compared with at 2 and 3 years, and at 8 years compared with at 1, 2 and 3 years. SL = longitudinal strain
Intra-observer variability for PW, TDI and STE parameters were <10%. CVs in all parameters were considered low.
LV mass
The results of LV mass are shown in Table 3. There were no significant differences detected throughout the study. Intra-observer variability for LV mass was 8.5%.
Results of left ventricular (LV) mass

Data are mean ± SD
SBP
The SBP results are shown in Figure 3. There were no significant differences detected in adulthood.
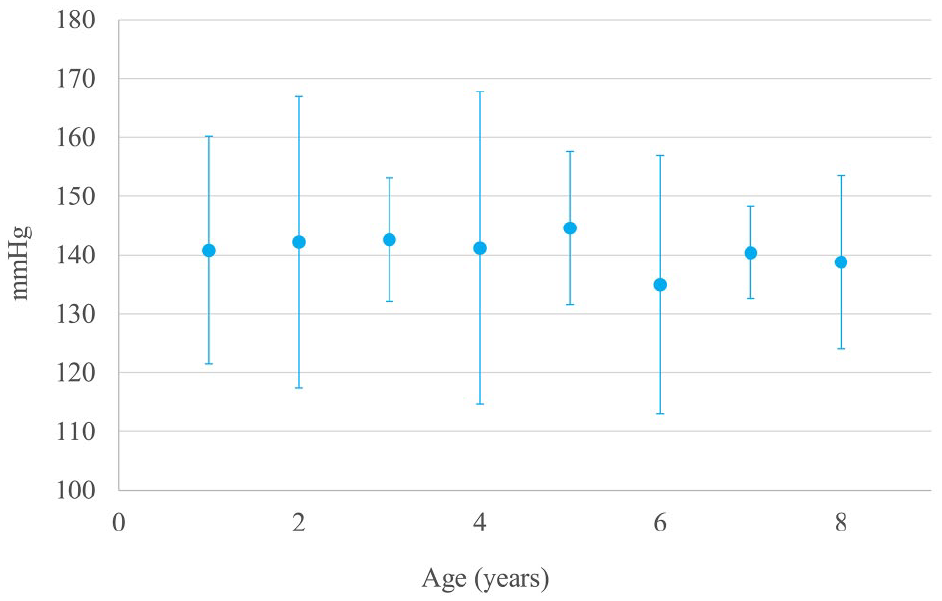
Results of systolic blood pressure (SBP) measurement. There were no significant differences in SBP
Discussion
To our knowledge, this is the first study to assesses chronological changes in cardiac function during adulthood in healthy cats. SrLe decreased from 6 years of age, while SrLs decreased at 8 years of age, although there were no changes in measures used in routine clinical practice, including LV fractional shortening, mitral inflow and the results of TDI. The relationship between age and cardiac function in cats has been investigated in previous reports using PW and TDI, but it is still controversial. Disatian et al 6 reported that there were weak correlations between age and E and A by classifying kittens (<1 year of age), adult cats (1–8 years of age) and older cats (>8 years of age). Simpson et al 8 reported that there was no relationship between age and myocardial velocities in older cats (>9 years of age). Conversely, Koffas et al 7 reported that there was a significant and moderate correlation between age and E′ in normal cats (aged 10 months to 14 years). In the current study E, A, S′ and E′ remained unchanged, although SrLs and SrLe were decreased. STE is a more sensitive method used to assess the cardiac function vs TDI. 13 Thus, it was considered that only STE parameters were able to detect these changes. Moreover, these changes were considered to be the early, minimal changes in systolic and diastolic functions that occur with age, and they would not have caused changes in the LA size and LV mass, valvular changes or changes in the subaortic LV thickness.
In human medicine, cardiac dysfunction as a result of ageing is associated with the development of congestive heart failure. 4 In cats with hypertrophic cardiomyopathy, the median age of cardiac death was reported to be approximately 8 years, 17 and the median age of diagnosis was during adulthood.17,18 The median age of cats at the time of the restrictive cardiomyopathy diagnosis was 6–10 years.19–21 The present study observed that cardiac functions began to decrease in middle age; this may be related to the progression of cardiac disease in cats.
Many cellular changes are observed in hearts with increasing age.22–24 Such changes then go on to induce functional and structural changes. LV filling occurs early and rapidly as a consequence of LV relaxation in young adult hearts; however, the hearts of older individuals fill with blood more slowly. 25 LA enlargement occurs as a result of age-related diastolic dysfunction and is most prevalent in human individuals over the age of 70 years. 26 Furthermore, by the age of 80 years, the diastolic filling rate of a human heart is reduced by up to 50%. 27 In this study, LA enlargement was not observed; however, diastolic function was found to have decreased with age. It was considered that the decline of diastolic dysfunction did not result in LA enlargement in our cats. LV systolic function is generally well preserved but deteriorates with age. 28 Similarly, in the present study, systolic function was decreased in 8-year-old cats, although diastolic function was decreased from 6 years of age.
An increase in cardiac mass and an increase in myocardial thickness along with atrial remodelling has been noted in ageing humans and beagles.26,29–32 It was reported that geriatric cats had a focal area of thickening at the level of the LV outflow tract. 33 Valvular changes in old age include myxomatous degeneration, and the deposition of collagen and calcium leading to sclerosis of the valves in human patients. 34 Despite this, visible structural changes were not detected in all cats during the study.
Vascular changes with ageing include increased calcium deposition within the aorta and peripheral arteries. 35 In cats, several studies have identified a correlation between SBP and ageing.9–11 Bijsmans et al 9 reported that SBP was significantly increased with ageing per annum for cats aged >9 years. Sansom et al 10 reported a higher BP in older cats if the cats were grouped according to age; 10 however, their results did not identify chronological changes. In this study, there were no significant differences identified in SBP throughout adulthood. It should be noted, however, that BP is sensitive to the influence of stress 36 and circadian variation. 37 The BP measurements collected during this investigation were taken at varying times and the cats were likely stressed; thus, further studies are warranted to evaluate BP.
The most important limitation in this study was that all participants were healthy, experimental cats. Moreover, the number of cats available for this study was limited. Furthermore, they were used for several experiments, many of which included medication or anaesthesia. Although echocardiography was performed prior to other experimental procedures, such exposure may affect the results. In cats with cardiomyopathy, cardiac function is decreased in early stages of the disease. 13 Therefore, the degree of change may differ between client-owned healthy cats and cats suffering from cardiac disease. Furthermore, the breed may influence echocardiographic parameters and the progression of cardiac disease. 18 Finally, although other studies have shown sex differences in structural changes, cardiac dysfunction and BP,10,38,39 we did not investigate sex differences in the present study.
Conclusions
This study revealed that some measures of cardiac function recorded from 1 to 8 years of age in six healthy cats were affected as age progressed; however, there were no structural changes or changes in measurements that are routinely assessed in clinical practice. It remains unclear whether such subtle changes can cause problems in cats, and these physiological, age-related processes should be considered when assessing cardiac function in these animals.
Footnotes
Acknowledgements
We gratefully acknowledge the work of past and present members of our laboratory.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of experimental animals and the study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.

