Abstract
Introduction:
Breast cancer (BC) is a major global health issue. Combination therapies using chemical compounds have shown potential benefits, including reduced toxicity, slowed cancer cell growth, and improved treatment outcomes, compared to single-drug treatments.
Objectives:
This study investigates the synergistic effects of doxorubicin (DOX) and Zm-093, a novel sulfonamide derivative with unique properties that offer potential advantages over existing sulfonamide compounds, including enhanced solubility and improved bioavailability, on apoptosis induction in BC cell lines.
Methods:
Zm-093 was synthesized, and its structure was confirmed through Fourier-transform infrared spectroscopy (FT-IR) and nuclear magnetic resonance (NMR) spectroscopy. Cell viability was assessed using the MTT assay, while the combination index (CI) was calculated with Compusyn software to evaluate the synergistic interaction between DOX and Zm-093 in MCF-7 cells. The apoptotic effects of these compounds were further analyzed using western blotting, flow cytometry, and TUNEL assays.
Results:
Notably, the combined treatment of DOX and Zm-093 at concentrations of 0.36 µM and 14.5 µM resulted in a 57% reduction in Bcl-2 expression compared to the control group. In contrast, expressions of pro-apoptotic proteins Bax and tBid, as well as caspase-3, increased significantly by 2.4, 3.3, and 5.7 times, respectively. Flow cytometry and TUNEL assay results indicated that the combination therapy significantly enhanced early apoptosis while minimizing necrosis.
Conclusion:
These findings confirm that DOX and Zm-093 exhibit synergistic effects on apoptotic pathways in MCF-7 cells, highlighting the potential of Zm-093 as a novel therapeutic agent with improved efficacy and reduced toxicity compared to existing sulfonamide compounds.
Introduction
BC ranks as the most prevalent cancer among women, with an estimated 2.4 million (around 11% of all cancer diagnoses) new diagnoses reported in 2022. This malignancy is recognized globally as a leading cause of cancer-related mortality. In total, the year 2022 witnessed nearly 20 million new cancer cases and approximately 9.7 million cancer fatalities. Epidemiological data suggest that roughly one in five individuals, regardless of sex, will receive a cancer diagnosis during their lifetime, while mortality rates indicate that about one in nine men and one in twelve women will succumb to the disease. 1 BC is diagnosed through a physical examination, followed by pathological, hormonal, and molecular analyses of tissue samples.2,3 Managing BC involves a variety of factors. To select an appropriate treatment for BC, a comprehensive understanding of tumor biology (estrogen and progesterone receptors, Ki-67, and Her2) is necessary. An important factor to consider when selecting a therapeutic approach is quality of life.4,5 Molecular characterization and tumor biology are used at present to determine prognosis and treatment. A molecular classification of breast tumors has been provided by multigene arrays and expression analyses. A and B luminal cells, Her2/neu, and basal-like cells are the most important subtypes. 6 Therapeutic modalities encompassing surgery, radiotherapy, hormone therapy, chemotherapy, molecular therapies, and photodynamic therapy have been identified as notable interventions in the field. 7 Combination therapy in cancer treatment holds potential benefits for patients, as it can enhance response rates and mitigate adverse effects associated with cancer therapies.8–10
Cancer treatment could be improved by inducing apoptosis,11–13 a natural mechanism of cell death. Cells and tissues undergo apoptosis to regulate and maintain their functions.14,15 Apoptosis is activated by intrinsic (mitochondrial) and extrinsic (death receptor) signaling pathways. 16 Several studies have shown that both intracellular and extracellular signals are involved in apoptosis. A variety of intracellular signals can be induced, such as DNA damage, a lack of growth factors, and a lack of cytokines. Death-inducing extracellular signals are released by cytotoxic cells of the immune system when they are damaged or infected. Pathways are converged by executioner caspases. The apoptosis pathway is generally inhibited in cancerous cells by the overexpression of antiapoptotic proteins (e.g. Bcl-2) and the decreasing expression of proapoptotic proteins (e.g. Bax).17,18
Cancer therapy relies heavily on anticancer compounds, which have been deemed vital to the fight against cancer. Currently, the U.S. Food and Drug Administration (FDA) has granted approval for more than 100 medications specifically designed for this purpose. 19 Nevertheless, drug resistance and adverse off-target outcomes influence the efficacy of cancer therapy. 20 Medicinal chemists are tasked with designing novel anticancer drugs that exhibit elevated specificity and potency while effectively mitigating off-target effects and drug resistance.21,22
Compounds containing a sulfonamide moiety are typically synthesized by reacting R-sulfonyl chloride with primary or secondary amines in a basic environment. 23 Recently, research has focused on molecules that incorporate sulfonamide fragments. This heightened interest can be attributed to the several possible uses of these compounds in various fields, such as coordination chemistry, 24 medicinal chemistry, and analytical chemistry. 25 In contrast, sulfonamide compounds are recognized for their intrinsic pharmacological characteristics, including antibacterial,25,26 anti-inflammatory, antiviral, antiproliferative, and angiogenic properties.27,28
Patients with hormone-sensitive tumors and Her2 overexpression may benefit from targeted therapies, such as hormonal therapy and trastuzumab. Cytotoxic chemotherapy remains the only treatment option for patients with triple-negative disease or those who progress after hormonal therapy. 29 The two most active chemotherapeutic agents for BC patients are docetaxel (Taxotere) and DOX.30,31 For BC, DOX is generally considered the agent of choice for first-line treatment. The response rate to DOX in first-line regimens ranges from 29% to 43%, and the median survival time is approximately 2 years. 32 The mechanism of action involves DNA interactions, namely, intercalation and inhibition of macromolecular formation, ultimately resulting in cellular death. However, DOX therapeutic efficacy is impeded by notable negative reactions and drug resistance development. 33 In addition to alopecia, myelosuppression, emesis, dermatitis, and other minor adverse effects, DOX has the potential to cause severe hypersensitivity reactions, cardiotoxicity, radiation recall, therapy-related leukemia, and palmar-plantar erythrodysesthesia. 34 Multiple studies have demonstrated that chemical coadministration of different pharmacological agents may successfully reduce DOX adverse effects while concurrently augmenting its pharmacological responsiveness.35–37
DOX primarily exerts its effects through DNA modification and interference. 38 Previous studies have identified a novel sulfonamide derivative, Zm-093: Figure 1, as a promising candidate for cancer therapy due to its anticancer properties and ability to induce apoptosis by altering intracellular apoptotic pathways. These studies have demonstrated that sulfonamide derivatives can modulate the expression of apoptotic factors, thereby enhancing cell death.39,40 This study investigated the anticancer and antiproliferative effects of DOX and Zm-093, as well as their potential synergistic effects on apoptosis induction in MCF-7 cells, a human BC cell line, using Western blotting, flow cytometry, and TUNEL staining. The combination of Zm-093 with DOX is expected to leverage these synergistic effects, potentially leading to improved outcomes in BC treatment.

Structure of Zm-093 based on sulfamethoxazole.
Materials and methods
Synthesis of azo compounds based on 2-amino-5-mercapto-1,3,4-thiadiazole (Zm-093)
Zm-093, an azo dye based on sulfamethoxazole, was synthesized through a high-yielding diazotization-coupling reaction. The diazonium salt was prepared by combining an equal mixture of sulfonamide derivative (2 mmol of sulfamethoxazole, 4 mL of acetonitrile, and a few drops of acetic acid) with HNO2 (generated from NaNO2 and HCl). The reaction mixture was then treated with β-naphthol to facilitate the synthesis of the diazonium salt.
The resulting diazonium salt was added dropwise to a mixture containing the coupling reagent (N-phenyl-2,2-iminodiethanol in ethanol). The reaction mixture was stirred vigorously for 2 h at a temperature range of 0–4°C. To maintain the pH within the physiological range, NaOH was added as needed. The progress of the reaction was monitored using thin-layer chromatography (TLC). After the reaction was complete, the crude product was filtered and washed with water three times. The resulting azo dye, Zm-093, was isolated through recrystallization. The structure of the obtained azo dye was confirmed using NMR and FT-IR spectroscopy, as shown in Supplemental Figures 1 and 2.39,41
Cell culture
The Pasteur Institute of Iran provided the MCF-7 cell line, derived from human BC. The materials used in cell culture were obtained from Bioidea in Iran. At 37°C, 5% CO2, and 95% humidity, cells were cultured in T-25 cm2 flasks with L-glutamine, 90% RPMI 1640, 10% fetal bovine serum (FBS), and 100 U/mL streptomycin/penicillin (1%). Fresh medium was added to the flask after approximately 24 h. Daily microscopic examinations were conducted to ensure that the cells were not contaminated or growing. 80%–90% confluence was reached after 3–5 days. Trypsin-EDTA (0.25%) was then used to treat the cells. A phosphate-buffered saline (PBS) solution was used to wash the cells adhering to the bottom of the flask twice after removing the culture medium. To disperse the cells throughout the flask, trypsin-EDTA was added. 3 min later, the cells were centrifuged for 5 min at 3000 rpm. After discarding the supernatant, the cell pellet was transferred to a flask for further treatment.18,42
Calculation of the IC50 values for DOX and Zm-093
MCF7 cells (5 × 105 cells/mL) were counted and transferred to 96-well plates. The cells were then incubated at 37°C, 5% CO2, and 95% humidity and cultured in medium supplemented with 90% RPMI 1640, 10% FBS, and 100 U/mL streptomycin/penicillin (1%). After 24 h, the medium was replaced with RPMI 1640 containing 1% FBS. The cells were then treated with different concentrations of DOX (prepared from the Faculty of Pharmacy at the University of Tehran) at 0, 0.091, 0.183, 0.367, 0.735, 1.47, and 2.94 µM and with Zm-093 at 0, 3.46, 6.92, 13.85, 27.71, 55.43, and 110.87 µM for 24 h. In the control group, no treatment was given. The cells were then tested for viability using a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay. Aseptic conditions were followed during all procedures. We repeated each concentration three times in each well, and the final volume was 200 µL.
To identify the optimal concentration range, a series of trials was conducted using a range of concentrations from 0 to 1000 µM. The concentrations that caused the least toxicity to cells were selected. The DOX and Zm-093 compounds were dissolved in 1% DMSO and 1% ethanol, respectively, in the final volume. Culture medium was then added to the mixture. The control group consisted of the solvent alone. Notably, previous studies have shown that 1% DMSO and 1% ethanol are non-toxic to cells, making them suitable solvents for this study.
MTT assays evaluated cell viability, proliferation, and cytotoxicity. 18 MTT experiments were conducted to determine the IC50 of DOX and Zm-093 and their optimal doses. Through spectrophotometry, the formazan form of the tetrazolium salt can be quantified after enzymatically reducing it.43,44 All wells were emptied at the end of the treatment period to perform the test. Fresh medium without FBS (which constituted 90% of the final volume of the wells) was mixed with a solution of MTT (5 mg/mL; Sigma-Aldrich) in a volume of 10 µL (which constituted 10% of the final volume of the wells). Incubation was performed at 37°C, 5% CO2, and 95% humidity for 3–4 h. A total of 100 µL was added to each well, and the supernatant was removed. For 30 min, 100 μL of dimethyl sulfoxide (DMSO; Sigma-Aldrich) was used to solubilize the formazan crystals that formed. By using a microplate reader (Bio-Rad Laboratories, CA, USA), we measured the optical density (OD) at 570 nm. The percentage of inhibition was calculated using the following formulas:
A graph was constructed based on the data calculated using the percentage of the inhibitory formula. Each graph featured a line with a slope (y = ax + b), which was used to calculate the IC50 value (Figure 2).

Determination of DOX and Zm-093 IC50 in MCF-7 cells (mean ± SD). (a) The IC50 of DOX was 1.21 µM. The growth inhibition rate increased with increasing dose. (b) The IC50 of Zm-093 was 51.24 μM.
Synergistic effects of DOX and Zm-093
The same numbers of cells were cultured under identical conditions. The synergistic effects of the DOX drug and the Zm-093 synthetic compound on MCF-7 cells were investigated. Next, the cells were treated with DOX (0, 0.367, 0.643, 0.919, and 1.28 µM) and Zm-093 (0, 10, 20, 30, and 40 µM) either alone or in combination with constant concentrations of 0.36 and 40 µM for DOX and Zm-093, respectively. These concentrations were approximately 30% of the IC50 values determined in Section 2.3. After 24 h, the MTT test was performed, and cell viability was calculated using the following formula 45 :
Combination indexes (CI) are quantitative measures used to assess the nature of pharmacological interactions, specifically synergistic, additive, or antagonistic interactions. Based on the equation below, CompuSyn software (version 1.0; Ting Chao Chou and Nick Martin, Paramus, NJ) was used to calculate the CI.46,47
In the CI formula, (Dx)1 and (Dx)2 represent the doses of DOX and Zm-093 in combination that were needed to achieve the same efficacy as that of DOX (D)1 and Zm-093 (D)2 when used alone.
A CI > 1 indicated an antagonistic interaction, and a CI < 1 indicated a synergistic interaction; a CI between 0.2 and 0.4 indicated a strong synergistic interaction, and a CI equal to 1 indicated an additive interaction.
The Drug Reduction Index (DRI) formula according to the CompuSyn software is used to assess the interaction between DOX and Zm-093, particularly in evaluating their combined effectiveness in inhibiting a certain effect (such as cell viability). The DRI is calculated using the following formula:
In the DRI formula, D1 and D2 represent the doses of DOX and Zm-093, respectively, needed to achieve a certain effect when each drug is used alone. A DRI value greater than 1 indicates a synergistic effect, suggesting that the combination of the two drugs is more effective than would be expected based on their individual effects. Conversely, a DRI value less than 1 indicates antagonism. 48 The result for this section is in the Supplemental Data (section 2).
Western blot analysis
Briefly, 5 × 105 MCF-7 cells/mL were seeded in 6-well plates and exposed to DOX (0.36 μM) and Zm-093 (14.5 μM) alone and in combination with each other (DOX + Zm-093: 0.36 + 14.5 μM) for 24 h. Subsequently, the cells were subjected to lysis utilizing a lysis solution supplemented with 1% proteinase inhibitors, namely, phenylmethylsulfonyl fluoride (PMSF), to ensure proteolytic inhibition. Following lysis, the total protein content was extracted. The concentrations were determined using a Thermo Scientific (United States)-manufactured BCA protein kit. By using Western blotting, Hua et al.49,50 analyzed tBid, Bax, Bcl-2, and caspase-3 expression patterns. Briefly, equal amounts of protein (25 μg) from each sample were loaded into gels and separated by SDS‒PAGE (12% w/v polyacrylamide gel with a 5% w/v stacking gel). The gels were then electroblotted onto a polyvinylidene fluoride transfer membrane (Millipore, Bedford, Massachusetts). Immunoblots were probed with primary antibodies against Bax, Bcl-2, caspase-3, and tBid (sc-7480, sc-492, and sc-7272 from SANTA CRUZ BIOTECHNOLOGY, INC; ab10640 from Abcam; United States), and GAPDH, a housekeeping protein, was used as a control and detected with monoclonal anti-GAPDH (6C5) (sc-32233 from SANTA CRUZ BIOTECHNOLOGY) and anti-mouse IgG-peroxidase conjugate (A2304, Sigma‒Aldrich) as primary and secondary antibodies, respectively. Additionally, the ladder protein used in the study was from Thermo Scientific (Product No. 84785). 3,3′-diaminobenzidine (DAB; Sigma‒Aldrich, D-7304) and H2O2 were used as substrates. The resulting images were captured using a ChemiDoc XRS imaging system (Bio-Rad, USA) in the presence of an enhanced chemiluminescence (ECL) solution.
Apoptosis assay via flow cytometry
For this experiment, 2 × 105 cells/mL MCF-7 cells were evenly distributed into individual wells of 6-well plates. The cells were then treated with 30% of the IC50 concentrations of DOX (0.36 μM), Zm-093 (22 μM), and a combination of both compounds (DOX + Zm-093: 0.36 + 22 μM) for 24 h. A fluorescein isothiocyanate (FITC) Annexin V Apoptosis Detection Kit (Elabscience, USA) was used to analyze cell death. 51 In the subsequent procedure, the cells were collected and subjected to two rounds of washing with PBS. After 3 μL of FITC Annexin V and 3 μL of propidium iodide (PI; 1 mg/mL) were added to the suspensions, the mixtures were thoroughly combined and subjected to a 20 min incubation period at room temperature under light-restricted conditions. A Becton-Dickinson FACSCalibur™ flow cytometer was used to assess apoptosis, and FlowJo software version 7.6.1 was used to process the data (FlowJo LLC, Ashland, OR). The proportions of cell populations in distinct quadrants were analyzed using quadrant statistics. This analysis focused on the proportions of viable cells (Annexin V−/PI−: Q4), early apoptotic cells (Annexin V+/PI−: Q3), late apoptotic cells (Annexin V+/PI+: Q2), and necrotic cells (Annexin V−/PI+: Q1).
TUNEL assay
Cell apoptosis rates were measured in various treatment groups using the TUNEL test. The cells were inoculated into 6-well plates at a concentration of 2 × 105 cells/mL and incubated overnight. 52 The cells were incubated at concentrations of 0.36 μM and 14.5 μM for 24 h for DOX alone or in combination with Zm-093, respectively. A TUNEL Assay Kit (Enhanced FITC, Elabscience: E-CK-A334) was used to determine the proportion of apoptotic cells. This procedure was performed following the manufacturer’s guidelines. To summarize, the slides were subjected to two rounds of xylene administration, each lasting 5 min, followed by immersion in 100% and 95% ethanol for 3 min. The slides were subjected to two rounds of washing, each lasting 2 min, utilizing a solution composed of Tween 20-PBS. To inhibit peroxidase activity, the slides were incubated for 10 min in a PBS solution containing 3% hydrogen peroxide (H2O2). Subsequently, the slides were washed three times with a Tween 20-PBS solution for 2 min per wash. Next, the slides were incubated for 40 min in Buffer Reaction Terminal Deoxynucleotidyl Transferase (TdT), followed by a 1 h incubation in Mixture Reaction TdT at 37°C. Subsequently, the slides were washed with buffer wash stop for 4 min and then subjected to three washes with Tween 20-PBS, each lasting 2 min. The slides were incubated with HRP-streptavidin for 20 min at ambient temperature, followed by three washes with Tween 20-PBS for 2 min each. Subsequently, the slides were subjected to a 1 min incubation period with DAB, after which the slides were further washed with water. The slides were immersed in a hematoxylin solution for 30 s. Following this step, the slides were rinsed with water and dehydrated in 95% and 100% ethanol for 3 min. After they were double immersed in xylene for 5 min each, the cells were mounted. Fluorescence microscopy (Olympus BX50; Japan) was used to obtain the images. The quantification of TUNEL-positive cells was performed using a microscope and ImageJ software. 53
Statistical analysis
This study used SPSS software (version 23.0) and GraphPad Prism (version 8.0) to analyze the numerical data. In each experiment, three replications were conducted, so all data are presented as means ± standard deviation (SD). For multiple comparisons, ANOVA and Duncan’s test were used to analyze significant differences between groups. Furthermore, the significance of the differences observed between the groups was evaluated using a one-sample t-test. The statistical significance was set at p < 0.0001.
Results
Antiproliferative activity against the MCF-7 cell line
MCF-7 cells were treated for 24 h with DOX and the synthetic compound Zm-093 to determine their cytotoxic effects (Figure 2). The survival rate of cancer cells was significantly lower in the DOX and Zm-093 treatment groups than in the control group. Cancer cell proliferation was inhibited by these compounds in a dose-dependent manner (p < 0.0001). The IC50 values for DOX and Zm-093 were calculated to be 1.21 and 51.24 μM, respectively (Figure 2(a) and (b)).
The synergistic effects of DOX and Zm-093 on MCF-7 cell viability
The quest for effective cancer treatments frequently involves the exploration of synergies between existing pharmacological agents. In the context of BC, DOX remains a cornerstone chemotherapeutic agent despite its associated challenges, notably the development of multidrug resistance (MDR) in treated cancer cells. The study under consideration evaluates the synergistic effect between DOX and Zm-093, a novel sulfonamide compound, on the viability of MCF-7 cells. It highlights the potential of this combination therapy to enhance therapeutic efficacy. To investigate the synergistic effect of DOX and the Zm-093 compound on the viability of MCF-7 cells, the cells were treated with different concentrations of DOX and Zm-093 alone and in combination with each other at a constant concentration of 0.36 µM (this concentration is approximately 30% of the IC50 that was studied) for DOX and 40 µM for Zm-093 for 24 h (Figures 3 and 4). The results revealed that increasing doses of either agent led to a significant reduction in cell survival rates, with p-values indicating a high statistical significance (p < 0.0001). Notably, the combination of DOX and Zm-093 resulted in a pronounced decrease in cell viability when compared to either drug used alone, suggesting a potential therapeutic advantage in co-administration (p < 0.0001; Figures 3(a) and 4(a)).

Survival and combination indexes (CI) at variable and constant concentrations of DOX and Zm-093 (mean ± SD). (a) Viability (%), (b) CI plot.
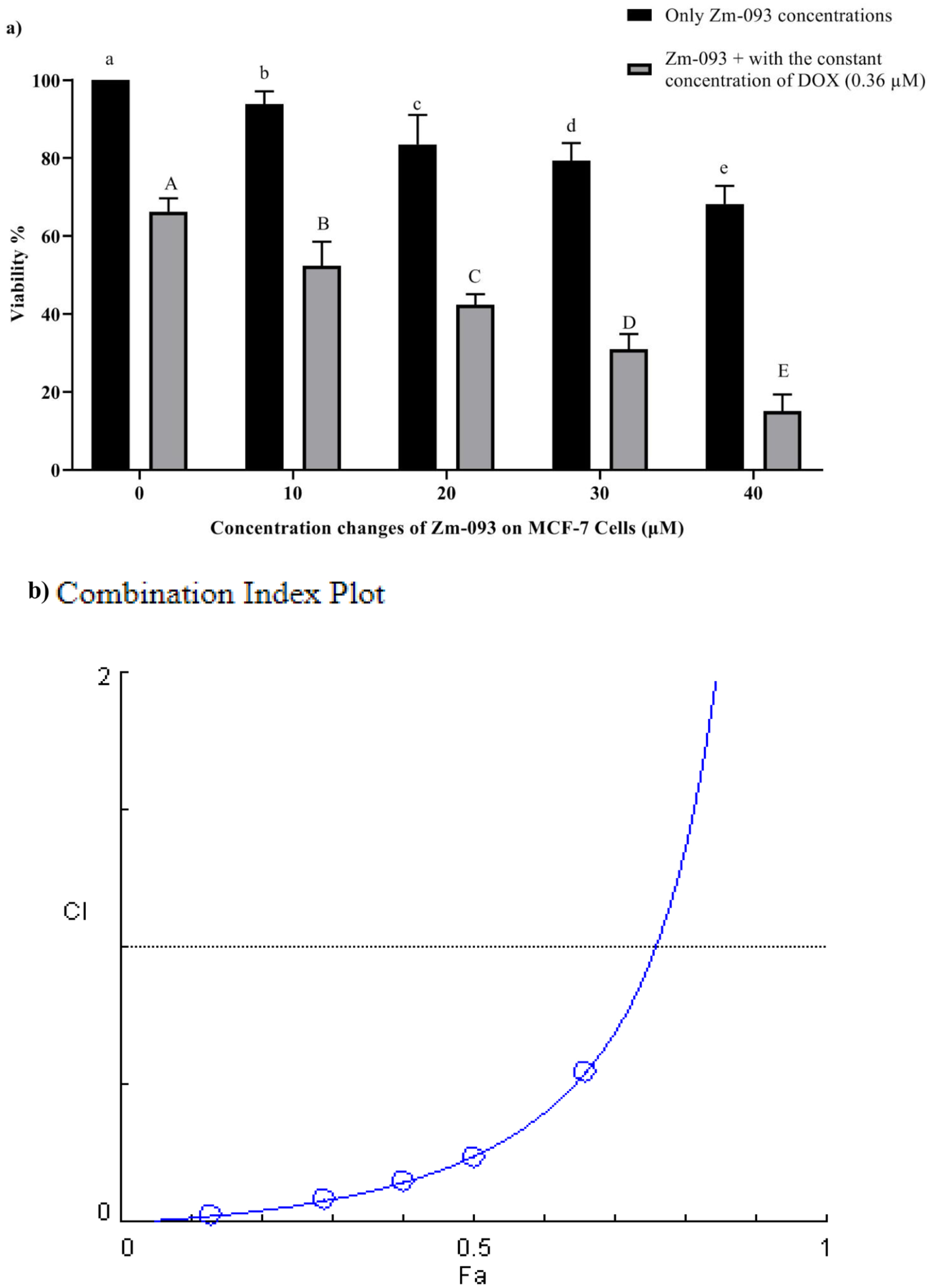
Survival and combination indexes (CI) at variable and constant concentrations of Zm-093 and DOX (mean ± SD). (a) Viability (%), (b) CI plot.
The OD was subsequently read by an Eliza reader spectrophotometer for DOX and the Zm-093 compound alone and in combination (Tables 1 and 2). To further underscore the degree of synergy observed, the study employed CompuSyn software to calculate the CI values across various concentrations (Figures 3(b) and 4(b)). The CI values less than 1 across all combinations confirm the synergistic interactions between DOX and Zm-093, as delineated by the methodology for synergy quantification established by Chou and Talalay. 48 Synergy, defined in this context as a greater effect than the simple addition of individual effects (Fa), suggests that the mechanisms of action of these two compounds may interact positively to enhance apoptosis in MCF-7 cells, a critical pathway often disrupted in cancer.
OD values for MCF-7 cells treated with different concentrations of DOX and Zm-093 alone (μM) for 24 h.

Zm-093: New sulfonamide derivative.
DOX: Doxorubicin.
The OD values for MCF-7 cells treated with different concentrations of Zm-093 and DOX alone (μM) after 24 h.

DOX: Doxorubicin.
Zm-093: New sulfonamide derivative.
Measurement of apoptotic and antiapoptotic protein expression by western blotting
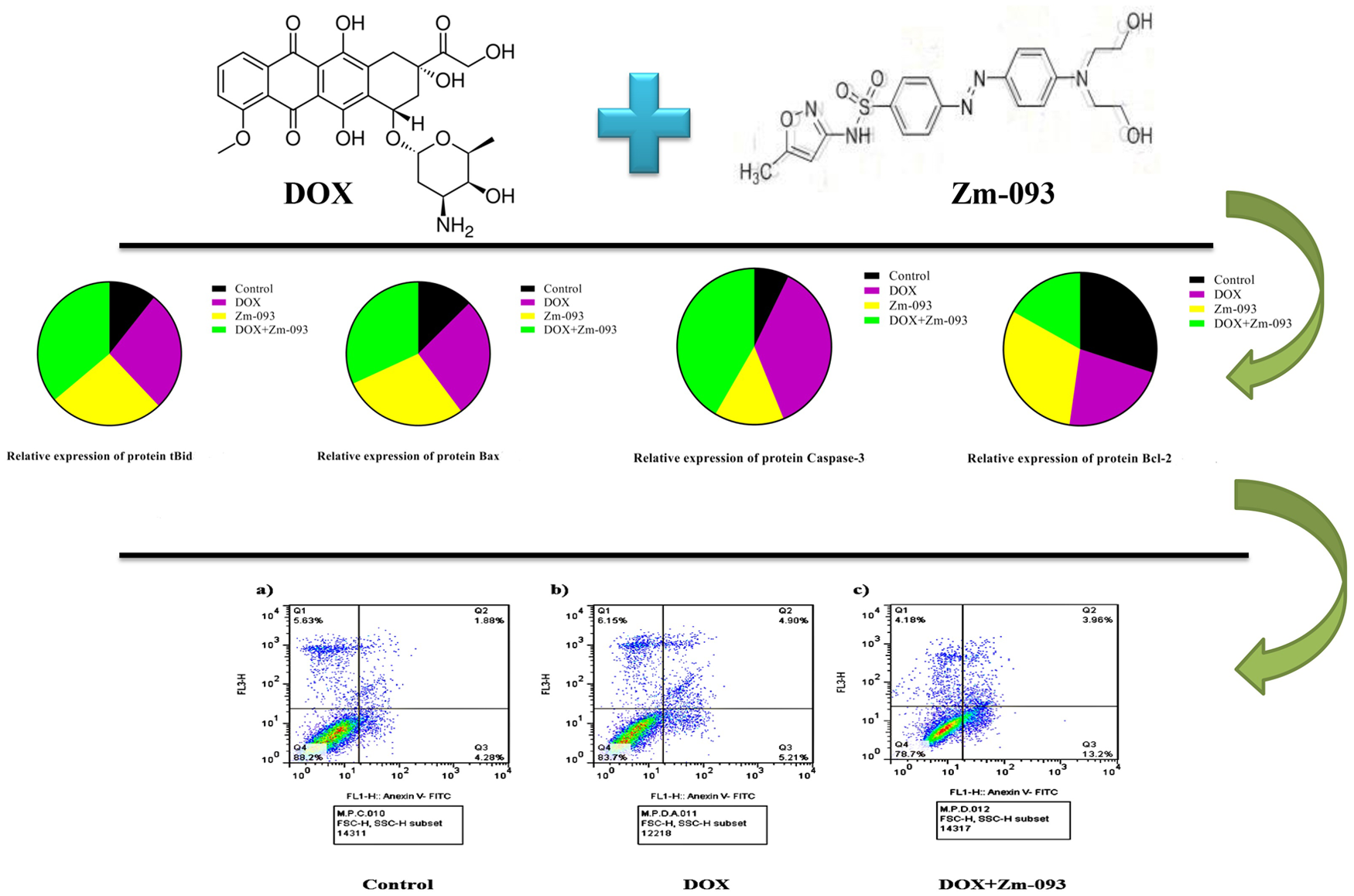
The apoptotic effects of DOX (0.36 μM) and Zm-093 (14.5 μM) alone or in combination (0.36 μM DOX + Zm-093 at 14.5 μM) on MCF-7 cells were examined. In total, the study included four groups: a control group (no treatment), a DOX group, a Zm-093 group, and a DOX + Zm-093 group. The levels of proapoptotic (tBid, Bax, and caspase-3) and antiapoptotic (Bcl-2) proteins were analyzed via western blotting (Figure 5). Original photos of the Western blot gels are available in Supplemental Figures S4 to S8.

Western blot analysis, which examines the relative expression of pro-apoptotic and anti-apoptotic proteins in MCF-7 cells that were treated. Panels (a) and (b) display tBid; panels (c) and (d) show Bax; panels (e) and (f) illustrate caspase-3; and panels (g) and (h) depict Bcl-2.
tBid
The DOX (2.56-fold) and Zm-093 (2.43-fold) groups exhibited similar changes in protein tBid relative expression, but there was a significant increase compared to the control group (p < 0.0001). Moreover, DOX + Zm-093 (3.39-fold) showed the highest level of tBid protein expression (p < 0.0001; Figure 5(a) and (b)).
Bax
There was a considerable increase in Bax protein expression compared to that in the control group, similar to the changes in the protein tBid in the DOX (2.16-fold) and Zm-093 (2.25-fold) groups. DOX + Zm-093 also increased Bax protein expression (2.53-fold, P < 0.0001; Figure 5(c) and (d)).
Caspase-3
When MCF-7 cells were treated with DOX or Zm-093, caspase-3 was in a procaspase-3 or cleaved caspase-3 state, respectively. Compared to those in the control group, all the treatment groups had lower pro-caspase-3 levels. Compared to those in the control group, the changes in expression increased after caspase-3 was cleaved. Therefore, we observed the highest expression of caspase-3 in the DOX + Zm-093 group (5.73-fold, p < 0.0001; Figure 5(e) and (f)).
Bcl-2
There was a difference in the Bcl-2 protein levels in the treated MCF-7 cells in the presence of the test compounds. In the Zm-093 group (1.03-fold), the changes in the Bcl-2 protein were similar to those in the control group. Compared to those in the control group, the DOX group produced less Bcl-2 protein (0.741-fold). The lowest level of Bcl-2 expression was also found in the DOX+Zm-093 group (0.562-fold, p < 0.0001; Figure 5(g) and (h)).
Apoptotic/necrotic ratios in MCF-7 cells
As shown in Figure 6, there were three groups according to flow cytometry: a control group (no treatment), a DOX group (0.36 μM), and a DOX + Zm-093 group (0.36 + 22 μM). The incidence of early apoptosis differed among the three treatment groups. Compared with control cells (4.28%) or DOX-treated cells (5.21%), cells treated with DOX + Zm-093 (13.2%) exhibited increased primary apoptosis. In addition, the DOX and DOX + Zm-093 groups exhibited greater rates of late apoptosis (4.90% and 3.96%, respectively) than did the control group (1.88%). In contrast, there was no significant difference in necrosis between the treated and control groups.

Analysis of apoptosis by flow cytometry with Annexin V/PI staining in MCF-7 cells.
TUNEL assay of treated MCF-7 cells
Apoptotic cells exhibit typical nuclear condensation as a morphological sign. As DNA strands are cleaved or nicked by nucleases, many 3′-hydroxyl ends are exposed. TUNEL assays were performed to detect cells that exhibited massive DNA fragmentation, a sign of late apoptosis (Figure 7). The study included four groups: a control group (no treatment), a DOX group (0.36 μM), a Zm-093 group (14.5 μM), and a DOX + Zm-093 group (0.36 + 14.5 μM). The amount of fragmented DNA in MCF-7 cells treated with the simultaneous combination of two compounds (DOX + Zm-093) increased compared to that in the control group and cells treated with each compound alone (DOX or Zm-093), indicating that the combination of the two compounds induces more apoptosis (p < 0.0001).

Assessment of the apoptosis rate in MCF-7 cells using the TUNEL test at 400x magnification. (a) Four groups were considered: 1. control cells without treatment, 2. DOX (0.36 µM), 3. Zm-093 (14.5 µM), and 4. DOX+Zm-093 (0.36 + 14.5 µM). Combining two compounds (DOX + Zm-093) increased fragmented DNA levels in MCF-7 cells. (b) A quantitative analysis of apoptotic nuclei was performed (mean ± SD).
Discussion
The goal of clinical treatment is to suppress and eliminate tumors, and chemotherapy is crucial for achieving this goal. The inherent limitations of single chemotherapy treatments lead to the use of combinational medications in clinical settings.54–56 Combining the right medicines is essential. DOX induces cellular apoptosis by inhibiting DNA replication and suppressing protein synthesis, making it an effective treatment option for a wide range of malignant tumors. In general, tumors respond to DOX when it is administered alone or in combination with other anticancer medicines. However, the use of this substance is limited by the occurrence of dose-limiting toxicities and increased drug resistance.57,58 Consequently, new combinations of drugs that can enhance or sustain effectiveness while mitigating toxicity and retarding the emergence of drug resistance must be explored. 59 In addition to their anticancer properties, sulfonamide-derived compounds exhibit a variety of biological actions. These compounds are heterocycles that can form hydrogen bonds, making them more soluble and likely to interact with biomolecular targets. The potential of sulfonamide derivatives as cancer medicines has been extensively studied.40,60,61 A study reported a series of indole-based benzenesulfonamides as potent inhibitors of carbonic anhydrases (CAs) and aimed to investigate the antitumor effects of these compounds on various cancer cell lines, including breast cancer (MDA-MB-231, MCF-7, and SK-BR-3), lung cancer (A549), and pancreatic cancer (Panc1). Among the 15 compounds tested, A6 and A15 exhibited potent cytotoxic and anti-migratory activities against MCF-7 and SK-BR-3 cells. To assess whether these compounds were synergistic with the common anticancer drug DOX, the cytotoxic effects of A6 and A15 in combination with DOX were analyzed using independent Chou-Talalay and Bliss methods. The data revealed that both A6 and A15 significantly enhanced the anticancer activity of DOX. 37
The purpose of this study was to determine whether DOX and a new sulfonamide derivative (Zm-093) can interact synergistically to induce apoptosis in MCF-7 cells. The IC50 values for DOX and Zm-093 were calculated to be 1.21 and 51.24 μM, respectively. Based on the obtained IC50 values, first, DOX was used at a constant concentration of 0.36 μM, while Zm-093 was tested at concentrations of 0, 10, 20, 30, and 40 μM. Second, a constant concentration of 40 μM was used for Zm-093, with DOX applied at concentrations of 0, 0.367, 0.643, 0.919, and 1.28 µM. The Chou-Talalay approach was employed to quantify the synergistic effects of DOX and Zm-093 utilizing the Compusyn program. The CI was plotted on the y-axis as a function of the effect level (Fa) on the x-axis to assess the potential synergistic effect. According to the Chou-Talalay technique, a CI value less than 1 indicates a synergistic effect, a value equal to 1 suggests an additive effect, and a value greater than 1 implies a possible antagonistic impact. 62 When DOX and Zm-093 were combined, cell survival decreased, and cell death increased in MCF-7 cells at all concentrations. A synergistic effect was observed when Zm-093 was combined with DOX at concentrations ranging from 10 to 20 μM, while the fixed DOX concentration was maintained at 0.36 μM. The same effect was noted with fixed Zm-093 (40 μM) concentrations and variable DOX concentrations, as demonstrated by a CI value of less than 1. Previous studies have also shown significant synergistic effects of combining DOX with other agents, such as ruthenium (II) complexes, on MCF-7 cell proliferation. 63 Synergism and antagonism are quantitatively defined by the CI theorem and the median-effect equation (MEE) in the mass-action law. These approaches provide straightforward, efficient, and cost-effective means of assessing the combined effects of medications, allowing them to be widely utilized. 64
The combined effects of several factors, including Bcl-2, Bax, tBid, and caspase-3, on the expression of proteins involved in the intrinsic apoptosis pathway were evaluated by Western blotting. Both the intrinsic and extrinsic pathways can trigger apoptosis, initiated by pro-apoptotic Bcl-2 family members such as Bax and Bak. As these proteins oligomerize and integrate within the mitochondrial outer membrane, they permeabilize the membrane, leading to the release of cytochrome C into the cytosol.65,66 This release activates the apoptosome and initiator caspase-9. Activated caspase-9 then cleaves downstream effectors, such as caspase-3 and caspase-7, leading to cell death.41,42 By administering DOX and Zm-093 simultaneously, Bcl-2 expression was reduced compared to the DOX and Zm-093 alone group or the control group. We focused on the pro-apoptotic property of Bax, which was significantly enhanced in cells treated with the combined drugs compared to the control. The Bax/Bcl-2 ratio, which regulates cytochrome C release, was elevated 2.91-fold with DOX alone, 2.18-fold with Zm-093 alone, and 4.5-fold in response to the combination treatment. This indicates the modulation of the Bax/Bcl-2 ratio through combined therapy. Additionally, the synergy in tBid expression suggested stronger activation of caspase-8 due to extrinsic apoptosis. 67 The DOX, Zm-093, and combined treatment groups (DOX + Zm-093) exhibited elevated caspase-3 levels compared to the control group (5.08-fold, 2.06-fold, and 5.73-fold, respectively). Moreover, MCF-7 cells treated with both drugs coexpressed active caspase-3 at a significantly higher level than those treated with Zm-093 alone.
In a related study by Shi et al., 68 the combined treatment of DOX and glycyrrhetinic acid in MCF-7 cells led to an enhanced upregulation of the Bax/Bcl-2 ratio as well as the active/inactive caspase-3 ratio compared to individual treatments. Additionally, a study investigating the synergistic effects of DOX and a mushroom extract (Hericium) demonstrated that combined treatments elicited a more pronounced upregulation of Bax and caspase-3 while downregulating Bcl-2 expression compared to monotherapy. 69 Furthermore, TUNEL staining revealed that combined treatment with phenethyl isothiocyanate and paclitaxel resulted in a significant increase in apoptosis in MCF-7 cells compared to individual treatments. Specifically, this combination treatment led to a 3.4-fold increase in apoptosis compared to phenethyl isothiocyanate alone and a 2.8-fold increase compared to paclitaxel alone. 70 In alignment with these findings, flow cytometry results demonstrated that the coadministration of DOX and Zm-093 significantly enhanced apoptosis in MCF-7 cells, indicating increased levels of early and late apoptotic cells compared to monotherapy. TUNEL staining showed that apoptosis rates increased by 3.8, 3.4, and 6.3 times for cells treated with DOX, Zm-093, and the combination, respectively. The percentage of apoptotic cells was 1.6 and 1.8 times greater in the combined treatment group compared to the groups receiving DOX and Zm-093 alone, further emphasizing the remarkable apoptotic effect of these treatments.
Despite the promising findings of this study, certain limitations must be acknowledged. The in vitro nature of the experiments limits the direct translation of findings to clinical settings, as cellular responses can differ significantly in the complex in vivo environment. Additional studies, including in vivo experiments, are necessary to assess the efficacy and safety of the DOX and Zm-093 combination in animal models. Furthermore, clinical trials involving human subjects will provide crucial insights into the therapeutic potential and tolerability of this combination therapy. Future research could also explore the mechanism of action of Zm-093 in greater detail, investigate its efficacy in different cancer types, and evaluate its potential in combination with other chemotherapeutic agents. Addressing these questions will help advance our understanding and improve treatment strategies against cancer.
Conclusions
The current investigation demonstrated that all CI values obtained were less than one, indicating a synergistic correlation between DOX and Zm-093 regarding their anticancer efficacy against MCF-7 cells. The results of the Western blot analysis illustrated the synergistic effect of DOX and Zm-093 on the protein expression levels of Bcl-2, tBid, and caspase-3, as well as on the Bax/Bcl-2 ratio. Furthermore, the concurrent administration of these two compounds initiated programmed cell death (apoptosis) in MCF-7 cells via the intrinsic apoptotic pathway. These findings were further validated through flow cytometry and TUNEL staining. The administration of substantial doses of DOX and Zm-093 individually is also limited by issues of cytotoxicity and solubility. By administering DOX and Zm-093 together, these undesirable effects can be minimized, and drug resistance can be mitigated.
Supplemental Material
sj-docx-1-iji-10.1177_03946320261422036 – Supplemental material for Apoptosis enhancement in MCF-7 cells: Synergistic effects of doxorubicin and the novel sulfonamide Zm-093
Supplemental material, sj-docx-1-iji-10.1177_03946320261422036 for Apoptosis enhancement in MCF-7 cells: Synergistic effects of doxorubicin and the novel sulfonamide Zm-093 by Shabnam Pedarpour, Sevda Zarei and Hossein Ghafouri in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgements
The authors are grateful to the University of Guilan for its support in conducting this study.
Author contributions
Consent to participate
All of the authors have read and approved the paper for publication.
Consent to publish
The authors declare that they have no known competing financial interests or personal relationships that seem to affect the work reported in this article.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.*
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The investigation did not include human or animal participants; all experiments were conducted in a controlled laboratory environment using the MCF-7 cell line obtained from the Pasteur Institute, ensuring compliance with the highest ethical research standards.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.




