Abstract
Objectives
The study objective was to investigate the prevalence and clinical characteristics of phenobarbitone-associated adverse effects in epileptic cats.
Methods
The medical records of two veterinary referral clinics from 2007 to 2017 were searched for cats fulfilling the inclusion criteria of a diagnosis of epilepsy, treatment with phenobarbitone and available follow-up information on the occurrence of adverse effects. Follow-up information was obtained from the medical records of the primary veterinarian and referral institutions and a questionnaire completed by the cats’ owners.
Results
Seventy-seven cats met the inclusion criteria. Fifty-eight were affected by idiopathic epilepsy and 19 by structural epilepsy. One or more of the following adverse effects were reported in 47% of the cats: sedation (89%); ataxia (53%); polyphagia (22%); polydipsia (6%); polyuria (6%); and anorexia (6%). Logistic regression analyses revealed significant associations between adverse effect occurrence and both phenobarbitone starting dosage and administration of a second antiepileptic drug (AED). For each 1 mg/kg q12h increment of phenobarbitone, the likelihood of adverse effects increased 3.1 times. When a second AED was used, the likelihood of adverse effects increased 3.2 times. No association was identified between epilepsy aetiology and adverse effect occurrence. An idiosyncratic adverse effect, characterised by severe neutropenia and granulocytic hypoplasia, was diagnosed in one cat. This resolved following phenobarbitone discontinuation.
Conclusions and relevance
The prevalence of phenobarbitone-associated adverse effects was 47%. Sedation and ataxia were most common. These are type A adverse effects and are predictable from phenobarbitone’s known pharmacological properties. In the majority of cases, adverse effects occurred within the first month of treatment and were transient. Idiosyncratic (type B) adverse effects, which were not anticipated given the known properties of the drug, occurred in one cat. Increased phenobarbitone starting dosage and the addition of a second AED were significantly associated with the occurrence of adverse effects.
Introduction
Phenobarbitone (PB) is considered the most effective first-line antiepileptic drug (AED) used in epileptic cats. 1 However, the evidence for PB’s efficacy and safety profile in the feline veterinary literature is weak. 1 Drug-related adverse effects (AEs) are categorised as type A or type B (also known, respectively, as type I and II AEs).
Type A AEs are those that can be explained by the known pharmacological properties of the agent and are therefore usually dose dependent and predictable. Type A AEs that may affect individuals receiving PB can be understood by considering the drug’s mechanism of action. PB exerts its antiepileptic effect by several different means. It is an agonist of the gamma-aminobutyric acid (GABA) A receptor and also modulates this receptor to increase GABA’s affinity for it. 2 This causes postsynaptic chloride channels to remain open for longer, allowing greater chloride influx into, and hyperpolarisation of, the postsynaptic neuron. Other purported mechanisms of action of PB include inhibition of the excitatory capacity of glutamate, 3 inhibition of voltage-gated calcium channels and binding of chloride channels. 4 The upshot is an increase in seizure threshold and a decrease in the spread of excitation to surrounding neurons. 5 While this provides efficacious seizure control, the overall effect of barbiturate administration is depression of the central nervous system. It can therefore be anticipated that type A AEs, such as sedation and ataxia, will occur with PB therapy. 4 Polyuria and polyphagia are also commonly reported during PB treatment; these are thought to arise through the inhibition by PB of antidiuretic hormone release. 5
Type B AEs are not dose related, are unpredictable and are unrelated to the known mechanism of action of the drug. They can be life threatening and often require the discontinuation of the offending agent to resolve.6,7
Sedation, ataxia, polyuria, polydipsia and polyphagia have all been reported as type A AEs in cats treated with PB, especially during the initial 2 weeks of treatment. 8 Accurate information on their prevalence, severity and median time to resolution is lacking. 9 In dogs, some or all of these dose-related AEs can persist and significantly affect quality of life. 10 It is unknown if similar phenomena occur in cats. Idiosyncratic (type B) AEs such as leukopenia, thrombocytopenia, lymphadenopathy, skin eruptions, pyrexia and coagulopathy have been described in a few feline case reports.11–15 The aim of this study was to investigate the prevalence and clinical characteristics of, and risk factors for, PB-associated AEs in epileptic cats that have not been previously evaluated in a large study population.
Materials and methods
This study was approved by the Ethics Committee of the Animal Health Trust (approval number AHT62-2016). The medical records of two referral veterinary clinics from 2007 to 2017 were retrospectively reviewed to identify cats diagnosed with epilepsy and treated with PB. Inclusion criteria were documentation of two or more unprovoked epileptic seizures (ie, reactive seizures were excluded) at least 24 h apart and availability of follow-up information on the occurrence of AEs.
The following information was retrieved from the medical records of the referral institution and primary veterinarian medical records: signalment; diagnostic investigation results; epileptic seizure aetiology (structural, idiopathic) and phenotype (focal, generalised, focal developing into generalised); the occurrence of status epilepticus or cluster seizures; date of PB treatment initiation; PB dosage and serum PB levels over time; duration of PB treatment; administration of a second AED; occurrence of AEs (type and clinical features); and date of AE resolution due to achievement of steady state or following dose reduction or discontinuation of PB. In this study, the 2015 International Veterinary Epilepsy Task Force definitions for idiopathic and structural epilepsy were applied, which are, respectively, ‘epilepsy in which the nature of the underlying cause is as yet unknown and with no indication of structural epilepsy’ and ‘epileptic seizures which are provoked by intracranial/cerebral pathology’. 16
The cats’ owners were invited to participate in this study by completing a questionnaire. An overview of the study and its aims were provided. The following information was requested via the questionnaire: date of questionnaire completion; PB dosage over time or reason for PB discontinuation; occurrence of any AEs (polyphagia, polydipsia/polyuria, lethargy, ataxia, pruritus, skin lesions, weight gain, changes in sleep pattern or other AEs to be specified) and their duration following PB initiation or dosage increase, dose reduction or discontinuation of PB due to AEs and subsequent outcome; date of initiation of a second AED and associated outcome; and details of epileptic seizure frequency. When assessed at the referral institutions, all cats were examined by a board-certified neurologist or neurology resident working under the supervision of a neurology diplomate.
Statistical analysis
Data were analysed using XL Stat (Addinsoft SARL) and Stata (IC v 15.0; Statacorp) and standard descriptive statistics were performed. The frequency of the occurrence of AEs (total number and specified individual AE) was assessed in cats meeting the inclusion criteria. The Mann–Whitney U-test was used to test for differences in PB dosage and serum concentration across cats with onset of AEs and without AEs in the first month. For the AEs occurring with the highest frequency, the influence of epilepsy aetiology (‘structural’ or ‘idiopathic’), the administration of a second AED (binary) and PB dosage (mg/kg) at the onset of AEs and PB serum levels (mg/l) on the likelihood of their occurrence was assessed in a mixed binomial/multinomial logistic regression model. As the PB serum levels were numerically higher by a factor of 10 than PB dose levels, and the model assesses odds per unit increase, the PB serum levels were divided by 10. Backward elimination was applied to the four variables initially modelled, with alpha ⩽0.1 the inclusion criterion for retention. 17
Results
Seventy-seven cats fulfilled the inclusion criteria. Information on sex, breed, epileptic seizure aetiology, including specific aetiology in cases of structural epilepsy, seizure phenotype and diagnostic tests performed are detailed in Table 1.
Sex, breed, epileptic seizure aetiology and phenotype, and diagnostic test information of 77 cats included in the present study
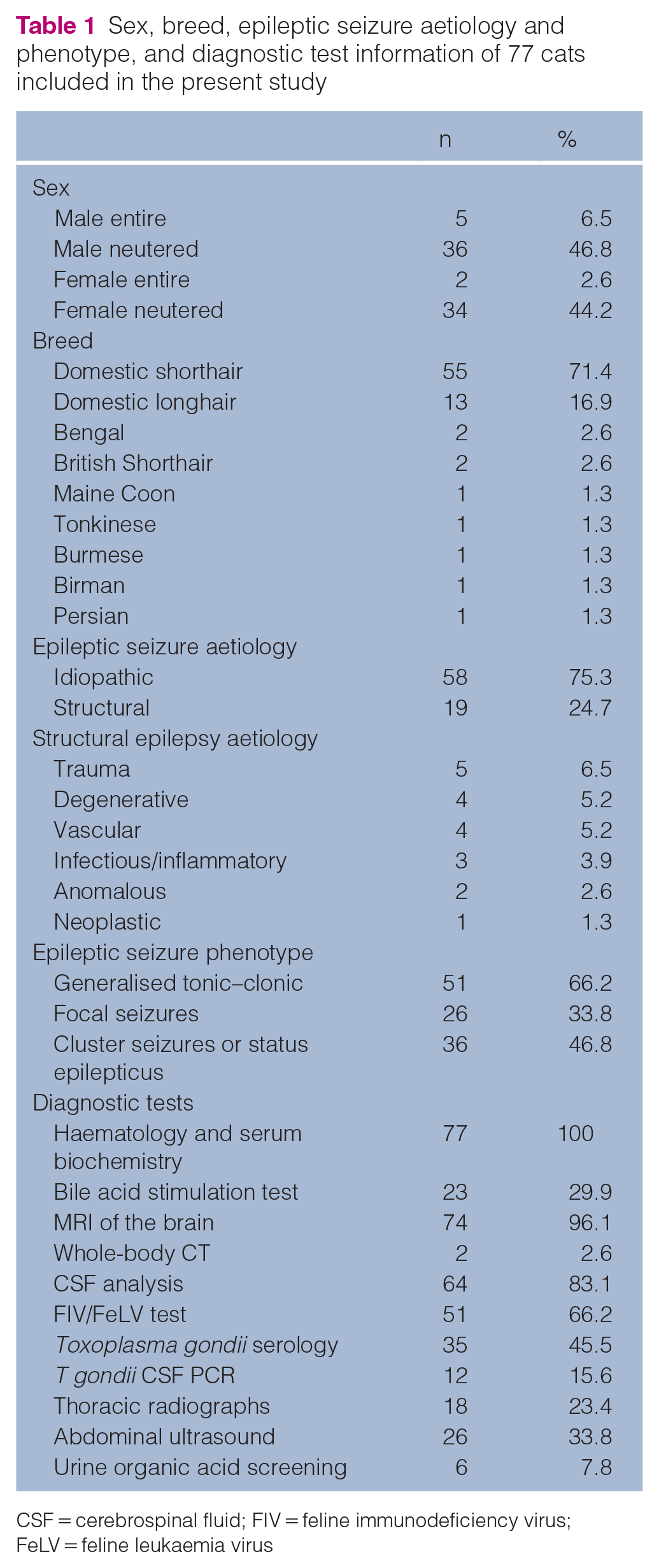
CSF = cerebrospinal fluid; FIV = feline immunodeficiency virus; FeLV = feline leukaemia virus
Seventy-six cats underwent advanced imaging of the brain (MRI in 74 cases and whole-body CT, including the head, in 2 cases). Of the 19 cases of structural epilepsy, three cats affected by infectious or inflammatory disease received specific treatment aimed at resolving the underlying disease. The remainder received AEDs in all cases, and three cats also received anti-inflammatory doses of prednisolone.
The median age of the cats at the time of referral was 68.4 months (range 4–206.4 months; mean ± SD 71.0 ± 46.3 months). The median age of the cats affected by structural epilepsy was 73 months (range 24–150 months; mean 75 months) and the median age of those affected by idiopathic epilepsy was 60 months (range 4–150 months; mean 66 months).
The median initial dosage of PB was 2.0 mg/kg q12h (range 0.6–4.4 mg/kg q12h; mean ± SD 2.0 ± 0.71 mg/kg q12h). PB loading was performed in five cats; the loading dose was 3 mg/kg q6h, four times, in two patients, and a total dose of 20 mg/kg was given in divided doses over 24 h in the other three cats. The median duration of PB treatment was 22 months (range 1.5–108 months; mean ± SD 30 ± 27 months). Thirty-six cats (46.8%) received a concurrent AED. This was levetiracetam in 34 cases and one case each of zonisamide and imepitoin.
Median follow-up time was 24 months (range 1.5–108 months; mean ± SD 30 ± 27 months). The primary veterinarians’ medical records from the time of referral to the time of data collection were obtained for 95% of the cats; the remaining 5% had a re-examination in one of the referral hospitals, which provided the information required for follow-up. The owners of 36 cats (46.8%) completed the follow-up questionnaire. The owners of 69.4% of the cats reported a reduction of seizure frequency of at least 50% with PB treatment. Seizure frequency was decreased by 25–49% in 8.3% of cats, was unchanged in 11.1% of cats and was unknown in 11.1%.
Type A AEs
Thirty-six cats (46.7%) experienced one or more type A AEs (Table 2).
Type A adverse effects in 36 cats

Several cats experienced more than one adverse effect, so the total is >100%
Twenty-five of these cats (69.4%) were affected by idiopathic epilepsy and 11 (30.5%) by structural epilepsy. Median PB dosage at the time of AE onset was 3 mg/kg q12h (range 1.25–5.0 mg/kg; mean ± SD 2.89 ± 0.89 mg/kg) in cats that developed AEs vs a median PB dosage of 2.0 mg/kg q12h (range 0.6–3.5 mg/kg; mean ± SD 2.09 ± 0.66 mg/kg) during the first month of treatment in cats that did not develop AEs. The difference in dosage between the two groups was statistically significant.
In cats experiencing AEs, median PB serum concentration at the time of onset of AEs was 36 mg/l (range 10.3–70 mg/l; mean ± SD 35.04 ± 16.78 mg/l). In cats that did not experience AEs, median PB serum concentration at the first available re-check (range 3 weeks to 3 months after PB treatment initiation) was 19.3 mg/l (range 6–40 mg/l; mean ± SD 21 ± 11.68 mg/l) (Figure 1). The difference in PB serum concentration between the two groups was statistically significant.

Phenobarbitone serum concentration at the first re-examination in 41 cats that did not develop adverse effects (green) vs serum phenobarbitone concentration in 36 cats with adverse effects (blue). The box represents median serum phenobarbitone concentration and interquartile range, and the cross represents the mean serum concentration
AEs were transient in 20 cats. Of these, 13 developed AEs during the initial 4 weeks of the PB treatment and seven developed transient AEs during ongoing PB treatment. Eight cats showed persistent AEs. It was not possible to determine the duration of the AEs in six cases and in two cases the patients were euthanased, reportedly owing to the severity of the AEs.
Twenty-two cats that developed AEs were administered a concurrent AED. This was levetiracetam in 21 cases (dose range 15–20 mg/kg; mean 29.3 mg/kg; median 20 mg/kg q8h) and imepitoin (20 mg/kg q12h) in one case. One cat received the alternative AED prior to commencement of PB. In five cases the concurrent AED was started at the initiation of PB treatment, in five cases between 2 and 4 weeks after PB initiation, in nine cases >1 month after PB was initiated and the initiation time was unknown in one case. The onset of AEs was up to 3 weeks after the initiation of both AEDs in six cases, weeks-to-months before the second AED was added in eight cases, months after the second AED was added in six cats and AE onset was unknown in two cases. The respective likelihood of any AE, sedation and ataxia increased by 3.2, 4.4 and 4.2 times, respectively, when a second AED was used.
Type B AEs
One cat showed lethargy and ataxia in all limbs 2 months after the initiation of PB treatment at 2.6 mg/kg q12h. No epileptic seizures had occurred since starting therapy. Haematology revealed moderate leukopenia with severe neutropenia (0.0 × 109/l; reference interval 2.5–12.5 × 109/l). Serum biochemistry was unremarkable. The serum PB level was 45 mg/l. Bone marrow evaluation (cytology and core biopsy) showed severe granulocytic hypoplasia suggestive of neutrophil aplasia or immune-mediated destruction of neutrophil precursors. PB was decreased by 25% every 4 days and discontinued after 2.5 weeks; levetiracetam (20 mg/kg q8h) and amoxicillin/clavulanic acid (20 mg/kg q12h) were commenced. Two weeks later the neutrophil count was 2.27 × 109/l and clinical signs had resolved. Six weeks and 3 months later, the neutrophil levels were 2.85 × 109/l and 6.40 × 109/l, respectively. This cat was considered to have type A AEs (lethargy and ataxia), as well as the type B haematological abnormalities.
One cat was reported to have consistently elevated alanine transaminase (ALT) 2 years after PB treatment initiation. Another cat was found to have generalised lymphadenopathy 2 months after starting PB. These cases were not classified as being affected by type B AEs as the aetiology of the clinical and biochemical abnormalities could not be definitively established from the available history.
Statistical analysis
Logistic regression models were run for outcome variables ‘AE’ (any type A AEs), ‘sedation’ and ‘ataxia’. The results obtained were very similar for the three models: the initial model significantly explained the data (χ2(4) ⩾10.6, P ⩽0.014) with ‘administration of a second antiepileptic drug’ and ‘PB dose on the occurrence of AEs’ being retained in the model (P ⩽0.011) but ‘aetiology’ and ‘PB serum level’ not making the selection criterion. The final models (χ2(2) ⩾16.2, P <0.0001) provided a good fit to the data (Hosmer–Lemeshow χ2(4) ⩽4.84, P ⩾0.304); the retrieved odds ratios are provided in Table 3. However, for cats suffering AEs and cats not suffering AEs, PB serum concentration at the time of the onset of AE (mg/l) data was not normally distributed (Shapiro–Wilk W ⩾0.904, P ⩽0.006). A Mann–Whitney U-test was used to identify potential significant differences in PB serum concentration between cats suffering AEs and cats not suffering AEs. PB concentrations were significantly higher for cats suffering AEs (U = 244, P <0.0001). PB dosage at the time of onset of AEs was significantly higher than that given during the first month of treatment to cats not affected by AEs.
Odds ratios for influence of phenobarbitone (PB) dosage and administration of a second antiepileptic drug (AED) on the occurrence of adverse effects (95% confidence intervals)

Discussion
This retrospective study is the first to report the prevalence and characteristics of PB-related AEs in a large population of epileptic cats. The prevalence of AEs was 47%, with sedation and ataxia being most common. These type A AEs can be expected with PB administration, given its known pharmacological properties. Increased PB starting dosage and use of a second AED were significant predictors of the occurrence of AEs. Serum PB concentration was significantly higher in cats suffering AEs, although logistic regression analysis determined that this could not be used as a predictor of AEs.
The prevalence of sedation (41.6%), ataxia (24.7%) and polyphagia (22.2%) associated with PB administration in the present study was greater than that reported in a recent systematic review, 1 which found prevalences of 22%, 11% and 11% for sedation, ataxia and polyphagia, respectively. The prevalence of polydipsia (our study 3%, review 2%) was similar. Additional type A AEs that have been previously reported at a prevalence of less than 10% in cats receiving PB include paraparesis, alkaline phosphatase elevation, weight loss and behavioural change. 1 These were not seen in our study population. One study of dogs treated with PB found that sedation/lethargy, ataxia, polyuria, polydipsia and polyphagia occurred in 50%, 55%, 35%, 40% and 30% of cases, respectively. 18 Therefore, type A PB-associated AEs appear with be slightly more common in dogs compared with our feline study population.
In the present study, one cat receiving PB and levetiracetam showed a persistently elevated serum ALT level that remained elevated, even after discontinuation of PB. It was not possible to determine, from the available information, the underlying aetiology of the ALT elevation, although this finding has been previously documented in cats receiving PB. 1
Type B AEs that have been reported in cats receiving PB include pseudolymphoma, thrombocytopenia, stomatitis, coagulopathy, cutaneous eruptions and hyperthermia.1,11–15 These are all rare, occurring in no more than 2% of cats treated with PB. One cat in the present study was reported to show generalised lymphadenopathy 2 months after starting PB; however, the aetiology of this was unknown so it was not classified as a type B AE. It could not be discounted that this cat was experiencing PB-induced pseudolymphoma, but information required for further confirmation of this, such as response to treatment modification, was not available. None of the other previously reported type B AEs was seen in our study population, in which the only type B AE was granulocytic hypoplasia, occurring in one cat (1.3% of the study population). The reported prevalence of haematological abnormalities in dogs treated with PB ranges from 4.2% to 22%.7,19 These are usually subclinical, 19 with only 4–5% of affected dogs showing clinical signs.7,19 Immune-mediated peripheral destruction or immune-mediated damage of precursor cells before their release into the peripheral blood are postulated mechanisms underlying PB-induced haematological abnormalities in dogs and humans.6,20 The mechanism for such changes in cats has not been investigated to date.
Some of the cats in the present study were reported to show PB AEs with low serum PB concentrations; five cases experienced AEs with a serum PB concentration of 10–15 mg/l. One of these had post-traumatic injury following a head trauma of unknown cause 8 months prior to the onset of seizure activity. The remaining five had idiopathic epilepsy. The neurological deficits shown by the cat affected by trauma may have been difficult to distinguish from PB AEs. Three of these five cats received levetiracetam in addition to PB, which may explain the occurrence of AEs in these individuals despite relatively low PB serum concentration. Alternatively, it is possible that the small number of cats in this study showing AEs with a PB serum concentration <15 mg/l reflects a sensitivity in these particular individuals to PB.
In the present study, 22/36 cats experiencing AEs were receiving a second AED in addition to PB (levetiracetam in 21 cats, and imepitoin in one case). The addition of a second AED was significantly associated with an increased risk of AEs. It is likely that PB and the second AED have an additive effect, so increasing AEs. It is challenging to determine, in some cases, whether reported AEs should be attributed to PB, the second AED or both. Lethargy, sedation and ataxia have been previously reported in cats treated with levetiracetam21–23 and imepitoin. 1 AEs of levetiracetam and imepitoin might, therefore, be indistinguishable from those caused by PB. However, the majority of cases in the present study treated with a second AED had AEs documented before the second AED was commenced (eight cases) or weeks to months after it was started (six cases), making a causative link impossible in the former scenario and unlikely in the latter scenario. It was impossible to determine the relative contribution of the second AED to AEs in some cases, especially when two different AEDs were commenced simultaneously. This problem has also been discussed in the human literature. 6 The likelihood of AEs may be increased when two different AEDs are commenced simultaneously, as AEs were reported in all five cases in which two AEDs were started at the same time in a previously untreated cat.
In the majority of cases, AEs occurred within a short time of PB treatment starting, being noticed in the first month after PB initiation in 26/36 cats. In four cases, AEs were reported a significant length of time after the initiation of PB treatment. In two cases, AEs were reported 6 months after starting PB, but in both of these cases AEs occurred following a dose increase due to poor seizure control and inadequate serum PB concentrations. In two cases, both with idiopathic epilepsy, AEs occurred 6 years after starting PB. In one case this was attributed to excess serum PB concentration (38 mg/l). The PB dose was reduced from 2.9 to 2.1 mg/kg q12h and AEs resolved. Another cat developed sedation and ataxia 6 years following the initiation of PB treatment but no further information on this case was available. Euthanasia, reportedly due to PB AEs, was a rare event, only occurring in two cases. The two cats that were euthanased both had structural epilepsy (following presumptive diagnoses of neoplasia in one case and degenerative disease in the other) and progression of these conditions may have been difficult or impossible to distinguish from AED AEs.
AEs were transient in 20/36 affected cats. Broadly, the transience of PB-induced AEs can be attributed to the development of pharmacokinetic and/or pharmacodynamic tolerance. 4 Pharmacokinetic tolerance occurs in dogs treated with PB whereby PB induces hepatic microsomal cytochrome P450 activity, increasing PB clearance. 24 This results in reduced serum concentration over time, despite the dosage remaining unaltered. Hepatic enzyme induction does not appear to occur in the cat, 25 possibly owing to low levels of CYP2C. 26 Pharmacodynamic tolerance is likely to account for the transience of AEs seen in the present study. Pharmacodynamic tolerance is determined by the interaction of the therapeutic molecule with receptors that can be altered over time by processes such as desensitisation, receptor loss and exhaustion of mediators. 27
The majority of the 19 cats affected by structural epilepsy did not receive specific treatment for their underlying disease. Only the three cats affected by inflammatory or infectious disease received specific, definitive therapy aimed at resolving the structural brain disease. The remainder of the cats with structural epilepsy received AEDs and in three cases also received anti-inflammatory doses of prednisolone. Some cats with structural epilepsy may have experienced progression of disease, which could affect mentation, gait and possibly appetite and thirst – signs that could be difficult to distinguish from PB AEs.
Our study found that 75.3% of seizuring cats were affected by idiopathic epilepsy and 24.7% had structural epilepsy. A previous study has classified the seizure aetiology in cats as reactive, structural and idiopathic in 22%, 50% and 25% of cases, respectively. 28 Other studies have found the prevalence of idiopathic epilepsy to range from 30% to 60%.8,29–32 The inclusion criteria of our study, which required participating cats to have experienced two or more seizures at least 24 h apart, meant that reactive cases were unlikely to be included. The ratio of idiopathic to structural aetiology was greater in our study than in previous investigations.28,31 Possible explanations for this are that our study population may be different to that of the other studies (reflective of different geographical or genetic backgrounds); the two centres involved in our study attract a different caseload, compared with other centres; or that the difference is simply due to chance.
While evaluating the efficacy of PB for seizure control in cats was not the primary purpose of this study, our data revealed that 69.4% of cases achieved a seizure frequency reduction of at least 50%. This parallels previous studies, which have shown that PB treatment is successful in the majority of epileptic cats.30,33 A limitation of this finding is that information on seizure frequency after treatment was only available for 31 cats. There was no obvious correlation between epilepsy aetiology and efficacy of treatment.
There were several limitations in this study, some of which are inherent in retrospective investigations. Diagnostic and treatment protocols and the dosage of both PB and any concurrent AED were not standardised. Different laboratories were used to analyse blood samples and measure serum PB concentrations. In many cases the rationale for the addition of the second AED was not available. The information and detail in medical records was variable. Only a minority of cases (5%) had follow-up assessment by a board-certified neurologist or resident working under the supervision of a specialist. Recall bias was possible when follow-up was conducted by interviewing owners or by questionnaire. In some cases of structural epilepsy, progression of the underlying disease may have been difficult to distinguish from PB AEs. While the timing of blood sampling to test serum PB concentration with respect to PB administration varied between cases, this may not be a limitation of the study. Following administration of PB to healthy cats, the serum concentration has been shown to increase for up to 2 h, then plateau for around 10 h. 34 The half-life of PB in cats has been reported to range from 43.4 h to 76 h,25,34 and in dogs it is 46.3–88.7 h.35,36 Ninety-one percent of epileptic dogs have been shown to have no therapeutically relevant changes in the serum PB concentration during the 12 h dosing interval, 37 and it is possible that the situation is similar in cats, meaning that timing of sampling for PB serum concentration may not be important in the majority of cases.
Conclusions
The prevalence of PB-associated AEs was 47%, with sedation and ataxia being most common. These are type A AEs and tended to occur within the first month of treatment before diminishing, likely with the development of pharmacodynamic tolerance. A type B AE occurred in only one cat. Increased PB starting dosages and the addition of a second AED were significantly associated with the occurrence of AEs. Owners should be informed of the likely AEs and their characteristics in cats treated with PB.
Footnotes
Acknowledgements
The authors would like to thank Danica Pollard for her invaluable assistance with the statistical analysis, as well as the owners and referring vets of all cats involved in this study.
Author note
This paper was presented, in part, as an oral abstract at the European College of Veterinary Neurology Congress, 2018, Copenhagen.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee was therefore not necessarily required.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work for the procedure(s) undertaken. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
