Abstract
Practical relevance:
Seizures are one of the most common neurological problems recognized in cats, affecting approximately 1–3% of the general population. Treatment options and prognosis are closely related to the underlying cause, so it is important that veterinarians are familiar with the diagnostic approach to cats with seizures and options for medical management.
Series outline:
This is the second of a two-part article series that reviews the diagnosis and treatment of seizures in cats. Part 2 describes chronic medical treatment options and prognosis for cats with recurrent seizures, and acute treatment of status epilepticus.
Audience:
This review of recurrent seizures in cats is intended for all veterinarians who are facing the challenges of seizure diagnosis and management in the feline patient.
Evidence base:
Recommendations for diagnosis and management of feline seizure disorders have historically been extrapolated from the canine and human literature. The information and guidance provided in this two-part series is based on a review of the recent published literature addressing seizure disorders and antiepileptic treatment in cats, as well as the authors’ clinical experience.

When and why do we treat?
Maintenance therapy with antiepileptic drugs (AEDs) is recommended for most cats with idiopathic epilepsy (IE) when seizures occur more frequently than once every 12–16 weeks, and in all cats whenever seizures occur in a cluster (>1 seizure/24 h), when status epilepticus occurs, or when seizure frequency increases over time (see box below).1,2 The recommendation to treat based on seizure frequency in cats with IE is controversial and should be somewhat flexible. Owner preferences must be taken into consideration and a seizure frequency of one seizure every 6 weeks may be tolerable if that can be maintained without having to administer twice daily AEDs. When the decision is made to treat chronic recurrent seizures with AEDs, a short period without drugs is often recommended to establish baseline seizure frequency before initiating treatment, but there is some evidence that each seizure event has the potential to increase epileptogenic foci in the brain and early treatment may result in better control.3–6
Chronic AED therapy should also be recommended whenever seizures occur post-trauma or when potentially progressive structural forebrain disease (neoplasia, inflammation, hippocampal necrosis) has been identified or is suspected as the reason for the seizures.3,4,7 Even when the primary cause of the seizures can be treated specifically, therapy for the seizures is necessary and may be lifelong. Chronic AED therapy is also recommended following initial AED loading and stabilization (as for status epilepticus) in cats presenting with an acute onset of repetitive seizures caused by a non-progressive intracranial disorder such as a cerebrovascular accident; in some of these cats chronic AED treatment can be gradually tapered and discontinued following weeks or months without seizure recurrence.


Protocol for AED therapy
Clients should be instructed to maintain a seizure log on a calendar, recording details such as length of each seizure, whether it was focal or generalized, and any unusual features or precipitating events. Most of the drugs used to treat seizures require initial veterinary monitoring to allow for dose adjustments and to evaluate for adverse effects. Treatment with one AED (monotherapy) is recommended initially to reduce possible drug interactions and adverse effects; 2 the AED chosen should be based on knowledge about efficacy and adverse effects, as well as owner constraints regarding cost and frequency of administration. Most cats are resistant to frequent administration of oral medication throughout the day, leading to a risk of poor owner compliance for many AEDs. 9
A standard dose of the chosen oral AED is administered and, when steady state serum concentrations have been achieved (based on five elimination half-lives), the serum concentration of the AED is measured if possible. 2 Once a cat is receiving a stable dose that maintains the AED serum concentration within an optimal therapeutic range, then the seizure log will be used to assess the effect of the AED(s) on seizure frequency. A drug is considered to be effective if it results in a decrease in seizure frequency of 50% or more.8,10
When therapy is not effective the patient and the treatment plan must be re-evaluated. Complete physical and neurologic examination and diagnostic testing are warranted in all non-responders to ensure that the cat does not have evidence of progressive intracranial disease. If there is still no evidence to support an underlying cause of seizures that should be treated specifically, and client compliance is good, then drug or dosage adjustments should be made. The dose of the AED being administered should be increased if the maximum therapeutic serum concentrations have not been achieved and if adverse effects are tolerable. If serious adverse effects from the original AED are present, or if the drug concentration is already in the high end of the therapeutic range, then another AED should be added (see box above). 3

Phenobarbital
Phenobarbital (PB) is the AED of choice in cats with recurrent seizures without a metabolic cause.2,3,8,10–12 This is an inexpensive drug that can be administered twice daily. It has a low incidence of serious adverse effects and reaches therapeutic serum concentrations relatively quickly. 10
Oral PB is rapidly absorbed and has high bioavailability, with peak plasma concentrations 1.0–1.5 h after oral administration.1,13 PB is metabolized primarily by the liver, with approximately one-third excreted unchanged in the urine.4,12,14 It has an elimination half-life of approximately 40–80 h in the cat after oral administration.1,11,13–15
The recommended oral dose for cats is 2.5–5 mg/kg ql2h but cats are very susceptible to the sedative effects of PB, so a lower dose (1–2 mg/kg ql2h) is often used initially.2,3 Serum PB concentrations reach steady state levels in approximately 9–15 days, so can be measured 2–3 weeks after starting PB and after each change in dose.1–3,8,16 The timing for collection of a blood sample after PB is administered is not critical because the dosing interval is shorter than the half-life and, with chronic twice daily administration of PB, serum concentrations are relatively stable. 10 PB is a potent inducer of hepatic microsomal enzyme activity; although it is common in dogs to require an escalating dose of PB over time to maintain a constant serum concentration, this phenomenon is negligible in cats.3,8,13,14
Ideal therapeutic serum concentrations of PB in cats are 20–30 µg/ml (90–129 µmol/l), which are slightly lower than the range for dogs, although many laboratories use the same therapeutic range as for dogs (15–45 µg/ml; 107–150 µmol/l) (see box on page 826 and Table l).2,3,8,10,14,16 Some cats will have good seizure control at subtherapeutic levels of PB, so chronic serum monitoring and dosage adjustments are usually only recommended in cats with unacceptable seizure control. Adverse clinical effects are more likely to occur when serum PB concentrations are above the recommended therapeutic range.
PB is a very effective AED in cats. 3 Finnerty et al reported good seizure control in >90% of cats with presumed IE or structural epilepsy with serum PB concentrations in the therapeutic range. 10 Other studies have reported good control during PB treatment in 60–80% of cats with IE, with approximately 40% of the cats becoming seizure-free;14,15 20–30% of cats in these studies were poorly controlled.5,17
One of the biggest obstacles with regard to PB therapy in cats is the necessity for twice daily pilling. This is a hardship for many owners and impossible for some. One study has shown that therapeutic serum PB concentrations could be achieved following chronic transdermal administration of PB gel, but absorption and actual serum concentrations attained are variable and this formulation is not yet commercially available for our patients.6,18
Adverse effects associated with chronic PB administration are minimal in most cats but can include sedation, ataxia, polyphagia and weight gain.3,5 Polyuria and polydipsia are less marked in cats compared with dogs being treated with PB. 8 Blood dyscrasias occasionally occur, leading to granulocytopenia or thrombocytopenia that is usually mild and reversible with discontinuation of PB therapy.1,5 Hepatotoxicosis progressing to hepatic failure, a rare but concerning complication most often identified in dogs with high serum concentrations of PB, has not been reported in cats.3,5,12 Mild increases in alanine aminotransferase and alkaline phosphatase activities are expected in 10–30% of cats treated with PB, but do not seem to be clinically significant. 10 Less common adverse effects include facial or generalized pruritus and immunemediated hypersensitivity reactions including fever, skin eruptions and lymphadenopathy.3,5,8,19 In one cat, dramatic generalized lymphadenopathy (pseudolymphoma) occurred after 3 weeks of PB treatment. 20 All of these adverse effects are reversible when administration of PB is discontinued. 8
Overall, PB is considered a safe and effective AED for cats. It is usually recommended that a complete blood count (CBC) and full biochemistry profile is performed every 12 months in cats receiving PB treatment. 3
Levetiracetam
Levetiracetam (LEV) is a novel AED that is reported to inhibit burst firing without suppressing normal neuronal excitability, thereby inhibiting seizure propagation. 21 It also has neuroprotective properties and may retard electrical kindling, thus decreasing the likelihood that seizure frequency will increase over time.3,8
LEV is rapidly absorbed following oral administration and has an oral bioavailability of nearly 100% whether administered to fasted or fed cats.3,22 Peak plasma concentrations are achieved within approximately 2 h, with an elimination half-life of 3 h; however, the effect of the drug seems to persist longer than its presence in the bloodstream.8,21,23 Most of the drug (70–90%) is eliminated by the kidneys through glomerular filtration so the dose should be decreased in cats with impaired renal function.3,21 As LEV does not undergo any hepatic metabolism it is a good choice for patients with hepatic disease. 23 Its protein binding is clinically insignificant (10%), hence it does not interfere or interact with other protein-bound drugs.3,22
The recommended oral dose of LEV is 20 mg/kg q8h (Table 1). The effective therapeutic range may be similar to that in humans (5–45 (µg/ml) but serum monitoring is rarely performed because there is little correlation between serum concentrations and seizure control.3,23 Because of its high safety margin some have advocated gradually increasing the daily dose in 20 mg/kg/day increments until the desired result is achieved. 38 When a patient receiving LEV is also receiving PB, higher doses of LEV may be necessary, as PB lowers the peak serum concentration of LEV by increasing the oxidative metabolism of LEV in extrahepatic tissues. 4
Oral antiepileptic drug doses in cats
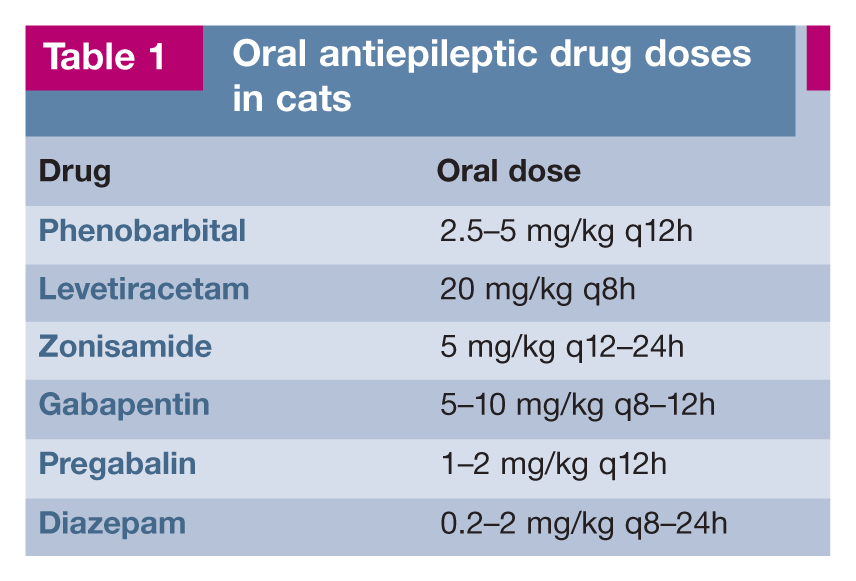
LEV is a good choice as an add-on AED in cats and may also have some utility as a single drug.3,8 When oral LEV was administered as an adjunct to chronic PB treatment, at least a 50% reduction in seizure frequency was reported in 70% of cats with IE. 23 In therapeutic trials, LEV was more successful than PB at controlling myoclonic seizures (93% vs 7%) in cats with audiogenic reflex seizures.24,25 LEV is very well tolerated in cats but three times a day dosing is difficult for most owners. An extended-release formulation of LEV is available and may be effective when administered once or twice a day, but is only formulated in large (500 mg) dosing units that cannot be split or crushed.6,9 Adverse effects of LEV administration, even at high doses, are uncommon and are limited to transient sedation, ataxia and inappetence.3,6,9,14,22
An injectable formulation of LEV is also available and is well tolerated in cats and dogs, with pharmacokinetics similar to the oral preparation.3,22 This drug can be administered (20–60 mg/kg intravenous bolus) to stop seizures in patients with acute seizures or status epilepticus. The anticonvulsant effect is rapid and is maintained for several hours.23,26,27 Patients do not become sedate, simplifying support and monitoring.3,22,26,27
Zonisamide
Zonisamide (ZNS) is a sulfonamide based drug that has been successfully used as an add-on AED and as monotherapy for partial and generalized seizures in people and dogs3,28,29 – and there are anecdotal reports of its use in cats. ZNS is well absorbed after oral administration and peak concentrations are achieved in approximately 4 h in cats.1,30 The drug is metabolized primarily by hepatic microsomal enzymes and has a long elimination half-life of 33 h in cats, reaching steady state within 7 days. The half-life may be significantly shortened in cats concurrently receiving PB or other drugs that increase the hepatic microsomal enzyme p450.8,30 ZNS exhibits relatively low protein binding, decreasing the risks of drug interaction.
The recommended dose for ZNS in dogs is 5 mg/kg ql2h, unless the dog is already being treated with PB, in which case 10 mg/kg ql2h.8,28,29,31,32 Therapeutic serum levels have not been established in cats or dogs, but a target range of 10–40 µg/ml has been advocated in dogs.28,29,31,32 Due to its long half-life, some have recommended once daily administration of a 5–10 mg/kg body weight dose for cats; however, further studies are needed.3,14,30,32
ZNS is primarily used as an add-on drug in dogs with poor seizure control despite treatment with PB and other AEDs, and a response rate of 58–82% has been reported, with a median reduction in seizure frequency of 70–81%.31,32 ZNS monotherapy has also been reported to be effective (>50% decrease in seizure frequency) in 60% of dogs with IE. 28 Three of five cats with refractory seizures when treated with PB alone had a >50% reduction in seizures when ZNS was added, at a mean dose of 11.54 mg/kg q24h. 33 There are also anecdotal reports of the clinical use of ZNS in cats. The authors have had success with a 5 mg/kg ql2h dose in a few cats.
ZNS has a wide therapeutic index but adverse effects reported in dogs include sedation, ataxia and loss of appetite.28,31,32 A dose of 20 mg/kg q24h caused adverse reactions in 50% of treated cats, including anorexia, diarrhea, vomiting, somnolence and ataxia. 30 Rarely, hepatotoxicity and renal tubular acidosis have been reported in dogs 29
Gabapentin/pregabalin
Gabapentin has been used as an adjunctive AED to treat partial seizures in humans.34,35 Gabapentin is excreted entirely by the kidneys in people; however, in dogs, 30–40% of the drug undergoes hepatic metabolism.8,14 One pharmacokinetic study in cats showed an elimination half-life of 2.5–3.5 h and suggested that administration of 8 mg/kg orally q6h would be expected to result in plasma concentrations similar to those recommended in humans.14,34 An oral dose of 5–10 mg/kg q8–12h has been used with limited success in cats (Table l).1,3,14
Pregabalin is a newer generation AED in the same class as gabapentin but with increased potency due to a greater affinity for the binding site 29 The recommended dose in cats is 1–2 mg/kg given orally ql2h (Table 1). The most common adverse effects include sedation and ataxia, and it is possible that these can be reduced by starting at a lower dose (0.5 mg/kg ql2h) with gradual increments over a few weeks. 29
Diazepam
Diazepam is a benzodiazepine that is a very effective AED for treatment of epilepsy in cats, whether administered as the only drug or as an add-on medication when PB is ineffective or contraindicated.3,5,11,18 Oral diazepam has a long elimination half-life (15–20 h) in cats; and, unlike dogs, cats do not appear to develop a functional tolerance to the drug. Diazepam monotherapy (0.5–2.0 mg/kg q8–12h) has been reported to be effective in cats with IE, with 40% of treated cats in one study becoming seizure-free and another 40% showing good control (>50% decrease in seizure frequency); 20% were resistant to treatment.3,5,11,18,19
The most common adverse effects are sedation, physical dependence and the potential for withdrawal seizures. 2 The administration of oral diazepam to cats has also occasionally been associated with potentially fatal idiosyncratic hepatotoxicosis.3,36,37 The two papers describing this reaction were published in 1996 and there have been no further reports since then, despite many veterinarians continuing to use oral diazepam as an anticonvulsant, behavior modifier or appetite stimulant. 38 Because of the potential for acute fatal hepatic necrosis, however, many clinicians, including the current authors, do not recommend the chronic administration of oral diazepam as an AED in cats.2,3,8 Hepatotoxicity was seen in some cats as early as 5 days after initiating therapy, so if a clinician does choose to administer oral diazepam to a cat they should monitor clinical signs and hepatic enzymes 5–7 days after starting the drug and then periodically during treatment for at least 6 months, with plans to discontinue the diazepam if any adverse effects occur.36,37
Despite the concerns surrounding administration of oral diazepam to cats, the intravenous formulation of diazepam is routinely used as the first-line treatment for active seizures and status epilepticus in both cats and dogs (see box on page 832).3,27 No hepatotoxic effects have been reported. 3 Many clinicians will also dispense the parenteral formulation of diazepam for owners to administer rectally to their cat if it has a seizure at home. This immediate treatment can decrease the number of cluster seizure events in a 24 h period and help prevent the development of status epilepticus. It also decreases the need for owners to transport their cat to an emergency clinic or veterinary hospital each time it has a seizure, dramatically decreasing the stress and cost associated with managing an epileptic cat. Recommendations are for the administration of 1–2 mg/kg rectally at the onset of the first seizure, to be repeated up to three times in 24 h.1,27 Diazepam is inactivated by light and adheres to plastic so the parenteral formulation should be dispensed in the original vial or screw top colored glass vial, with instructions for the owner to draw up the required amount into a syringe and then attach a teat cannula or soft catheter to the end of the syringe for rectal administration. 1
Potassium bromide
Bromide was initially thought to be a safe and desirable AED in cats due to its lack of hepatic metabolism and its long elimination half-life (approximately 11 days), allowing for once daily dosing.2,3,8,11,39 Administration of potassium bromide (KBr) at 30 mg/kg q24h or 15 mg/kg ql2h controls seizures in approximately 35–50% of treated cats.1,39
Mild adverse effects can occur in cats treated with KBr, including polydipsia, vomiting and sedation. 39 Unfortunately, nearly 40% of cats receiving KBr have been shown to develop clinical signs similar to bronchial asthma, with chronic cough, mild to marked peribronchial infiltrates and inflammatory bronchoalveolar lavage with eosinophilic predominance.3,39,40 The respiratory signs can be severe, and do not always resolve when KBr is discontinued, progressing to severe bronchiolitis/pulmonary fibrosis and death in some cats.3,11,35,36 The current authors agree with others that, given the relative lack of efficacy of KBr, the availability of other effective AEDs, and the high rate of occurrence of this serious respiratory adverse effect, the use of KBr is contraindicated in cats.3,8,11,38

Status epilepticus
Status epilepticus is usually defined either as seizure activity that lasts for more than 5 mins continuously or two or more seizures lasting less than 2–3 mins each but occurring without complete recovery between seizures.27,45 Most cases of status epilepticus involve generalized motor seizure activity and, therefore, are easy to recognize (convulsive status epilepticus). Focal (partial) seizures that continue for longer than 30 mins are considered simple focal status epilepticus when normal mentation is maintained, and complex focal status epilepticus when mentation is abnormal. Rarely, non-convulsive status epilepticus is recognized in cats, manifested by episodes of stupor, coma or bizarre behavior. 46
Status epilepticus will increase arterial blood pressure, body temperature, heart rate, cerebral blood flow, cerebral oxygen consumption and cerebral metabolic rate. It will also decrease blood pH (due to lactic acidosis) and may decrease arterial oxygenation due to hypoventilation. If status epilepticus continues, metabolic deterioration, increased intracranial pressure, acidosis, hyperthermia, cardiac dysrhythmias and cerebral ischemia develop. Neurons begin to die, resulting in permanent neurologic damage, cerebral edema and further increases in intracranial pressure, often leading to brain herniation and death. Regardless of the reason for their seizures, cats presenting in status epilepticus commonly die.45,47
The most common reasons for cats with IE to present in status epilepticus would include inadequate seizure control and abrupt withdrawal of antiepileptic medications (or missed doses). Non-epileptics present in status epilepticus due to a variety of metabolic disorders, intracranial disorders and intoxications. It is important to learn from the history if there have been changes in behavior or other neurologic abnormalities in the weeks or months preceding the seizure, if there is potential for toxin exposure, and if the cat is at risk for thiamine deficiency. It is also imperative to perform quick diagnostic tests to investigate for metabolic causes of seizures that should be managed specifically (especially hypoglycemia, hypocalcemia and electrolyte disturbances, with blood ammonia if available).27,45 If a metabolic or toxic cause of seizures is identified, specific treatment should be initiated immediately.
The goals of treatment are to stabilize the cat, stop the seizure activity, protect the brain from further damage and allow recovery (see box on page 832).27,45,47 Oxygen is administered, as well as fluid therapy and supportive care including active cooling and blood pressure management. Diazepam is administered intravenously if a patent intravenous catheter is in place, or rectally if there is no intravenous access. If diazepam boluses are effective at stopping the seizure, but seizures recur, a diazepam constant rate infusion can be administered.27,47 A bolus of LEV (20–60 mg/kg IV or SC) can also be very effective at halting seizures in cats with status epilepticus; and, given the high safety profile of LEV and synergy between LEV and diazepam, there is some support for administering both AEDs (when available) as first-line (emergency) therapy.27,47
A longer acting drug, usually PB, is then administered (loading dose – see box on page 832) to help prevent seizure recurrence and to establish therapeutic serum concentrations to aid long term control. There will be a delay of approximately 30 mins before the PB can be expected to have much effect, so if none of the previous treatments have been effective at halting seizure activity, more aggressive treatment will be required to end the seizures; this involves a propofol or pentobarbital infusion to effect as a bolus to induce general anesthesia followed by a continuous rate infusion.27,45,47 This treatment does require careful monitoring and occasionally intubation and ventilation.27,45 Ongoing treatment with longer acting parenteral AEDs (PB ± LEV) should continue even once the seizures have stopped. When the cat is conscious and stable it can be transitioned to standard oral doses of these AEDs for chronic therapy27,45,47
Many cats in status epilepticus have been seizuring for 30 mins or longer before presenting for evaluation or treatment, so should be considered to have the potential for progressive brain injury and increased intracranial pressure. 48 Whenever status epilepticus has been prolonged or brainstem reflexes (pupil size/response and eye movements) suggest brain injury, aggressive treatment is warranted to maintain cerebral perfusion and oxygenation.45,47,48 Supplemental oxygen should be provided by flow-by (100 ml/kg/min). 48 Intravenous crystalloid fluids should be administered to correct acid–base and electrolyte abnormalities and to maintain normovolemia.47,48 Colloid fluids are added if necessary to correct systemic hypotension. Systemic hypertension in an obtunded animal further raises concern for increased intracranial pressure.47,48 Hyperosmolar therapy with mannitol or hypertonic saline is recommended for all patients with presumed intracranial hypertension to decrease intracranial pressure and improve cerebral perfusion pressure.45,47,48

Additional treatments may be warranted in cats with status epilepticus. Thiamine deficiency can occasionally occur and cause status epilepticus in cats on commercially available diets; even if a cat is not thiamine deficient the increased metabolic rate associated with seizure activity can increase the requirement for thiamine. Cats presenting in status epilepticus should therefore be routinely treated with parenteral thiamine (100 mg/cat ql2h). 49 Corticosteroid administration may be warranted in an attempt to decrease inflammation and brain edema when there is a high index of suspicion for intracranial neoplasia or encephalitis. Hyperosmolar therapy may also transiently decrease increased intracranial pressure in some cats with structural intracranial disease.
Key Points
Early treatment with antiepileptic drugs AEDs may result in improved seizure control.
Monotherapy with AEDs is recommended, whenever possible, to reduce the risk of drug interactions and adverse effects, and to improve owner compliance.
Phenobarbital is the AED of choice for most cats with recurrent seizures without a metabolic cause.
Levetiracetam is a safe and rapidly acting AED that is often effective as an add-on drug in cats but can also be used as monotherapy.
Potassium bromide should not be administered to cats due to its potential for causing serious respiratory adverse effects.
Cluster seizures and status epilepticus are life-threatening situations and owners should be able to recognize them as such. The main goals of treatment are to stop the seizure activity and protect the brain from further injury.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
