Abstract
Objectives
The aim of this study was to describe the clinical features, diagnostic imaging findings, treatment and outcome in cats with thoracic vertebral canal stenosis (TVCS).
Methods
Medical records and imaging studies of cats with TVCS were retrospectively reviewed. Outcome was acquired from patient records and from owners or referring veterinary surgeons via a telephone questionnaire. For each case, breed-, age- and sex-matched controls were identified with CT imaging of the thoracic vertebral column. For each cat, vertebral canal height was determined at three levels for each thoracic vertebra. Vertebral canal heights were compared between control cats of different breeds and between affected and control cats of the same breed.
Results
Nine TVCS cases were included. British Shorthairs and male neutered cats were over-represented (P <0.05). Median age at presentation was 9 years. All cats were presented for a chronic, progressive, painful, ambulatory, T3–L3 myelopathy. Five cats were treated conservatively, three surgically and one was euthanased. Two cats treated surgically demonstrated improvement of clinical signs and one demonstrated initial improvement followed by deterioration. Of the conservatively treated cats, three deteriorated and two improved. Compared with controls, affected cats had a lower vertebral canal height at multiple thoracic vertebral levels, being most prominent for British Shorthairs and domestic shorthairs (P <0.05). Unaffected British Shorthairs had a lower thoracic vertebral canal height at multiple levels than control domestic shorthairs (P <0.05).
Conclusions and relevance
TVCS should be considered a differential diagnosis in middle-aged to older cats presenting with a chronic, progressive, painful, T3–L3 myelopathy. The predisposition of British Shorthairs could be explained by a narrower vertebral canal in this breed.
Introduction
Over recent years, our knowledge of feline spinal disease has increased, with more patients undergoing advanced diagnostic investigations and imaging procedures, such as MRI. Infectious and neoplastic disorders, such as feline infectious peritonitis and lymphoma, have previously been considered to represent the most common feline spinal disorders.1,2 However, a more recent study indicated that non-lymphoid neoplasia and intervertebral disc disease were also common causes of spinal disease in cats. 3 Thoracic vertebral canal stenosis (TVCS) was recently documented to be among the 10 most common causes of spinal disease in the cat, 3 and a recent report described the successful surgical management of two cats with articular process hypertrophy causing TVCS. 4 However, there is little known about the clinical presentation, imaging findings and outcome of cats with this disease.
Vertebral canal stenosis is an abnormal narrowing of the vertebral canal, resulting in compression of the spinal cord or nerve roots. It can occur focally, segmentally or generalised throughout the vertebral column and can be classified by aetiology into congenital, developmental and acquired causes. 5 Congenital stenosis results from malformations present at birth while developmental stenosis is caused by an active underlying process that remains present through the growth period, until the vertebrae reach maturity. Acquired stenosis may result from a variety of pathologies such as hypertrophied ligaments, intervertebral disc herniation and degenerative articular changes. Vertebral canal stenosis can further be classified into absolute and relative vertebral canal stenosis. A vertebral canal diameter that results in compression of neural elements directly is termed absolute stenosis, whereas a diameter that is less than normal but not causing compression of neural elements is termed relative stenosis. 6 The latter condition results in decreased available space for the spinal cord to compensate for extradural space-occupying conditions. Relative vertebral canal stenosis therefore predisposes animals to develop clinical signs when relatively mild space-occupying pathologies, such as age-related intervertebral disc protrusion or ligamentous hypertrophy, occur.5,7
Several studies have reported the clinical characteristics and treatment of TVCS in dogs.8–11 However, there is a paucity of information about this disease in feline patients. The aims of this study were therefore to describe the clinical and diagnostic imaging characteristics, treatment and outcome in a group of cats with TVCS. We hypothesised that cats diagnosed with TVCS would demonstrate relative vertebral canal stenosis. Furthermore, it was hypothesised these cats would have a characteristic clinical presentation and would demonstrate a favourable response to surgical management.
Materials and methods
Ethics approval was granted by the Royal Veterinary College (RVC) Ethics and Welfare committee (reference number: SR2018-1654).
Criteria for inclusion
The digital medical records of the University of London, Royal Veterinary College (RVC), Small Animal Referral Hospital from 1 January 2010 through to 1 September 2018, were reviewed to identify cats with a diagnosis of TVCS. For inclusion in the study, cats were required to have MRI and neurological examination findings consistent with a diagnosis of TVCS and available follow-up information. A diagnosis of TVCS was made based on the findings of reduced dimensions of the thoracic vertebral canal with associated compression of the spinal cord with or without adjacent spinal cord intraparenchymal signal intensity (ISI) changes. Cats were not included if a clear degenerative or anatomical change was present that would likely cause clinical signs in the absence of reduced vertebral canal size. All diagnostic imaging studies and medical records were reviewed by a board-certified veterinary neurologist (SDD) to evaluate study eligibility. Information recorded for each cat included signalment, duration and type of clinical signs, treatment prior to referral and response, and general physical and neurological examination findings. Diagnostic tests performed and results, treatment received, duration of hospitalisation and presence of complications were also recorded. Cats were excluded if the medical files or imaging records were not available, or if other abnormalities were detected that could have caused or contributed to the cat’s clinical signs.
For comparative reasons, a control group was established. For each cat identified with a diagnosis of TVCS, it was aimed to include 10 breed- and sex-matched controls. For inclusion, control patients were to be skeletally mature and have undergone full CT imaging of the thoracic vertebral column for reasons unrelated to gait abnormalities, spinal disease or trauma. The signalment of each cat and reason for CT imaging were recorded from the medical records.
Diagnostic imaging
For all included cats with a diagnosis of TVCS, MRI was performed using a 1.5 T unit (Intera; Philips Medical Systems). MRI was performed under general anaesthesia and included a minimum of T2-weighted (T2W) and T1-weighted (T1W) sagittal and transverse images. T1W sagittal and transverse images were acquired after administration of gadolinium-based contrast medium (0.5 ml/kg IV). For each cat, the site(s) and suspected anatomical cause of TVCS were recorded, as well as any associated ISI changes relative to normal spinal cord parenchyma. ISI changes were recorded if a T2W hyperintensity was seen, with or without a corresponding hypointense ISI change on T1W images. For all included case-controls, CT imaging was performed with a 16-slice helical CT scanner (PQ 500, Universal Systems, Solon; GE Healthcare) under sedation or general anaesthesia. Sagittal reconstructions were made after the transverse images were acquired.
Measurements
All CT and MRI measurements were made by a veterinary neurology specialist-in-training (SG) under the supervision of a board-certified veterinary neurologist (SDD) on commercially available DICOM viewing software (Horos, version 1.1.7, www.horosproject.org). For cats with a diagnosis of TVCS, measurements were made on mid-sagittal T1W magnetic resonance images. For the control group, measurements were made on mid-sagittal CT images of the thoracic vertebral canal in the bone window (helical scan mode, slice thickness 2 mm, interslice interval 1 mm, collimation pitch 16 × 1.5 mm, 120 kVp, 100 mA and a 512 × 512 matrix). The accuracy of the digital measurement tool was limited to 0.01 mm.In each cat, for vertebrae T1–T13, vertebral canal height was determined at the cranial (VCHcr), middle (VCHm) and caudal (VCHcd) aspects of the vertebral body as previously described. 12 To improve visualisation of the dorsal margin of the vertebral body, a reference line was drawn to connect the most craniodorsal and most caudodorsal points. Vertebral canal height measurements were made perpendicular to this reference line. The VCHcr was measured from the most craniodorsal point of the vertebral body to the lamina. The VCHm was measured from the point that corresponded to half the length of the vertebral body to the lamina. The VCHcd was measured from the most caudodorsal point of the vertebral body to the lamina.
Outcome assessment
Outcome information for cats with a diagnosis of TVCS was acquired from a combination of medical records of re-examination visits at the RVC Small Animal Referral Hospital, and telephone interviews with referring veterinarians and owners. Referring veterinarians were first contacted and asked a series of questions regarding the patient’s clinical status, current medications, neurological deficits present and progression after commencement of treatment. For cases that were deceased, the date, cause of death and last documented neurological status were recorded. Conforming to local ethics and welfare guidelines, the owners of cats known to have died were not contacted further. Owners of cats last known to be alive were mailed a letter that included the study details and a standardised questionnaire that had been reviewed and approved by a local ethics and welfare committee. Telephone interviews were conducted with one investigator (SG) based on the questionnaire, which included information on activity levels, paresis, incontinence, pain levels, type of treatment received, response to treatment and quality of life (see the supplementary material). Treatment outcome was defined as improved, stabilised or deteriorated based on change in the original neurological signs. Improvement was defined as an increase in pelvic-limb function score without occurrence of urinary or faecal incontinence or pain. Deterioration was defined as a decrease in pelvic-limb function score, occurrence of urinary or faecal incontinence, or continued pain.
Statistical analysis
Data analysis was performed using a statistical software package (SPSS Statistics for OSx, Version 24.0; IBM). Data were tested for normal distribution using the Kolmogorov–Smirnov test and presented as mean ± SD. Independent t-tests were used to compare the cranial, middle and caudal vertebral canal heights of each vertebra between unaffected cats of different breeds and between affected and control cats of the same breed. A χ2 test was used to compare the prevalence of sex and breeds that were included more than twice in the list of affected breeds (domestic shorthair [DSH], British Shorthair [BSH]) with the general hospital population seen over the same period. For all comparisons, P values <0.05 were considered significant.
Results
Nine cats with TVCS and 81 control animals were included in the study.
Of the 12 cats with TVCS identified from the database, three cats were excluded owing to incomplete medical or imaging records. The nine cats with TVCS included in the study consisted of three BSHs, three DSHs, and one each of Bengal, Exotic Shorthair and Maine Coon. Eight of the cats were male neutered and one cat was female neutered. Median age was 9.0 years (range 5.0–14.0 years). Compared with the general hospital population, BSHs (P <0.0001) and male neutered cats (P = 0.017) were significantly over-represented.
Eighty-one control cats were included, comprising 30 BSHs, 30 DSHs, 10 Bengals, five Exotic Shorthairs and six Maine Coons. For Exotic Shorthair and Maine Coon breeds, five and six control cases were included, respectively, owing to insufficient CT studies being available. Ten female neutered and 71 male neutered cats were included, with a median age of 8 years (range 1.2–16.8 years). Control cats underwent CT for variety of reasons, including respiratory disease (n = 20), abdominal disease (n = 14), neoplastic disease (n = 40) and further investigation of immune-mediated disease (n = 7).
Clinical presentation and diagnostic findings in cats with TVCS
Duration of clinical signs prior to presentation ranged between 1 day and 9 months, with eight cats demonstrating progressive clinical signs of at least 2 weeks’ duration. One cat demonstrated an acute onset of clinical signs following a minor trauma. All cats demonstrated ambulatory paraparesis and proprioceptive ataxia in the pelvic limbs, lateralising in four cases. All cats demonstrated hyperaesthesia on spinal palpation and none had a history of faecal or urinary incontinence. Neuroanatomical localisation was to the T3–L3 spinal cord segments in all cases.
Imaging
MRI studies demonstrated a single site of TVCS in eight cats and three sites of TVCS in one cat. In all cases, the thoracic vertebral canal was subjectively narrowed with secondary, mild changes to adjacent structures contributing to stenosis. In six cases (and at eight locations), vertebral canal stenosis was secondary to a combination of ventral spinal cord compression due to mild intervertebral disc protrusion and dorsal compression caused by the dorsal lamina and ligamentum flavum (n = 4) or articular processes (n = 2). The T3–T4 and T11–T12 intervertebral disc spaces were most often affected (n = 3 sites of spinal cord compression for each), followed by T4–T5 (n = 1) and T8–T9 (n = 1). In two cases, stenosis was secondary to mild hypertrophy of the dorsal lamina and ligamentum flavum alone, at T2 and T5. In one case, marked dorsoventral compression of the spinal cord was present at T9 secondary to a subjectively narrowed vertebral canal, in the absence of an appreciable anatomical abnormality. ISI changes at the site of spinal cord compression were characterised by an ill-defined, T2W hyperintense and T1W isointense lesion in eight cats and a focal T2W hyperintense and T1W hypointense lesion in one cat. Abnormal contrast uptake was not noted in any cat. In all cases, intervertebral disc protrusion was mild and anatomical changes to the vertebrae or associated structures were difficult to appreciate, despite obvious spinal cord compression and adjacent ISI changes (Figure 1). Additional CT studies were available for two cats and demonstrated no obvious anatomical abnormalities leading to TVCS.
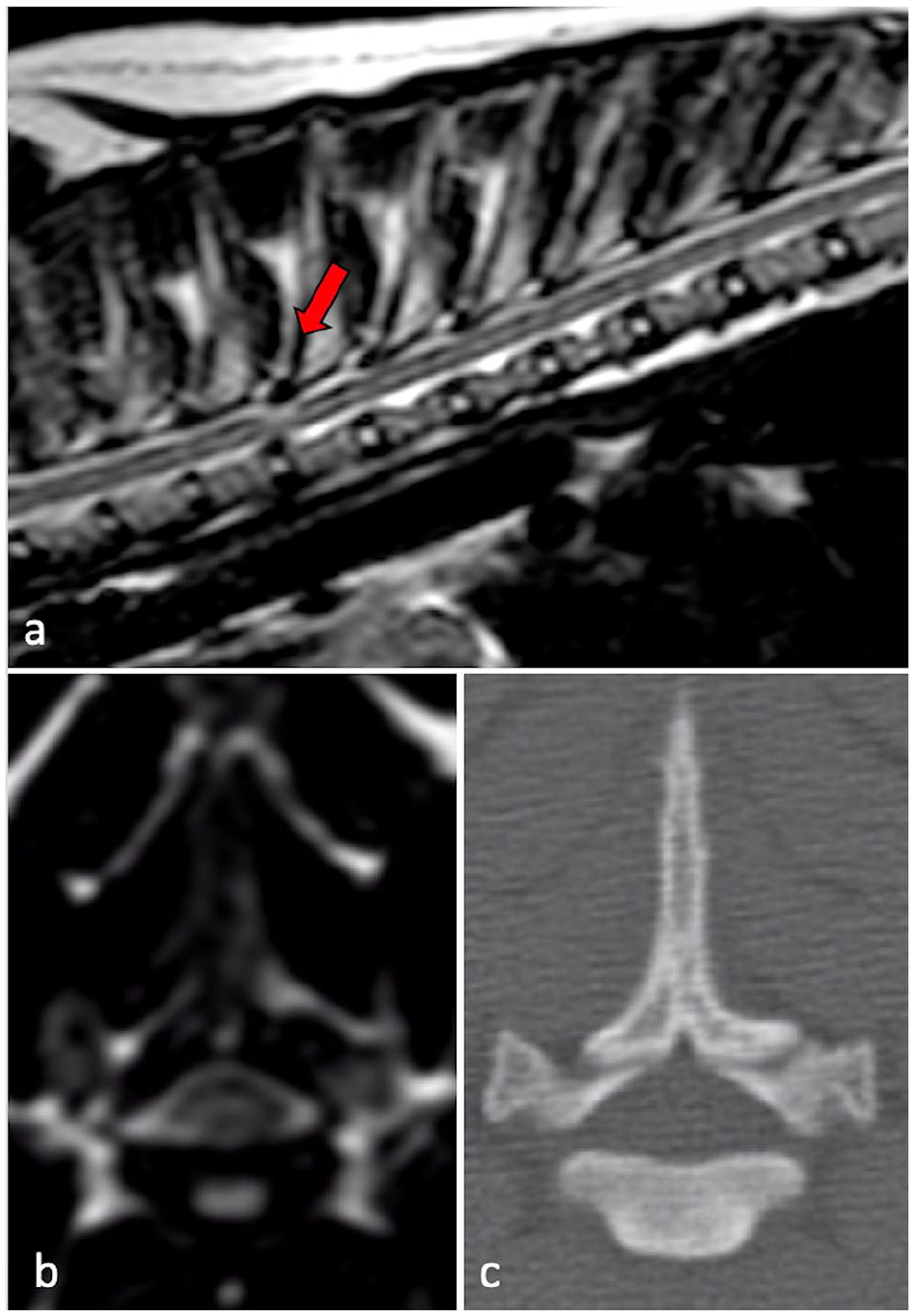
T2-weighted (a) sagittal and (b) transverse magnetic resonance images of the thoracic spine at the level of T3–T4 (red arrow), demonstrating subjective thoracic vertebral canal narrowing and an intramedullary hyperintensity. (c) Corresponding CT image at the same level demonstrating a lack of obvious anatomical abnormality
Ancillary diagnostics
Haematology and serum biochemistry results were available for all nine cats and were unremarkable in all cases. Feline immunodeficiency virus and feline leukaemia virus status were available for four cats, all of which were negative. Toxoplasma serology was performed in three cases and was negative in all. Four cats had cerebrospinal fluid (CSF) analysis performed from the lumbar cistern. Total nucleated cell count (TNCC) and total protein (TP) concentrations were considered normal in three cases (TNCC <5 mm3 and TP <0.45 g/l). In one case, there was evidence of mild albuminocytological dissociation with a TP concentration of 0.75 g/l (reference <0.45 g/l). In one cat, toxoplasma and feline coronavirus PCRs performed on CSF were negative.
Treatment
Owners were informed of treatment options by a neurology specialist or specialist in training and treatment undertaken was based on owner preference. In one cat presenting acutely, medical management was preferentially recommended. Eight cats underwent medical (n = 5) or surgical (n = 3) treatment of the condition. One cat was euthanased shortly after diagnosis owing to deteriorating neurological signs after which the owners declined further treatment.
Surgical treatment
Two cats underwent a left-sided hemilaminectomy for removal of an articular facet joint perceived to be contributing to vertebral canal stenosis. The procedure was performed at the T11–T12 and T3–T4 intervertebral disc spaces, respectively. These two cats demonstrated no postoperative deterioration and were ambulatory at the time of discharge. One cat with stenosis at T4–T5 underwent initial medical management with meloxicam, gabapentin and restricted exercise. Owing to continued progression of paraparesis and proprioceptive ataxia over 4 weeks, a T3–T5 dorsal laminectomy was performed. Surgery revealed a subjectively narrow vertebral canal without obvious anatomical abnormalities. This cat demonstrated deterioration postoperatively and was non-ambulatory at the time of discharge with good pelvic limb movement. All cats were hospitalised for 6 days postsurgery and were discharged with instructions for 4 weeks’ restricted exercise and meloxicam (0.1 mg/kg PO q24h) for 7–14 days. At re-check examinations between 1 and 2 months following surgery, all cats that underwent surgical treatment were ambulatory and comfortable with a mild improvement in neurological function compared with original presentation. None of the cats were considered neurologically normal.
Follow-up information was available for all three cats treated surgically. Two cats were alive at the time of follow up (11.5 months and 74.3 months, respectively). One cat underwent a hemilaminectomy and was considered neurologically normal 74.3 months after surgery. The other cat underwent a dorsal laminectomy and had serial neurological examinations every 2–3 months at the study institution. It demonstrated signs of slow deterioration (increased paraparesis and faecal incontinence) approximately 8 months after surgery, after an initial period of improvement. The MRI study at the time of diagnosis demonstrated a combination of intraspinal T2W hyperintensity and T1W hypointensity at the site of spinal cord compression (Figure 2). The third cat had been euthanased 51 months after hemilaminectomy surgery (at 14 years of age) due to the development of non-ambulatory paraparesis. This cat demonstrated an initial, sustained improvement in neurological signs and lived with a mild paraparesis and proprioceptive ataxia for approximately 4 years, prior to a chronic deterioration. Unfortunately, neither cat demonstrating a period of stabilisation and subsequent deterioration underwent further investigations and the exact cause of neurological deterioration remains unknown.

(a) T2-weighted (T2W) and (b) T1-weighted (T1W) sagittal images at the level of T4–T5 (red arrow) demonstrating a focal T2W hyperintense and T1W hypointense intraparenchymal lesion at the site of spinal cord compression. These imaging changes may be considered consistent with a cystic or cavitary type lesion
Medical treatment
Three cats received meloxicam (0.05 mg/kg PO q24h) alone and one cat received a combination of meloxicam and gabapentin (10 mg/kg PO q8h). One cat was started on oral prednisolone at an initial dose of 0.6 mg/kg PO q24h, tapering down over a period of 3 months.
Follow-up information was available for all five cats treated medically. One cat was alive at the time of follow-up (32 months) and had been tapered off oral medication (prednisolone). This cat was reported to be neurologically improved but still had a mild ambulatory paraparesis and proprioceptive ataxia. One cat demonstrated improvement and was reported to be neurologically normal before death in a road traffic accident 15 months after treatment. This cat had demonstrated an acute onset of clinical signs following a minor trauma. Three cats had been euthanased at 1, 35 and 48.5 months after starting treatment due to deteriorating paraparesis, with one cat becoming non-ambulatory. One of these three cats also developed urinary and faecal incontinence.
Vertebral canal measurements
One MRI study was of insufficient quality for accurate measurements to be performed. Therefore, imaging studies from eight cats with TVCS were further evaluated.
Unaffected BSHs were found to have a significantly smaller VCHcr, VCHm and/or VCHcd at all levels when compared with DSHs (P <0.05). More specifically, at T4, T5, T9 and T11 VCHcr, VCHm and VCHcd were all significantly smaller in BSHs compared with DSHs (Table 1). Unaffected Bengals demonstrated a significantly smaller cranial and middle sagittal vertebral canal height at T5, and unaffected Maine Coons did not demonstrate a significantly different vertebral canal height at any level, when compared with DSHs.
Mean values of cranial (VCHcr), middle (VCHm) and caudal (VCHcd) vertebral canal heights in control British Shorthair (BSH) and control domestic shorthair (DSH) cats

Significant difference between values in BSH and DSH cats (P <0.05)
Compared with controls, cats with TVCS had significantly smaller thoracic vertebral canal heights at multiple levels. This was most notable in the DSH and BSH breeds (P <0.05, Table 2). More specifically, at T2–T7, T11 and T12, VCHcr, VCHm and/or VCHcd were significantly smaller in affected BSHs compared with control BSHs. At T1, T3, T5 and T8–T13, VCHcr, VCHm and/or VCHcd were significantly smaller in affected DSHs compared with control DSHs. VCHcr was significantly smaller at T9 in affected Bengals compared with control Bengals, and VCHca was significantly smaller at T3 in affected Maine Coons compared with control Maine Coons.
Mean values of cranial (VCHcr), middle (VCHm) and caudal (VCHcd) vertebral canal heights in affected British Shorthair (BSHa) and control British Shorthair (BSHc) cats
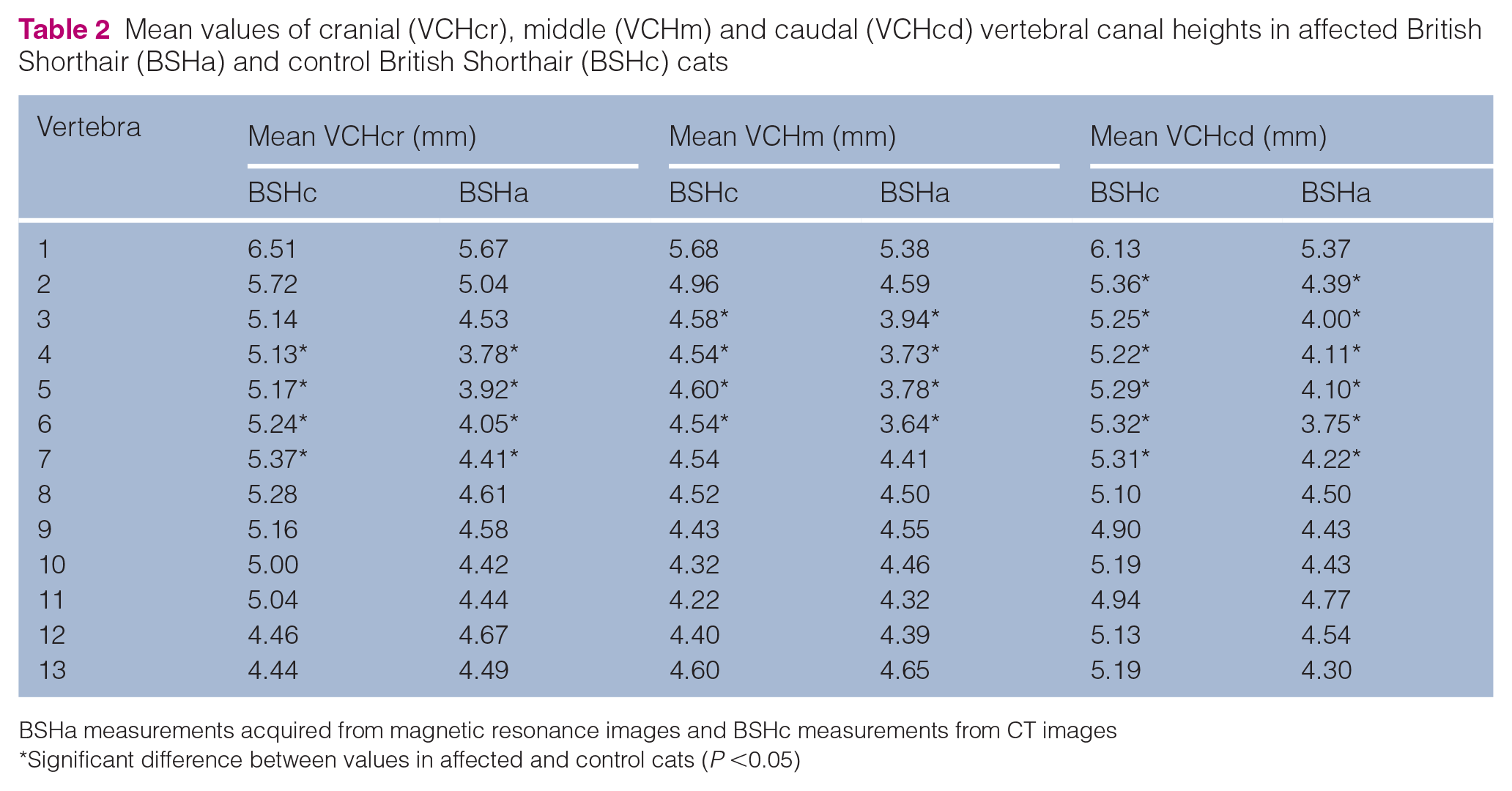
BSHa measurements acquired from magnetic resonance images and BSHc measurements from CT images
Significant difference between values in affected and control cats (P <0.05)
Discussion
Thoracic vertebral canal stenosis is poorly characterised despite being a common cause of spinal disease in the cat. 3 This study demonstrated TVCS to be most frequent in middle-aged to older male neutered cats presenting with a chronic, progressive, painful T3–L3 myelopathy. Imaging studies typically demonstrated dorsoventral spinal cord compression and ISI changes in the absence of a marked anatomical abnormality. Surgical and medical treatment of this disease appears to carry a variable prognosis.
In dogs, TVCS typically occurs in the cranial thoracic segments of young, large-breed dogs with a conformation characteristic of Molosser breeds. Lateral and dorsolateral spinal cord compression results from enlargement and malformation of the articular facet joints, with a developmental aetiology considered likely.8–11 In contrast, TVCS in cats represents a different disease process. In this study, all cats were middle-aged to older at presentation and typically presented with chronic, progressive clinical signs. Imaging studies demonstrated no evidence of marked anatomical abnormality to the vertebrae or associated structures despite obvious spinal cord compression and adjacent ISI changes. Any changes seen at the site of compression were considered mild, expected to be age related and would not be anticipated to cause clinical signs in the presence of normal vertebral canal dimensions. Given the clinical presentation and imaging findings, we hypothesised that these cats may have a pre-existing relative vertebral canal stenosis. Reduced ‘free space’ for the spinal cord may then lead to the development of an absolute stenosis and clinical signs secondary to mild age-related degenerative changes, such as intervertebral disc protrusion, ligamentous hypertrophy or articular process hypertrophy.
A recent case report described thoracic vertebral canal stenosis in two cats, secondary to bilateral articular process hypertrophy. 4 It is unclear if this report describes a different disease process to that seen in the current study, given the appreciable articular process hypertrophy on imaging. However, in agreement with the current study, both cats were middle-aged or older at presentation and demonstrated a chronic, progressive T3–L3 myelopathy. In contrast to the present study, both cats had evidence of ventral spondylosis deformans at the site of vertebral stenosis, suggesting chronic vertebral instability and secondary articular process hypertrophy as the pathophysiology. 4
Overall, 6/9 cats affected by TVCS in the present study were of purebred descent and results suggested a breed-related predisposition in BSHs. Interestingly, this breed was previously reported to be more commonly diagnosed with thoracolumbar intervertebral disc disease. 13 Although it is unclear why BSHs may be predisposed to both TVCS and thoracolumbar intervertebral disc disease, it is possible that pre-existing relative vertebral canal stenosis contributes to the development of clinical signs with age-related degenerative changes.
The most frequent sites of stenosis were the T3–T4 and T11–T12 intervertebral disc spaces, consistent with the sites affected in the aforementioned case report. 4 It is unclear why the sites of stenosis varied and it may be expected for degenerative changes to primarily occur in the caudal thoracic vertebral column, which shows a higher degree of flexibility and is subject to increased biomechanical forces compared with the cranial thoracic compartment.14,15 Equally, it is possible relative stenosis is limited to selected regions of the vertebral canal.
Susceptibility of the spinal cord to compression depends on vertebral canal dimensions in both the transverse and sagittal planes. 16 These data should thus be interpreted with caution given that measurements were only acquired in the sagittal plane. However, our results suggest that BSH cats without TVCS have a relatively smaller thoracic vertebral canal height at multiple levels than DSH cats without TVCS. This might provide an explanation for a predisposition to development of TVCS in this breed. Correspondingly, TVCS-affected cats demonstrated a significantly lower vertebral canal height at multiple thoracic levels, reflecting the sites of stenosis on MRI studies.
Although well established in dogs,7,17–19 there is little known about normal vertebral canal dimensions in cats. Breed and body size are known to influence morphometric dimensions of the vertebral column in dogs. Large breed dogs and Dachshunds have lower vertebral canal dimensions than small breeds at multiple thoracic and lumbar vertebral levels. Sites of reduced vertebral canal dimensions appear consistent with those sites most commonly clinically affected by spinal cord compression. 17 Similarly, it may be speculated that cats affected by TVCS have lower thoracic vertebral canal dimensions than normal. However, further morphometric studies are required in cats to determine normal vertebral canal dimensions and possible breed or size-related variations, as seen in dogs.
There is little information available detailing treatment and prognosis for TVCS in cats. A good outcome 6 months after surgery has been reported for two cats with TVCS secondary to articular process hypertrophy. 4 Similarly, of the three cats that underwent surgery in the present study, all demonstrated a good outcome 1–2 months postsurgery and two demonstrated a good long-term outcome.
The cat that underwent a T3–T5 dorsal laminectomy demonstrated a progressive deterioration 8 months after surgery. This cat was initially treated medically without success and demonstrated a focal T2W hyperintense and T1W hypointense intraparenchymal lesion at the site of spinal cord compression. These imaging changes may be considered consistent with a cystic or cavitary type lesion. 20 Such parenchymal signal intensity changes have previously been associated with neurological deterioration and histopathological evidence of segmental chronic myelomalacia and gliosis in two dogs with disc-associated cervical spondylomyelopathy. 21 Equally, there is growing evidence in people that these signal intensity changes represent pathologically irreversible spinal cord damage and have been associated with a poor prognosis in cases of cervical spondylotic myelopathy.22–24 It is unknown why the cat in this report demonstrated progressive deterioration after initial clinical improvement was observed. Although it is possible that the specific intraparenchymal intensity changes seen at the time of diagnosis reflected irreversible spinal cord damage, it is also possible that the delay in surgical treatment associated with initial medical management affected prognosis. This cat did not undergo repeat imaging at the time of neurological deterioration. Therefore, it cannot be excluded that an additional site of spinal cord compression or another spinal problem occurred.
In 3/5 cats treated with medical management alone, the condition continued to slowly progress to the point of euthanasia. This likely reflects the chronic, progressive nature of degenerative changes in the vertebral canal. Notably, the one cat that presented acutely made a full and rapid recovery with medical management alone. It may be hypothesised that the already compromised spinal cord suffered an acute contusive injury in the region of stenosis.
The present study was limited by the small number of cats with TVCS and by its retrospective nature, which necessitated reliance on the accuracy of the medical records and did not allow for standardised follow-up assessment. Not all control cats underwent neurological examination and thus mild spinal disease could not be completely excluded. Owing to an inadequate number of CT studies in TVCS-affected cats, MRI measurements were compared with CT measurements in control cats. Although this can be considered less accurate, a previous study demonstrated good agreement for vertebral canal height measurements between low-field MRI and CT. 25 Equally, imaging measurements represented true measurements found on cadaveric specimens. In the present study, only sagittal vertebral canal measurements could be acquired owing to a lack of transverse studies of the entire thoracic vertebral column in TVCS-affected cats. Linear ratios have previously been suggested unreliable in predicting relative vertebral canal stenosis in dogs12,26 and people,27,28 and therefore absolute measurements were determined and compared. Relative vertebral canal stenosis depends on the transverse and sagittal vertebral canal dimensions, as well as the dimensions of the spinal cord. 12 Cross-sectional area measurements on transverse images may be considered a more reliable determinant of relative vertebral canal stenosis in future studies.
Conclusions
TVCS should be considered a differential diagnosis in middle-aged to older cats presenting with a chronic, progressive, painful, T3–L3 myelopathy. Its prevalence appears to be higher in BSH and male neutered cats. Further studies are required to determine optimal treatment in these cats and it is possible that outcome may vary according to the anatomical structures contributing to stenosis. Medical management typically resulted in a slow progression of clinical signs. Surgical management resulted in a good short-term outcome and variable long-term outcome. It is suspected these cats may have a pre-existing relative stenosis of the thoracic vertebral canal and develop absolute stenosis secondary to age-related degenerative changes of the vertebral column and associated structures. It remains unknown if vertebral stenosis is generalised throughout the vertebral canal or limited to the thoracic region and, equally, if there is variation between different breeds and size of cat. Although further studies are necessary, the predisposition of BSHs could potentially be explained by a narrower vertebral canal in this breed compared with other breeds.
Supplemental Material
Supplemental Material
The questionnaire used for conducting telephone interviews
Footnotes
Supplementary material
The following file is available online:
The questionnaire used for conducting telephone interviews.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animal(s) only (owned or unowned), and followed established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care. Ethical approval from a committee was therefore not necessarily required.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work for the procedure(s) undertaken. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
