Abstract
Objectives
The aim of this study was to describe the pharmacokinetics of oral transmucosal (OTM) detomidine gel in healthy cats and assess its effects on sedation and hemodynamic variables.
Methods
Eight adult cats weighing 4.12 kg ± 0.72 received 4 mg/m2 detomidine gel onto the buccal mucosa. Level of sedation, heart rate (HR), blood pressure (BP) and respiratory rate (f R) were assessed at predetermined intervals following administration. Blood samples for plasma detomidine concentrations and venous blood gas variables were collected from a medial saphenous catheter. Plasma detomidine concentrations were analyzed using ultra-high-pressure liquid chromatography with mass spectrometry detection, and pharmacokinetic estimates were obtained with compartmental methods. Data were analyzed using ANOVA and paired t-test or appropriate non-parametric tests.
Results
Sedation occurred in all cats, and was increased from baseline at 30 mins (P <0.001). Decreases in HR occurred from 15–60 mins, ranging from 140 to 165 beats per min (P <0.001). Blood glucose increased from 101 ± 12 mg/dl to 168 ± 27.3 mg/dl at 60 mins (P = 0.004). Systolic blood pressure decreased from baseline (139 ± 14.8 mmHg) to 103 ± 23.0 mmHg at 60 mins (P = 0.023). All changes abated by 120 mins. Emesis occurred in 7/7 cats within 2 mins of gel administration. Geometric mean (coefficient of variation) for clearance was 220.7 ml/min/kg (35.3 ml/min/kg), volume of distribution was 14.9 l/kg (39.9 l/kg) (both a function of bioavailability) and elimination half-life was 46.9 mins (16.0 mins). Maximum plasma concentrations of 10.5 ng/ml (35.5 ng/ml) detomidine occurred at 36.9 mins (21.5 mins).
Conclusions and relevance
OTM detomidine gel produced moderate sedation with minimal undesirable side effects in healthy cats, although emesis occurred in all cats. The pharmacokinetic profile supports short-term, minimally invasive sedation in this species. Further studies are warranted to assess its safety and feasibility for use in debilitated cats, or prior to general anesthesia.
Introduction
Alpha (α)2-adrenergic agonists are among the most frequently used medications in veterinary medicine today and are known for their potent sedative and anxiolytic effects. 1 Detomidine is an α2-adrenoceptor agonist commonly used to provide sedation and analgesia in horses.2,3 The injectable formulation has been administered via the sublingual route in horses, as well as an oral transmucosal (OTM)-administered gel formulation (Dormosedan Gel; Zoetis) to facilitate sedation. 4 This formulation has been used via the buccal or sublingual routes in multiple species, including dogs, ferrets and rabbits.5–10 In addition to its use in equine medicine, it has been shown to have favorable sedative and anxiolytic effects in dogs.6,7,10
Stress-free handling of feline patients at veterinary clinics is a concern for veterinarians and owners alike. Cats can become stressed during transportation to the clinic, as well as during the examination period. Different medications can be used for sedation or anxiolysis in feline patients but can be associated with limitations in time of onset, duration and side effects. For most of these medications, owners would need to have their cat ingest a capsule/tablet such as gabapentin or trazodone,11,12 or veterinarians must give them an injection of a sedative. Onset and degree of sedation can be unreliable with orally administered medications, and injections can be difficult to administer to already stressed cats and are painful. This can make these options less desirable for owners and can lead to non-compliance and fewer veterinary visits. A medication that only requires placement into the oral cavity may help to reduce stress and injuries to both patients and owners, and provide easily administered sedation for minor procedures.
Given the sedative effects previously noted in horses, dogs and ferrets, the use of OTM detomidine gel for sedation in cats is a reasonable option for a sedative drug and route of administration that would help veterinarians perform minor procedures, such as examinations and venipuncture, to otherwise highly stressed cats. The current study’s aims were to determine the sedative and hemodynamic effects of OTM detomidine gel in cats and to describe its pharmacokinetics in healthy cats. We hypothesized that OTM detomidine gel would produce sedation and result in transient bradycardia and hypertension but would have no detectable effects on ventilation or blood gas variables, and would exhibit systemic absorption with a short half-life in cats.
Materials and methods
Animals
Eight healthy adult domestic shorthair research colony cats were used in this study: three females and five male (sterilized) with a mean ± SD weight and age of 4.18 ± 0.75 kg and 8.9 ± 1.0 years, respectively. This study was approved by the University of Tennessee Institutional Animal Care and Use Committee (protocol number 2346-0515). All cats were considered healthy based on previous complete blood count and serum biochemical analysis, as well as physical examination on the day of the study. Food was withheld from the cats for 12 h prior to each study day, but was returned to the cats after they had fully recovered from any treatment-induced sedative effects. Cats were housed in standard kennel housing. Each cat was provided a litter box, as well as a cardboard box for privacy. Water was available ad libitum aside from times of sedation and food was offered twice daily, with small treats of canned food following recovery from sedation each day. The cats were monitored by direct supervision from approximately 7 am to 5 pm by the investigators. They were rechecked at 10 pm on each evening of study dates.
Treatments
Cats were administered detomidine hydrochloride as a commercially available gel formulation (Dormosedan Gel) in the buccal pouch at a dose of 4 mg/m2. Oral cavity pH was measured in each of the cats on the first day of the experiment using a strip of pH paper (pH Hydrion; Micro Essential Laboratory) placed against the buccal surface of the cats, prior to any drug administration.
Sample collection
One day prior to drug administration, cats were anesthetized using isoflurane in 100% oxygen via mask for the placement of a medial saphenous vein sampling catheter (MILACATH Guidewire, Single Lumen, 19 Ga [3.5 Fr] × 25 cm [10”]). The catheter was aseptically placed to facilitate blood collection at multiple time points. Blood samples (approximately 1.25 ml) were obtained through this catheter at baseline (immediately prior to drug administration) and at 15, 30, 45 and 60 mins, followed by 2, 4, 6, 8 and 12 h after dosing with OTM detomidine gel. Blood samples were immediately placed in lithium heparin tubes, placed on ice and centrifuged within 60 mins of collection at 1509 × g for 10 mins. Plasma was collected from these samples and stored at –80ºC until analysis by liquid chromatography–tandem mass spectrometry (LC-MS).
Heart rate was monitored using direct thoracic auscultation and digital palpation. Indirect blood pressure measurements, obtained during the same time period as blood samples, were measured using a Doppler ultrasonic probe (Parks Medical Electronics Model 811-B Doppler and 9 MHz ultrasonic probe) with a size 3 cuff on the right forelimb. Five readings were taken over the course of 1 min. Of these readings, the highest and lowest readings were removed and the mean of the remaining three readings were used as the blood pressure. 13
Blood samples taken at baseline and 60 mins after OTM detomidine administration were used for venous blood gas analysis (VetScan i-STAT 1 Analyzer point of care monitor and CG8 + cartridges [Abaxis]) and measurement of blood lactate (Nova Biomedical Lactate Plus). Cats were observed throughout the entire study period for adverse events, such as emesis, diarrhea, hypotension, anemia and dysrexia.
Sedation scoring
Sedation scoring was performed at every blood sampling time point with previously used numerical rating scale, where a score of 0 = no sedation, 1 = cat can stand but is wobbly/staggers when walking, 2 = sternal recum-bency, 3 = recumbent but able to lift head and 4 = fast asleep with no response to electric hair clippers.14,15 The sedation score was performed before any other data or samples were collected.
Plasma detomidine analysis
Detomidine was quantified in feline plasma by LC-MS with a method previously validated for equine and canine plasma.5,7 A partial validation was performed with feline plasma used as a matrix. The response for detomidine was linear and gave a correlation coefficient (R 2 ) of ⩾0.99. Interday and intraday precision and accuracy of the assay were determined by assaying detomidine quality control samples in replicates (n = 6). Accuracy (% nominal concentration) was 93%, 95% and 96% for 0.3, 5 and 45.0 ng/ml, respectively. Precision (% relative SD) was 8%, 4% and 1% for 0.3, 5 and 45.0 ng/ml, respectively. The assay was optimized to provide a limit of quantitation of 0.1 ng/ml and a limit of detection of 0.05 ng/ml.
Pharmacokinetic analysis
Plasma pharmacokinetics of detomidine following OTM administration in cats was analyzed with a commercial software program (WinNonlin 6.3; Certara). Only data points (plasma concentrations) equal to or above the limit of quantitation (0.1 ng/ml) were included in the analysis. A compartmental approach was used, and the model was chosen on the basis of visual inspection of observed vs predicted data, residuals plot and Aikake’s information criteria. 16 Standard compartmental equations were used to generate pharmacokinetic parameter estimates. 17
Statistical analysis
All data were tested for normality with a Shapiro–Wilk test and were presented as mean ± SD (for normally distributed data). ANOVA was performed on normally distributed data, while an ANOVA on ranks was performed on any non-parametric data. Pharmacokinetic parameter estimates are presented as geometric mean and coefficient of variation. A Bonferroni or Dunnet’s post-hoc test was performed for multiple comparisons when significance was noted. The alpha value was set at P ⩽0.05. Blood gas variables were compared from baseline to 60 mins using either a paired t-test or a Wilcoxon signed rank test. Categorical data, including sedation scores, were compared between groups by means of a Mann–Whitney U-test. All analyses were carried out with a commercially available statistical program (Graphpad Prism 6).
Results
Eight cats were enrolled in this study; however, owing to complications with venous access, one cat was excluded prior to any drug administration. The remaining seven cats completed the study. The mean ± SD dose and volume of detomidine gel administered to the cats was 251.0 ± 13.8 µg/kg and 0.135 ± 0.015 ml, respectively. The average oral pH was 8.36 ± 0.24.
Plasma concentrations of detomidine were detectable at the first post-drug administration sampling time (15 mins) and reached maximum levels of approximately 11 ng/ml at 45 mins following drug administration. Concentrations of detomidine were not detectable at 360 mins after administration in 3/7 cats. Only one cat had detectable levels at 480 mins following drug administration (Figure 1). The remainder of the pharmacokinetic results are presented in Table 1.
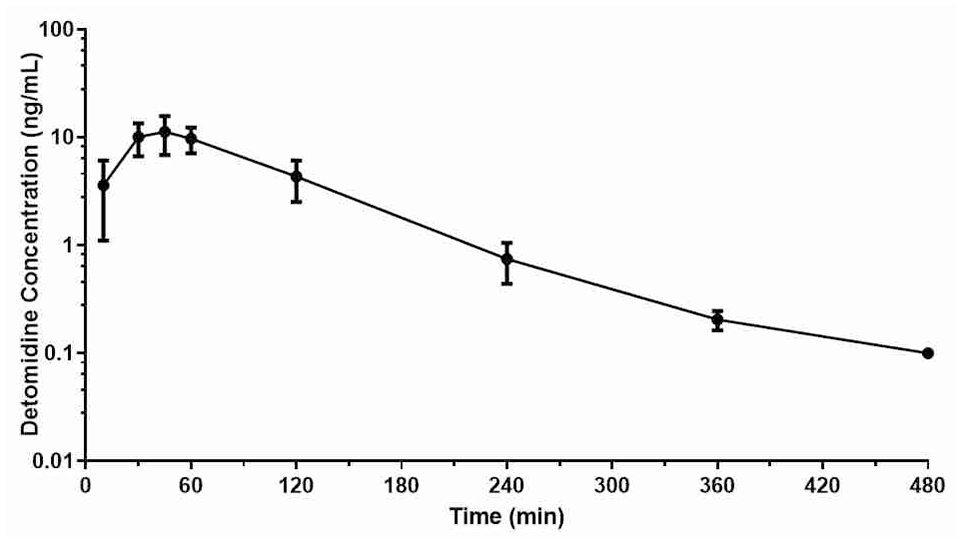
Mean ± SD of plasma concentrations of detomidine in seven cats following buccal administration of commercially available formulation at a dose of 4 mg/m2
Compartmental pharmacokinetic parameters following oral transmucosal administration of 4 mg/m2 detomidine gel to healthy cats (n = 7)

AUC = area under the curve; K01 and K10 = absorption and elimination rate constants; T½ = half-life; Cmax = maximum concentration; Tmax = time to maximum concentration; Vss = steady state volume of distribution; Cl = total body clearance; F = bioavailability
All cats exhibited increased sedation scores (Figure 2) following buccal detomidine gel administration. Sedation scores were significantly (P <0.001) increased at 30 mins after OTM detomidine, after which time the cats’ scores began to return to baseline. By 120 mins, all sedation scores were 0 (no sedation). Doppler blood pressure was significantly lower at the 60-min time point (103 ± 23 mmHg) when compared with baseline (139 ± 15 mmHg; P = 0.023). Doppler blood pressure varied throughout the study, and individual measurements ranged from 78 to 183 mmHg. Heart rate was significantly lower compared with baseline (206 beats per min [bpm]) from 15 to 60 mins post-drug administration (P <0.001; Figure 3). Respiratory rates were not significantly different compared with baseline, and were highly variable during the experimental period.

Sedation scores of seven individual healthy cats following oral transmucosal detomidine gel administration.
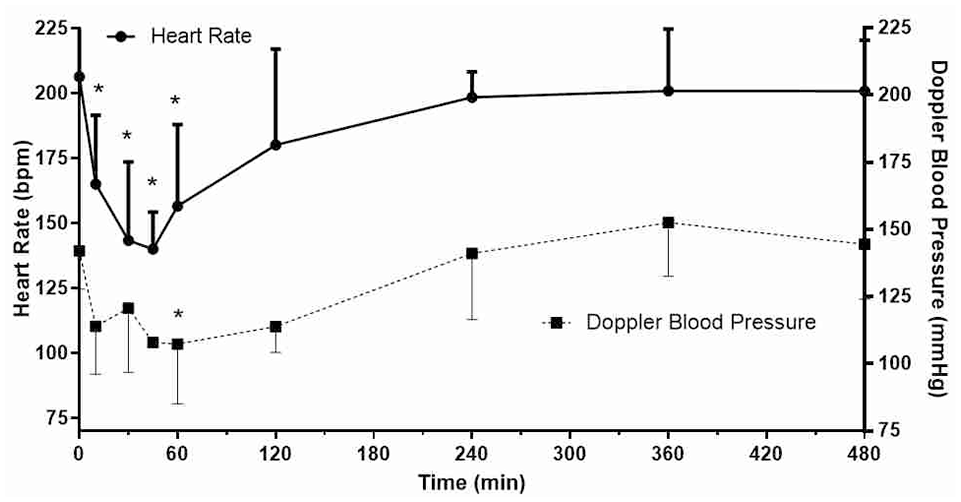
Heart rate (mean ± SD) and Doppler blood pressure (mean ± SD) in cats (n = 7) at baseline and following oral transmucosal detomidine gel administration.
Venous blood data collected at baseline and 60 mins post-detomidine administration are reported in Table 2. Statistically significant differences were found in normal mixed venous oxygen tension (PvO2), which decreased from a baseline value of 36.1 ± 3.9 mmHg to 28.7 ± 4.2 mmHg at 60 mins (P = 0.016) and blood glucose, which increased from 101 ± 12.0 mg/dl to 168 ± 27.3 mg/dl at 60 mins following administration (P = 0.004). The remainder of the hematological and blood gas values were unremarkable and within normal reference intervals (RIs) for cats. 18
Venous blood gas variables prior to sedation, and 60 mins after 4 mg/m2 oral transmucosal detomidine gel in healthy cats (n = 7)

Data are mean ± SD
Significantly different (P ⩽0.05) from baseline
PvO2 = normal mixed venous oxygen tension; PvCO2 = partial venous carbon dioxide tension; HCO3 = bicarbonate; TCO2 = total carbon dioxide; Na = sodium; K = potassium; iCal = ionized calcium
Following detomidine administration, all cats displayed signs suggestive of nausea, which included hypersalivation and lip smacking; 7/7 cats vomited, which occurred within 2 mins of drug administration. By 15 mins post-dose, vomiting was no longer noted. All episodes of vomiting occurred before the onset of sedation in all cats. No other adverse effects were observed.
Discussion
There are few published reports on the use of detomidine in cats,19,20 and heretofore there are no studies on the use of OTM detomidine gel in cats. While it is more common for clinicians to use other injectable α2 agonist drugs such as dexmedetomidine in cats, the concentrated gel formulation of detomidine is attractive as it allows for small volume OTM-administered sedation in this species. Detomidine is a less selective and less potent α2 agonist than dexmedetomidine and is most commonly used in horses as a sedative and analgesic.3,5 Detomidine gel has been used extra-label for successful sedation in dogs and ferrets.8,10 Some cats can be challenging to handle for examinations and minor procedures, and injectable sedation can be painful; thus, reliable transmucosal or oral sedative options are desirable.
The pharmacokinetic profile following OTM delivery (buccal or sublingual) of detomidine has been previously described in dogs and horses,5,7,21 and reported similar results as the current study in cats. Detomidine was absorbed quickly into systemic circulation, but the absorption rate was highly variable among cats, which is to be expected given the extravascular route of administration and sparse sampling times during the absorption phase. Additionally, the drug was apparently cleared rapidly, given the short terminal half-life of 47 mins and inability to detect the drug in most cats after 480 mins. To determine true drug clearance, an intravenous route of administration must be used, which was not performed in this study; thus, clearance is reported per fraction absorbed. Bioavailability is also not known, but based on studies in other species with the gel and with other injectable α2 agonists administered transmucosally to cats, one would anticipate bioavailability to be low (ie, <30%).22,23 Despite these limitations, we demonstrated both systemic absorption with mild sedative and hemodynamic effects at a dose of 4 mg/m2 (approximately 0.24 mg/kg) in this study, which was chosen based on pilot work performed by the authors.
All cats exhibited varying levels of sedation following detomidine administration, which was statistically significant compared with baseline at 30 mins after administration. At this time, plasma concentrations of detomidine were approximately 10 ng/ml. No cat scored a 4 (fast asleep with no response to electric hair clippers), and only one cat had a score of 3 (recumbent but able to lift head), which occurred at 15 mins post-dose. Sedation scores >0 were recorded in some cats up to 60 mins after administration, but by 120 mins all cats had scores of 0 (no sedation); thus, sedative effects are generally short-acting and mild, allowing a veterinarian to perform a physical examination while mitigating undue stress on the cat and without concern for oversedation. We used a simple numerical score of 0–4 for ease owing to concurrent pharmacokinetic sampling and hemodynamic measurements; thus, we could not perform more extensive scoring of sedation, which might be considered a limitation of our study. In addition, our observer was not masked to treatment, which could have influenced the results.
Blood pressure and heart rate were both affected by OTM detomidine gel; a significant decrease in Doppler blood pressure vs baseline was observed only at 60 mins after administration, which corresponded to detomidine plasma concentrations of 9.7 ng/ml. Heart rate was significantly decreased from baseline for 15–45 mins after the administration of detomidine, with the lowest heart rate being 140 bpm (at 45 mins; detomidine plasma concentration 11.3 ng/ml). Similar findings were reported by Grove and Ramsey, 19 where bradycardia was present but no differences in respiratory rates, rectal temperature or peripheral capillary oxygen saturation (SpO2; the latter two were not measured in our study). While Doppler blood pressure was decreased from baseline, most values were considered normal for cats. 13 In two cats, hypotension was observed at 60 mins (mean Doppler blood pressure was recorded as 78 and 79 mmHg, respectively). No intervention occurred, and at the next recording the blood pressure had increased to 99 in both cats. There was no correlation between sedation and degree of hypotension in this study. Lactate remained unchanged (1.0) in both of these cats. The decrease in Doppler blood pressure was most likely secondary to decreased heart rate. If a clinician is concerned about pre-existing or prolonged hypotension, detomidine gel should probably be avoided as a sedation option if possible. Similarly, if clinicians are specifically evaluating a cat with cardiovascular disease or there is a concern for hypertension secondary to other conditions (hyperthyroidism or kidney disease), detomidine gel might not be a desirable sedative choice due to its effects on blood pressure and heart rate. Based on the findings in this study, blood pressure should be monitored in cats sedated with OTM detomidine gel.
The only changes in venous blood gas variables were decreased PvO2 and increased blood glucose. The normal RI for PvO2 is 33.9–56.3 mmHg in healthy, awake cats, 18 and our baseline value was 36 mmHg, which is near the low end of this reported RI. RIs differ between methodology (I-stat vs other) and might account for differences in these values. Nevertheless, PvO2 is not a useful variable to assess oxygenation and many factors can influence its value. As all cats recovered uneventfully, we believe the decreases in PvO2, while statistically significant, are not clinically significant. Future studies should include measurement of arterial blood gas variables following sedation with detomidine gel, which would offer a better indication of any effects on oxygenation. Hyperglycemia is well documented following α2 agonist administration in cats, 24 and occurs as a result of activity at pancreatic beta cells to transiently inhibit insulin production. The degree of hyperglycemia was slightly outwith the RI (78–158 mg/dl) 18 and clinicians should be aware that transient mild hyperglycemia following detomidine gel administration is expected. We assessed venous samples at only 60 mins post-administration, which did not coincide with maximum levels of sedation or hemodynamic effects, which could be considered a limitation. However, no long-lasting systemic effects of sedation or detomidine gel on the majority of venous blood gas variables were observed.
Emesis occurred in 100% of the cats following OTM detomidine administration. Emesis is commonly reported after the administration of α2 agonists to cats; in fact, xylazine is the drug of choice to induce vomiting in cats.19,25,26 Similar to xylazine, emesis occurred before the sedative effects, but, nevertheless, a client or clinician should expect vomiting and potential sequelae (esophageal irritation or aspiration). The cats in our study were fasted, which would be recommended prior to using this drug for sedation. A clinician could also consider the use of OTM detomidine gel to induce vomiting in cats depending on the availability of other options. Dose-dependent emesis is a known side effect in cats following the administration of α2 agonists. 25 , 26 Previous studies have reported the emetogenic effects of these drugs can be blocked by reversal of the α2 agonist, and atipamezole is commercially available as a reversal agent labeled for use in dogs, but is used extra-label in cats. 27 We did not assess reversal in this study. While nausea and vomiting might be ameliorated with reversal, sedation would also be reversed, thus negating the purpose of medicating. Additional complications such as aspiration pneumonia were not reported in these previous studies but would certainly be a concern in cats that experience rapid-onset sedation. All cats in the current study were noted to have emesis but, similarly to previous studies, had no long-term side effects following these incidents.
Ease of OTM administration was not specifically assessed in this study, although 6/7 cats were subjectively easy to medicate. One cat was extremely difficult to medicate (resistant to gentle restraint for administration; attempted to bite investigators), which is an important clinical consideration for the use of this product in cats. While owners could be taught to administer the gel (in instances where the cat does not exhibit aggressive or fearful behaviors toward owners), it may be prudent for veterinary professionals to administer the gel, or for owners to administer under veterinary supervision (ie, in an examination room) owing to the previously discussed side effects of emesis and possible hypotension and bradycardia.
A potential limitation of this study was the use of purpose-bred cats from a dedicated research colony. These cats may have been more compliant to handling, which could have affected the sedation scores. Future studies should involve a larger number of cats, which would ideally include healthy, client-owned cats enrolled in a double-masked, placebo-controlled clinical trial, which would be more representative of the intended patient population and provide better assessment of the pharmacodynamic efficacy of this this medication.
Conclusions
Detomidine gel produced moderate, short-acting sedation in healthy cats in this study, but transient hyperglycemia, mild bradycardia, possible hypotension and emesis should be expected after administration of OTM detomidine gel. Further studies are warranted to assess its safety and feasibility for use in client-owned or debilitated cats, or prior to general anesthesia.
Footnotes
Author note
This study was presented, in part, in abstract form, at the 2016 International Veterinary Emergency & Critical Care and American College of Veterinary Anesthesia and Analgesia Annual Symposium, which was published as an abstract in Veterinary Anaesthesia and Analgesia in 2017, and has been reused with permission from the publisher.
Conflict of interest
While this study was funded solely by the Department of Molecular Biomedical Sciences at North Carolina State University, KMM has received research grants, speaking honoraria and consulting fees from several sources, including Zoetis, Bayer, Piedmont Animal Health, Innovate Biopharmaceuticals, Jurox and Ellevet.
Funding
This study was financially supported by the Department of Molecular Biomedical Sciences at North Carolina State University.
Ethical approval
This work involved the use of experimental animals. The study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
