Abstract
Objectives
Leishmaniosis is a vector-borne disease and in European countries is caused by Leishmania infantum. Cats are considered secondary reservoirs of the infection in endemic areas. The objective of this retrospective study is to describe the clinical findings, diagnosis, treatment and outcome of feline leishmaniosis (FeL) in 16 cats in Spain.
Methods
Medical records of cats diagnosed with leishmaniosis were retrospectively reviewed for cases that met the following inclusion criteria: identification of Leishmania organisms and/or DNA on cytological and/or histological specimens and/or a high anti-Leishmania antibody titre, compatible clinical findings and pathological abnormalities.
Results
Sixteen cats met the inclusion criteria, all of which were living in areas endemic for canine leishmaniosis. Systemic signs were present in 11 cases (68.8%). The most common clinical signs on presentation included cutaneous lesions in 12 cats (75%), ocular disease in six cats (37.5%) and anorexia in six cats (37.5%). A polyclonal gammopathy was noted in 12 cats (85.7%). Non-regenerative anaemia and renal abnormalities were present in six (37.5%) and five patients (31.3%), respectively. In nine cats (56.3%), immunosuppressive conditions/comorbidities were identified. The diagnosis was made in eight of the cats (50%) by cytology, but a combination of diagnostic tests was needed for definitive diagnosis in the remaining patients. Twelve cats (75%) were treated specifically for leishmaniosis. Five of the 12 cats (41.7%) did not improve with treatment. The median survival time in the group of patients treated specifically for leishmaniosis was 17 months. Median survival of patients treated with concomitant diseases was 13 months vs 41 months in those without, although this was not statistically significant (P = 0.557).
Conclusions and relevance
Presentation of FeL appears to be similar to canine leishmaniosis but with some specific features: ulcerative and nodular skin lesions are the predominant cutaneous signs; cats with immunosuppressive conditions or coexisting diseases were more commonly present than typically seen in dogs (mainly feline immunodeficiency virus). A combination of diagnostic tests may be needed for definitive diagnosis.
Introduction
Leishmaniosis is a zoonotic parasitic disease caused by Leishmania species worldwide. Leishmania infantum is the protozoan responsible for this disease in European countries and it is transmitted by a vector of the genus Phlebotomus.1–3 Dogs are considered the main reservoir host and cats are considered secondary reservoirs of this disease.1,4–8
Since 1977, 53 natural cases of feline leishmaniosis (FeL) have been described worldwide.9–16 Most cases of FeL have been reported in countries in the Mediterranean basin, although it has also been reported in southern USA, Central and South America, Brazil and Iran.4,17–22 Different studies have identified that the prevalence of Leishmania infection in cats in endemic areas varies from 0% to 68.5%.4,23–26
Most infected cats are asymptomatic. Common clinical manifestations of the disease previously reported involve cutaneous and mucocutaneous lesions, with or without visceral signs.4,9–13,24,27–35 Nodules and ulcerations are the most common cutaneous and mucocutaneous findings. 4 Ocular lesions have been described in a third of cases.4,10,12,14,36–40
To the authors’ knowledge, the published literature on FeL includes case reports and a few retrospective case series on clinical signs and skin lesions. The objective of this study was to describe the clinical findings, diagnosis, treatment and outcome in 16 cats with leishmaniosis diagnosed in Spain.
Materials and methods
Cats with a diagnosis of leishmaniosis were reviewed at the Universitat Autònoma de Barcelona Hospital (UAB) between 2000 and 2015. More cases were recruited from private practices over Spain via an electronic survey through the Small Animal Spanish Veterinary Association (AVEPA) feline medicine working group and online forums (dermatology, feline medicine and internal medicine). Sixteen cats were enrolled: six cats were diagnosed at UAB, four cats at Ars Veterinaria Hospital (Barcelona) and the other six cats at different practices in the Barcelona area, Mallorca and Valencia. Inclusion criteria for this study were as follows: identification of Leishmania organisms and/or DNA on cytological and/or histological specimens and/or a high anti-Leishmania species antibody titre, along with compatible clinical findings and pathological abnormalities. Cats with only positive antibody titres and lacking additional clinical details were excluded. Sixteen cases met the criteria for case selection. Data collected included signalment, lifestyle, clinical signs, physical examination findings, clinicopathological abnormalities, diagnostic tests, retroviral status, concurrent diseases and/or immunosuppressive conditions, specific treatment, treatment response, outcome and survival.
Survival data and curves were generated by the Kaplan–Meier method, and survival plots were compared by use of the log-rank test. Kaplan–Meier survival curve construction comparing cats treated with concomitant diseases and those without concomitant disease was performed. For this analysis, any cat that died or was euthanased was classified as dead, and any cat still alive at the time it was lost to follow-up was censored.
Statistical analyses were performed with SPSS Statistics for Macintosh version 25.0 (IBM) and descriptive statistics were used to report baseline data. P values <0.05 were considered to be significant.
Three cases included in this study had been previously published as case reports, one in Veterinary Opthalmology, 37 one in Clínica Veterinaria de Pequeños Animales (AVEPA journal) 9 and the final as a poster at the Southern European Veterinary Conference in 2016. 41
Results
Seven cats were male (all neutered), eight were female (seven neutered and one intact) and in one case sex was not recorded. Fourteen cats were domestic shorthairs and two were Siamese. Age at diagnosis was known for 13 cats; mean age was 7 years (range 3–21 years). Seven cats were outdoor, one was indoor and the lifestyle was not known in the remaining cases.
Systemic signs were present in 11/16 cats (68.8%) (Table 1). Seven of these 11 cats also had cutaneous signs (63.6%).
Clinical abnormalities reported for 16 cats at the time of diagnosis

Cutaneous lesions were present in 12/16 cats (75%) (Figures 1 and 2). Skin disease without systemic signs was seen in 5/16 cases (31.3%). The skin lesions observed included the following: nodules in the facial area and extremities (one cat); nodules solely in the facial area (three cats); ulcerated nodules in the extremities (one cat); ulcerative lesions affecting the paws (two cats); nodule on one footpad (one cat); ulcers in peri-ocular area and pressure points (one cat); single ulcer on the bridge of the nose (one cat); multifocal ulcers over trunk, face and extremities (one cat); exfoliative dermatitis (three cats); and focal ventral alopecia (three cats).
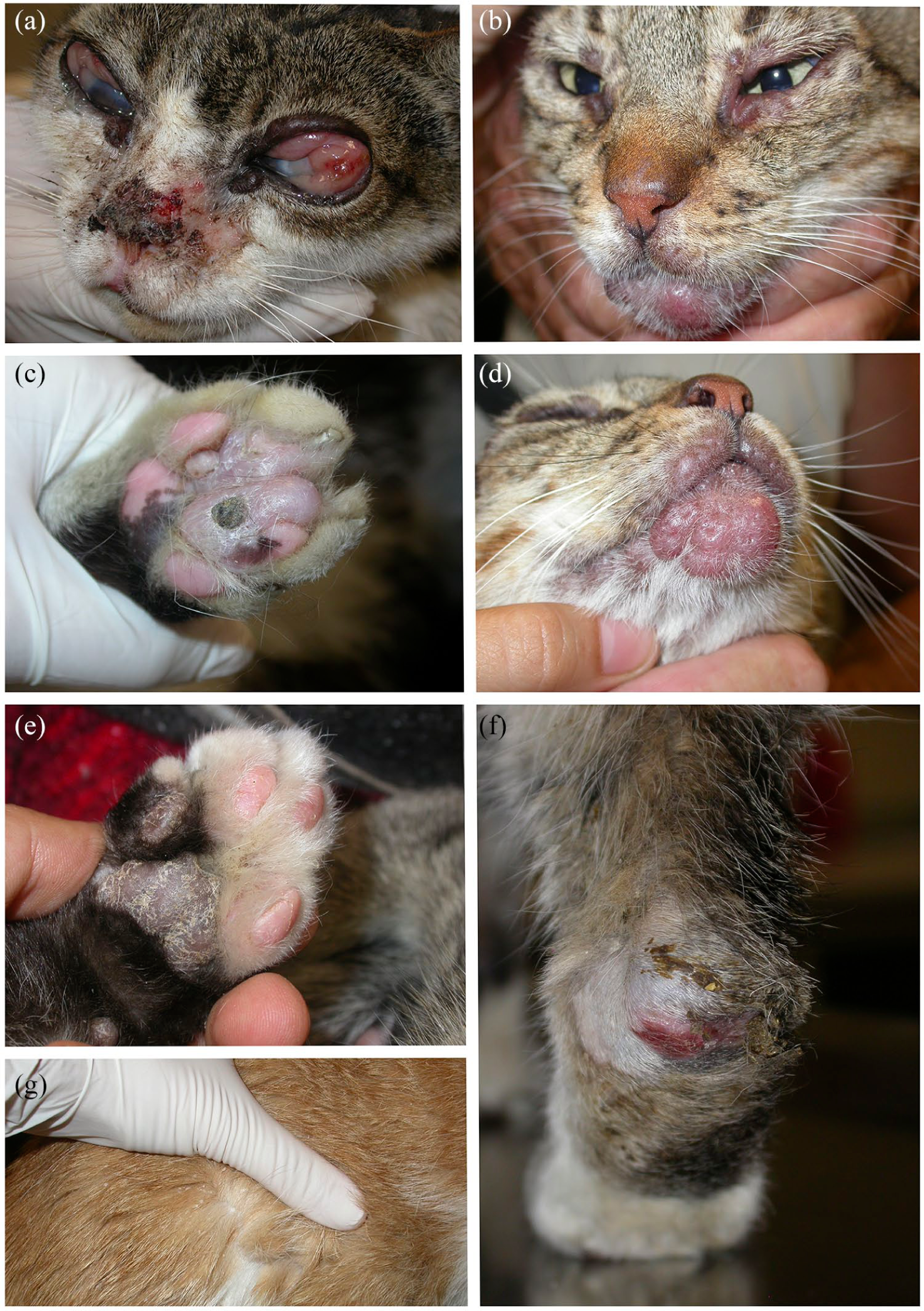
Skin and ocular lesions in patients with feline leishmaniosis. (a) Patient 7. Papules on the eyelids. Corneal oedema and severe chemosis and proliferative conjunctivitis. Ulcers and crusts on the bridge of the nose and dorsal planum nasale. Focal alopecia. (b) Patient 6. Papules on the eyelids and chin. (c) Patient 8. Nodule with a central crust with an underlying ulcer on a digital footpad. (d) Patient 6. Papules on the dorsal lips and a plaque on the chin. (e) Patient 6. Mild footpad hyperkeratosis. (f) Patient 7. Pressure point ulcer, alopecia and crusts on the hock. (g) Patient 4. Generalised scaling

Skin lesions in patient 1: (a) exophytic nodule on the chin; (b) nodule on the left carpal area; and (c) ulcer on the left metatarsal footpad
Ocular disease was present in 6/16 (37.5%) cats. Systemic signs were seen in 4/6 cats with concurrent ocular signs. Corneal oedema and panuveitis were present in three cats, each presenting additional problems, including: melting keratitis and corneal perforation (one cat); chorioretinitis alongside exophthalmus (one cat); and chemosis with proliferative conjunctivitis (one cat). Chemosis, proliferative conjunctivitis and palpebral nodules with no other lesions were present in one cat. Conjunctival and palpebral nodules were seen in another cat, which also had multiple oral nodules and glossitis. Conjunctivitis and uveitis were present in one cat.
Stomatitis was present in 3/16 cats (18.8%), one of them with oral dysphagia. Glossitis was identified in 2/16 cats (12.5%), one cat had both stomatitis and glossitis, and the other cat had multiple oral nodules. These two cats presented oral dysphagia. Two cats (one with stomatitis and one with both glossitis and stomatitis) had been previously treated with long-term glucocorticoids, but the remaining three cats had not received any recent glucocorticoid treatment. Hepatomegaly was present in 2/16 cats (12.5%), splenomegaly in 1/16 cats (6.3%) and renomegaly in 1/16 cats (6.3%). One cat presented with neurological signs suspected to be secondary to diffuse central nervous system disease.
Complete blood count and serum biochemistry were available for all cats (Table 2). No abnormalities were found in 4/16 (25%) cats. Platelet counts were normal in all cases. Polyclonal gammopathy was present in 12/14 cats.
Clinicopathological abnormalities in the 16 cats infected with Leishmania species
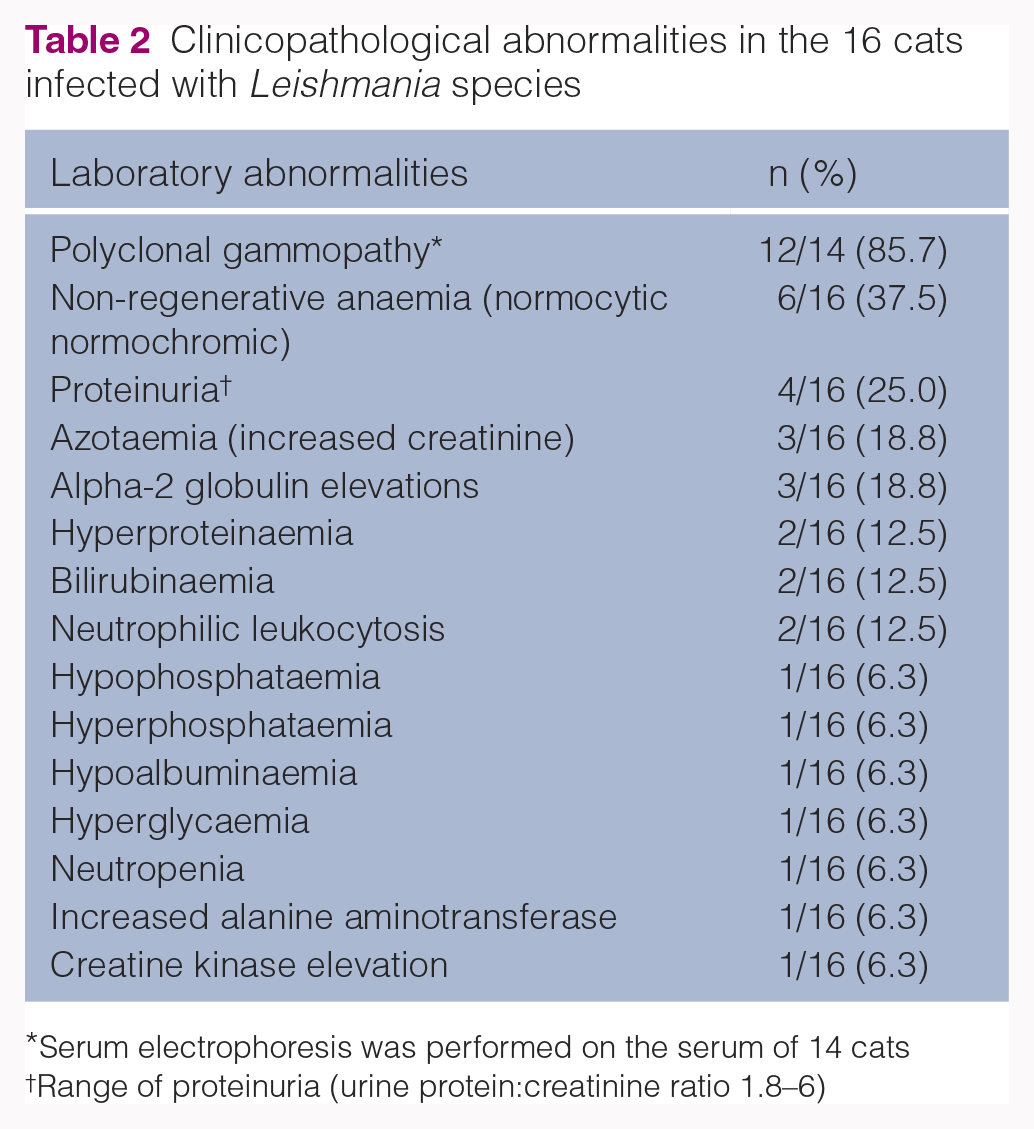
Serum electrophoresis was performed on the serum of 14 cats
Range of proteinuria (urine protein:creatinine ratio 1.8–6)
In 9/16 (56.3%) cats, immunosuppressive conditions or coexisting diseases were identified (Table 3). Feline leukaemia virus (FeLV) and feline immunodeficiency virus (FIV) testing (in-house ELISA to detect antibodies against FIV or FeLV antigen in blood) was conducted in all but two cats. Five of 14 (35.7%) cats were FIV positive; two of them were suspected to be in an advanced state of immunosuppression due to the presence of infectious or opportunistic diseases (Table 3). All cats tested negative for FeLV. Four of 16 cats were receiving high doses and/or long-term glucocorticoids; one for chronic bronchial disease (also FIV positive), which received oral and inhaled glucocorticoids; and three for chronic gingivostomatitis, which were receiving a combination of subcutaneous and oral glucocorticoids (see specific doses in Table 3). One of these three was also FIV positive. One of these cats developed type 2 diabetes mellitus secondary to oral and subcutaneous glucocorticoids. One cat was pregnant at the time of diagnosis and one cat was 21 years old at the time of diagnosis; both conditions may be associated with immunosuppression.
Signalment, concomitant diseases, diagnosis, specific treatment for leishmaniosis and outcomes of this group of patients
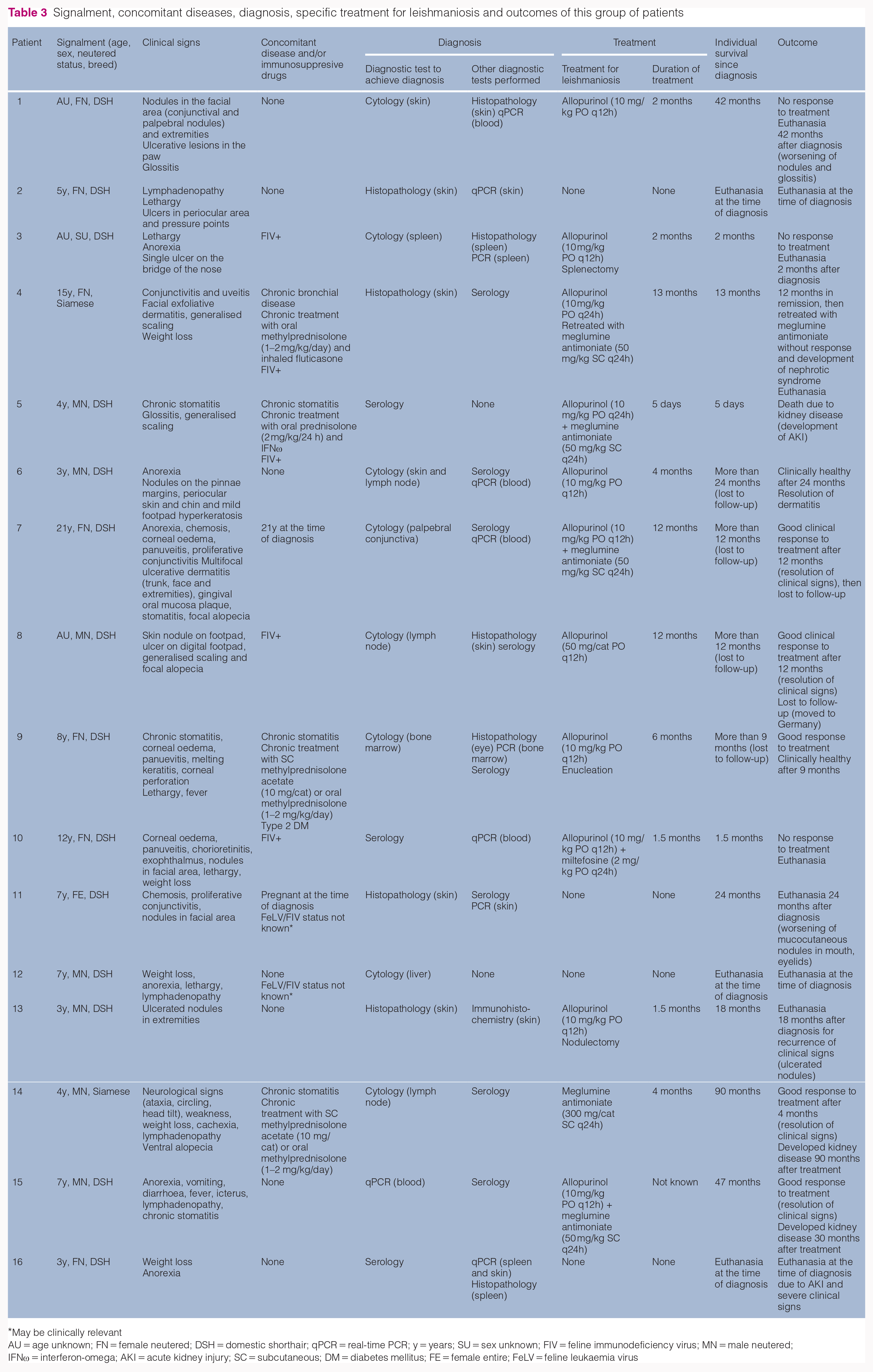
May be clinically relevant
AU = age unknown; FN = female neutered; DSH = domestic shorthair; qPCR = real-time PCR; y = years; SU = sex unknown; FIV = feline immunodeficiency virus; MN = male neutered; IFNω = interferon-omega; AKI = acute kidney injury; SC = subcutaneous; DM = diabetes mellitus; FE = female entire; FeLV = feline leukaemia virus
Diagnosis was obtained in 8/16 (50%) cats by cytology of the following: skin lesions (n = 1), lymph nodes (n = 2), skin lesion and lymph node (n = 1), palpebral conjunctiva (n = 1), bone marrow (n = 1), spleen (n = 1) and liver (n = 1). Histopathology was used to obtain a diagnosis in four cats (skin) and to confirm cytology results in five cats (two skin, two spleen and one eye). Leishmania species antibody titres were performed in 11/16 cats (immunofluorescence or ELISA), with high positive antibody titres in six cats, medium or low positive titres in four cats and negative titres in one cat. Antibody titres were the principal test to obtain a diagnosis in 3/11 cats and were used to confirm a diagnosis in 8/11 cats that had previous histological or cytological detection of Leishmania species. Immunohistochemistry was used in one cat to detect Leishmania species in a skin sample. PCR was performed in 10/16 cats in different tissues (Table 3). In three cases, qualitative PCR was performed and in seven cases a quantitative real-time PCR (TaqMan assay) was performed. PCR results were positive in all cases. Only in one case was blood PCR the test used to obtain the diagnosis (Table 3).
Four of 16 cats died without attempting any specific treatment (three were euthanased at the time of diagnosis and one cat lived for 2 years and was euthanased owing to worsening of cutaneous nodules). The remaining 12 cats were treated specifically for leishmaniosis and three of them had surgical interventions as part of their treatment (Table 3, Figure 3).

Treatment and outcome of the 16 cats in the study
Five of 12 cats did not improve with the treatment and three of them died or were euthanased during the initial treatment period (Table 3 and Figure 3).
The median survival time in the group of patients treated specifically for leishmaniosis was 17 months. In the treatment group, a comparison of median survival time was made between cats with concomitant diseases or known immunosuppression and those cats without any concomitant diseases or immunosuppression (Figure 4). Median survival of the patients treated with concomitant diseases was 13 months vs 41 months for the treated patients without concomitant diseases, although this was not statistically significant (P = 0.557).
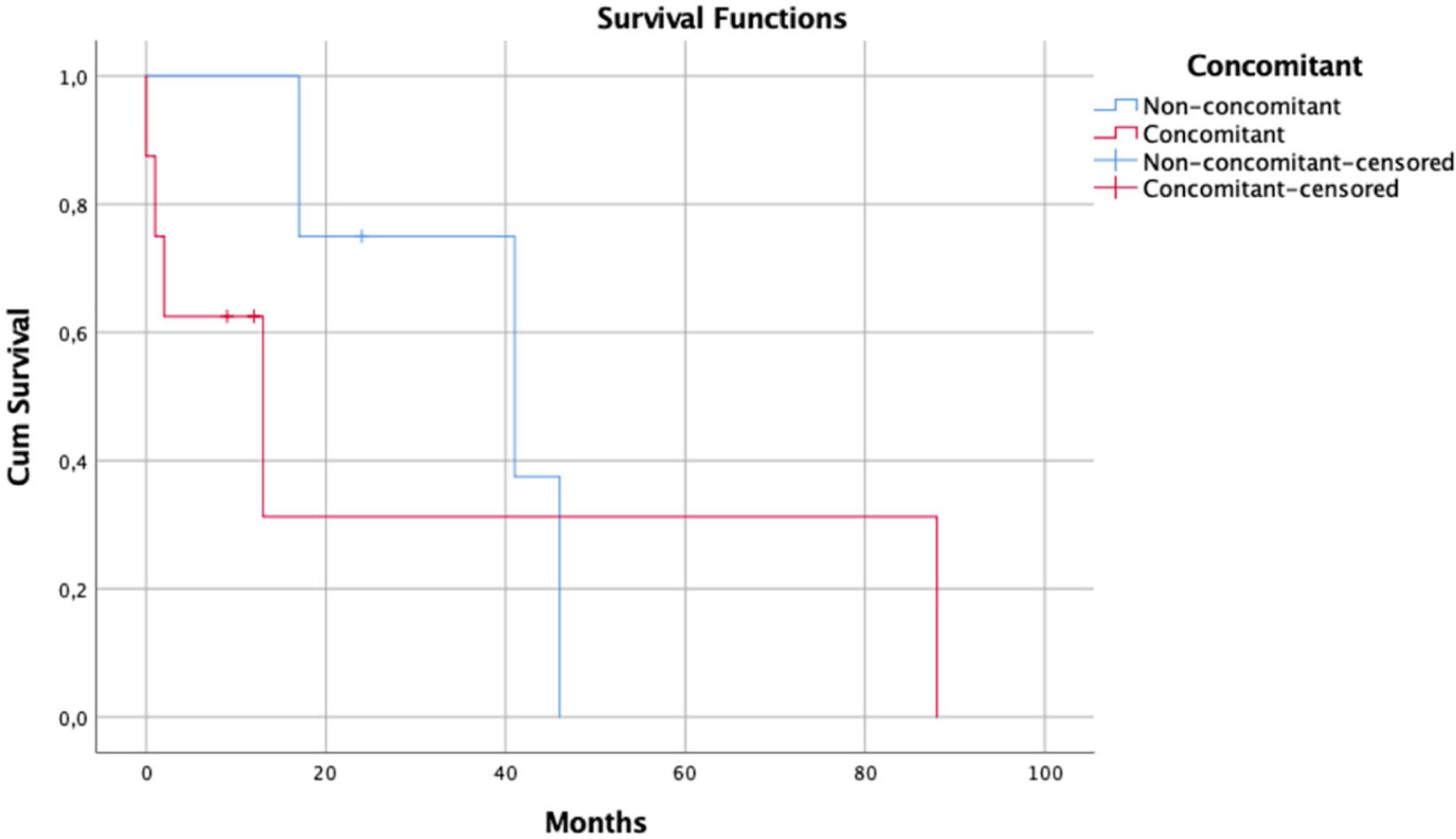
Kaplan–Meier survival curve for cats treated specifically for leptospirosis, comparing those with concomitant diseases or immunosuppression (red line) and those with no concomitant diseases (blue)
In the group of patients without specific treatment (four cats), only one cat was not euthanased at the time of diagnosis, so median survival time could not be calculated for these patients.
Discussion
The most common clinical signs reported in this group included skin or mucocutaneous lesions in 75% of patients. Of these, 50% of cats also presented with systemic signs (including anorexia, weight loss and lethargy). It is also noteworthy that most of the ocular signs in this study appeared with systemic signs (66.7%). Our results are similar to previously reported cases, in which some cats showed only dermatological lesions,4,26–30 while others demonstrated a combination of skin or ocular lesions with systemic signs.9–13,24,31–35
Based on the combined findings of this study, in conjunction with previously published cases, FeL seems to be characterised predominantly by cutaneous lesions, including cases of nodular, alopecic, scaling and ulcerative dermatitis.24,29,31,33,39,42–47 This study identified that the most common presenting sign was ulcerative dermatitis, followed by nodular dermatitis, exfoliative dermatitis and alopecia. The presentation of leishmaniosis in cats may differ from the canine presentation based on our data and also based on previous literature.24,29,31,33,39,42–49 In canine leishmaniosis, exfoliative dermatitis is the most common presentation followed by ulcerative dermatitis and nodular dermatitis.48,50–73
Each presentation may reflect a different host–parasite relationship.48,56,74 In dogs, susceptibility to infection and disease progression is mediated predominantly by a non-protective T helper 2- and a T helper 1-oriented immune response, which stimulates phagocytosis by macrophages and consequent phagocyte-based parasite intracellular elimination. The association between clinical presentation and immune response has not been fully investigated in feline patients, but species-specific differences in the feline innate and adaptive immune responses might account for the observed lower prevalence of L infantum infection, as well as clinical leishmaniosis, in cats vs dogs. Recently, it has been described that cats from endemic areas are able to activate a cell-mediated adaptive immune response. 75 However, other authors have suggested that the humoral immune response is protective in FeL,1,76 highlighting the potential differences in the immune response between these two species. In some dogs, the simultaneous presence of more than one presentation could be due to other factors, such as skin vulnerability to mechanical trauma and/or to vascular compromise,48,77 and that might be the case in some cats.
In this group of patients, ocular signs were the second most common presentation described, observed in 37.5% of cases. Corneal oedema and panuveitis were the most common reported findings, although chorioretinitis, chemosis, conjunctivitis and melting keratitis were also seen in this population of cats. Ocular manifestations are frequently found in dogs and cats affected by leishmaniosis. Ocular signs occur in 16% to 80% of affected dogs.78,79 Blepharitis, keratoconjunctivitis and anterior uveitis were described as the most frequent signs in canine patients.78,79 Ocular lesions have been reported in approximately one-third of affected cats. 4 In cats, the most common ocular signs observed based on previous case reports were unilateral or bilateral uveitis, with occasionally a pseudotumoral granulomatous pattern and panopthalmitis.12,14,36–38 Blepharitis and conjunctivitis have also been observed in many reports of feline cases.10,39,40 In our case series, results were similar to those published in the literature, with a wide range of different clinical presentations. These results may reflect a considerable variability in the prevalence and type of eye lesions observed in our study, as happens in the canine population. 78
Most of our patients (n = 14/16) presented with cutaneous/mucocutaneous and/or ocular involvement, but a minority (n = 2/16) presented with non-specific clinical signs such as weight loss, anorexia, lethargy and lymphadenopathy. It is also interesting that FeL presentation may range from mild to severe and from acute to chronic.
Information regarding clinicopathological abnormalities seen in FeL is scarce and mainly based on case reports.4,14,49,80 In our case series, a normocytic normochromic non-regenerative anaemia was the most frequent haematological abnormality. Mild-to-severe normocytic normochromic non-regenerative anaemia is also the most frequent haematological abnormality reported in clinical cases.4,49 Hypergammaglobulinaemia was present in 87.5% of the cases in this study; however, it is remarkable that hyperproteinaemia was only present in 12.5% of the cases. Hyperproteinaemia with hypergammaglobulinaemia has also been described in many FeL cases, as also found in canine leishmaniosis.14,49,80 Polyclonal gammopathy occurs in many infectious and inflammatory diseases and is not specific for FeL. Despite this, it may be useful to evaluate the response to treatment or disease status in FeL as in dogs, but this is speculative and has not been evaluated to date. In this case series, the presence of renal disease appears to be similar to dogs. This presentation may be acute or chronic, or it may even appear with the course of the disease.
Proliferative and ulcerative chronic inflammation of the oral mucosa associated with FeL can be included in the list of possible causes of the feline chronic gingivostomatitis syndrome (FCGS). 4 This immune-mediated disease is considered multifactorial and has been associated with infectious and non-infectious agents.81–84 Infectious agents such as FeLV, FIV, feline calicivirus and feline herpesvirus-1, along with a wide variety of bacteria, have been isolated in cats with FCGS. These suspected pathogens can also be present in healthy animals, making it less consistent with a clear causal relationship.81,85–94 It is remarkable that stomatitis has been reported in around a quarter of FeL cases.4,19,48,78,80 In this case series, the prevalence of this syndrome is similar to previous reports. However, owing to the retrospective and multicentric nature of this study, there was no consistent information on the severity of the stomatitis and precise location of the lesions. Leishmaniosis may have been the cause of disease in this group of patients; however, other concomitant diseases could not be ruled out based on our data. Only by histopathological identification of the parasite in oral lesions would it be possible to differentiate between both diseases. Owing to the retrospective nature of this study, this could not be performed.
In previous studies, the clinical disease of FeL has been associated with an impaired immunocompetence due to several factors, including retroviral infections (FIV and FeLV), immunosuppressive treatment and concomitant debilitating diseases such as malignant neoplasia or diabetes mellitus.6,21,25,26,95–101 In our group of patients, possible immunosuppressive conditions (eg, pregnancy, age) or concurrent diseases were identified in 9/16 (56%) cases. In this case series, one-third of the cats were FIV positive, making it the most frequent concomitant disease found. Prevalence rates of FIV and FeLV in the region where these cats live have been previously reported to be 2.6–7.4% and 6.0–8.5%, respectively.102,103 Both FIV and/or FeLV infections have been referred as FeL predisposing factors explained by the ensuing immunosuppression.1,104–106 Supporting studies found a high positivity (~70%) of cats to both leishmaniosis and FIV, 104 and even a statistically significant correlation with FeL and both FIV 99 and FeLV.1,98 However, other studies failed to corroborate this finding.1,6,31,44,101,107–112 The cause–effect relationship between various aetiological and pathogenic factors is not always easy to establish.4,13 Full screening for other pathogens was not performed in all cases, but no other diseases were identified in the study population.
Most diagnostic techniques for Leishmania species infection currently used in cats are the same available for dogs. Diagnosis of FeL is based on serological, cytological, histological or PCR methods. 4 It is remarkable that diagnosis was obtained in 50% of our cases by cytology. This technique represents a rapid, inexpensive and simple procedure to achieve diagnosis in many cases; additionally, it is a highly specific and non-invasive technique. However, histopathology was the diagnostic method in 4/16 cases. In 2/4 of these cases, serology was also performed in order to support histopathology results. This may indicate the importance of serology as a screening test when leishmaniosis is suspected as a differential diagnosis, avoiding more invasive tests in many cases. However, serology may not be enough to reach a diagnosis in negative or low positive cases, 4 and so a combination of diagnostic tests may be needed for definitive diagnosis. Discrepancies can be seen in cats, as occurs in dogs, when serological and molecular tests are used at the same time.4,80,113 The sensitivity and specificity of serological and molecular tests may be influenced by many factors, and this may result in a lack of consistency between tests results.
PCR was performed in 10/16 cases in different tissues and was positive in all of them. PCR may be more sensitive than cytology and histology, but some investigations have shown that animals with increased titres of anti-Leishmania antibodies presented decreased positivity in PCR, whereas the greatest identification of genetic material through PCR occurred more frequently in cats with reduced antibody titres.1,76,105 This suggests that the immune response in cats differs from that observed in dogs, which might explain the high number of asymptomatic infected cats as well as the variable clinical manifestation of the disease. 1
In this case series, median survival time was greater than a year (17 months) in the group that received treatment. However, median survival time could not be calculated in the non-treated patients owing to the small sample size of the group, since all but one of the cats were euthanased at diagnosis. In the five patients that showed no improvement with leishmaniosis treatment, survival time ranged from 5 days to 3.5 years (median 60 days). In the group of patients that showed a clinical response to treatment, survival times ranged from 9 months to 7.5 years (median 407 days). Cats positive for FIV and cats on chronic corticosteroids treatment (considered concomitant immunosuppressive conditions) were present in both groups.
According to the retrospective nature of this study and variability in the treatment of each case, it is difficult to establish the best treatment and an accurate prognosis. In a recent study, 114 prognosis was not influenced by therapy or the retroviral status of the patients.
Treatment of cats with clinical FeL is still not based on scientific evidence, but on clinical experience from published case reports and on the off-label use of the most common drugs prescribed to dogs.4,114–120 This means that the efficacy and safety of these protocols have never been evaluated in controlled studies. Interestingly, median survival time in the group of animals treated specifically for leishmaniosis without concomitant diseases was longer than in the group with concomitant diseases (Figure 4); however, no statistical differences were seen between groups. Owing to the relatively small number of cases in each group a definitive conclusion could not be made with the information available.
The main limitations of this study, as with all retrospective studies, are its variability the management and diagnosis of each case. Extensive clinical information was not available for all cases and there was inconsistency in the follow-up periods and evaluations of the cases, making it more difficult to draw solid conclusions.
Conclusions
The most common clinical signs reported in this study were cutaneous lesions followed by ocular abnormalities, in which some cats showed a combination of skin or ocular lesions with systemic signs. Immunosuppressive conditions or coexisting diseases were identified in more than half of the cases, with FIV coinfection having the greatest prevalence. It should be taken into consideration that FeL clinicopathological abnormalities may be non-specific. Diagnosis in FeL was made by serological, cytological, histological or PCR methods, or a combination of these, but the diagnosis was obtained in 50% of the cases by cytology. Owing to the retrospective nature of this study and variability in the treatment of each case, it is difficult to establish the best treatment and provide an accurate prognosis, although median survival time in the group of animals treated specifically for leishmania without concomitant disease was longer than in the group with concomitant diseases, but not significantly so. The combination of cutaneous lesions and/or ocular lesions with other clinical signs in an endemic area should increase the suspicion of leishmaniosis.
Footnotes
Acknowledgements
We would like to thank Dr Jordi Puig for his help with the statistical analysis.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animal(s) only (owned or unowned), and followed established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care. Ethical approval from a committee was therefore not necessarily required.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work for the procedure(s) undertaken. For any animals or humans individually identifiable within this publication, informed consent (either verbal or written) for their use in the publication was obtained from the people involved.
