Abstract
Objectives
The aim of this study was to describe the clinicopathologic findings and associated diseases found in a population of hypocholesterolemic cats referred to two tertiary care facilities.
Methods
An electronic medical record search was performed at two veterinary university referral centers to identify cats with serum cholesterol values below the reference interval between January 2004 and 2016. Clinicopathologic data were reviewed for each case and cats were classified into specific disease categories based on clinical diagnosis. Median cholesterol values were compared between disease categories, as well as between survivors and non-survivors.
Results
In total, 106 hypocholesterolemic cats were included. The median age of the cats was 6 years (range 0.24–18 years). The most common disease categories were gastrointestinal (25.9%), hepatobiliary (19.8%), hematologic (14.8%) and urogenital (14.8%). Though median serum cholesterol values did not differ significantly between survivors and non-survivors, cats with concurrent hypoalbuminemia were at higher risk (odds ratio 15.6, 95% confidence interval 5.2–46.6; P <0.0001) of not surviving to discharge than cats with normal serum albumin concentrations.
Conclusions and relevance
Taken together, our data suggest that while the degree of hypocholesterolemia did not appear to influence survival rates, the concurrent presence of hypocholesterolemia and hypoalbuminemia was associated with a worse prognosis.
Introduction
Cholesterol provides a wide range of metabolic, as well as structural, functions in mammals. The clinical significance of low serum cholesterol levels has been extensively studied in human medicine, where it has been associated with malnutrition and a variety of other clinical conditions, including surgery, sepsis, liver dysfunction and acute hemorrhage. 1 Multiple prospective studies have demonstrated a correlation between hypocholesterolemia and higher rates of multiple organ dysfunction, longer hospitalization times and higher mortality in people.2–4 In addition, experimental data suggest that hypocholesterolemia might not only serve as a marker of disease severity, but might also clinically exacerbate some diseases by increasing the risks of endotoxemia and adrenal insufficiency of critical illness. 5
In spite of the established importance of dyslipidemias in morbidity and mortality in critically ill people, the prevalence, causes and consequences of hypocholesterolemia in veterinary medicine remain poorly characterized. Low serum cholesterol has been commonly reported in dogs with hepatic (acute liver failure, portosystemic shunts),6–8 gastrointestinal (chronic enteropathies, protein-losing enteropathies)9–12 and inflammatory (eosinophilic intestinal granulomas, immune-mediated anemia) diseases,13,14 as well as some neoplasia (histiocytic sarcomas). 15 As a predictor of morbidity and mortality, cholesterol levels have been shown to be lower in non-survivor populations of dogs suffering from heat shock, parvoviral enteritis, aflatoxicosis and sepsis.6,16–18 In contrast to dogs, low serum cholesterol concentrations have only been incidentally described in published reports of specific feline gastrointestinal, endocrine or neoplastic diseases.19–22 No studies currently exist describing the clinical findings and disease associations seen in a large population of hypocholesterolemic cats.
The objectives of this descriptive retrospective study were to assess the prevalence of feline hypocholesterolemia, describe associated clinicopathologic abnormalities and disease processes, as well as clinical outcomes in a tertiary referral population.
Materials and methods
Case selection
A multicenter retrospective observational study was performed with clinical case data from the Iowa State University (ISU), Lloyd Veterinary Medical Center and The Ohio State University (OSU) Veterinary Medical Center. Cases were recruited from the clinical pathology laboratory databases at both institutions by a search for cats with serum cholesterol levels below the institutional reference interval (RI) between January 2004 and January 2016. Data from 2009 were excluded from ISU owing to implementation and validation of a new chemistry analyzer during that year. Twenty normocholesterolemic cats seen during the same time frame as that of the study were selected from each institution as controls for comparison of the different chemistry analyzers being used to determine if significant inter-laboratory variation was present.
For inclusion in the study, a complete medical record, including physical examination, complete blood count (CBC) and serum biochemistry was required. Cats were excluded if medical records were incomplete or if full bloodwork had not been completed within 24 h of hospital admission. In addition, for cases seen for multiple hospital visits, only the initial visit was retained for analysis.
Clinical diagnosis
Based on data available from the medical records, case diagnoses were categorized by the authors according to their primary disease: gastrointestinal, hepatobiliary, hematologic, urogenital, respiratory, cardiac, dermatologic, neurologic and unknown.
Statistical analysis
Descriptive statistics were performed on age, weight and clinicopathologic parameters and presented as median (range) as applicable. Normality was assessed with a D’Agostino and Pearson’s test; all quantitative data were not normally distributed. A Wilcoxon rank sum test was performed to compare cholesterol concentrations between institutions; no differences were identified. A Wilcoxon rank sum test was used to compare serum cholesterol concentrations between cats that survived to hospital discharge and cats that died naturally or were euthanized. A Kruskal–Wallis ANOVA was performed to evaluate serum median cholesterol concentrations across disease categories. For hypoalbuminemia specifically, a Fisher’s test was used to compare the frequency of hypoalbuminemia between survivors and non-survivors, and a univariate logistic regression analysis was performed to see if hypoalbuminemia was predictive of survival to hospital discharge. Significance was set at P <0.05 for all comparisons. Statistical comparisons were performed with commercially available software (GraphPad Prism 6; GraphPad Software).
Results
Signalment
A total of 106 cats (49 from ISU and 57 from OSU) met the inclusion criteria for the study. Over the period of time assessed, the prevalence of hypocholesterolemia in the general population of feline patients at ISU was 0.4% (n = 49/12,696) and at OSU was 0.3% (n = 57/18,798). The median age of the cats was 6 years (range 0.24–18 years). The population was composed of 56 (52.8%) neutered males, three (2.8%) intact males, 41 (38.7%) spayed females and six (5.7%) intact females. The majority of cats in the study (n = 87/106 [82.1%]) were of mixed breed. The remaining cats were four Maine Coons, two each of Manx, Persian and Ragdoll, and one each of Exotic Shorthair, Himalayan, Scottish Fold, Selkirk Rex and Sphynx. The breed was not noted in four cases. Median weight on admission, available for 102 of the cats, was 4.6 kg (range 1.5–8.7 kg). The body condition score (scale 1–9 or scale 1–5) was recorded for 69 cats. When considered together, 35 (51%) were below, 20 (29%) were above and 14 (20%) were of ideal body condition.
Clinicopathologic abnormalities
The most common hematological abnormality in hypocholesterolemic cats was lymphopenia, present in 44 cats (41.5%). In addition, anemia was noted in 42 cats (39.6%), being microcytic in three cases, normocytic in 30 cases and macrocytic in nine cases (Table 1). Neutrophilia was present in 24 cats (22.6%). The most common electrolyte abnormalities noted were the presence of hypochloremia and hyponatremia, which were present in 50 (47.2%) and 40 (37.7%) cats, respectively. Biochemical changes consistent with impaired hepatic function or hepatic injury were frequently noted. Hyperbilirubinemia, hypoalbuminemia, low blood urea, and increased alanine aminotransferase and alkaline phosphatase were present in 30 (28.3%), 26 (24.5%), 14 (13.2%), 28 (26.4%) and 15 (14.2%) cases, respectively. Hypoglycemia was present in only four (3.8%) cases. Serum triglyceride values were low in two (13.3%) and high in one (6.7%) of the 15 cats in which they were measured.
Complete blood count (CBC) and serum chemistry results in hypocholesterolemic cats (n = 106)
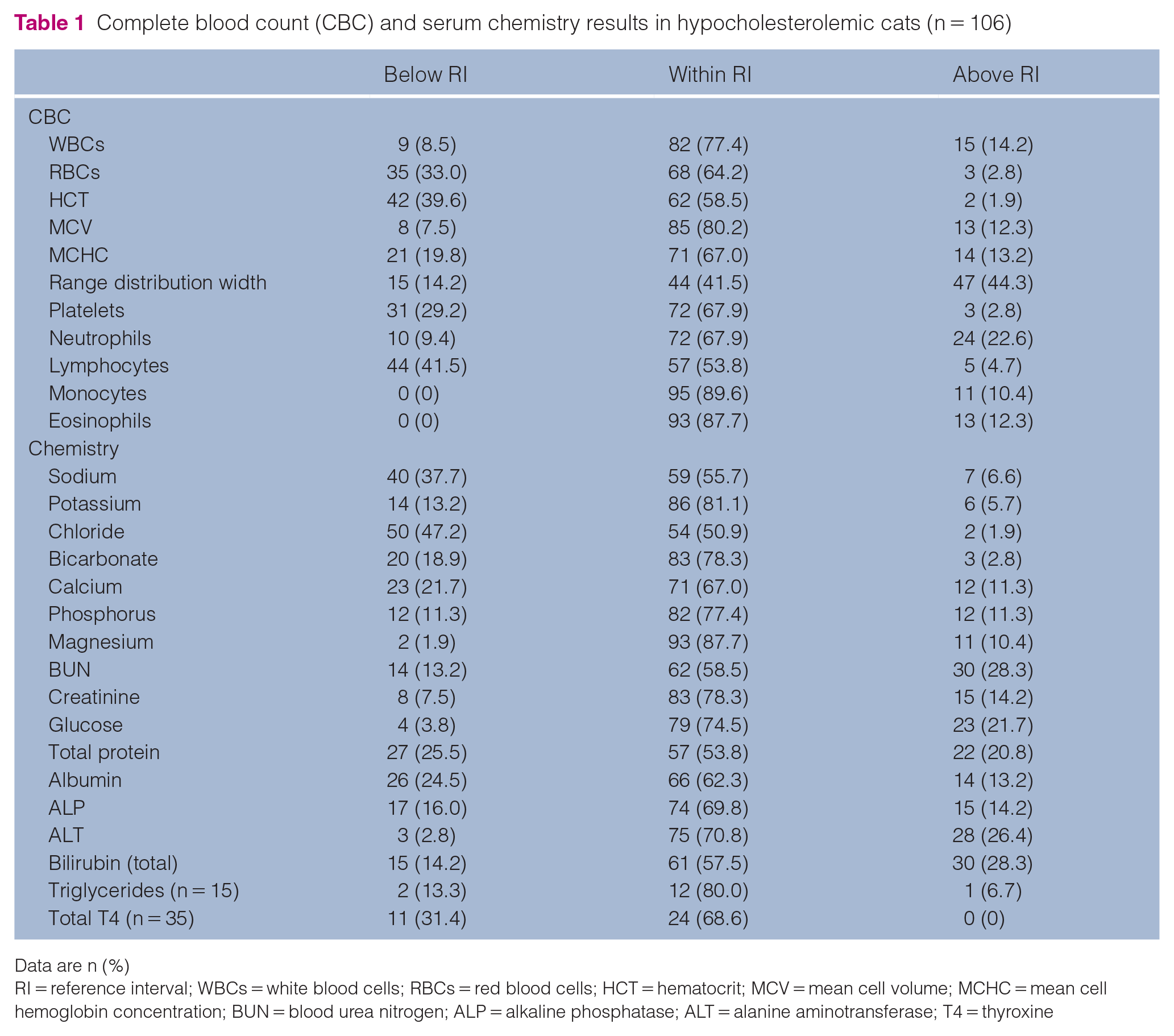
Data are n (%)
RI = reference interval; WBCs = white blood cells; RBCs = red blood cells; HCT = hematocrit; MCV = mean cell volume; MCHC = mean cell hemoglobin concentration; BUN = blood urea nitrogen; ALP = alkaline phosphatase; ALT = alanine aminotransferase; T4 = thyroxine
In the 35 cats that had measurement of total serum thyroxine levels performed, values were normal in 24 (68.6%) and low in 11 (31.4%) cases. Increased thyroxine was not noted in any of the cases.
Urinalysis data were available for 85 cats. The median urine specific gravity (USG) reported was 1.039 (range 1.007–1.060), with two (2.4%) being hyposthenuric (USG <1.008), seven (8.2%) isosthenuric (USG 1.008–1.012) and 27 (31.8%) poorly concentrated (USG 1.013–1.035). The remaining 49 (57.6%) were concentrated (USG >1.035).
Disease categories
Underlying diseases identified were categorized based on the body system affected (Figure 1). Classification was based on appropriate radiologic, cytologic or histopathologic findings. Excluding the 25 cases where no definitive diagnosis was established, the four most commonly affected systems were gastrointestinal (n = 21), hepatobiliary (n = 16), hematologic (n = 12) and urogenital (n = 12). The median serum cholesterol concentration of cases diagnosed with dermatologic disease was higher than the median cholesterol concentration in urogenital cases (69 mg/dl vs 53 mg/dl, respectively, P = 0.014) (Figure 2). However, no differences in median serum cholesterol concentrations were observed between any other disease categories.

Distribution of hypocholesterolemic cats by disease category
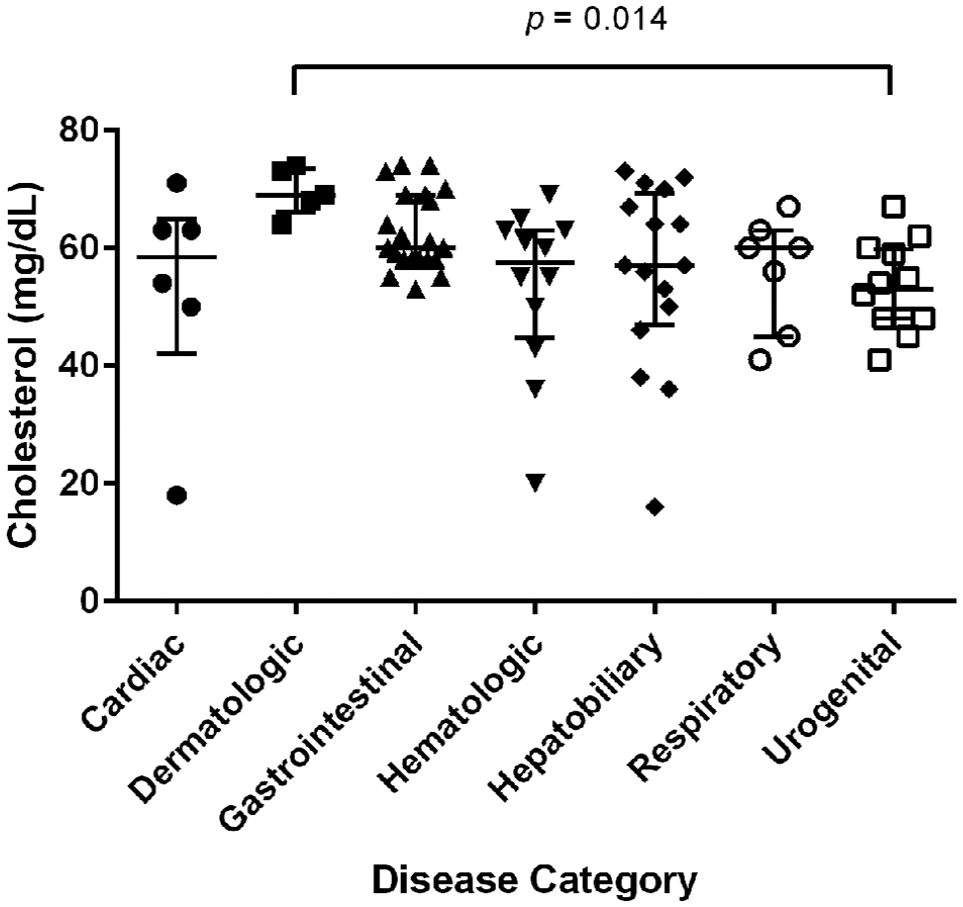
Cholesterol values by disease category. The horizontal bars represent the median value. The T-bars represent the interquartile range from the 25th to the 75th percentile
Prognosis
In total, 82 cats (77.4%) survived to discharge from hospital. Of the 24 (22.6%) cats that died in hospital, 21 were euthanized. No significant difference was noted between median serum cholesterol values of animals that survived to discharge, were euthanized or died naturally (61 and 57 vs 67 mg/dl, respectively, P = 0.0625) (Figure 3). However, logistic regression analysis revealed that hypoalbuminemic cats had an increased risk of death or being euthanized with an odds ratio of 15.6 (95% confidence interval 5.2–46.6; P <0.0001).

Cholesterol values for each outcome group. The horizontal bars represent the median value. The T-bars represent the interquartile range from the 25th to the 75th percentile
Discussion
The results of our study indicate that while hypocholesterolemia is a rare biochemical abnormality in cats, it is encountered in a wide range of clinical diseases. In addition, though survival to discharge did not appear to be influenced by the degree of hypocholesterolemia, cats with low serum albumin levels were at increased risk of not surviving to discharge.
The association of hypoalbuminemia with decreased survival in our study is an interesting one. However, owing to our study design, we cannot determine whether hypoalbuminemia is a predictor of mortality only in combination with hypocholesterolemia or whether it is an independent predictor of mortality. Indeed, hypoalbuminemia has been identified as a negative prognostic factor in cats with other critical illnesses, including acute kidney injury and hepatic lipidosis.23,24 Albumin has diverse and vital roles in maintaining vascular integrity and oncotic pressure, and it would appear logical that decreased concentrations would be associated with more severe illness. Given that gastrointestinal and hepatobiliary diseases were the most encountered categories in our study, it is likely that cats with concurrent hypocholesterolemia and hypoalbuminemia had more severely compromised gastrointestinal integrity or hepatic function.
It is surprising that our data failed to identify a significant difference in the serum cholesterol values between survivor and non-survivor cats. Though no previous studies exist investigating cholesterol’s prognostic value in cats, most reports in dogs have found cholesterol to be a reliable predictor of mortality. In dogs with specific conditions such as aflatoxicosis, heat shock and parvoviral enteritis, non-survivors were shown to have lower cholesterol levels than survivors.6,16,17 This association was also confirmed in a broader population of dogs in a referral center intensive care unit. 25 Conversely, in septic dogs, cholesterol was found to differ significantly between survivors and non-survivors only when non-survivors were defined as patients that died naturally and were not euthanized. 18 Given the fact that 87.5% of the cats that died in our study were euthanized, it is possible that non-medical factors such as financial constraints contributed to the mortality rate and skewed our results. The lack of a significant difference in cholesterol concentrations between survivors and non-survivors might also be due to the fact that our case numbers were simply insufficient to achieve significance. Though most of the previously mentioned canine studies investigated cholesterol in the context of specific pathologies, our study evaluated a much broader population with a range of disease etiologies and severities. This likely resulted in a greater variation in the magnitude of hypocholesterolemia in our population.
The lack of any significant difference in serum cholesterol values between survivors and non-survivors or between disease categories, with the exception of dermatologic diseases, suggests that serum cholesterol values might have limited prognostic value in cats. However, our study was limited to single cholesterol measurements and there are substantial data suggesting that trends in cholesterol, rather than single measurements, might have greater clinical significance.2–4,26 Considerable inter-individual variation of resting cholesterol levels exists owing to multiple factors such as diet, genetics and cholestasis. 1 Consequently, the prognostic value of cholesterol might not be in the comparison of an absolute value to a population-based RI, but rather in the comparison to an individual’s own RI. This is likely more important in patients with severe inflammatory or infectious diseases. Indeed, as serum cholesterol decreases in response to pro-inflammatory cytokines such as tumor necrosis factor alpha and interleukin-6, 27 it tends to act as a negative acute phase reactant, decreasing with worsening inflammation. In support of this concept, it has been shown that while serum cholesterol levels in people measured at the time of admission to the intensive care unit did not differ between survivors and non-survivors, the latter group did show progressive declines in serum cholesterol values over the course of hospitalization.2,3
Interestingly, the only significant difference in cholesterol values between disease categories in our study was between dermatologic and urogenital cases. Though this finding might be due to some bias given that the dermatologic category comprised only five cats, it can also be explained by the fact that cats with dermatologic conditions had less severe disease than those with urogenital disease. Indeed, as low serum cholesterol values have been shown to correlate with disease severity and higher critical illness scores (MODS and APACHE), 28 it is not surprising that cats with more confined diseases have higher cholesterol values than patients with more severe systemic disease, such as urologic disease.
Owing to its retrospective nature, this report has several limitations. First and foremost, this study was purely descriptive and did not include a control group. The incorporation of a normocholesterolemic critically ill control group could have allowed further characterization of the underlying diseases and prognostic importance of hypocholesterolemia. The relative importance of the disease categories reported in our study is similar to what has previously been reported in critically ill cats.29,30 However, the lack of a statistical comparison with a population from the same institutions precludes the drawing of any conclusions as to a possible unique breakdown of diseases associated with hypocholesterolemia. Though our inclusion criteria dictated that all cases required a full physical examination, as well as CBC and serum chemistry, the lack of other diagnostics such as hepatic function testing or cobalamin measurements hindered our ability to fully assess the involvement of synthetic liver failure or malabsorptive gastrointestinal disease as being the direct causes of hypocholesterolemia. In addition, only 15 cats in our study had concurrent triglyceride measurements performed. Given that concurrent hypocholesterolemia and hypertriglyceridemia has recently been shown to be a strong negative prognostic factor in hypocholesterolemic dogs, 25 the evaluation of triglycerides in a larger population could help further characterize the prognostic importance of dyslipidemias in cats. Finally, the case histories of the cats included in this study were limited and factors such as the presence and duration of anorexia were not consistently documented. Perhaps more significantly, medication histories were not consistently noted in the medical records. This is of importance as many commonly used medications, most notably glucocorticoids, can alter lipid metabolism.
Conclusions
Though gastrointestinal and hepatobiliary diseases accounted for almost half of our cases, hypocholesterolemia was present in diseases affecting many other body systems. While the magnitude of hypocholesterolemia did not appear to be a prognostic factor for survival in cats, the concurrent presence of hypoalbuminemia was a substantial negative prognostic factor.
Footnotes
Author note
Some of these data were presented as a poster at the annual forum of the American College of Veterinary Internal Medicine, National Harbor, MD, USA, June 2017.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals only (owned or unowned), and followed established internationally recognized high standards (‘best practice’) of individual veterinary clinical patient care. Ethical approval from a committee was not necessarily required.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work for the procedure(s) undertaken. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
