Abstract
Aim:
The overarching purpose of the 2019 AAFP Feline Zoonoses Guidelines (hereafter referred to as the ‘Guidelines’) is to provide accurate information about feline zoonotic diseases to owners, physicians and veterinarians to allow logical decisions to be made concerning cat ownership.
Scope and accessibility:
The Panelists are physicians and veterinarians who worked closely together in an attempt to make these Guidelines a document that can be used to support the International One Health movement. This version of the Guidelines builds upon the first feline zoonosis panel report, published in 2003 (catvets.com/guidelines), and provides an updated reference list and recommendations. Each of the recommendations received full support from every Panelist. Primary recommendations are highlighted in a series of ‘Panelists’ advice’ boxes.
Introduction
The American Association of Feline Practitioners (AAFP) first published a feline zoonoses panel report in 2003, followed by a panel report on feline bartonellosis in 2006.1,2 Those documents were extensively referenced and this version will focus on new information published since 2003. The aim of these Guidelines is to offer practical recommendations to help physicians and veterinarians provide accurate information to owners concerning health risks associated with cat ownership. (See

The recommendations of the Panelists are based on published data when available, and recommendations of other public health affiliated groups are taken into consideration. Information from the Centers for Disease Control and Prevention (CDC; cdc.gov/healthypets/index.html), the Companion Animal Parasite Council (CAPC; capcvet.org), the World Small Animal Veterinary Association (WSAVA) One Health Committee (wsava.org/educational/one-health-committee) and the American Association of Food Safety and Public Health Veterinarians (AAFSPHV; aaphv.org) was consulted and referenced within this Guidelines document.

For each recommendation in the draft documents, the Panelists were individually asked whether they agreed or disagreed with the recommendation. All of the final recommendations were supported by each Panelist. After the Panelists had what was considered a final document, select members of the CAPC board and the WSAVA One Health Committee were asked to make comments, which were considered for inclusion in this document. Select members of the International Society of Feline Medicine (ISFM) also reviewed the document.
Zoonotic diseases are defined as being common to, shared by, or naturally transmitted between humans and other vertebrate animals. There are multiple agents that can infect cats and their owners, and these are summarized in Tables 1–5.1,3 Humans are infected with zoonot-ic agents from direct contact with infected cats, contact via contaminated food or water, from shared vectors and from the shared environment. Direct contact with feline feces (enteric zoonoses), respiratory secretions, urogenital secretions, or infected skin and exudates, as well as bites and scratches, can result in human infections. Some zoonotic agents are transmitted between cats and people by shared vectors such as fleas, ticks or mosquitoes. Anaplasma phagocytophilum (ticks), Borrelia burgdorferi (ticks), Ehrlichia species (ticks), Bartonella species (fleas, ticks), Rickettsia felis (fleas) and Dirofilaria immitis (mosquitoes) are examples of vector-borne zoonoses.4,5 With these agents, the cat potentially brings the vector of the organism into the human environment, resulting in exposure. 6 Some zoonotic agents, including Histoplasma capsulatum, Coccidioides immitis, Blastomyces dermatitidis, Cryptococcus neofor-mans and Aspergillus species, do not usually infect humans through direct contact with the infected cat but are acquired from the same environmental source. Other agents like Sporothrix species can be acquired from infected cats or the environment.7,8
Potential enteric zoonotic agents of cats

Ancylostoma braziliense is the most likely to cause cutaneous larva migrans in the USA. Infective larvae develop after passage of the eggs into the environment; hence, direct transmission through contact with cats is less likely than exposure through environmental contamination
Most cats are infected by Campylobacter upsaliensis; this host-adapted species is rarely found in humans
Most cats are infected by C felis, and this host-adapted species is rarely found in humans
Host-adapted and zoonotic assemblages exist. Cats can harbor zoonotic assemblages, but whether levels of infection result in reinfection of humans is not established
Most Helicobacter species found in cats are host-adapted species. When Helicobacter pylori is detected in a cat it is likely from reverse zoonotic transmission
Sporulation of oocysts occurs after passage into the environment; hence, direct transmission by contact with cats is less likely than exposure through environmental contamination
Potential scratch, bite or exudate associated zoonotic agents of cats

Bartonella henselae, Bartonella koehlerae and Bartonella clarridgeiae are transmitted among cats by Ctenocephalides felis and so are also listed under flea-borne disease (Table 5). There are other Bartonella species with zoonotic implications. Cats generally develop a higher level of bacteremia than dogs and so are epidemiologically linked more frequently to human disease. The vectors are unknown for some Bartonella species
F tularensis can be acquired by direct contact with a bacteremic cat but is also vector borne
Potential ocular or respiratory zoonotic agents of cats
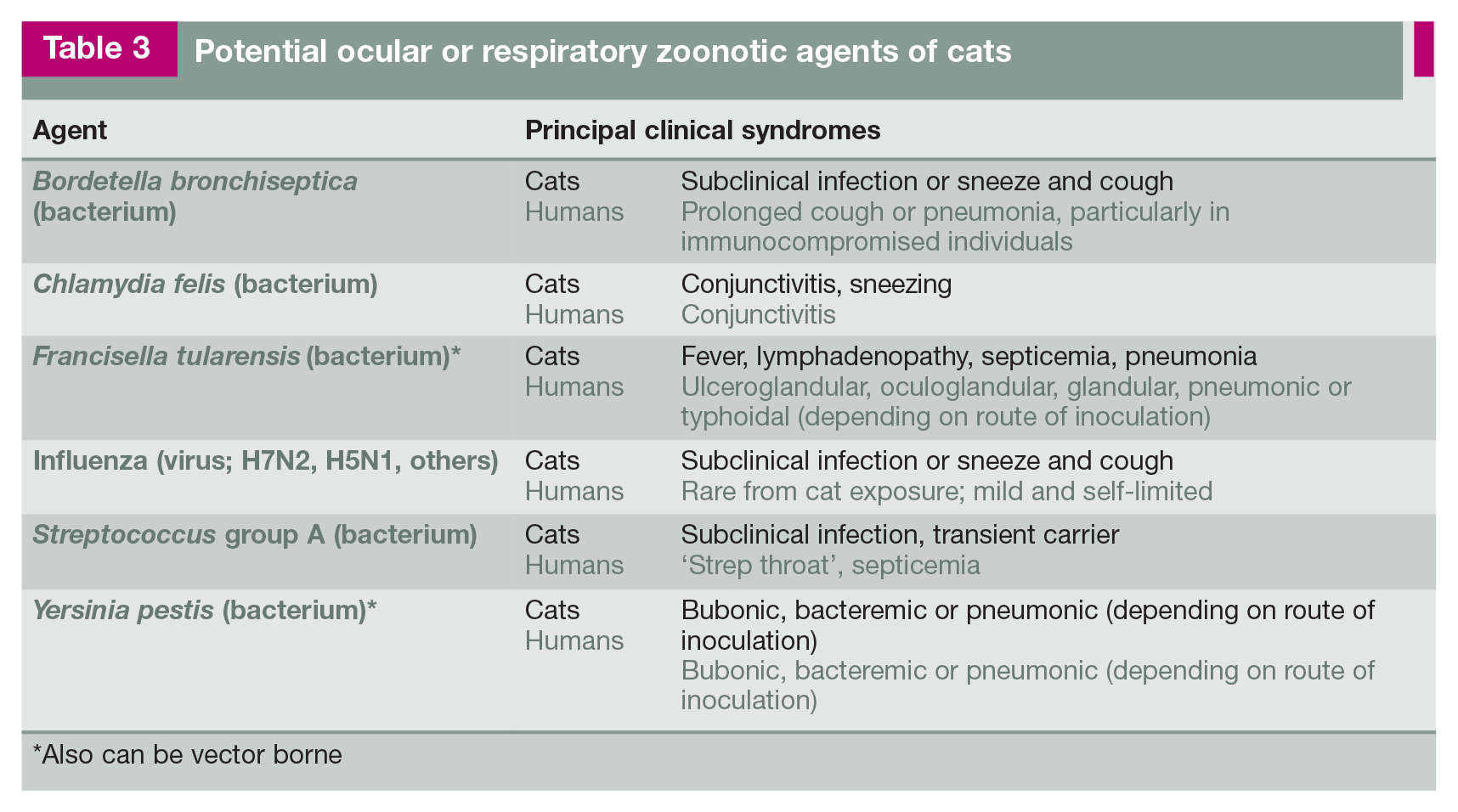
Also can be vector borne
Potential urogenital zoonotic agents of cats

Common flea- and tick-borne zoonotic agents of cats
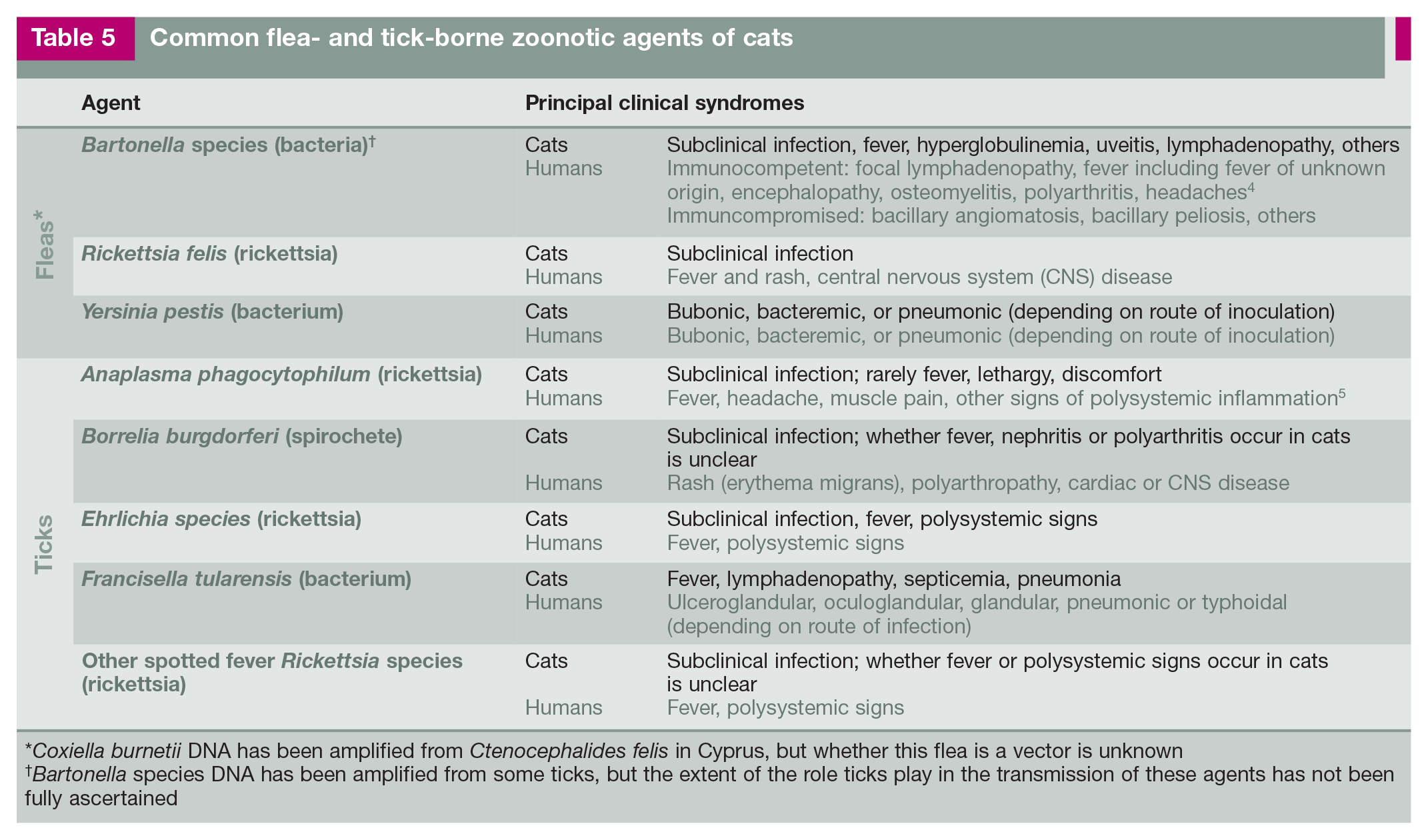
Coxiella burnetii DNA has been amplified from Ctenocephalides felis in Cyprus, but whether this flea is a vector is unknown
Bartonella species DNA has been amplified from some ticks, but the extent of the role ticks play in the transmission of these agents has not been fully ascertained
Most of the agents discussed in these Guidelines can infect and cause disease in anyone, but disease is generally more prevalent or more severe in those with immunodeficiency-inducing disorders. 9 Humans with AIDS are discussed most frequently, but there are many more individuals with immunodeficiencies, including the very old, the very young, individuals receiving chemotherapy or glucocorti-coids for immune-mediated diseases, organ transplant recipients and cancer patients. Humans are unlikely to contract zoonotic diseases from direct contact with their healthy cats and the mental health benefits from pet ownership can be considerable.10,11 Pet ownership is known to improve general sense of wellbeing and there is some evidence that happiness influences immunological factors. 12
General recommendations to help prevent zoonotic transfer of disease for owners and veterinarians are presented in the box on page 9. In each of the subsections that follow, additional recommendations are provided based on the route that humans are exposed to feline zoonotic agents.
Enteric zoonoses
There are multiple infectious agents of the gastrointestinal tract that can be shared between cats and humans (Table 1). Since some enteric zoonotic agents (eg, some Campylobacter species, Salmonella species, Yersinia enterocoliti-ca) are infectious when passed in feces, direct contact with infected cats can result in human infection and disease.13,14 Some enteric agents of cats that are infectious immediately in feces, like Giardia species or Cryptosporidium felis, are not considered significant zoonotic agents; when these infections occur, the strains are generally cat-specific.15–18
Other infectious agents, such as Ancylostoma species, Toxocara cati and Toxoplasma gondii, require a period of time out of the host prior to becoming infectious. Thus, many enteric zoonoses result from ingestion of the infectious agent in contaminated food, water or other environmental sources. (See

Raw meat can harbor enteric pathogenic bacteria like Campylobacter species, entero-toxigenic Escherichia coli and Salmonella species.19–21 Freezing meat does not consistently kill all bacteria and it is recognized that feeding raw meat to pets can result in amplification of potentially pathogenic bacteria in animal feces. (See

Some Ancylostoma species of cats are associated with cutaneous larva migrans and T cati is associated with ocular and visceral larva migrans (see Table 1). Toxocara species eggs were found on the fur of dogs and cats in one study, but were non-viable.
23
While hookworm and roundworm egg shedding is generally highest in kittens, adult cats can also shed eggs that become infectious in the human environ-ment.24–26 (See

Cats (and other felids) are the only definitive host for T gondii and shed millions of oocysts in feces after primary infection.
27
Once these oocysts have sporulated (Figure 1), infection of humans can occur. Human exposure can also occur by ingestion of T gondii tissue cysts and transplacentally if a previously uninfected mother ingests sporulated oocysts or tissue cysts during pregnancy. It is now known that humans are commonly infected by ingestion of sporulated oocysts.
28
(However, see
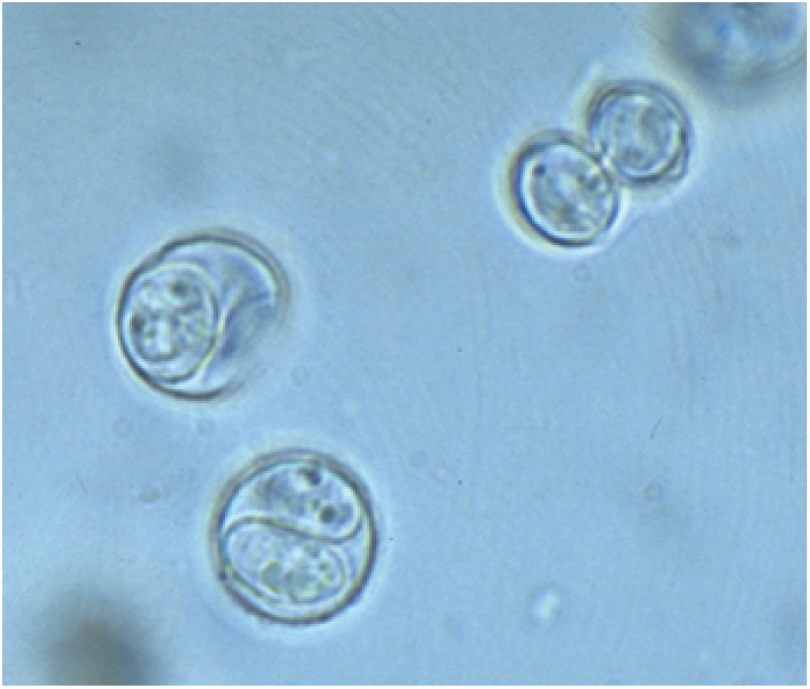
Toxoplasma gondii sporulated oocysts (8 μm × 10 μm), each containing two sporocysts. This form of the oocyst is zoonotic to humans

Most studies evaluating cat ownership as a risk factor for human toxoplasmosis have shown minimal associations, including one study of HIV-infected individuals.
29
In addition, in one study, veterinary staff members that worked frequently with cats had low sero-prevalence rates, suggesting exposure to cats did not increase their risk of acquiring T gondii infection.
30
Cats that are infected by T gondii usually do not shed or shed lower numbers of oocysts on secondary exposure,
31
and in one experimental study did not have repeat oocyst shedding after being administered ciclosporin.
32
(See


Enteric zoonotic agent prevalence rates that have been reported in several studies of cats are generally higher in young cats with diar-rhea.24–26,33 However, most of the agents can still be present even if the stool is normal. These findings emphasize that diagnostic work-ups for enteric infections are indicated due to potential human health risks. (See


Gastrointestinal signs of enteric bacterial infections generally resolve with supportive care such as use of therapeutic diets and probiotic administration. Antibiotics should only be considered if these cats have fever or other evidence of bac-teremia or sepsis; and, if believed to be necessary, should only be administered parenterally.
Scratch, bite or exudate exposure zoonoses
Approximately 1% of emergency room visits per year in the USA are to evaluate people bitten by animals.
36
Most of the aerobic and anaerobic bacteria associated with bite or scratch wounds (eg, Pasteurella species, Staphylococcus species) cause cellulitis in immunocompetent individuals. Approximately 28–80% of cat bites become infected, and severe sequelae including meningitis, endocarditis, septic arthritis, osteoarthritis and septic shock can occur.
37
Immunodeficient humans or humans exposed to Pasteurella species, Capnocytophaga canimorsus or Capnocytophaga cynodegmi more consistently develop systemic clinical illness.38–40 Splenectomized humans, as well as those with non-functional spleens, such as in sickle cell disease, are at increased risk of developing overwhelming sepsis/purpura fulminans with Capnocyto-phaga species infection.
41
(See

Bartonella species infection of humans can be associated with bites and scratches, and these agents are also vector-associated zoonoses (Tables 2 and 5; also see box on page 9). It is known that Bartonella species (particularly Bartonella henselae), the cause of cat scratch disease, peliosis hepatis, bacillary angiomato-sis, bacterial endocarditis and a number of other human inflammatory syndromes such as polyarthritis, are present in the oral cavity, on the skin and on the claws of cats with Ctenocephalides felis infestations.4,44–46 Veterinary healthcare providers may be at greater risk of development of Bartonella species-associated syndromes from exposure to cats or infected C felis.
47
Consistent use of flea control products has been shown in a B henselae (cat scratch agent) model to block transmission of the pathogen among cats.48,49 (Thus, see


Francisella tularensis (tick-borne agent) and Yersinia pestis (rodent fleas) infections can also be associated with bite wounds, but are not as common in humans as Bartonella species-associated infections (Tables 2 and 5, also see box on page 9).51–53
Of the many fungal agents that infect both humans and animals, Sporothrix species (Figure 2) and the dermatophytes (Figure 3) appear to be the most common to infect humans upon direct exposure.7,8,54 Histoplasma, Blastomyces, Coccidioides, Aspergillus and Cryptococcus species infections of humans and animals can occur in the same household, but infection of humans generally results from a common environmental source rather than direct contact with an infected animal. (See

Cutaneous sporotrichosis in a young adult cat

Characteristic cutaneous ringworm lesion on the forearm of a veterinarian

Cats can harbor meticillin-resistant Staphylococcus aureus (MRSA) and Staphylococcus pseudintermedius, and transmission between animals and humans is likely to occur within households.55–57 Unlike other infections discussed in these Guidelines, MRSA in cats in generally acquired from humans and is considered a ‘reverse zoonosis’ or ‘humanosis’. (See

Although uncommon in cats, rabies is still the only significant small animal viral zoono-sis in the USA and is associated with bite wounds.59,60 (See

Ocular or respiratory zoonoses
Bordetella bronchiseptica and Chlamydia felis cause mild respiratory disease in cats (Table 3). 62 Cough is most common with B bron-chiseptica infection and conjunctivitis with C felis infection. It is believed that C felis may be associated with conjunctivitis in people.63,64 Most people with Bordetella species infections are infected by Bordetella pertussis, but some individuals, particularly immunocompro-mised people, can be infected with B bronchi-septica.65,66
Cats with cough and systemic evidence of bacterial infection such as fever might occasionally be infected with Y pestis and/or F tularensis, if living in endemic areas; these agents can be transmitted from cats to humans in respiratory secretions.53,67,68 (See

Humans are the principal natural hosts for Streptococcus group A (Streptococcus pyogenes) bacteria, which cause ‘strep throat’ in people. Cats in close contact with infected humans can develop transient, subclinical colonization of pharyngeal tissues and can transmit the infection to other humans. Older data suggested this was a common occurrence; however, with improved diagnostics it has been shown to be an uncommon event. Early studies used crude antimicrobial data on bacitracin susceptibilities rather than genetic subtyping.69,70 Later, when Lancefield typing was performed, the true prevalence in household pets was found to be only 0–3% and not correlated with the presence of infection in the owner, and hence the risk for transfer of infection from pet cats to humans is considered low. 71
Avian influenza viruses occasionally infect cats.72,73 Rarely, cats infected with an influenza virus are associated with clinical disease in humans.
74
Recently, an H3N2 vaccine for dogs marketed in the United States was approved for use in cats (Merck Animal Health, New Jersey, USA) and a different canine vaccine was shown to induce H3N2 immune responses in cats.
75
Whether these vaccines are indicated for use in cats to lessen human exposure in the USA is currently unknown. (See

Urogenital tract zoonoses
Coxiella burnetii (Table 4) is a rickettsial agent found throughout the world that is associated with Q fever.76,77 Many ticks, including Rhipicephalus sanguineus, are naturally infected with C burnetii and so this agent is considered to be a shared vector zoonosis. While abortion can occur, cats infected by C burnetii usually do not show clinical signs of disease. C burnetii DNA was amplified from 8.5% of uterine biopsies taken after elective ovario-hysterectomy in a small study of cats in Colorado, USA.
78
Human illness associated with close contact with infected cats occurs after aerosol exposure to the organism passed by parturient or aborting cats; clinical signs develop 4–30 days after contact.
79
Humans commonly develop acute clinical signs similar to those associated with other rickettsial diseases, including fever, malaise, headache, pneumonitis, myalgia and arthralgia. (See

The major agents associated with urine that could be a direct feline zoonosis are the Leptospira species.
80
These spirochetes can be transmitted in urine to humans and result in clinical disease. While Leptospira species antibodies and DNA have been detected in cats, the role these agents play in leptospirosis of people is unclear.81–83 (See

Vector-borne zoonoses
There are multiple infectious agents potentially transferred from cats to humans by fleas and ticks (Table 5). C felis from cats contain multiple pathogens, with Bartonella species and R felis being the most common.46,84,85 (See the scratch, bite or exudate exposure section on pages 4–6 for more information about Bartonella species.) R felis can cause mild clinical signs of disease including fever and malaise.
86
While C felis and ticks collected from cats or dogs are commonly positive for R felis DNA, it appears the dog is the better reservoir.
87
It is possible that C burnetii and Y pestis could be transmitted to humans from cats with fleas. Humans can also be infected by Dipylidium caninum by accidental ingestion of infected fleas, which typically is reported in young children that spend time on the carpet where fleas usually reside when not on the host.
88
(See

Recently, it was documented that pet ownership increases the risks for humans of exposure to ticks.
6
Cats have been shown to be infected experimentally and naturally with many tick-borne agents that could infect humans including B burgdorferi and A phagocy-tophilum.89–92 (Thus, see

There are many more vector-borne diseases of potential significance in cats of the USA, including West Nile virus (mosquitoes) and Leishmania species.93,94 However, in contrast to the flea- and tick-associated zoonoses, cats are unlikely to influence exposure of these agents to owners in the USA. However, travel history from other countries should always be considered. For example, cats can harbor both Old World and New World Leishmania species. International guidelines (leishvet.org/fact-sheet-feline-leishmaniosis) are available to provide information about leishmaniosis and control for cats imported from endemic areas.

Summary Points
While humans are rarely infected with a zoonotic agent by exposure to a healthy cat, there are many potential infections that can occur.
Disease is generally more prevalent or more severe in people with immunodeficiency-inducing disorders, the very old, very young, individuals receiving chemotherapy or glucocorticoids for immune-mediated diseases, organ transplant recipients and cancer patients.
Cats should have consistent deworming and should be prescribed vector control.
Cats with clinical signs of disease should be assessed by a veterinarian to determine the risk of zoonotic disease transmission and to have the clinical abnormalities treated.
Supplemental Material
Supplementary_Material_Feline_Zoonoses_Client_Brochure – Supplemental material for 2019 AAFP Feline Zoonoses Guidelines
Supplemental material, Supplementary_Material_Feline_Zoonoses_Client_Brochure for 2019 AAFP Feline Zoonoses Guidelines by Michael R Lappin, Tom Elston, Lisanne Evans, Carol Glaser, Lorraine Jarboe, Peter Karczmar, Cathy Lund and Michael Ray in Journal of Feline Medicine and Surgery
Footnotes
Appendix: Client brochure
Acknowledgements
The Panelists thank Ms Kimberley Kern for helping with literature reviews. The final draft of the document was reviewed by the Companion Animal Parasite Council Board (capcvet.org). The members of the WSAVA One Health Committee (![]() ) provided comments on the document.
) provided comments on the document.
Conflict of interest
The Panelists have no conflicts of interest to declare.
Funding
The Panelists received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
