Abstract
Objectives
Subclinical bacteriuria (SBU) is the presence of bacteria in urine with no clinical evidence of lower urinary tract disease. The aims of this study were to investigate if being overweight and/or obesity predispose cats to SBU, to investigate previously reported risk factors and to determine the prevalence of SBU in a prospectively sampled cohort of middle-aged and elderly cats.
Methods
Cats aged ⩾6 years presenting to the University Hospital for Companion Animals in Copenhagen from 2015–2019 for causes unrelated to the lower urinary tract were eligible for enrolment. Body condition scoring was performed on a 9-point scale. Overweight was defined as a body condition score (BCS) ⩾6 and obese as a BCS ⩾8. The correlation between SBU and the variables of sex, healthy/diseased, age, BCS and comorbidities (chronic kidney disease, diabetes mellitus, hyperthyroidism, hepatic disorders and gastrointestinal disease) were analysed by binominal logistic regression.
Results
In total, 179 cats ranging from 6–20 (median 10) years of age were included. SBU was identified in 11/179 cats (6.1%). Being overweight was not a significant risk factor (overweight/obese odds ratio [OR] 0.3, 95% confidence interval [CI] 0.06–1.6, relative risk [RR] 0.3 [95% CI 0.05–1.3] vs lean; P = 0.2) and neither was obesity compared with lean and overweight cats (P = 0.99). Female sex (OR 6.2 [95% CI 1.3–30], RR 4.7 [95% CI 1.5–12] vs male; P = 0.02) and the presence of hepatic disease (OR 7.5 [95% CI 1.4–39], RR 5.3 [95% CI 1.3–12]; P = 0.02) were significant risk factors.
Conclusions and relevance
The prevalence of SBU in cats is low, and being overweight/obese was not identified as a predisposing factor. The increased risk associated with hepatic disease has not been previously reported, and further studies are needed to confirm this finding.
Introduction
Subclinical bacteriuria (SBU) is defined as the culture-confirmed presence of bacteria in the urine of an animal with no clinical signs of lower urinary tract disease. 1 The term SBU was first introduced by the 2011 International Society for Companion Animal Infectious Diseases guidelines on urinary tract infection (UTI) management in cats and dogs, in which veterinarians were encouraged to discriminate between SBU and manifest UTIs as epidemiology, therapeutic needs and prognosis may vary between the conditions. 2 Prior to that time, research in feline UTI generally did not discriminate between cystitis and SBU, and to date there are only a limited number of studies investigating feline SBU as a separate entity.
The prevalence of SBU in the overall cat population is unknown. The condition appears to be uncommon in young (median age 4 years) healthy cats, with a reported prevalence of 0.9%. 3 A higher prevalence of SBU has been reported in studies investigating mixed populations of cats (median age 12–14 years) presenting to veterinary facilities (prevalences of 7.9% and 28.8%),4,5 and in populations of mainly elderly cats (median age 12–13 years) with diabetes mellitus (DM; 11%), hyperthyroidism (HT; 12%) or chronic kidney disease (CKD; 17–23%).6,7 As is the case for cystitis, female sex and increasing age are the most commonly reported risk factors for SBU in cats.4–6,8,9
In people, obesity has been implicated as a risk factor for asymptomatic bacteriuria (ABU), the analogous human condition of SBU. An increased frequency of SBU has also been found in morbidly obese dogs (body fat percentage >45%). 10 The pathophysiological link between obesity and SBU remains obscure, but there are several mechanisms by which the risk of SBU or UTIs may be increased in obese animals. Obese animals are more likely to have a reduced mobility, which may lead to less frequent urination, thereby providing the bacteria with a better opportunity to adhere to the bladder mucosa. 11 It is also more likely for obese animals to have skin fold infections in the perineal areas. This may lead to the ascension of bacteria, and most bacterial UTIs occur as a consequence of the ascending migration of pathogens. 12
An association between being overweight and SBU has not been investigated in cats, despite obesity being reported as the second most common health problem in domestic cats in Western countries.13–16
The primary objective of this study was to investigate if being overweight and/or obesity predispose cats to SBU. Secondary objectives were to investigate point prevalence and other potential risk factors for SBU (female sex, old age, presence of comorbidities) in a prospectively collected population of middle-aged to older cats.
Materials and methods
Recruitment of cats
The study was designed as a prospective cross-sectional study of middle-aged to elderly cats. Cats were recruited at the University Hospital for Companion Animals, University of Copenhagen, from October 2015 to January 2019. The study protocol was approved by the Ethical Administrative Committee of the University Hospital for Companion Animals, and all cat owners signed an informed consent form. Client-owned cats ⩾6 years of age presenting for reasons other than lower urinary tract disease were eligible for inclusion. Cats were included if they had a complete blood count (CBC), serum chemistry and urinalysis of urine collected by cystocentesis performed as part of their diagnostic work-up or health check. Cats were excluded if they had a history of clinical signs of lower urinary tract disease (dysuria, pollakiuria, stranguria, haematuria, periuria) within the past 4 months, if they had been treated with antibiotics within the past week or if they had already participated in the study.
Cats fulfilling the inclusion criteria were retrospectively categorised as either healthy or diseased. Cats presenting for inclusion in other ongoing research projects, 17 or for a health examination were included in the healthy category if no clinically relevant abnormalities were observed upon clinical examination, CBC, serum chemistry and urinalysis. Diseased animals (included either through other research projects as described above or through the hospital’s internal medicine or emergency services) were subcategorised into the following six groups: CKD (cats diagnosed with CKD of all stages according to the International Renal Interest Society guidelines); HT (cats with HT defined as thyroxine >42.5 nmol/l and the presence of clinical signs of the disease); DM (cats with DM defined as hyperglycaemia, glycosuria and clinical signs consistent with the disease or receiving insulin at the time); hepatic disease (HD) (cats diagnosed with a primary HD based on elevated liver enzymes [alanine aminotransferase, alkaline phosphatase], elevated fasting bile acids and/or hyperbilirubinaemia for which no extrahepatic cause was identified); gastrointestinal diseases (GIDs) (cats with gastrointestinal signs and no extragastrointestinal disease identified based on CBC, serum chemistry and urinalysis); or miscellaneous (cats diagnosed with a disease presented in <10 cats each). Some of the cats included were diagnosed with more than one disease. These cats were included in the group representing their primary complaint at the inclusion time.
Body condition scoring
All cats included in the study were body condition scored on a scale from 1–9. 18 Being overweight was defined as a body condition score (BCS) ⩾6 and obesity as a BCS ⩾8. Body condition scoring was performed by the principal investigators (FSM, INK) or their students (PVB, LMP) for cats admitted during the daytime. For cats admitted out of hours, the attending clinician performed the body condition scoring. Approximately 25% of cases were out-of-hours cases with scoring performed by the attending clinician.
Urine aerobic culture and susceptibility testing
After collection by cystocentesis, excess urine from the urinalysis was stored refrigerated (4ºC) for a maximum of 24 h prior to culture. Quantitative aerobic urine culture was performed by streaking 1 µl and 10 µl of urine on each half of a blood agar plate, respectively, followed by incubation at 37ºC for 24 h. Plates displaying no bacterial growth were re-incubated and evaluated again after 48 h. Samples were considered culture positive (ie, significant bacteriuria) if one or more colonies were detected upon incubation. Samples suspected of contamination (eg, growth of colonies outside streaking lines) were evaluated by a specialised veterinary microbiologist (PD) to determine whether or not the corresponding cats had significant bacteriuria. Culture-positive agar plates were brought to the microbiology laboratory, Sund Vet Diagnostik (University of Copenhagen), for bacterial species identification and susceptibility testing. All colony types were identified to the species level by matrix-assisted laser desorption/ionisation–time of flight (MALDI–TOF) mass spectrometry (Vitek MS RUO; BioMérieux). Antimicrobial susceptibility was tested for all isolates using the broth microdilution method (Sensititre COMPAN1F; TREK Diagnostic System) according to the Clinical and Laboratory Standards Institute.19,20
Sample size calculations
Sample size calculations were performed using an online calculator 21 and applying a significance level of alpha = 0.05 and beta = 0.2. Estimating the prevalence of SBU in healthy cats to be 1% 2 and the prevalence in cats with comorbidities to be 13%,5,6 the minimum number of cats required were estimated to be 140 in total.
Statistical analysis
Binomial logistic regression with multiple independent variables was performed with culture (positive vs sterile) as the dependent variable, and sex, group (disease vs healthy), age and BCS (⩾6 vs <6) as the independent variables. Subsequently, relative risk (RR) transformation of odds ratios (ORs) was calculated. Statistical analysis was performed using the stats and sjstats packages in R version 3.4.3 (30 November 2017). The same variables were included in a second binomial model with multiple independent variables, in which only the cut-off for BCS was changed to ⩾8, to analyse if obesity was a risk factor.
In addition, for each of the five disease groups (CKD, HT, DM, HD and GID) a separate binomial logistic regression model with multiple independent variables was also created. In each model, the dependent variable was culture (positive vs sterile), while the independent variables were sex, age, BCS (⩾6 vs <6) and disease group (eg, CKD vs non-CKD, etc).
Statistical significance was defined as P <0.05, and 95% confidence intervals (CIs) were calculated.
Results
A total of 179 cats were enrolled, including 83 females and 96 males (Table 1). Age ranged from 6–20 years (median 10 years). Sixty-eight cats were scored as overweight (BCS ⩾6), and, of these, 16 cats were scored as obese (BCS ⩾8). In total, 111 cats were scored as lean or underweight, with a BCS <6. Fifty-seven cats were considered healthy and 122 diseased.
Distribution of cats included in the study
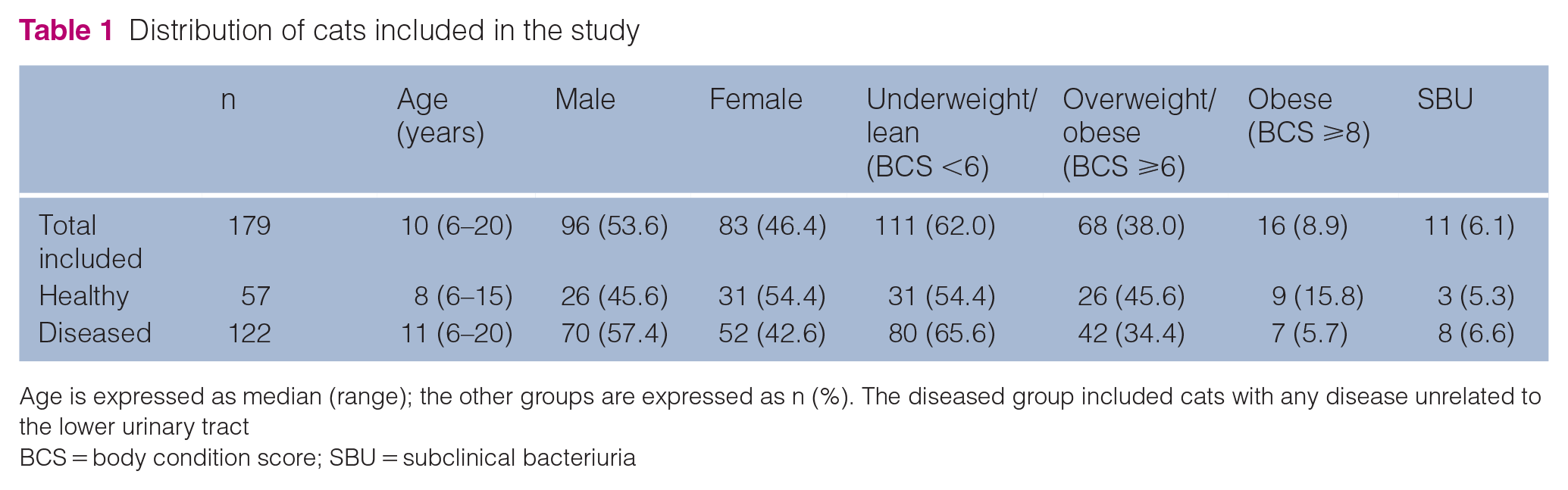
Age is expressed as median (range); the other groups are expressed as n (%). The diseased group included cats with any disease unrelated to the lower urinary tract
BCS = body condition score; SBU = subclinical bacteriuria
Of the 122 diseased cats, 21.3%, 21.3%, 13.1%, 9% and 8.2% were diagnosed with CKD, DM, GID, HD and HT, respectively (Table 2). The remaining 27% were diagnosed with miscellaneous diseases, with each disease represented by <10 cats.
Distribution of comorbidities for the cats in the diseased group

Age is expressed as median (range); the other groups are expressed as n (%)
BCS = body condition score; SBU = subclinical bacteriuria; GID = gastrointestinal disease; HD = hepatic disease; CKD = chronic kidney disease; DM = diabetes mellitus; HT = hyperthyroidism
SBU was detected in 11 cats (6.1%). Of the 11 culture-positive cats, nine (81.8%) were female and two (18.2%) were male. Of the female culture-positive cats, 1/9 was diagnosed with GID, 3/9 with HD, 1/9 with DM, 1/9 with a miscellaneous disorder and 3/9 were healthy. Of the two culture-positive male cats, one was diagnosed with CKD and one was diagnosed with DM. All positive cultures displayed growth of ⩾1000 colony forming units (CFU)/ml and were monocultures with the following species distribution: Escherichia coli (n = 3), Streptococcus species (n = 3), Staphylococcus species (n = 2), Corynebacterium species (n = 1), Enterococcus faecalis (n = 1) and Micrococcus luteus (n = 1).
Female cats had a significantly increased risk of SBU compared with male cats (Table 3). An increased risk was not found for cats with BCS ⩾6 (overweight/obese) or for obese cats (BCS ⩾8) vs the remaining population. Age and overall health status (healthy vs diseased) were not significant variables in the model.
Results for the risk factors being overweight/obesity, sex, group and age expressed as odds ratio (OR) and relative risk (RR) with a 95% confidence interval (CI)

Statistically significant
Of the comorbidities tested (CKD, HT, DM, HD and GID), only HD was found to be a significant risk factor for SBU (Table 4).
Results for the evaluated comorbidities as risk factors for subclinical bacteriuria expressed as odds ratio (OR) and relative risk (RR) with a 95% confidence interval (CI)

GID = gastrointestinal disease; HD = hepatic disease; CKD = chronic kidney disease; DM = diabetes mellitus; HT = hyperthyroidism
Statistically significant
Discussion
The main objective of the present study was to investigate if being overweight and/or obesity are associated with an increased risk of SBU in cats, an association that we could not demonstrate in our study population.
In dogs, Wynn et al 10 found an increased prevalence of subclinical bacteriuria in individuals that were morbidly obese, as determined by dual-energy X-ray absorptiometry (DEXA) (fat percentage >45%), vs those that were lean, overweight or moderately obese, 13 indicating that the increased risk is linked to being severely overweight. There are notable terminological and methodical differences between our study and the one in dogs, the most important being that BCS was used in our study for assessment and graduation of obesity, as compared to DEXA scanning used in the study by Wynn et al. 10 Although these differences preclude direct comparison between the studies, it is worth mentioning that, in our study, obesity (BCS ⩾8) was found in only 8.9% of the cats (n = 16/179), the majority of which were male (n = 11/16). In comparison, 60% of morbidly obese dogs (body fat >45%) were female in the study by Wynn et al. 10 It is possible that in our study, a potentially increased risk of SBU posed by being severely overweight (BCS ⩾8) may have been hidden by the decreased risk of SBU associated with male sex.
The predominance of male cats in the obese population is in agreement with prior reports showing that neutered male cats are at an increased risk of obesity. 22 This is in contrast to dogs, where obesity is more often associated with female sex. 22
We did not investigate if being underweight (as determined by a low BCS) was associated with an increased risk of SBU in our population, as this was not our a priori hypothesis. However, some studies have found a reverse association between weight and risk of SBU/UTI in cats. In a retrospective study including 155 cats with cystitis or SBU and 186 control cats, decreased body weight was found to be a significant risk factor for SBU and/or cystitis, 23 and similar results were found in the more recent retrospective study of feline SBU by Puchot et al, 5 in which lower body weight was associated with a positive quantitative bacterial culture. Specifically investigating the association between being underweight and SBU and/or UTI in cats could be of interest in future prospective studies.
In our study, diseased cats had an increased OR of 1.6 vs healthy cats, but this finding did not reach statistical significance. In earlier reports, SBU was reported in 11–23%6,7 of cats suffering from HT, DM or CKD, suggesting that those conditions may pose an increased risk of SBU. However, age-matched controls were not included in these reports, and a point prevalence of SBU of 10–13% was found in a recent longitudinal study of cats with a similar age distribution. 8 In our study, 34.6% of cats had either HT, DM or CKD, but an increased OR was only demonstrated for cats with DM, and this finding did not reach statistical significance. Other studies of mixed populations of cats have also failed to demonstrate a statistically significant increased risk associated with these conditions, except for the study by Puchot et al, 5 which showed an increased risk of SBU in cats with CKD vs a combined control group of cats, including those with lower urinary tract disease. 5
Unexpectedly, we found HD to be a significant risk factor for SBU. To our knowledge, this has not been reported in previous studies of cats. However, in humans, several studies have reported a high prevalence of asymptomatic bacteriuria and UTI in cirrhotic patients.24–27 The pathophysiology behind this association has not yet been elucidated. Investigating several associations carries the innate risk of committing a type I error, and it cannot be ruled out that the association between HD and SBU in our study is a random finding rather than a true association.
We found female sex to be a significant risk factor for SBU, which is a consistent finding in all studies.4,5,7,8 Most likely, this is owing to female cats having a relatively short and wide urethra anatomically located close to the anus. 28 The finding of females being more likely to have SBU is also in accordance with studies of SBU in dogs,29,30 and asymptomatic bacteriuria in humans. 31
Age was not a significant risk factor for SBU in this study. A possible explanation for this may be the selection of middle-aged to older cats (⩾6 years) masking an association between increasing age and SBU. Results of previous studies regarding the impact of age on SBU in cats are conflicting. Litster et al 4 found culture-positive cats to be significantly older than culture-negative cats; likewise, increasing age was found to be a significant risk factor for SBU in female cats in a study of cats with CKD. 7 In contrast, more recent studies, including a large retrospective study of a mixed cat cohort 5 and a prospective longitudinal study of elderly cats, 8 have not identified increasing age as a significant risk factor for SBU. However, as in our study, the latter study by White et al 8 selected animals above a certain age (⩾7 years), whereby a potential impact of increasing age could have been masked.
The prevalence of SBU in our study population was 6.1%, which is fairly similar to the prevalence of 7.9% found in the recent retrospective study of 368 cats by Puchot et al. 5 However, it is in contrast to earlier studies investigating SBU in mixed cat populations (containing healthy and sick individuals). The Australian medical record study of 135 cats by Litster et al reported a substantially higher prevalence of 28.8%, 4 and a more moderate point prevalence of 10–13% was reported in the longitudinal cohort study of 67 elderly colony cats by White et al. 8
While there is no single obvious explanation for the huge variation in reported prevalence between studies, several differences in study design and study populations may contribute. Although our study population is a convenience sample of cats visiting a teaching hospital, the selection bias may be slightly lower than in the other studies. This was a prospective enrolment study with bacterial urine culture performed for the purpose of the study and not at the discretion of the attending clinician. Twenty-eight percent (n = 50/179) of our study population were cats actively recruited from the outside community to participate in other ongoing studies. Differences in study population not related to study design include a lower median age of 10 years in our study vs 14 years and a female/male ratio of 0.86 vs 1.4 in the Australian study. 4 Finally, there could be differences in sample handling and interpretation of positivity vs contamination. However, this is unlikely as all positive culture samples were characterised by substantial growth of ⩾1000 CFU/ml in all of the mentioned studies.4,5,8
This study is the largest prospective study on SBU in cats to date. However, owing to proportional differences between cats at risk and cats not at risk being lower than expected, the study lacks power for several of the risk factors investigated, and this constitutes the most important limitation of the study. Continuation of the study to obtain adequate power was not a feasible option based on post-hoc sample size calculations performed at the end of the study period. Applying a significance level of alpha = 0.1 and beta = 0.2, we estimated it would require the inclusion of 540 cats to prove that the decreased OR and RR for SBU in overweight cats found in our study are true. To prove an increased risk of SBU in cats with comorbidities (disease) and cats with DM, it would require 8985 and 1867 cats, respectively, numbers that were not realistic to obtain within the current study set-up.
Another limitation of this study is the body composition being evaluated by BCS rather than a more objective measurement such as DEXA, and the BCS being performed by different investigators. BCS is a rela-tively subjective parameter, and, accordingly, variation between investigators can be expected. However, the majority of cats included were body condition scored by the principal investigators (FSM, INK) or two of the co-authors (PVB, LMP).
Conclusions
The prevalence of SBU in middle-aged and elderly cats was lower (6.1%) than previously reported for similar mixed populations. Being overweight or obese were not identified as predisposing factors. Female sex and HD were the risk factors significantly associated with SBU. The latter has not been previously reported, and further studies are needed to confirm the association.
Footnotes
Author note
The study was performed at the Department of Veterinary Clinical Sciences, University of Copenhagen, Denmark. This paper was presented in part, as a poster, at the 2018 ECVIM congress. 32 The data set analysed during the current study is available from the corresponding author upon request.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animal(s) only (owned or unowned), and followed established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care. Ethical approval from a committee was not necessarily required.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work for the procedure(s) undertaken. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
