Abstract
Objectives
The aim of this study was to assess the effects of famciclovir administration in cats with spontaneously acquired acute upper respiratory tract disease.
Methods
Twenty-four kittens with clinical signs of acute upper respiratory tract disease were randomly allocated to receive doxycycline (5 mg/kg PO q12h) alone (group D; n = 12) or with famciclovir (90 mg/kg PO q12h; group DF; n = 12) for up to 3 weeks. Clinical disease severity was scored at study entry and daily thereafter. Oculo-oropharyngeal swabs collected at study entry and exit were assessed using quantitative PCR for nucleic acids of feline herpesvirus type 1 (FHV-1), feline calicivirus (FCV), Chlamydia felis, Bordetella bronchiseptica and Mycoplasma felis.
Results
The median (range) age of cats was 1.5 (1–6) months in group D vs 1.6 (1–5) months in group DF (P = 0.54). Pathogens detected in oculo-oropharyngeal swabs at study entry included FCV (n = 13/24; 54%), M felis (n = 8/24; 33%), FHV-1 (n = 7/24; 29%), C felis (n = 7/24; 29%) and B bronchiseptica (n = 3/24; 12%). Median (range) duration of clinical signs was 11.5 (3–21) days in group DF and 11 (3–21) days in group D (P = 0.75). Median (range) total disease score at the end of the study did not differ between groups (group D 1 [1–1] vs group DF 1 [1–3]; P = 0.08).
Conclusions and relevance
This study revealed no significant difference in response to therapy between cats treated with doxycycline alone or with famciclovir; cats improved rapidly in both groups. However, identification of FHV-1 DNA was relatively uncommon in this study and clinical trials focused on FHV-1-infected cats are warranted to better evaluate famciclovir efficacy.
Introduction
Feline herpesvirus type 1 (FHV-1), an enveloped, double-stranded alphaherpesvirus, is an important cause of morbidity, and sometimes mortality, as a result of upper respiratory tract disease in cats. Clinical signs vary in severity, but include fever, depression, anorexia, ocular or nasal discharge, sneezing, conjunctival hyperemia and sometimes keratitis. Secondary bacterial infections are also common. Acutely infected cats and those that are latently infected but undergoing viral reactivation are the most important sources of infection. 1 Other pathogens involved in feline upper respiratory tract infections include feline calicivirus (FCV), Bordetella bronchiseptica, Chlamydia felis and Mycoplasma felis and influenza virus.2–5 Herpetic disease and viral shedding are of particular importance within multi-cat environments, especially shelters, where in one study 53% of cats shed FHV-1 within 1 week of admission. 2 Even in home-based foster care programs, clinically affected cats are more likely than healthy cats to shed FHV-1. 6
Diagnosis of herpetic disease is often presumptive based on clinical signs. When a laboratory diagnosis is sought, PCR is typically preferred over traditional techniques, such as virus isolation, owing to its higher sensitivity and the rapidity with which results are available.7–10 Two recent studies have evaluated sampling sites, showing that sampling multiple sites increases the rate of PCR detection of FHV-1 DNA, 11 and that there is no difference in detection rates for FHV-1 among sites, including the tongue, oropharynx, conjunctiva and nares. 12 Interpretation of PCR results is, however, complicated by the fact that clinically normal cats shed FHV-1. 8 Quantification of FHV-1 correlated with severity of histopathologic changes in nasal epithelium from shelter cats with acute rhinitis, suggesting that quantitative PCR (qPCR) might be useful in differentiating between cats with latent or active FHV-1 infections. 13 Conversely, another study of cats with signs of acute ocular and upper respiratory infections showed that FHV-1 qPCR results were not predictive of response to therapy with cidofovir ophthalmic drops. 14 qPCR has been used to assess the efficacy of some antiviral drugs. 15
Management of acute infectious upper respiratory disease in cats traditionally relies upon supportive care and treatment of secondary bacterial infections. 1 The efficacy of some specific antiviral therapies has also been reported. 16 Famciclovir is an oral prodrug of penciclovir, a nucleoside deoxyguanosine analogue with excellent efficacy against FHV-1. In cats experimentally infected with FHV-1, oral administration of famciclovir was associated with significant reductions in clinical disease scores, histologic conjunctivitis scores, serum FHV-1 antibody titers, serum globulin concentrations and conjunctival FHV-1 DNA and RNA loads. 17 Cats treated with famciclovir also showed increases in body weight. 17 Famciclovir administration has also been described in retrospective studies of cats with spontaneous disease believed to be due to FHV-1 infection.18,19 However, these were uncontrolled studies where FHV-1 infection was not confirmed and famciclovir has not been prospectively evaluated in naturally infected FHV-1 cats. Few adverse effects have been associated with the use of this drug in cats and it is regarded as generally safe.17,18,20
This study was designed to assess clinical and virologic effects of famciclovir administration to cats with naturally acquired infectious upper respiratory disease. We hypothesized that using famciclovir would reduce clinical disease severity and the duration of clinical signs in FHV-1-infected cats.
Materials and methods
Approval for this study was obtained from the Institutional Animal Care and Use Committee (#19021) and owners provided informed written consent regarding their cat’s involvement. Cats with clinical signs of infectious upper respiratory disease of less than 30 days’ duration were prospectively recruited to this study from local rescue organizations that use home-based foster care programs in northern California, USA. Therefore, most cats in this study briefly transited through local shelters prior to study entry. More than one kitten from affected litters could be enrolled. Cats previously treated for upper respiratory tract infection were included if they had not responded to prior treatment and any treatment was of less than 7 days’ duration. Any concurrent health conditions were documented and cats excluded if these were severe enough to require hospitalization. Estimated age, breed, sex, neuter status and vaccination history were recorded on study entry. When known, the presence of feline immunodeficiency virus (FIV) antibodies or feline leukemia virus (FeLV) antigens was also recorded.
At study entry (day 0), all cats were randomly assigned to one of two treatment groups. Randomization was performed using Microsoft Excel (2018) for the generation of random numbers; odd numbers were assigned to group D and even numbers to group DF.Cats in group D received 5 mg/kg doxycycline monohydrate suspension (5 mg/ml Doxycycline for Oral Suspension USP; Lupin Pharmaceuticals) and placebo (lactose powder), each administered orally q12h. Cats in group DF received 5 mg/kg doxycycline suspension and 90 mg/kg famciclovir, each administered orally q12h. The intent was to treat all cats for at least 7 days beyond the resolution of clinical signs. Doxycycline monohydrate suspension was reconstituted with water according to the manufacturer’s instructions. According to the package insert, this suspension was stable for 14 days at room temperature. Famciclovir was prepared by grinding commercially available tablets (Famciclovir 250 mg tablets; Teva Pharmaceuticals Industries) into a fine powder that was then formulated into capsules containing 22.5, 45, 90, 135 or 180 mg famciclovir. Lactose powder (placebo) was delivered using identical capsules filled to the same volume as those containing famciclovir. Owners of cats in both groups were asked to mix the contents of the capsule their cat was assigned with doxycycline suspension in a small container and administer the total volume orally immediately after mixing. Owners were masked to capsule contents, with unmasking performed at study end or if any adverse effects were reported. Owners of cats in group D were offered the opportunity to have their cats cross over into group DF if clinical signs persisted for more than 7 days.
At study entry and exit, each cat underwent a complete physical examination performed by one of two investigators (LK or LRJ), body weight and body condition score were recorded and an oculo-oropharyngeal swab was obtained using a sterile cotton-tipped applicator (Fisherbrand polyester-tipped applicators; Fisher Scientific). One drop of 0.5% proparacaine hydrochloride ophthalmic solution was applied to both eyes. The swab was first rolled gently in the conjunctival sac of both eyes and then in the oropharynx. Swabs were refrigerated for up to 48 h prior to submission to a commercial laboratory for assessment using qPCR specific for nucleic acids of FHV-1, FCV, C felis, B bronchiseptica, M felis and H7N2 influenza A virus (Feline Upper Respiratory Dis-ease RealPCR Panel, test code 2512; IDEXX Laboratories).At study entry and exit, owners completed a questionnaire regarding severity, duration and history of clinical signs of upper respiratory tract disease (see the Appendix in the supplementary material). This questionnaire used a semiquantitative scoring system ranging from 1 (mild) through 10 (severe) modified from one published by this group in related studies. 21 Throughout the study, disease scores were assessed once daily by owners using a published scoring system. 22 Owners were trained in disease scoring, including provision of a visual scale with examples of each score for ocular and nasal signs. After study exit, there was no ongoing follow-up of included cats.
For data analysis, an adequate response to therapy was defined as a reduction in owner-perceived severity of clinical signs by at least one grade (3 points on the scale). A priori power analysis conducted assuming initial client scores in the moderate-to-severe range and treatment-related reduction to a mild-to-moderate score 23 suggested that 25 cats per group would afford a power of 0.80 with probability of a type I error of 0.05 (www.statisticalsolutions.net). However, this analysis did not consider differential rates of FHV-1 detection and likely underestimated the number of cats required to demonstrate significance.Categorical responses were compared between treatment groups at the onset and end of the study using Fisher’s exact test. Clinical scores were assessed for normality using D’Agostino and Pearson omnibus normality testing. For normally distributed data, Student’s t-test was used to assess differences between groups. Non-parametric data were compared using the Mann–Whitney U-test. Prevalence of positive vs negative qPCR results for upper respiratory pathogens was compared between each treatment group at each time point using Fisher’s exact test. The number of pathogens detected on PCR was correlated with day 1 clinical scores and number of days in treatment by use of linear regression. Statistical analyses were performed using a commercially available software package (GraphPad Prism version 5.0f) and significance was set at P <0.05.
Results
A total of 32 cats were entered into the study. However, five were subsequently excluded from data analysis owing to incomplete data (one cat from group DF) or failure to return for clinical assessment (three cats from group D and 1 cat from group DF). One cat from group D died 4 days after study entry despite resolution of clinical signs. The cause of death was unknown and a necropsy was not performed. Two cats in group D failed to show a response to therapy within 7 days, group assignment was unmasked, and they were moved into group DF. Their data were considered separately to those of the other 24 cats. The remaining 24 cats were randomized equally into the two treatment groups. Enrolment was discontinued early when interim analysis did not reveal significant differences in response to therapy between treatment groups.
Considering all cats jointly, 12/24 (50%) were intact females, 8/24 (33%) were intact males, 3/24 (13%) were castrated males and 1/24 (4%) was a spayed female. The majority (96%) were domestic shorthair (n = 21), mediumhair (n = 1) or longhair (n = 1) cats; the remaining cat was a Himalayan cross. At study entry, median (range) age for all cats was 1.5 (1–6) months, body weight was 0.7 (0.2–2.3) kg and body condition score was 4/9 (3/9–5/9). No significant differences in demographic data were detected between cats assigned to group D or DF (Table 1). Antibodies to FIV were not detected in the five cats tested and FeLV antigens were not detected in the 11 cats tested. The majority of cats (n = 16/24; 67%) had been vaccinated subcutaneously using a modified live vaccine against FHV-1, FCV and feline parvovirus at a median (range) time of 7 (1–31) days prior to study entry.
Summary data for 24 cats with spontaneous acute infectious upper respiratory disease receiving 5 mg/kg doxycycline suspension and 90 mg/kg famciclovir administered PO q12h (group DF; n = 12) or 5 mg/kg doxycycline suspension and placebo (lactose powder) administered PO q12h (group D; n = 12)

qPCR = quantitative PCR
Five cats had received orally administered doxycycline at an unknown dose for 24 h prior to study entry; these cats were assigned to group D (n = 3) or group DF (n = 2). Three cats had received oxytetracycline–polymyxin B ophthalmic ointment for 24 h prior to study entry; these cats were assigned to group D (n = 2) or group DF (n = 1). One cat had received polymyxin B, bacitracin and neomycin ophthalmic ointment for 24 h prior to study entry; this cat was assigned to group D.
Common reasons for presentation included sneezing (n = 24/24; 100%) and ocular (n = 23/24; 96%) or nasal (n = 22/24; 92%) discharge. Other reasons included hyporexia (n = 1), lethargy (n = 3) or corneal ulceration (n = 1). No cat presented for poor response to prior treatment. Information on disease duration was available for 16 cats, and the median (range) disease duration prior to study entry was 7 (1–24) days, with no significant difference detected between group D and group DF (Table 1). Using the 10-point scale provided, median (range) owner-rated disease severity score at study entry for the 24 included cats was 7 (1–9) for ocular discharge, 6 (1–10) for sneezing, 6.5 (1–10) for nasal discharge and 4 (1–9) for difficulty or noisy breathing. No significant differences were detected between disease severity scores at study entry for cats in group D or group DF (Table 1).
Median (range) study period for included cats did not significantly differ for cats in group DF (14 [11–21] days) or those in group D (19.5 [4–21] days; P = 0.92). According to owner-completed disease score sheets, cats demonstrated clinical signs for a median (range) of 11.5 (3–21) days in group DF and 11 (3–21) days in group D (P = 0.75). Using the 10-point scale provided, median (range) owner-rated disease severity score for all cats at study exit was 1 (1–3) for ocular discharge, 1 (1–1) for sneezing, 1 (1–1) for nasal discharge and 1 (1–1) for difficulty or noisy breathing in group D; and was 1 (1–2) for ocular discharge, 1 (1–4) for sneezing, 1 (1–1) for nasal discharge and 1 (1–3) for difficulty or noisy breathing in group DF. Median (range) total disease score at the end of the study did not differ between cats in group D (1 [1–1]) and those in group DF (1 [1–3]) (Figure 1; P = 0.08).

Comparison between median total clinical disease scores at study entry, day 7 and study end in 24 cats with spontaneous acute infectious upper respiratory disease receiving 5 mg/kg doxycycline suspension and 90 mg/kg famciclovir PO q12h (group DF; n = 12) or 5 mg/kg doxycycline suspension and placebo (lactose powder) each administered PO q12h (Group D; n = 12). No difference was detected between groups at study entry, day 7 or study end (P >0.05)
At study exit, median (range) body weight for all cats was 1 (0.5–2.8) kg with no significant difference detected between treatment groups (P = 0.40; Table 1). Likewise, median (range) body weight gain did not significantly differ between cats in group D (0.38 [0.20–0.55] kg) and those in group DF (0.32 [0.24–1.12] kg; P = 0.75).
The frequency with which known respiratory pathogens were detected using qPCR did not significantly differ for cats from each treatment group (Table 1). Considering all cats collectively, pathogens most commonly detected in oculo-oropharyngeal swabs at study entry were FCV (13/24 cats; 54%), M felis (n = 8/24; 33%), FHV-1 (n = 7/24; 29%), C felis (n = 7/24; 29%) and B bronchiseptica (n = 3/24; 12%). Influenza virus was not detected in any cat. Combinations of infectious agents detected are shown in Table 2. The median (range) number of organisms detected per cat was 1 (0–4); 45.8% of cats had more than one infectious agent detected.In the seven cats in which FHV-1 was detected at study entry, median (range) calculated number of viral copies was 1433 (0.9–8168) thousand per swab. At study exit, FHV-1 DNA was detected in swabs from 2/24 (8%) cats: one from group DF in which the calculated viral copy number decreased from 2761 thousand per swab at entry to 0.1 thousand per swab at exit; and one from group D in which the calculated viral copy decreased from 2323 thousand per swab at entry to 0.3 thousand per swab at exit. At study exit, FCV RNA was detected in two cats negative at study entry and M felis DNA in two cats negative at study entry. There was no correlation between the number of pathogens isolated and day 1 clinical disease score (Figure 2; P = 0.78) or number of days of treatment until clinical resolution of signs (P = 0.75) or total number of days of treatment (Figure 3; P = 0.68).
Combinations of infectious agent nucleic acids detected by quantitative PCR (qPCR) assessment of oculo-oropharyngeal swabs collected at study entry from 24 cats with spontaneous acute infectious upper respiratory disease
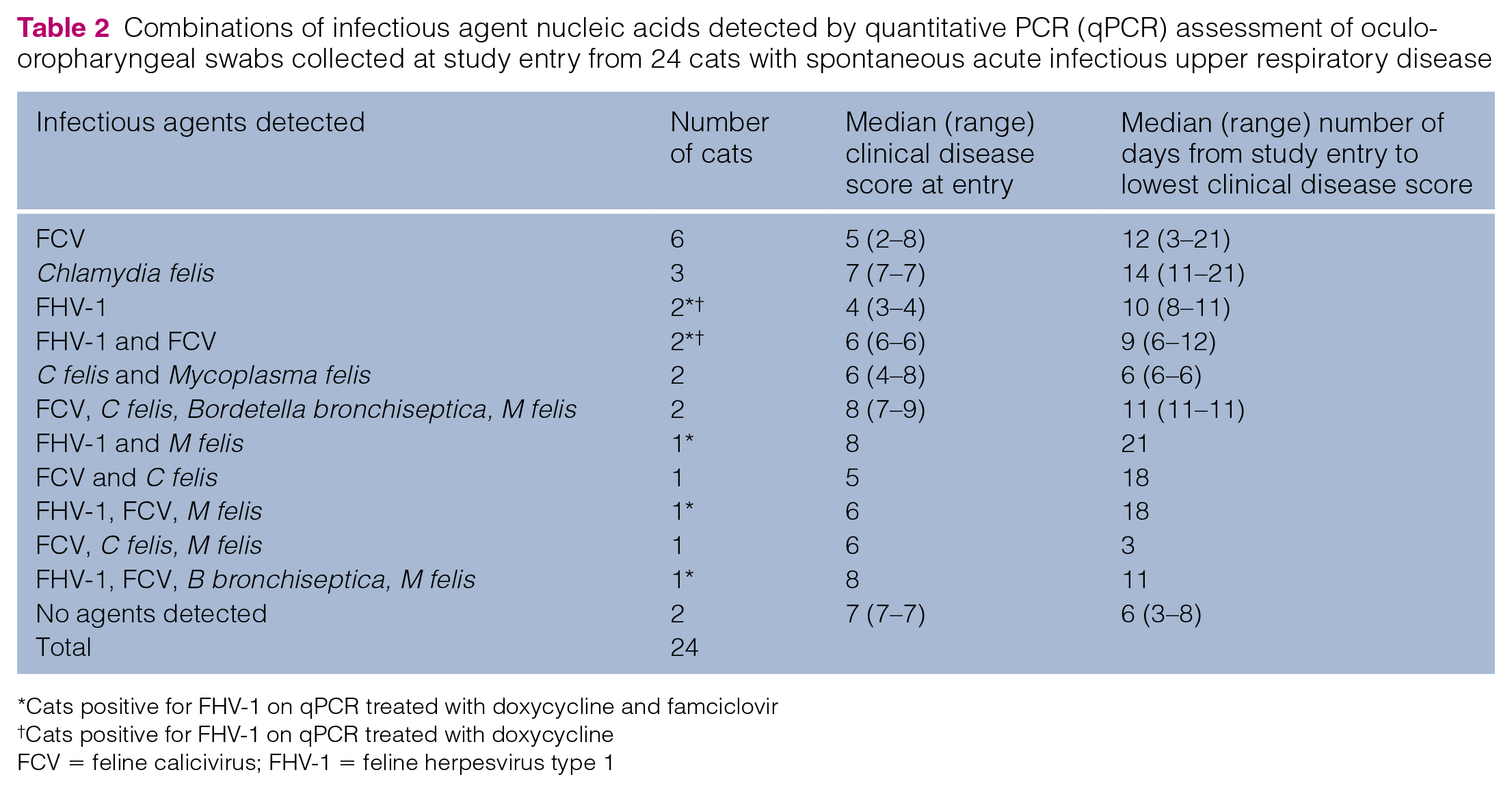
Cats positive for FHV-1 on qPCR treated with doxycycline and famciclovir
Cats positive for FHV-1 on qPCR treated with doxycycline
FCV = feline calicivirus; FHV-1 = feline herpesvirus type 1
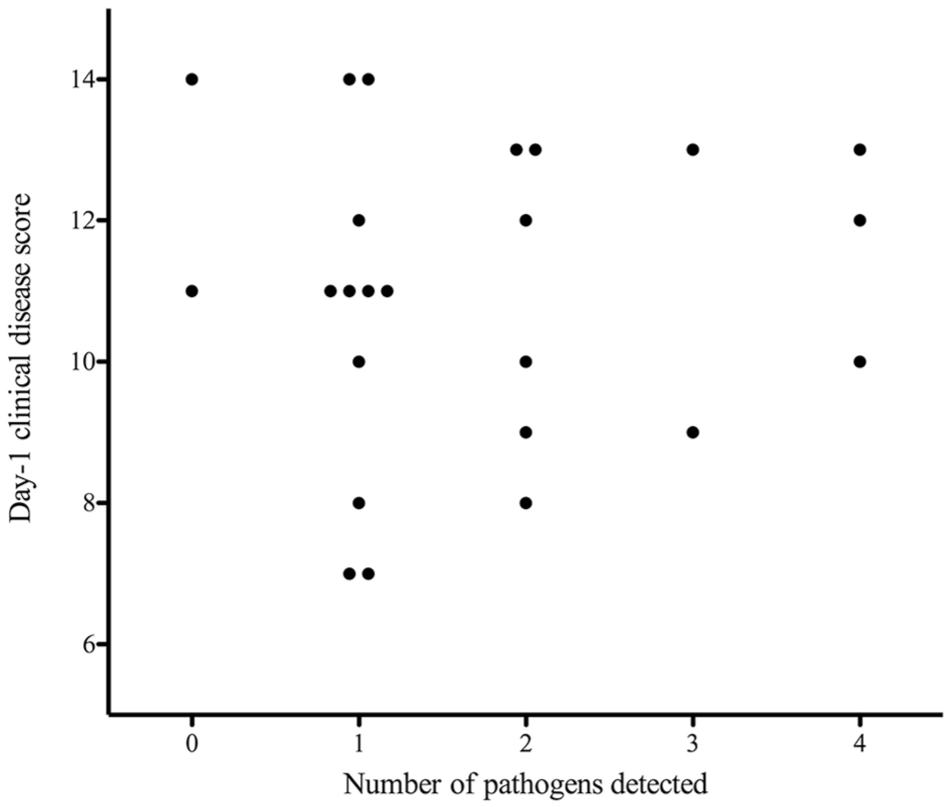
Clinical disease score and number of pathogens detected in an oculo-oropharyngeal swab from 24 cats with spontaneous acute infectious upper respiratory disease at study entry

Duration of treatment and number of pathogens detected in an oculo-oropharyngeal swab at study entry from 24 cats with spontaneous acute infectious upper respiratory disease
Two cats crossed over from group D to group DF owing to poor response to 7 days of therapy. Both were 6-week-old domestic shorthair cats – one female intact and one male castrated. Oculo-oropharyngeal swabs collected from these two cats contained nucleic acids of FCV and M felis for one cat and FCV only for the other. Clinical disease scores by day for these two cats are presented in Figure 4.

Total clinical disease score by day for two cats that crossed over from group D to group DF on day 8 (cat 1) and day 7 (cat 2). Arrows indicate the days on which each cat crossed over
Discussion
The present study did not identify significant differences in outcome when cats with clinical signs of spontaneous infectious upper respiratory disease were treated with doxycycline and famciclovir or with doxycycline and placebo. Because we considered it unethical to have an untreated group, our data might reflect the potentially large proportion of young cats with infectious respiratory disease that improve quickly regardless of therapy. 24
Famciclovir has been found to be effective for treatment of cats following experimental inoculation with FHV-1. 17 In that study, famciclovir-treated cats had lower disease scores and higher percentage changes in weight than cats not receiving famciclovir. 17 The difference in findings in the present study, where cats had naturally acquired, acute infectious upper respiratory disease, could be due to detection of more than one infectious agent in many cats, lower prevalence of FHV-1 detection in these cats, or both. Interestingly, two cats negative for FHV-1 DNA did not respond to doxycycline alone, but did improve rapidly after starting famciclovir. This could reflect false-negative qPCR results for FHV-1 or that these cats required more time to improve rather than a change in therapy.
In a retrospective study of cats treated with famciclovir for ocular, respiratory or dermatologic disease consistent with FHV-1 infection, cats receiving famciclovir at 90 mg/kg PO q8h had a shorter median time to improvement and greater magnitude of improvement than cats receiving 40 mg/kg PO q8h. 21 However, given that a pharmacokinetic study showed that administration of oral famciclovir at a dose of 40 or 90 mg/kg in healthy cats resulted in similar maximum concentrations and time to maximum concentrations, a lower dose of famciclovir could have been considered in the present study. 25 In the retrospective study of cats treated with famciclovir, cats had clinical signs for a longer period before starting famciclovir (median 40 days) 21 than did cats in the present study (median 7 days). This suggests famciclovir might be more likely to benefit cats with more chronic or recurrent clinical syndromes. Prospective evaluation of famciclovir administration to cats with more chronic clinical signs might better elucidate its efficacy in such cases.
Although cats entered into the present study represented a clinically relevant population (ie, young cats presenting with spontaneous signs of acute infectious upper respiratory disease), FHV-1 DNA was detected using qPCR performed on an oculo-oropharyngeal sample in only 29% of cats. Although PCR is more sensitive than other tests for FHV-1, such as indirect immunofluorescence and virus isolation, 9 false-negative tests could have contributed to decreased prevalence in our study population. 11 Conversely, several PCR assays detect vaccine virus and cannot discriminate it from wild-type FHV-1 in cats. 8 This might be relevant in the present study because most cats were vaccinated a median of 7 days before study entry, although this is also affected by vaccine type. In one vaccine challenge study, kittens receiving a modified live vaccine against FHV-1 did not have FHV-1 DNA detected on PCR, whereas some kittens vaccinated using a modified live intranasal vaccine against FHV-1 did. 26 In another study conducted in home-based foster care programs, there was no difference in the detection of FHV-1 DNA between cats that had been recently vaccinated and those that had not. 6 The effect of false-positive qPCR results for FHV-1 in the present study is unknown.
A previous study evaluated upper respiratory tract pathogen prevalence in cats from four different environments. 6 The prevalence with which infectious organism nucleic acids were detected in cats with clinical signs in home-based foster care programs was similar to that presented here for FCV (42% vs 54% in the present study), FHV-1 (23% vs 29%), C felis (24% vs 29%) and B bronchiseptica (18% vs 12%). However, the proportion of cats from which M felis DNA was detected was notably lower in our population (33%) than the other study (59%). 6 It is worth noting that qPCR failed to detect nucleic acids from any tested pathogen in swabs obtained from the two cats with the highest clinical disease scores at study entry. Given that their clinical signs were compatible with acute infectious upper respiratory disease, it is possible that this reflects a falsely negative qPCR result or that these cats were infected with an organism not detected by this assay. Because the time of study entry preceded the time at which retroviral testing was routinely performed by home-based foster care organizations in this study, retroviral status was unknown for some cats evaluated. As a result, it was not possible to determine the impact of retroviral status on the prevalence of detection of infectious organism nucleic acids or response to treatment with famciclovir.
Conclusions
This study in cats with acute infectious upper respiratory disease revealed no significant difference in response to therapy with doxycycline alone vs doxycycline with famciclovir, with cats in both groups improving rapidly. However, FHV-1 DNA was detected relatively uncommonly in this study and clinical trials focused on FHV-1-infected cats are warranted to better evaluate effects of famciclovir. In addition, clinical trials focusing on cats with more chronic FHV-1 infections are warranted.
Supplemental Material
Appendix
Owner survey at study entry
Footnotes
Acknowledgements
The authors are grateful to Dr Ann Cooper for formulating the visual scale of nasal and ocular signs used by owners in this study.
Supplementary material
The following file is available online: Appendix: Owner survey at study entry.
Conflict of interest
One of the authors (CML) works for IDEXX Laboratories. The authors declared no other potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by a residency research grant provided by the Center for Companion Animal Health, School of Veterinary Medicine, University of California, Davis.
Ethical approval
This study involved the use of client-owned animals only, and followed internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care. The study had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal guardian of all animals described in this study for the procedures undertaken.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
