Abstract
Objectives
The aim of this study was to conduct a comprehensive assessment of feline infectious upper respiratory tract infection (URTI) and disease (URTD) in Australian cats.
Methods
Laboratory data demonstrating URTI from feline URTD multiplex PCR panel (feline herpesvirus 1 [FHV-1], feline calicivirus [FCV], Bordetella bronchiseptica, Chlamydophila felis, Mycoplasma felis and H1N1 influenza) submissions in Australia (2013–2015) were obtained. For comparison, reports of feline URTD during the same time period were sourced from a voluntary companion animal disease surveillance system.
Results
A total of 3126 samples were submitted for testing; 1533 (49%) were positive. Of these, the most commonly detected agents were M felis (21.5%) and FCV (16.0%) alone, followed by FCV and M felis (13.4%) together as a respiratory infection complex, then FHV-1 (7.0%) alone. During the study period, there were 262 reports of 320 clinical feline URTD cases. Most cases (69%) were reported from New South Wales, <1 year of age (41%) and equally distributed between the sexes. Infection was more common in entire cats (69%) and most cases (55%) involved domestic shorthair cats. Of the 90 reports that had a known vaccination status, 63 had a vaccination history, 40 of which were recently vaccinated. Most (72%) feline URTD cases recovered from clinical disease. Both feline URTI and URTD were more common during winter months.
Conclusions and relevance
Feline URTI and URTD cause substantial impact in Australia, being most commonly associated with M felis and FCV infection. This information can be used by veterinarians to educate clients about prevention and management of this important infectious disease of cats.
Introduction
Feline upper respiratory tract disease (URTD) is most prevalent in stressful and crowded feline populations. Of the reported pathogens, the five most prevalent aetiological agents are feline herpesvirus-1 (FHV-1), feline calicivirus (FCV), Bordetella bronchiseptica, Chlamydophila felis and Mycoplasma felis. 1 In contrast, H1N1 influenza virus is rare in the feline population. However, recently, a feline influenza A virus (H7N2) was identified to cause respiratory disease in approximately 500 cats in animal shelters in New York. 2
FHV-1 is a DNA virus that causes rhinotracheitis in felid species worldwide. 3 Infected cats are lifelong carriers because the virus remains latent in the trigeminal ganglia post-recovery.4,5 Cats can develop conjunctivitis, corneal ulceration, nasal or ocular discharge and, less commonly, oral ulcerations.3,6 During stressful periods, carriers can re-shed the virus via nasal, oral and ocular secretions.
Previous epidemiological studies have estimated the prevalence of FHV-1 to range from 3–38%, depending on the population sampled.7–10 Vaccination against FHV-1 and FCV reduces the severity of clinical signs but does not prevent infection. 11 In multi-cat households, 20% of unvaccinated cats and 15% of vaccinated cats develop signs of URTD associated with FHV-1 and FCV or other respiratory agents. 12
FCV is an RNA virus that characteristically causes oral ulcerations. 6 Unlike FHV-1, cats can shed the virus for weeks to years post-infection in ocular, respiratory and oral secretions.5,10,13 The estimated prevalence of FCV ranges from 26–50% and while morbidity is high, mortality rates are generally low.4,7,8,14
M felis is a commensal organism of the feline upper respiratory tract. 14 Its role as a primary pathogen is incompletely understood. Opportunistic infection following respiratory epithelial damage caused by a primary pathogen is more typical.15,16 However, M felis has been isolated from cats with URTD in which no primary pathogen was detected, demonstrating a possible primary pathogenic role.15–17 Of cats presenting with URTD, an M felis prevalence of 46% has been estimated. 14 However, owing to its commensal origin, M felis has also been isolated in up to 20% of cats without URTD.7,14–16
B bronchiseptica is increasingly recognised as a URTD pathogen in cats. Bacteria are shed through oronasal secretions, and shedding can be intermittent with no clinical signs. 3 Clinical signs are non-specific and can range from mild (sneezing and nasal discharge) to severe (pneumonia).12,18 Unlike dogs, coughing is not a characteristic sign of infection in cats. Previous studies have estimated the prevalence of B bronchiseptica to range from 0–30%.7,12
C felis infection causes mild respiratory signs. It is less commonly implicated in feline URTD. Bacteria are shed in ocular discharge and clinical signs include conjunctivitis, hyperaemia and blepharospasm.3,12 Its prevalence has been estimated to range from 0–15%.7,12,19
The aim of the current study was to determine potential pathogens that might be responsible for feline upper respiratory tract infection (URTI) in Australian cats. To provide context, reports of clinical feline URTD during the same period were evaluated. This comprehensive assessment will allow veterinarians to educate clients about prevention and management of this disease.
Materials and methods
Animal ethics
No animals were used in this study. All data used were secondary data.
Data collection
Case data for this study was obtained from laboratory testing and from a passive surveillance system. The laboratory testing database provides a diagnostic result (positive or negative) for each submitted sample for the pathogens included in the multiplex quantitative PCR panel, namely FCV, FHV-1, B bronchiseptica, M felis, C felis and H1N1 influenza virus between January 2013 and November 2015. 20 Test results obtained only demonstrated infection (not disease) because no information on clinical signs was available. Each case had an accession number, assay code, assay name, case date, result (positive, negative, state, postcode and breed.
Disease Watchdog (DWD) is an Australian national surveillance system that allows for online data collation of diagnosed companion animal diseases.5,21 The DWD database was searched for cases of FCV and FHV-1 (together referred to as feline URTD) reported between 2013 and 2015. Available information for cases includes date, location, breed, age, sex, neutered status, case outcome, case diagnostics and vaccination history. Each record was included as one report (containing one or more cases). Subsequent reports of the same feline patient (eg, revisit appointment) for the same diagnosis were considered to be one report only. Owing to the infectious nature of feline URTD, cases reported in kittens as a litter were also considered to be one report only.
Data management
The data were managed in spreadsheets (Excel 2013; Microsoft). The laboratory testing data were sorted to identify the number of case submissions and the number of cats positive for an upper respiratory tract pathogen, as a proportion of the total number of submissions. Pivot tables were created to identify all cases that were positive for more than one pathogen, and the number of positive URTD cases by Australian postcodes. These same pivot tables were created for the surveillance dataset for the same timeframe. Cases from both data sources were assigned to a season based on the reported case date (summer: December–February; autumn: March–May; winter: June–August; spring: September–November).
Each report in the surveillance data set was checked for incorrect spelling, irrational numerical values, duplicate entries (cross-matching case date, patient name, clinic name, postcode and state), incorrect categorisation and incomplete information. Records with incomplete data fields were categorised as unreported or unknown for the purpose of this study, unless otherwise indicated. Cats that were reported to be strays were excluded. The variables extracted from the surveillance database were age, sex, neuter status, breed, vaccination status (at least one lifetime vaccination, unvaccinated or unknown) and case outcome. Vaccinated cats were further categorised as recent (vaccinated within 365 days of the case date) or incomplete (vaccinated at least once, >365 days from the case date).
Data analysis
Laboratory testing cases were grouped according to the following variables: accession number, assay code, assay name, case date, result, state, postcode and breed. Surveillance cases were summed and grouped according to the following variables: case outcome, age, sex, neuter status, breed and vaccination status. The case fatality rate was calculated by using the number of cats reportedly euthanased or died (numerator) as a proportion of the total number of reports: clinically recovered, euthanased or died (denominator).
Results
During the period between January 2013 and November 2015, 3126 samples were submitted for diagnostic testing; 1533 cats (49.0%) tested positive for at least one respiratory pathogen. During the same period, there were 262 reports (320 cases) of feline URTD made within the passive surveillance system.
Of the 1533 cases that tested positive for respiratory pathogens, the most commonly detected agents were M felis (21.5%) and FCV (16.0%) alone (Figure 1). This was followed by FCV and M felis (13.4%) together as a respiratory coinfection, then FHV-1 (7.0%) alone. Detection of B bronchiseptica and C felis alone was uncommon, and H1N1 influenza was not detected.
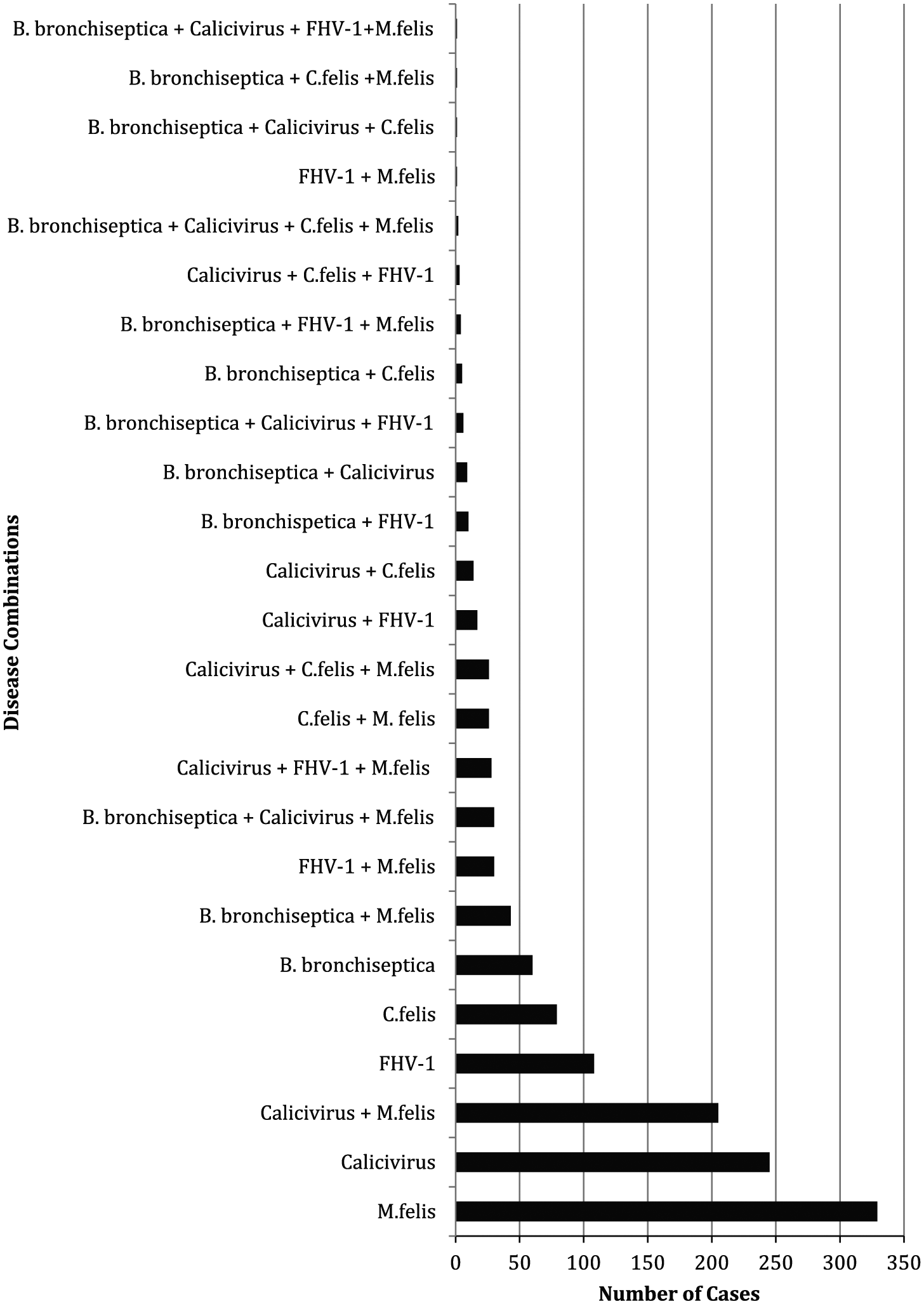
Common feline upper respiratory tract disease combinations in Australia, based on laboratory testing (RealPCR Panel) data, 2013–2015 (n = 1533). The results are based on the IDEXX Feline Upper Respiratory Disease RealPCR Panel. The pathogens tested by conventional PCR include feline herpesvirus-1 (FHV-1), feline calicivirus (FCV), Bordetella bronchiseptica, Chlamydophila felis, Mycoplasma felis and H1N1 influenza virus
Feline URTD reports during the same period as laboratory testing were mostly (n = 102/249; 41%) patients <1 year of age. Approximately equal proportions (48% vs 52%) of male and female cats were reported. Infection was more common (n = 180/262; 69%) in entire cats and diagnosis was mainly (n = 251/262; 96%) based on clinical presentation. Of the 104 reports that provided a vaccination status, 63 (61%) had a known vaccination history. However, only 40 (38%) were recently vaccinated, with disease being reported 0–14 days (11 cases), 15–28 days (three cases) and 29–365 days (26 cases) post-vaccination. Most (77/139; 55%) reports involved domestic shorthair (DSH) cats, and most (102/141; 72%) recovered from clinical disease.
The seasonality of feline URTI and URTD was similar. The proportion of samples submitted and tested positive for at least one respiratory pathogen was 21%, 27%, 31% and 21% in summer, autumn, winter and spring, respectively. These proportions for feline URTD reports were 18%, 29%, 34% and 19%, respectively.
Discussion
During the study period, the most common reports of infection were associated with M felis and FCV. Based on passively reported cases of feline URTD, this disease syndrome predominantly affects young, entire and DSH breed cats, most of which recover from clinical disease. Feline URTD is reported in previously vaccinated cats, and both infection and disease occurs more commonly during the winter months.
M felis was the most prevalent single pathogen identified in laboratory testing between 2013 and 2015. Assuming that veterinarians mostly submitted samples for testing from cats with clinical signs of URTD our findings are consistent with previous studies suggesting that M felis can be a primary URTD pathogen.15,16,22 However, this could be an overestimate of disease prevalence – M felis may have been an ‘innocent bystander’; other infectious pathogens not included in the PCR panel, or included in the PCR panel but not detected, could have been causing disease, or some cats might have had a non-infectious cause of URTD such as chronic rhinosinusitis, neoplasia, foreign body- or immune-mediated disease.15,16,22 Therefore, results should be interpreted with caution. Despite these caveats, M felis likely plays an important pathogenic role in feline URTD in Australia. Access to data on clinical history (unavailable in this study) would further help to clarify the role of M felis in feline URTD.
Following M felis, FCV (245/1533; 16.0%) was the second most commonly detected pathogen, with no other concurrent pathogens. Given the high prevalence of FCV, this finding is consistent with the virus’s stability and long survival in the environment. 13 In addition, FCV and M felis were detected as co-pathogens in 13% (n = 205/1533) of feline URTI cases. This might suggest that primary infection with FCV followed by secondary infection with M felis is a relatively common occurrence leading to the presentation of cats to veterinarians. However, some cats shed FCV continuously for months to years after infection with field or vaccine strains of virus.
Based on our surveillance data, feline URTD rarely causes death; however, euthanasia can be an outcome. Factors that influence an owner’s decision for euthanasia include animal welfare, finance, human–animal bond, infectiousness of recovered carriers and prognosis. 5 This reflects the significance of feline URTD in Australia’s domestic cat population.
Respiratory pathogens such as FCV, FHV-1, M felis, C felis and B bronchiseptica can be present in a subclinical carrier state; therefore, PCR results need to be interpreted with care.1,23 A positive PCR result does not confirm the viability of the respiratory pathogen and cannot distinguish between clinical illness, reactivated shedding, shedding due to vaccination or by healthy carrier cats.23,24 The use of qPCR allows for the stages of FHV-1 infection to be characterised. 23 A previous study concluded that there is a correlation between qPCR results and FHV-1 virus titres during early infection; however, the qPCR signal decreases with negative virus isolation during the final stages of infection. 25 A positive qPCR result is indicative of either active (high viral load) or latent (low viral load) FHV-1 infection depending on the range of virus detected per swab.20,25 A high viral load indicates active viral replication; however, vaccination with a modified live vaccine weeks prior to testing can also be detected, as can reactivated shedding during stress.
In the testing and surveillance databases, feline URTI and URTD were reported more commonly in winter. FHV and FCV, specifically, survive for longer periods of time on fomites in cool and moist conditions.13,26 As a result, indirect transmission is more likely to occur in winter. Other contributors to disease may include stress and reactivation of latent infection as a result of colder temperatures in winter.27,28
Approximately two thirds of surveillance reports recorded a vaccination history, with 38% reporting vaccination within the 12 months prior to presentation for feline URTD. The majority (65%) of recently vaccinated cats developed disease more than 28 days − but less than a year − after vaccination. Vaccinating against FHV-1 and FCV does not provide sterilising immunity, and disease in cats vaccinated 0–14 days previously might be the result of an adverse reaction, vaccination failure (due to the vaccine itself or an insufficient immune response) or an incomplete vaccination course. 29 Further studies are necessary to understand the association between feline URTD and vaccination status, particularly in cats vaccinated >14 days and <1 year prior to disease.
There were several limitations to this study. Ancillary data (eg, age and history) associated with laboratory testing were unavailable. For this reason, passive surveillance data were also sourced to provide more context to the epidemiology of feline URTD in Australia. However, the latter relies on voluntary reporting by veterinary clinics. This database contained substantially fewer feline URTD reports than the laboratory testing database but provided more detail on each case. Reports were almost exclusively diagnosed via clinical presentation, representing a different cohort of the feline population under veterinary care in Australia between 2013 and 2015. Diagnosis of feline URTD based on clinical signs alone lacks accuracy when compared with laboratory testing. 1 Under-reporting and incomplete reporting are also sources of bias in this study. Finally, the population at risk (cats presented to veterinarians for URTD) was unknown.
Conclusions
Feline URTI frequently involves more than one pathogen. Despite being vaccinated, cats can still present with URTD. However, client education about feline URTD and increased awareness of vaccination programmes might decrease the occurrence of feline URTD, especially those cases that involve FHV-1 and FCV. Future surveillance studies should focus on the role that environmental conditions play in the occurrence of feline URTD and its seasonal presentation.
Footnotes
Acknowledgements
The authors thank the veterinary clinics who reported data to the Disease Watchdog database, and also IDEXX laboratories for provision of the PCR data for this study.
Author note
This study represents a BVSc Honors dissertation submitted by the lead author to the Sydney School of Veterinary Science, The University of Sydney.
Conflict of interest
Disease Watchdog is owned by Virbac Animal Health Australia. However, no funding was received from Virbac Australia for this study. No funding was received from IDEXX.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
