Abstract
Objectives
The current study was designed to evaluate the prevalence and prognostic significance of multiorgan dysfunction syndrome (MODS) in cats with sepsis.
Methods
Cats hospitalised in the intensive care unit of a veterinary university hospital with a diagnosis of sepsis were prospectively enrolled and divided according to disease severity and outcome (survivors; non-survivors). The feline acute patient physiological and laboratory evaluation (APPLE) scores were calculated upon admission, as previously described. Specific criteria to identify selected organ dysfunction (hepatic, renal, respiratory, cardiocirculatory, haemostatic) were adapted from the available human and veterinary literature, and evaluated at baseline and at the end of hospital stay. MODS was defined as the presence of at least two dysfunctional organs simultaneously. Non-parametric statistics were used for comparisons. Univariate and multivariate regression analyses to evaluate significant risk factors for death were carried out. Correlations between variables were assessed by the Spearman’s rank correlation coefficient. Significance was set at P <0.05.
Results
A total of 43 cats with heterogeneous sources of sepsis were included. MODS was identified in 25/43 cats upon admission and in 32/43 cats at the end of hospital stay. Regression analyses showed a significantly elevated odds ratio for mortality for the presence of MODS, renal and cardiovascular dysfunction upon admission, as well as for the number of dysfunctional organs. The latter was the only variable retained by the model from the multivariate binary logistic regression analysis. Significant correlations were documented between the number of dysfunctional organs and the APPLE scores.
Conclusions and relevance
MODS is a frequent complication of feline sepsis, and is associated with worse outcomes. In particular, renal and cardiovascular dysfunction significantly increase the odds for death. Hence, systematic screening for organ dysfunction is advocated in cats with sepsis.
Introduction
multiorgan dysfunction syndrome (MODS), the progressive dysfunction of organ systems following an acute threat to systemic homeostasis, is a common complication of critical illness and a leading cause of mortality in human intensive care units (ICUs).1,2 Among the spectrum of critical care conditions, sepsis represents the major inciting cause for MODS. The inter-relationship between sepsis and MODS is supported by the recent re-definition of the sepsis syndrome as ‘the life-threatening organ dysfunction caused by a dysregulated host response to infection’. 2 Progression from sepsis to MODS has been investigated in dogs; similar to human findings, the number of dysfunctional organs has been associated with an increase in the mortality risk.3–5
Frequency and clinical manifestation of sepsis and MODS are less documented in cats; however, the reported mortality rate is elevated. 6 Biomarkers of feline sepsis, including meta-rubrocytosis, high circulating muscle enzyme activities (aspartate transaminase; creatine kinase), high band neutrophil percentage and elevated serum amyloid A (SAA) concentrations, have been investigated in this species.7–10 However, selected organ dysfunctions have been rarely characterised in septic cats,9–11 and studies systematically evaluating MODS in the course of feline sepsis are lacking.
The aim of the present study was to investigate and characterise organ dysfunctions and MODS in feline sepsis. We hypothesised that MODS is a complication of feline sepsis, and that its occurrence is associated with increased mortality.
Materials and methods
Animals
Critically ill cats with sepsis presented to the Veterinary University Hospital of Bologna and hospitalised in the ICU between October 2015 and September 2017 were prospectively included. The study was approved by the local Institutional Animal Care and Use Committee.
Cats were diagnosed with sepsis if they fulfilled at the time of presentation at least 3/4 feline systemic inflammatory response syndrome criteria (rectal temperature >39.7ºC or <37.8ºC, heart rate >225 beats/min [bpm] or <140 bpm, respiratory rate >40 breaths/min, and white blood cell count >19,500/µl or <5000/µl or band neutrophil fraction >5%), as previously reported, 7 and had a documented underlying infection. Presence of infection was confirmed by means of cytology, microbiology or molecular diagnostic methods. Cats discharged against medical advice or euthanased for financial reasons were excluded.
Data collection
Attending ICU clinicians were responsible for the clinical management of the patients included in the study. For each enrolled cat, the following data were recorded: signalment and body weight, lifestyle, vaccination history, presence of comorbidities, prior medical treatments, therapies administered during ICU stay, need of fluid resuscitation (types and volumes of intravenous fluids), blood products, vasopressors and positive inotropes, and duration of hospitalisation.
Upon admission, clinical findings, non-invasive blood pressure measurement (Minidop ES-100VX; Hadeco), pulse oximetry (Dash 3000 Patient Monitor; GE Medical Systems Information Technologies) and ultrasonography (Z5 Vet; Shenzhen Mindray Bio-Medical Electronics) for the evaluation of body cavity fluid scores, as previously reported, 12 were registered. Blood was collected by venepuncture with vacuum system according to standard operating procedures, and analysed within 1 h of ICU admission. The following analyses were performed: venous blood gas, including electrolytes and lactate (ABL 800 FLEX blood gas analyser; Radiometer Medical), complete blood cell count (ADVIA 2120; Siemens Healthcare Diagnostics) and microscopic evaluation of the blood smear, chemistry profile – including measurement of serum creatinine, bilirubin and SAA (Beckman Coulter-Olympus AU 480, LZ Test Eiken SAA [Eiken Chemical]) – prothrombin time (PT) and partial thromboplastin time (PTT) (BFT II; Siemens). The feline acute patient physiological and laboratory evaluation (APPLE) scores, APPLEfast and APPLEfull, respectively, were calculated, as previously described. 13
Cats were classified according to their outcome as survivors (alive to discharge) and non-survivors (died despite medical treatment or humanely euthanased because of moribund conditions). Cats euthanased for other reasons were excluded.
Organ dysfunction criteria
Criteria to define organ dysfunction were adapted from the available human and canine literature.2,3,14,15 Respiratory dysfunction was defined in presence of SpO2 <95% in room air, or if oxygen therapy or mechanical ventilation were needed. Hepatic dysfunction was defined as serum bilirubin >0.7 mg/dl in absence of haemolysis or biliary obstruction. Acute kidney injury (AKI) was defined as serum creatinine (sCr) >1.8 mg/dl and/or increase in sCr of ⩾0.3 mg/dl from baseline and/or oliguria (urine output <1 ml/kg/h over 6 h), and graded (I–V) according to the International Renal Interest Society (IRIS) staging system, proposed by Cowgill and accepted by the IRIS group. 15 Cardiovascular dysfunction was defined as hypotension (systolic blood pressure <90 mmHg) in volume-replete patients requiring vasopressors therapy. Haemostatic dysfunction was defined as PT >15 s, and/or activated PTT (aPTT) >20 s and/or platelet count <100,000/mm3 in the absence of platelets clumps at blood smear evaluation. Cut-off values for specific variables (serum creatinine, PT and aPTT) were based on the upper bound of the reference intervals of our clinical pathology laboratory. MODS was defined as presence of at least two dysfunctional organs simultaneously. The number of dysfunctional organs upon admission and at the end of hospital stay was recorded.
Statistical analysis
Statistical analysis was performed using a medical statistic software (MedCalc 16.8.4). Data were assessed for normality with the D’Agostino-Pearson test and evaluated by descriptive statistics. Most data were non-parametric; therefore, data are presented as median and range (minimum–maximun). The Mann–Whitney U-test was used to compare continuous variables between groups. Univariate and multivariate logistic regressions were used to identify variables able to predict outcome; results are presented as odds ratio (OR) and 95% confidence interval (CI). Correlations between variables were assessed by the Spearman’s rank correlation coefficient. Significance was set at P <0.05.
Results
Forty-three cats met the criteria for inclusion in this study. Median age was 5.2 years (range 0.2–15.7) and median body weight was 4.0 kg (range 0.4–6.6). There were 19/43 female cats (eight sexually intact and 11 spayed) and 24/43 males (eight sexually intact and 16 castrated). Comorbidities were recognised in the majority of the patients (n = 28): polytrauma (n = 7), chronic enteropathy (n = 6), diabetes mellitus (n = 4), feline lower airway disease (n = 4), chronic kidney disease (n = 2), hypertrophic cardiomyopathy (n = 2), neoplasia (n = 2) and feline lower urinary tract disease (n = 1). Upon presentation, 8/43 patients were receiving immunosuppressive drugs (steroids, n = 6; steroids plus melphalan, n = 2) and 17/43 were treated with antimicrobial agents by the referring veterinarian.
Major causes of sepsis were the following: pyothorax (n = 13), feline panleukopenia (n = 11) and septic peritonitis (n = 9). Other sources of sepsis were urinary tract infection (n = 3), bacterial cholangiohepatitis (n = 2), endocarditis (n = 1), myocarditis (n = 1), septic arthritis (n = 1), pyelonephritis (n = 1) and ocular abscess (n = 1). Main clinical and clinicopathological findings in the overall study population at presentation are shown in Table 1.
Descriptive statistics for selected variables measured at the time of admission in cats with sepsis (n = 43)

Values for each analyte are presented as median (range). RI = reference interval; NA = not applicable; rpm = respirations per min; bpm = beats per min; SBP = systolic blood pressure; APPLE = acute patient physiological and laboratory evaluation; WBCs = white blood cells; SAA = serum amyloid A; PT = prothrombin time; PTT = partial thromboplastin time
Seventeen of 43 cats required fluid resuscitation upon admission. Isotonic crystalloid solutions were used by the primary clinician for fluid resuscitation in the whole population with a median value of 20 ml/kg (range 10–85). Tetrastarch (6% hydroxyethyl starch 130/0.4; Voluven) or 7.5% hypertonic saline were administered in a minority of the patients at the following doses: 2 ml/kg (1–8; n = 4) and 4 ml/kg (n = 1), respectively. Ten of 17 cats were not responsive to fluid resuscitation and required vasopressors. Non-responders received a significantly higher volume of crystalloid solution compared with responders (20.5 ml/kg [range 10–85] vs 15.0 ml/kg [range 10–20]; P = 0.02). Vasopressor and inotropic support in cats with cardiovascular dysfunction in the whole study period (n = 18/43) included noradrenaline alone (n = 18/18) or in combination with dobutamine (n = 14/18). During the ICU stay blood products were administered in 12/43 cats, with 10/12 receiving fresh frozen plasma, 3/12 packed red blood cells and 2/12 fresh whole blood. Twenty-six of 43 cats were survivors, whereas 17/43 were non-survivors (9/17 cats were humanely euthanased because of moribund conditions and 8/17 died despite medical treatment).
The following variables evaluated at presentation were significantly different between survivors and non-survivors: body temperature (38.4°C [range 33.2–40.9°C] vs 35.6°C [range 32–40.5°C]; P = 0.04); APPLEfast score (26 [range 18–40] vs 34 [range 12–46]; P = 0.02), leukocyte count (12.185 cells × 103/mm3 [range 0.25–81.48 cells × 103/mm3] vs 4.01 cells × 103/mm3 [range 0.05–60.91 cells × 103/mm3]; P = 0.03); total amount of intravenous fluids administered during hospitalisation (47 ml/kg/day [range 5–93 ml/kg/day] vs 87 ml/kg/day [range 30–148 ml/kg/day]; P = 0.001). None of the latter variables were retained from the univariate regression analysis.
Organ dysfunction: prevalence and associations with outcome
At presentation, 25/43 cats had evidence of MODS and frequencies of dysfunctional organs were distributed as follows: haemostatic dysfunction (n = 24), respiratory dysfunction (n = 23), hepatic dysfunction (n = 15), renal dysfunction (n = 14) and cardiovascular dysfunction (n = 10). During ICU stay, 37/43 cats developed MODS with the following frequencies: 32 had haemostatic dysfunction, 26 had hepatic dysfunction, 26 had respiratory dysfunction, 18 had cardiovascular dysfunction and 18 had renal dysfunction. Upon admission, the majority of the cats with renal dysfunction (n = 11/14) had an increased sCr and were classified as IRIS stage 2 (n = 6) and stage 3 (n = 5), respectively, whereas only 3/14 cats had an IRIS stage of 1 and a sCr within the normal range.
Results of the univariate logistic regression showed a significantly elevated OR for mortality for the presence of MODS, renal and cardiovascular dysfunction, as well as for requirement of fluid resuscitation and for the number of dysfunctional organs recorded upon admission and at the end of ICU stay. When the multivariate binary logistic regression analysis was performed, the number of dysfunctional organs recorded upon admission was the only variable retained by the model (Table 2).
Univariate and multivariate binary logistic regression of variables that were associated with outcome (survivors/non-survivors) in 43 cats with sepsis

P value indicates difference between survivors and non-survivors in the overall population of septic cats; bold indicates significance (P <0.05).
RC = regression coefficient; SEM = standard error of the mean; OR = odds ratio; CI = confidence interval; ICU = intensive care unit
The Spearman’s rank correlation coefficient between the number of dysfunctional organs and the APPLEfast score at presentation was 0.66 (P <0.0001; Figure 1). The Spearman’s rank correlation coefficient between the number of dysfunctional organs and the APPLEfull score at presentation was 0.56 (P = 0.011).
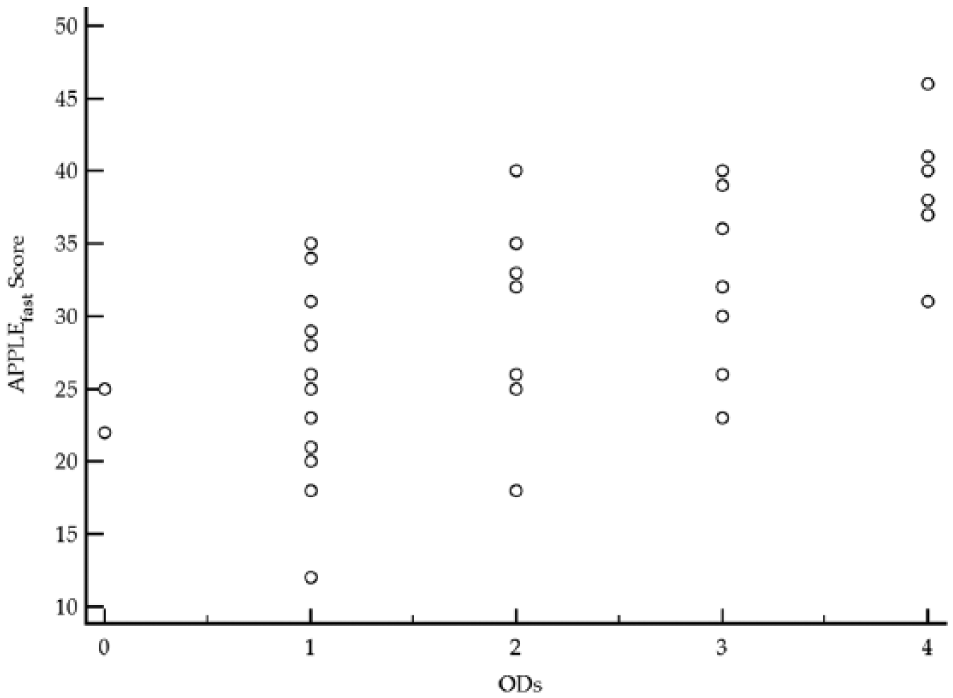
Scatter diagram showing the relationship between the number of dysfunctional organs (ODs) and the feline acute patient physiological and laboratory evaluation (APPLE) score, APPLEfast, in cats with sepsis (n = 43). The Spearman’s rank correlation coefficient was 0.66 (P <0.0001)
Discussion
The present study investigated the prevalence and the prognostic significance of MODS in a population of cats with sepsis. A focused organ-by-organ assessment was prospectively performed in the enrolled cats, both at the time of presentation and at the end of the ICU stay. Organ dysfunction occurred frequently in septic cats and was related to higher APPLE scores and worse outcomes. Specifically, presence of MODS, renal and cardiovascular dysfunction at presentation significantly increased the risk of death. The overall number of dysfunctional organs was the strongest predictor of mortality in the current population. These results are novel in feline critical care medicine, resemble the data reported in humans and dogs with sepsis, and further corroborate the link between this syndrome and the development of organ dysfunction.3–5,16–19 Moreover, the number of dysfunctional organs in this population was positively correlated with the feline APPLE scores. The latter are outcome predictors validated in critical feline patients, 13 but only the APPLEfast score was significantly higher in non-survivors compared with survivors, even though it was not prognostic according to the univariate logistic regression. Although the latter result was not completely expected, the feline APPLE scores lack an extensive validation in sepsis; thus, their potential prognostic significance in this clinical setting needs to be confirmed.
Renal dysfunction was defined as the presence of AKI according to the recent IRIS guidelines established in veterinary patients. 15 AKI is a common complication of human sepsis, being related to inflammatory mediators, tubular injury and renal microcirculatory alteration.16,17,20 There is a paucity of studies concerning sepsis-related AKI in veterinary medicine, 21 and standardised criteria for the early identification of this syndrome have been only occasionally used in cats. 22 The prevalence of AKI was elevated in this population of septic cats at the time of presentation. In this regard, two of the azotaemic cats had primary renal involvement due to urosepsis (one cat with pyelonephritis, one cat with septic uroabdomen). Additionally, CKD was suspected in other two cases owing to history and imaging findings. These results could have influenced the real frequency of sepsis-related AKI in our population. However, the presence of AKI at presentation was a strong predictor of mortality in our study. This result could support the use of the application of the IRIS staging system to aid early identification of renal dysfunction in septic cats.
Cardiovascular dysfunction was defined as hypotension in volume-replete patients requiring vasopressor support. Despite hypotension and relative bradycardia being described in cats with sepsis, epidemiological data for feline sepsis-induced cardiovascular dysfunction are lacking.6,7 Cardiovascular dysfunction occurred frequently in this population, with septic peritonitis and feline panleukopenia being the main diseases associated with it (data not shown). According to the univariate logistic regression analysis, the presence of cardiovascular dysfunction upon admission was associated with elevated odds for mortality. This result parallels the data available from human and small animal studies, as persistent hypotension despite fluid-resuscitation is considered the most serious manifestation of sepsis, with a reported mortality rate as high as 80–90% in both species.2–5,19,23,24 As such, the presence of cardiovascular dysfunction might reflect greater disease severity and be associated with worse outcomes. The negative impact of hypotension on systemic organ perfusion and oxygen supply has to be considered as a further mechanism leading to worsening clinical pictures and, eventually, death. 25
Hepatic, respiratory and haemostatic dysfunction occurred with a variable frequency in the current study population, but no association with outcome was noted. Icterus and hyperbilirubinaemia have been described as markers of sepsis in cats,7,9 and seem to have a potential prognostic role. 10 Prevalence of hepatic dysfunction was elevated in the study reported herein, and raised during hospitalisation. Whether hyperbilirubinaemia reflected true hepatocyte dysfunction or sepsis-induced cholestasis remains unknown.
Despite the recent definition of criteria to identify acute respiratory distress syndrome in small animals,11,26 respiratory dysfunction is commonly identified as the need for oxygen therapy, mechanical ventilation or hypoxaemia.3,14 However, such criteria are subjective (eg, opinions may differ over the need for supplemental oxygen therapy), and, owing to technical reasons, arterial PaO2 assessment through blood gas analysis was only rarely performed herein. The prevalence of respiratory dysfunction was elevated in our study, but mainly documented in cats diagnosed with pyothorax (n = 10/23) and not necessarily associated with lung disease. Additionally, the presence of respiratory dysfunction was not related to outcome in this study population. The favourable survival rate documented in the majority of the cats with pyothorax might partially explain the latter result. Moreover, it is possible that the degree of respiratory dysfunction was only moderate in this population of septic cats.
Abnormalities in the haemostatic profile were common in our population, representing the most frequently reported organ system dysfunction at the time of inclusion and during ICU stay. No association between haemostatic dysfunction and outcome was identified. Potential limitations in the criteria used for the assessment of haemostatic dysfunction in the present study must be considered, not allowing specific characterisation of either the severity or the nature of the dysfunction itself (presence of a hyper- or hypocoagulable state). Blood product administration was reserved to cats considered at risk of bleeding (n = 10/12) or with signs of active bleeding (n = 2/12), and could have potentially affected their outcome.
Additional clinical variables were different between survivors and non-survivors in the present study. Of note, non-survivors received a significantly greater amount of intravenous fluids, and requirement for fluid resuscitation carried an elevated OR for death. Whether intravenous fluid therapy reflected higher illness severity or concurred to worsen prognosis and sepsis-related complications cannot be clarified from our data. However, this result parallels similar findings in human and canine ICU patients,27,28 and need to be addressed by further studies.
There are some limitations to consider when interpreting our results. The small size of the population and the different sources of sepsis might have influenced the results of the study. In addition, organ dysfunction was not graded according to severity, but only deemed as present or absent on the basis of criteria adapted from canine medicine. This latter approach, despite being questionable, was inevitable owing to the lack of established standards in cats. The intrinsic limitations of such criteria might have biased some of our findings especially in respect to prognosis, as already discussed; moreover, neurological and gastrointestinal dysfunctions were not assessed for the current study. Finally, organ dysfunction was deemed present regardless of the location of the primary septic focus, potentially increasing its prevalence in some cases.
Conclusions
The results of the present study document a high prevalence of organ dysfunction in cats with sepsis. The presence of MODS is associated with higher APPLE scores and worse outcomes, with renal and cardiocirculatory dysfunction markedly increasing the odds of death. Screening for dysfunctional organs has to be advocated early in the management of feline sepsis, in order to identify the most critically ill patients and maximise supportive care. Further larger studies aimed at characterising MODS in feline sepsis and to investigate its prognostic significance are warranted.
Footnotes
Acknowledgements
The authors would like to acknowledge Dr Valentina Stocchetti for the technical help in case recruitment and sample collection.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
