Abstract
Objectives
The aim of this study was to evaluate the efficacy and safety of lispro insulin for the treatment of feline diabetic ketoacidosis (DKA). Times to resolution of hyperglycaemia, ketosis and acidosis were compared between cats treated with continuous rate infusion (CRI) of lispro insulin and cats treated with CRI of regular insulin.
Methods
Client-owned cats with naturally occurring DKA, newly diagnosed with diabetes mellitus (DM) or already receiving treatment for DM, were included. Diagnosis of DKA involved the presence of at least two clinical signs consistent with DKA (eg, polyuria/polydipsia, anorexia, severe lethargy, vomiting and dehydration), blood glucose (BG) concentration >13.9 mmol/l (>250 mg/dl), blood beta hydroxybutyrate (BHB) concentration >2.5 mmol/l and venous pH <7.3 or bicarbonate <15 mEq/l. Cats were treated with a standard protocol of an intravenous (IV) CRI of regular insulin (group R) or lispro insulin (group L). The time to resolution of DKA was defined as the time interval from when the IV CRI of insulin began until marked hyperglycaemia (BG >13.9 mmol/l [>250 mg/dl]), ketosis (BHB concentration >1 mmol/l) and acidosis (venous pH <7.3 and/or bicarbonate <15 mEq/l) resolved.
Results
Eighteen DKA cases (nine per group) were enrolled into the study. There were no significant differences in the median time to resolution of three variables (hyperglycaemia, ketosis and acidosis) between the two groups. Two cats in group R developed hypoglycaemia during the CRI of insulin. One cat in group L and three cats in group R developed hypophosphataemia, which required phosphate supplementation.
Conclusions and relevance
IV CRI of lispro insulin has few side effects and appears to be as effective as IV CRI of regular insulin in the treatment of cats with DKA.
Introduction
Diabetic ketoacidosis (DKA) is the most common complication of naturally occurring diabetes mellitus (DM) and is characterised by a biochemical triad of hyperglycaemia, ketosis and acidosis.1–5 Treatment of DKA comprises intravenous (IV) fluid resuscitation, correction of acid/base and electrolyte derangements, insulin therapy and targeted therapy for comorbid conditions. 5
During DKA, regular insulin is usually administered intramuscularly or intravenously in cats and dogs; 6 in humans, it is also injected subcutaneously. 7 Nevertheless, the dehydration and shock state of patients with DKA leads to erratic and inconstant absorption of intramuscular and subcutaneous (SC) insulin. 7 For this reason, IV infusion of regular insulin has been the mainstay of treatment of DKA as it causes a more predictable fall in blood glucose and it allows for rapid adjustments. 8
Lispro insulin is a genetically engineered analogue of human insulin in which proline at position B28 and lysine at position B29 are inverted in their sequence, reducing the formation of insulin dimers and hexamers. This structural change ensures more rapid absorption and elimination from the SC injection site, resulting in the rapid onset and a short duration of hypoglycaemic activity.9,10 Furthermore, one study in human medicine comparing the end-organ metabolic effects of IV lispro insulin, regular insulin and glulisine insulin showed that all these insulins have similar effects on the suppression of endogenous glucose production, glucose uptake and free fatty acid, glycerol and lactate levels. 11
The success of lispro insulin, as well as other insulin analogues, has gradually reduced the use of regular insulin, as demonstrated by Eli Lilly’s financial report. 12 Assuming that the production of regular insulin may be discontinued, a valid alternative for treating DKA in dogs and cats should be found. Two studies demonstrated that IV continuous rate infusion (CRI) of lispro and aspart insulin is safe and appears to be as effective as an IV CRI of regular insulin for the treatment of canine DKA.13,14
The aim of this study was to evaluate the efficacy and safety of lispro insulin for the treatment of feline DKA by comparing the times to resolution of hyperglycaemia, ketosis and acidosis between cats treated with CRI of lispro insulin and cats treated with CRI of regular insulin.
Materials and methods
Client-owned cats admitted to the University Veterinary Hospital of Bologna (Italy) between May 2009 and March 2017 with naturally occurring DKA, either newly diagnosed with DM or with known DM, were considered for inclusion. The diagnosis of DKA involved the presence of at least two clinical signs consistent with DKA (eg, polyuria/polydipsia, anorexia, severe lethargy, vomiting and dehydration), blood glucose concentration >13.9 mmol/l (>250 mg/dl), blood beta hydroxybutyrate (BHB) concentration >2.5 mmol/l and venous pH <7.3 or bicarbonate <15 mEq/l. 15 Cats with DKA, admitted between May 2009 and February 2012, and treated with a protocol for insulin therapy adapted from a published protocol using an IV CRI of regular insulin (Humulin R; Eli Lilly) 16 were used as part of the control group of this study. From March 2012 to April 2014 cats with DKA admitted to the University Veterinary Hospital were treated with the same insulin protocol, but using lispro insulin (Humalog; Eli Lilly), until the number of cats was the same in both groups. Between May 2014 and March 2017, cats admitted for DKA were alternately treated with regular insulin or lispro insulin.
Cases were divided according to whether they received an IV CRI of regular insulin (group R) or IV CRI of lispro insulin (group L). Cats with multiple hospitalisations for DKA management during the study period were included in the analyses, with each hospitalisation event treated as a separate case.
Cases were excluded from the study if they had unavailable or missing medical records and if they died or were euthanased prior to administration of insulin therapy. The trial was approved by the Scientific Ethics Committee, University of Bologna, Italy. Owners signed the written informed consent before enrolment in the study.
At the time of admission to the hospital, history, physical examination findings and results of blood gas analysis, complete blood count, serum biochemistry profile, urinalysis and bacterial culture from urine collected via cystocentesis were performed in each cat in order to confirm DKA and identify any concurrent disorder. An abdominal ultrasound was performed in order to detect any abnormalities (eg, acute pancreatitis, neoplasia). Thoracic radiographs or other diagnostic tests were also performed according to the clinician’s discretion.
Definitions of ‘resolution time’, time of SC insulin administration and length of hospitalisation
The ‘resolution time’ for the variables hyperglycaemia, ketosis and acidosis was calculated starting from ‘time zero’, which was the time at which the IV CRI of insulin treatment was initiated. The time to resolution of pronounced hyperglycaemia was defined as the time interval between ‘time zero’ and the time at which the blood glucose concentration fell to <13.9 mmol/l (<250 mg/dl). The time to resolution of ketosis was defined as the time interval between ‘time zero’ until BHB was ⩽1.0 mmol/l for two consecutive measurements 1 h apart. The time to resolution of acidosis was defined as the interval between ‘time zero’ and the time at which venous pH was ⩾7.3 and/or bicarbonate ⩾15 mEq/l. The time to resolution of ketoacidosis was defined as the time interval between ‘time zero’ and the time at which ketosis and acidosis had both resolved. The IV CRI of insulin was stopped when ketosis and acidosis had resolved and the cat was eating well.
The time to SC insulin administration was defined as the time interval from the resolution of ketoacidosis (when the transition from the IV to the SC insulin administration occurs) up to the hospital discharge. The length of hospitalisation (LOH) was defined as the time interval between ‘time zero’ and discharge from the hospital.
Monitoring protocol
Blood glucose was monitored hourly for the first 24 h with a hand-held glucometer, previously validated for use in cats (Optium Xceed, Optium Glucose Test Strips; Abbott Laboratories), 17 and then every 2–3 h during the entire time that the cat received an IV CRI of insulin. Blood BHB was measured every 4 h using a portable ketometer, previously validated for cats (Optium Xceed, Optium β-ketone Test Strips; Abbott Laboratories) 17 until BHB was ⩽1.0 mmol/l; in this case the BHB was measured 1 h later and if a BHB ⩽1.0 mmol/l was confirmed then ketosis was deemed to be resolved. A blood gas analysis (including pH, base excess, serum bicarbonate, sodium, potassium, ionised calcium and lactate) was performed with a point-of-care analyser (IDEXX VetStat; IDEXX Laboratories) every 8 h during the first 24 h, and then every 12 h until ketosis was resolved.
Insulin-induced hypoglycaemia was defined as a blood glucose concentration <4.4 mmol/l (<80 mg/dl); hypokalaemia was defined as serum potassium <3.6 mEq/l. Hypophosphataemia was defined as serum phosphate <0.5 mmol/l (<1.5 mg/dl).
Treatment protocol
Upon admission, all cats were treated with IV crystalloids (Ringer’s lactate or acetate or 0.9% NaCl) prior to and while receiving insulin treatment. The initial rate of fluid administration was determined by the attending clinician to meet the specific rehydration needs of each cat. IV insulin CRI was initiated from 2–8 h after fluid administration had been started, depending on the clinician’s perception that severe dehydration had resolved.
The insulin solution, which was administered in a separate line from the fluids, consisted of 48 ml 0.9% NaCl to which 1.1 units per kg body weight of lispro insulin or regular insulin were added. 16 To saturate binding of insulin to the IV tubing, the insulin solution was allowed to stand in the line for 30 mins and then run through the IV line. 18 At this point, the insulin solution was re-prepared and the infusion was started. The initial dosage for the insulin CRI was based on the cat’s blood glucose concentration when the CRI was started (‘time zero’) (Table 1). 6 The insulin CRI was adjusted every 1–2 h based on the cat’s blood glucose. Adjustments to the insulin CRI and the addition of dextrose were implemented at each clinician’s discretion based on previously published guidelines (Table 1). 6 Long-term insulin was initiated when ketoacidosis was resolved and the cat was eating and appropriately hydrated.
Sliding scale for adjustment of intravenous continuous rate infusion of insulin treatment and dextrose supplementation for cats with diabetic ketoacidosis

Insulin solution composed of 1.1 U/kg of regular or lispro insulin added to 48 ml 0.9% NaCl
2.5% dextrose composed of 25 ml dextrose 50% added to 475 ml 0.9% NaCl or Ringer’s
5% dextrose composed of 50 ml dextrose 50% added to 450 ml 0.9% NaCl or Ringer’s
Serum potassium concentration was corrected as previously described. 16 If serum phosphate concentration decreased to <0.5 mmol/l (< 1.5 mg/dl) it was corrected by administration of an IV CRI of potassium phosphate at a rate of 0.01–0.03 mmol phosphate/kg/h for 6 h and then phosphataemia was re-evaluated. Supplementation with potassium was taken into account when giving potassium phosphate for correction of hypophosphataemia. Antimicrobials were administered to all cats for the duration of hospitalisation; additional drugs, including gastroprotectants, antiemetics and analgesics, were administered as deemed appropriate by the attending clinician according to the clinical condition and concurrent disorders.
Statistical analysis
Statistical analysis was performed using commercially available software (GraphPad Prism 5). Owing to the small number of cases in each group, continuous variables were considered to be non-parametric and descriptive statistics are reported as median (minimum–maximum). The Mann–Whitney U-test was used to compare variables between the two insulin groups. The Wilcoxon signed rank test was used to compare changes from baseline of the continuous variables within each insulin group. A P value <0.05 was considered significant. In order to compare the different variables between cats with newly diagnosed DM and cats with known DM, regardless of the type of insulin used, the Mann–Whitney U test was used.
Results
Twenty-four DKA cases were evaluated in the study period. Four cases were excluded because cats died or were euthanased before initiating insulin CRI therapy, and two cases were excluded because of incomplete medical records. A total of 18 cases in 15 cats were included in the study; one cat had three DKA events (in two events it received lispro insulin and in another event received regular insulin) and another cat had two DKA events during the study period (one received lispro insulin and the other received regular insulin). Nine cases were managed with lispro insulin (group L) and nine cases were managed with regular insulin (group R). In 15 cases cats were discharged from the hospital; one cat from group L died and two cats from group R were euthanased; these three cats were newly diagnosed with DM.
History, clinical signs and physical examination findings
There was no significant difference between groups with regard to median age, body weight, breed and sexual status (neutered or intact; Table 2). The median age among all 18 cases was 10.4 years (range 7.7–16.5 years). The median body weight of all 18 cases was 4.5 kg (range 2.6–8 kg) and median body condition score was 6.5 (range 2–8). All 15 cats included in the study were European shorthair cats except one Persian cat in group R and one Birman cat in group L. Eight cats were neutered males, five were neutered females, one was an intact male and one an intact female.
Baseline data and blood glucose and beta hydroxybutyrate (BHB) concentration at ‘time zero’ in cats with diabetic ketoacidosis treated with an intravenous (IV) continuous rate infusion (CRI) of lispro insulin (group L) and treated with an IV CRI of regular insulin (group R)
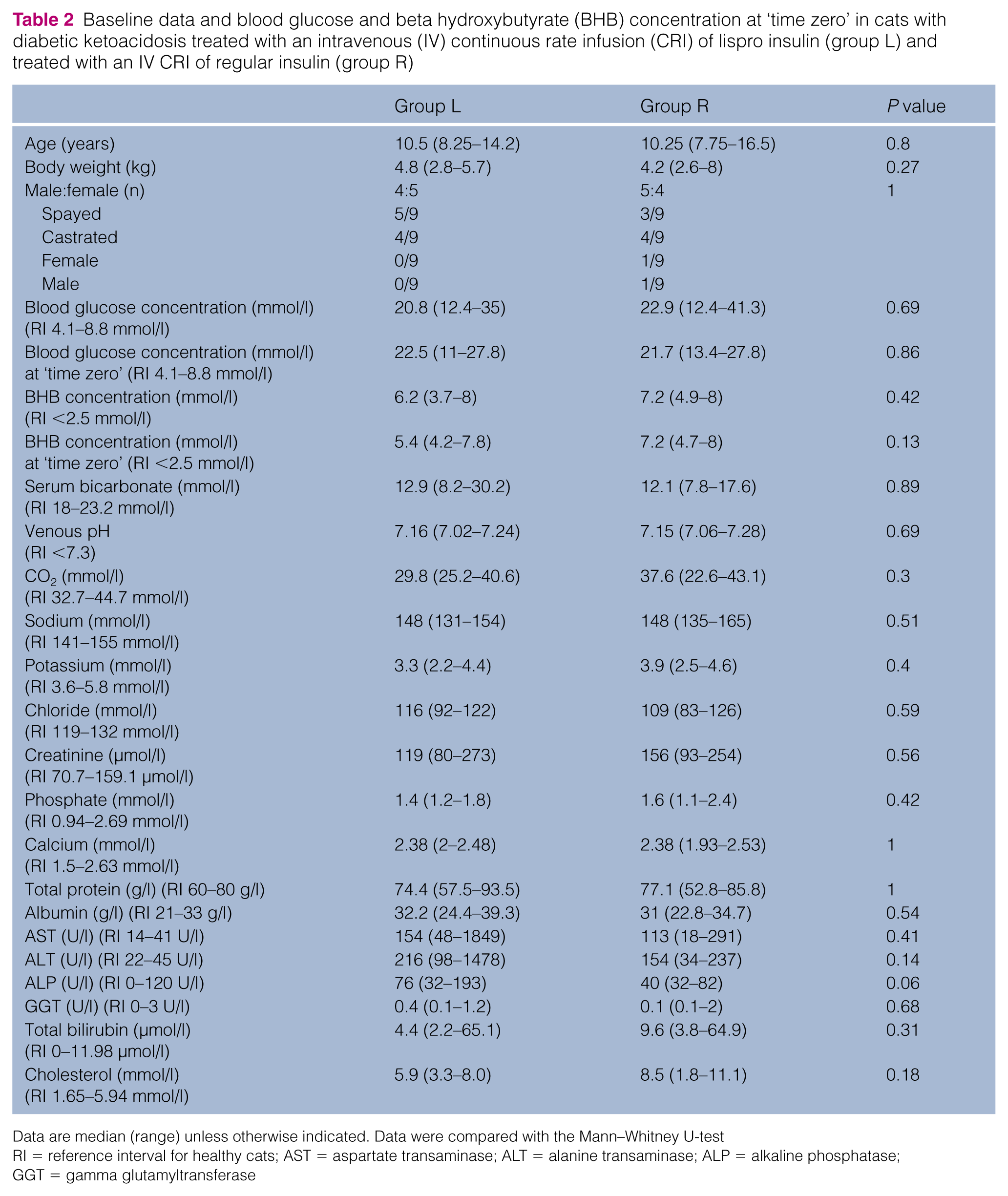
Data are median (range) unless otherwise indicated. Data were compared with the Mann–Whitney U-test
RI = reference interval for healthy cats; AST = aspartate transaminase; ALT = alanine transaminase; ALP = alkaline phosphatase; GGT = gamma glutamyltransferase
In 10 cases cats were newly diagnosed with DM at the time of enrolment into the study. In eight cases (seven cats), cats had previously been diagnosed with DM (five in group L and three in group R), a median of 8 months (range 1–12 months) prior to enrolment into the study and they were all receiving glargine insulin (Lantus, 100 U/ml glargine; Aventis Pharmaceuticals). Insulin dosage at the time of enrolment into the study was 0.5 U q12h in one cat, 1 U q12h in four cats, 2 U q12h in two cats and 2.5 U q12h in one cat.
Clinical signs observed by the owners prior to admission into the hospital included lethargy (observed in 15/18 [83%]), anorexia (15/18; 83%), polyuria and polydipsia (10/18; 56%), vomiting (6/18; 33%), weight loss (5/18; 28%), asthenia (4/18; 22%) and diarrhoea (3/18; 17%). Medications administered to the cats at the time of admission into the hospital included insulin (7/18; 39%), tylosin (2/18; 11%), methimazole (Tapazole; 1/18; 6%), marbofloxacin (Aristos; 1/18; 6%) and enrofloxacin (Baytril; 1/18; 6%).
At the time of admission, the most common abnormalities included some degree of dehydration (observed in 17/18 [94%]), dull or depressed mentation (17/18; 94%), hypothermia (8/18; 44%), overweight body condition (7/18; 39%), underweight body condition (5/18; 28%), pale mucous membranes (5/18; 28%), jaundice (4/18; 22%), muscle atrophy (3/18; 17%), heart murmur (2/18; 11%) and palpable thyroid nodule (1/18; 6%).
Clinicopathological findings
At the time of admission into the hospital, median blood glucose concentration, BHB concentration, venous pH and serum bicarbonate concentration did not differ significantly between the lispro insulin and regular insulin-treated group (Table 2).
The median blood glucose concentration in group L and group R was 20.8 mmol/l (range 12.4–35 mmol/l; 374 mg/dl [224–630 mg/dl]) and 22.9 mmol/l (range 12.4–41.3 mmol/l; 413 mg/dl [224–744 mg/dl]), respectively. At ‘time zero’, the median blood glucose concentration was 22.5 mmol/l (range 11–27.8 mmol/l; 405 mg/dl [198–500 mg/dl]) and 21.7 mmol/l (range 13.4–27.8 mmol/l; 391 mg/dl [241–500 mg/dl]) in group L and group R, respectively. No significant differences were detected in blood glucose concentration between the two treatment groups at the time of admission (P = 0.69) or at ‘time zero’ (P = 0.86). The rate of decrease in blood glucose concentration was <5.6 mmol/l/h (100 mg/dl/h) in all 18 cases during the entire study.
The median BHB concentration in group L and group R was 6.2 mmol/l (range 3.7–8 mmol/l) and 7.2 mmol/l (range 4.9–8 mmol/l), respectively. At ‘time zero’, the median BHB concentration was 5.4 mmol/l (range 4.2–7.8 mmol/l) and 7.2 mmol/l (range 4.7–8 mmol/l) in group L and group R, respectively. No significant differences were detected in BHB concentration between the two treatment groups at the time of admission (P = 0.42) or at ‘time zero’ (P = 0.13).
At the time of admission, there were also no significant differences between the two treatment groups with respect to any of the biochemical parameters analysed (Table 2).
The median time interval between the time at which fluid therapy was initiated until ‘time zero’ was 4 h in the lispro insulin group (range 2–8 h) and 4.5 h (range 1–8 h) in the regular insulin group; there was no significant difference between the two groups (P = 0.62).
Adverse drug reactions
No local or systemic adverse effects associated with IV insulin administration were noted in either group. Two cats in group R developed hypoglycaemia during the CRI of insulin (4.39 mmol/l [79 mg/dl] and 2.22 mol/l [40m g/dl], respectively), but these cats did not show clinical signs compatible with hypoglycaemia. In all 18 cases, cats developed transient hypokalaemia during the study. Median minimum potassium concentrations did not differ significantly between the lispro (2.8 mmol/l; range 2.2–3.7 mmol/l) and regular (2.6 mmol/l; range 2.2–3.5 mmol/l) insulin treatment groups (P = 0.82). One cat in group L and three cats in group R developed hypophosphataemia, which required supplementation during the study.
Resolution time of hyperglycaemia, acidosis and ketosis, time of SC insulin administration and LOH
Severe hyperglycaemia resolved in all 18 cases, acidosis resolved in 15 cases (seven in group L and eight in group R) and ketosis resolved in 16 cases (eight in group L and eight in group R). One cat in group L died prior to resolution of acidosis and ketosis. Acidosis did not resolve in one cat in group L and one cat in group R that had suffered an acute kidney injury at the time of admission; ketosis did not resolve in one other cat in group R, possibly owing to the insulin resistance secondary to a concurrent carcinoma.
There were no significant differences in the median time to resolution of three variables (hyperglycaemia, ketosis and acidosis) between the two groups when evaluated separately; there was no significant difference in the median time to resolution of ketoacidosis (Table 3).
Time to resolution of hyperglycaemia, ketosis, acidosis and ketoacidosis, time of subcutaneous (SC) insulin administration and length of hospitalisation in cats with diabetic ketoacidosis treated with an intravenous (IV) continuous rate infusion (CRI) of lispro insulin (group L) and treated with an IV CRI of regular insulin (group R)

Data are expressed as median (range)
The median times to resolution of severe hyperglycaemia in group L and group R were 8 h (range 0–25 h) and 9 h (range 0–24 h), respectively (P = 0.72). Median time to resolution of ketosis was 29 h (range 16–94 h) in group L and 26.5 h (range 21–53 h) in group R (P = 0.83). Median time to resolution of acidosis in group L and group R was 8 h (range 8–32 h) and 20 h (range 8–48 h), respectively (P = 0.26). Median time to resolution of ketoacidosis in group L and group R was 33 h (range 16–94 h) and 28 h (range 21–53 h), respectively (P = 1).
There were no significant differences between newly diagnosed and previously diagnosed diabetic cats with respect to median time to resolution of hyperglycaemia and ketosis (analysed separately), and ketoacidosis (Table 4). However, the median time to resolution of acidosis in the newly diagnosed diabetics (12 h; range 8–24 h) was significantly shorter than in previously diagnosed diabetics (24 h; range 8–48 h; P = 0.02).
Time to resolution of hyperglycaemia, ketosis, acidosis and ketoacidosis, time of subcutaneous (SC) insulin administration and length of hospitalisation in cats with diabetic ketoacidosis, comparing cats with newly diagnosed diabetes mellitus (DM) and cats with known DM

Data are expressed as median (range)
Bold indicates statistical significance
Venous pH decreased during the first hours of treatment, before it began to rise, in 5/18 cases (one case in group L and four cases in group R). The lowest pH for these five cases was reached at a median of 8 h (range 8–16 h) from the time at which fluid infusion had begun. The median lowest pH for the five cases in which this initial decline occurred was 7.07 (range 6.94–7.25) and did not differ significantly (P = 0.06) from the pH of the same patients at admission (median 7.15; range 7.02–7.28). Also, there was no significant difference in LOH between cases in which pH decreased before it began to rise and cases in which this did not happen.
The median time to administration of SC insulin, in the 15 cases that were discharged, did not differ significantly between group L (76 h; range 34–168 h) and group R (89 h; range 48–244 h [P = 0.25]). Likewise, the median duration of hospitalisation for these 15 cases did not differ significantly between group L (110.5 h; range 74–268 h) and group R (146 h; range 94–294 h; P = 0.18) (Table 3).
No significant differences were found in median time to administration of SC insulin and in median duration of hospitalisation between newly diagnosed and previously diagnosed diabetic cats (Table 4).
The lispro insulin-treated cat that died had been hospitalised for 45 h at the time of death; the two cats treated with regular insulin that were euthanased had been hospitalised for 106 and 129 h, respectively, at the time of euthanasia.
Evaluation for presence of concurrent disorders
Based on the diagnostic protocol, concurrent disorders were identified in 11 cases (five in group L and six in group R). In group L, two cases had inflammatory bowel disease, one cat had pancreatitis, one cat had concurrent pancreatitis, lipidosis and acute kidney injury, and one cat had pulmonary neoplasia. In group R, one cat was diagnosed with a bacterial urinary tract infection (based on urinary culture), one cat was diagnosed with pancreatitis and inflammatory bowel disease, one cat had pancreatitis and polycystic kidney disease, one cat had hyperthyroidism and herpesvirus infection, one cat had chronic kidney disease and one cat had a giant cell tumour.
The diagnosis of pancreatitis was based on abdominal ultrasound (enlarged, irregular, hypoechoic pancreas surrounded by hyperechoic mesentery, and mild-to-moderate ascites) and positivity to a feline pancreatic lipase immunoreactivity test.
Discussion
Lispro insulin was developed to resolve the problems associated with the use of regular human insulin (peak of activity reached too late, hypoglycaemic effect possibly lasting too long) by SC injection. 19 The major difference between lispro insulin and regular insulin is the rate of self-disassociation, which causes differences in the rate of absorption from the injection site. However, this difference may not exist with IV administration. A study on rabbits showed that the hypoglycaemic response profiles after IV administration of lispro insulin and regular human insulin were very similar in pattern and confirmed that their biological activities are equivalent. 20
The aim of this study was to evaluate the efficacy and safety of lispro insulin for the treatment of feline DKA.
The need to test a new insulin, which could provide an alternative to regular insulin, has arisen because insulin analogues are widely used for the management of DM and treatment of uncomplicated DKA in human medicine, although some patients with severe comorbidities still require intensive care and IV insulin administration.8,21–24 It is possible that the production of regular insulin may be discontinued in the future, and as only regular insulin is currently indicated for the treatment of DKA with the constant low-dose IV insulin infusion technique in cats, a viable alternative to regular insulin is needed to manage these patients.
In a prospective randomised study, Sears et al compared the efficacy and safety of an IV CRI of lispro insulin with that of regular insulin in a population of 12 dogs with DKA. 13 They observed comparable improvement in glycaemia, ketosis and acidosis between the two groups and the time to resolution of ketoacidosis was significantly shorter in the lispro insulin group, although the LOH did not differ significantly. They concluded that an IV CRI of lispro insulin is safe and appears to be as effective as an IV CRI of regular insulin for the treatment of canine DKA.
In our study on feline DKA, the time of resolution of hyperglycaemia, ketosis, acidosis and ketoacidosis was similar in cases treated with lispro insulin and those treated with regular insulin; the time of SC insulin administration and the LOH also did not differ significantly between the two groups. Nevertheless, the time to resolution of acidosis and the LOH were both shorter in the group of cases treated with lispro insulin, although these differences were not significant. A number of variables, including concurrent disorders, may have contributed to these findings, and it is reasonable to suppose that studying a larger group of cases in the future could reveal that each of these times is significantly shorter with lispro insulin treatment.
The median time to resolution of acidosis in newly diagnosed diabetic cats was significantly shorter than in previously diagnosed diabetics; this result may reflect a different efficiency in the buffering system and a difference in acid–base status between newly and previously diagnosed diabetic cats.
In the first hours after the onset of insulin therapy, venous pH decreased before it began to rise in only one cat in group L and in four cats in group R; these results could be attributed to the more rapid action of lispro insulin compared with regular insulin. Furthermore, hyperglycaemia resolved in all 18 cases, acidosis resolved in 7/9 cases in group L and in 8/9 cases in group R, and ketosis resolved in 8/9 cases in group L and group R. On the basis of these results an IV CRI of lispro insulin appears to be as effective as an IV CRI of regular insulin for the treatment of cats with DKA.
With regard to safety, in our results, side effects were seen less frequently in cases treated with lispro insulin than in cases treated with regular insulin; in fact, in group R on two occasions cats developed hypoglycaemia and three cats developed hypophosphataemia, whereas only one cat in group L developed hypophosphataemia. Despite supplementation, transient hypokalaemia occurred in all cats during the IV infusion of insulin, regardless of the type of insulin used. Although hypokalaemia that develops during DKA rarely becomes symptomatic, in our opinion it would be more appropriate to use higher rates of supplementation than those normally reported in textbooks during the first hours of insulin therapy; this was also suggested by Nelson, 5 but only in those patients with normal urinary production and if frequent assessments of kalaemia are possible.
In this study there were no significant differences in blood glucose concentration or BHB concentration between the two treatment groups at the time of admission or at ‘time zero’, when insulin therapy began. On the contrary, a decrease in blood glucose concentration during the first hours of fluid therapy has been reported in human medicine and in some studies in veterinary medicine, and has been attributed to rehydration-induced renal excretion of glucose, decreased concentrations of the counter-regulatory hormones, or improved perfusion and delivery of endogenous insulin.13,14,25,26 This reduction was not observed during our study, probably owing to the conservative fluid therapy, which was not too ‘aggressive’. On that note, one of the current study’s limitations is the lack of standardised criteria for the evaluation of the degree of dehydration (determined subjectively) to determine when to start insulin therapy.
Another important limitation is the small number of cases enrolled, which influenced the power of statistics. It is likely that some differences between groups were not detected because of this bias. Furthermore, some cats were included more than once and this is also a possible bias; in fact, it is possible that a cat responds in a similar manner with repeated treatment as a different cat, or that a cat can be more severely affected with subsequent visits because of progression of concurrent illness or even less severely affected because owners recognised the signs earlier.
Finally, further limitations are the absence of randomisation and the heterogeneity of the population with regard to the presence of concurrent disorders. However, our population’s characteristics were very similar to those in other studies of feline DKA,27–31 suggesting that this small population is representative of cats with spontaneous DKA.
Conclusions
The results demonstrate that an IV CRI of lispro insulin treatment did not show severe side effects in cats of this study and appears to be as effective as an IV CRI of regular insulin treatment in managing cats with DKA.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
