Abstract
Objectives
Feline gingivostomatitis (FGS) is a painful chronic inflammatory disease of the oral cavity. The purpose of this study was to examine the frequency of detection of certain common feline bacteria and viruses to determine any potential associations with FGS.
Methods
A multicentre case-control study design was conducted. In total, 72 control cats and 32 cats with FGS were included in the study. Oral swabs were cultured for bacterial identification and a PCR assay was carried out to examine the infection of feline calicivirus (FCV), feline herpesvirus-1 (FHV-1), Chlamydia felis, Mycoplasma felis and Bordetella bronchiseptica.
Results
There was a significant difference in age distribution between the control and the FGS group. Based on a PCR assay, the positive rate of FCV was significantly higher in FGS cats than control animals. For other infectious pathogens, including FHV-1, C felis and M felis, there was no significant difference. Bacterial culture of oral swabs revealed that Pasteurella multocida was most frequently detected, but the detection rate was significantly lower in FGS cats. In FGS cats, the incidence of Enterococcus faecalis and anaerobic bacteria were more frequently isolated than in control cats.
Conclusions and relevance
This study indicates that the positive rate of FCV was significantly higher in cats with FGS, and the microflora of the oral cavity of cats with FGS might be disrupted, although additional studies are required to compare the oral microbiome in cats of a variety of ages.
Introduction
Feline gingivostomatitis (FGS) is characterised by focal or diffuse chronic inflammation of the oral mucosa and gingiva. The incidence is estimated to be about 6–7% of all cats.1,2 Typical clinical signs are characterised by oral pain, ulceration of oral mucosa, halitosis, dysphagia, anorexia and weight loss.3,4 Hyperglobulinaemia is also an important and common presenting sign. Cats may be affected at an early age and lifelong therapy may sometimes be required because the disease is difficult to cure completely. Histopathological examination of oral lesions typically shows abundant infiltration of plasma cells, and moderate but variable numbers of lymphocytes, neutrophils and macrophages. 5 Some studies suggest that nutritional factors, physiological or environmental stresses, and genetic predisposition may be the cause of FGS.3,4
As immune responses are present in FGS lesions in cats, it has been proposed that infectious agents are associated with the development of the disease. Viral infections, including feline immunodeficiency virus (FIV), feline leukaemia virus (FeLV), feline calicivirus (FCV) and feline herpesvirus-1 (FHV-1), might be the cause of FGS.6–8 Further, a variety of other bacteria may play an important role in FGS, although few studies have demonstrated this link.9,10 Importantly, these infectious agents have been isolated not only from FGS cats, but also from control animals.9,11 Therefore, a causal relationship is difficult to prove in each case of FGS in cats.
Corticosteroids, antibiotics, interferon, ciclosporin and chemotherapeutic agents have been used to treat FGS.3,4 Unfortunately, these treatments may become less effective with time. Extraction of teeth has shown better results than drug therapy, although clinical cure has been achieved in up to 57% of cases. 12 Then, more invasive treatment such as full-mouth or near-full mouth extractions can be selected. Thus, novel treatment for FGS with prolonged effect and less invasiveness needs to be developed. In order to develop a possible therapeutic therapy, we examined the frequency of detection of certain common feline bacteria and viruses to determine any potential associations with FGS.
Materials and methods
Sample collection
The study was conducted at eight private veterinary hospitals in Osaka prefecture and one in Hiroshima prefecture, Japan, between October 2014 and May 2016. FGS was diagnosed by the combined assessment of the history and clinical presentation of each cat. In total, 104 client-owned cats were included (72 control cats, 32 cats with FGS). From each cat, two oral swab samples were collected. One sample was preserved in Amies gel with charcoal transport media (CultureSwab Plus Transport Systems; Becton Dickinson) for culture. Swab samples were immediately transported to a commercial laboratory (Japan Clinical Laboratories) and bacterial culture was performed the same day as sampling. Then, bacterial species were identified. Another swab sample was frozen in liquid media (BD Universal Viral Transport System; Becton Dickinson) at −80°C until PCR analysis was performed. The blood samples were tested for retrovirus infection by the SNAP FIV/FeLV Combo Test (IDEXX Laboratories).
PCR assays
PCR analysis was performed at a commercial laboratory (Veterinary Diagnostic Laboratory; Marupi Lifetech). For FCV detection, RNA extraction was performed with a commercial kit (QIAamp RNA Mini Kit; Qiagen Science) according to the manufacturer’s instructions. PCR analysis of RNA samples was performed with the QIAGEN OneStep RT-PCR Kit, and then amplified PCR products were used for nested PCR with AmpliTaq Gold (Applied Biosystems). For the analysis of FHV-1, Chlamydia felis, Mycoplasma felis and Bordetella bronchiseptica genomic DNA was extracted with the QIAamp Viral DNA Mini Kit and amplified with AmpliTaq Gold. All primer sets were as mentioned in previous studies,13–17 and are summarised in Table 1. Electrophoresis of the resulting PCR products was performed in 2% agarose gel, which was stained with ethidium bromide and visualised under ultraviolet light.
Primers used

The Cali1 and Cali2 set was used for reverse transcription PCR, and the Cali3 and Cali4 set was used for nested PCR of feline calicivirus
bp = base pair
Statistical analysis
The age distributions between control and FGS cats were compared by Mann–Whitney U-test. The rates of positive test results for PCR assays (FCV, FHV-1, C felis, M felis and B bronchiseptica) were compared between control and FGS cats by Fisher’s exact test. For bacterial cultures from oral swabs, the positive rates of each identified bacterial species between control and FGS cats were with Fisher’s exact test. In this study, a P value <0.05 was considered to be statistically significant.
Results
Characteristics of the study population
In total, 72 control cats and 32 cats with FGS were included in the study. The total population contained entire males (25 in the control group and eight in the FGS group), neutered males (12 in the control group and seven in the FGS group), entire females (25 in the control group and 10 in the FGS group) and neutered females (10 in the control group and seven in the FGS group). Median age of control and FGS cats was 0.9 years (range 0.6-14.5 years) and 8.5 years (range 1.0-20 years), respectively. There was a significant difference in age distribution between the control and FGS groups (P <0.01).
Bacterial species detected by microbial culture
The bacteria identified from oral swabs are described in Table 2. In total, 12 bacteria were identified from the sample of control cats. In particular, Pasturella multocida was frequently detected (54/72 cats; 75%). The second most frequently found bacteria were Escherichia coli and Branhamella catarrhalis (4/72; 5.6%), but the incidence was much lower than that of P multocida. From the samples of cats with FGS, P multocida was also the most frequently identified (15/32; 46.9%), but the rate was significantly lower than that of control cats (P <0.01). Enterococcus faecalis was the second-most frequently found bacteria, and the incidence (8/32; 25%) was significantly higher than that of control animals (2/72; 2.8%) (P <0.01). Other enteric bacteria were found at lower frequencies: E coli (6/32; 18.8%) and Proteus mirabilis (4/32; 12.5%). Anaerobic bacteria were detected in 1/72 (1.4%) control cats and in 4/32 (13%) FGS cats. The rate was significantly lower in control cats than in FGS cats (P <0.05).
Isolated bacteria from oral swab samples of both control and feline gingivostomatitis (FGS) cats
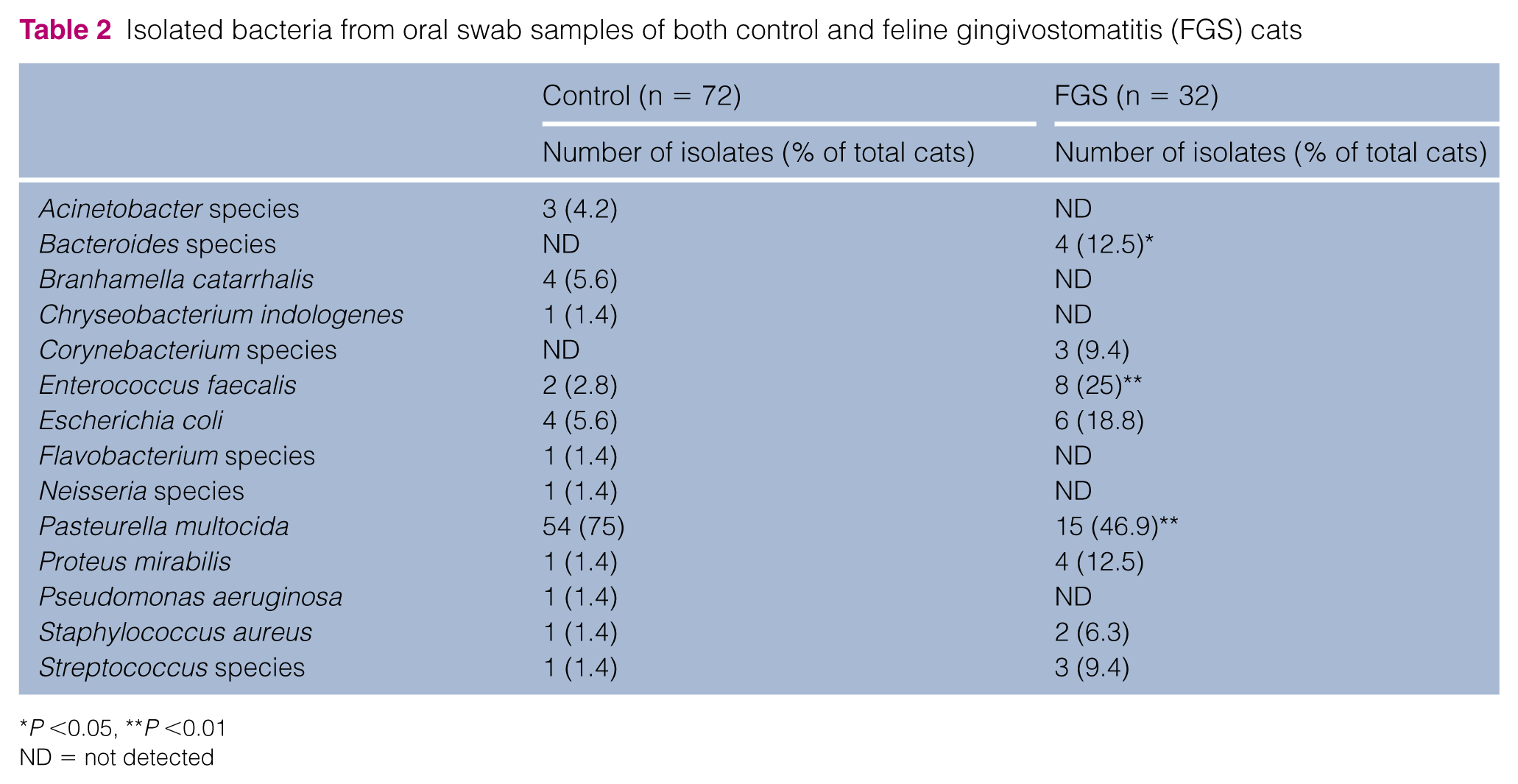
P <0.05, **P <0.01
ND = not detected
Prevalence of microbial pathogens detected by PCR
In the present study, we examined the vaccination history from the medical records and the clinical history provided by the owner. Two FGS cats showed positive results for FIV infection (both cats were not vaccinated for FIV) and there were no cats infected with the FeLV virus. The detailed results of PCR analysis are presented in Table 3. From the analysis, positive FCV results were identified in 28/72 (38.9%) control and 20/32 (62.5%) FGS cats. The rate of positive results was significantly higher in FGS cats (P <0.05). For other infections, including FHV-1, C felis and M felis, there were no significant differences in positive rates between control and FGS cats. B bronchiseptica was not isolated from any cat examined in this study.
Rate of positive PCR results for feline calicivirus (FCV), feline herpesvirus-1 (FHV-1), Chlamydia felis and Mycoplasma felis in control cats and cats with feline gingivostomatitis (FGS)

Bordetella bronchiseptica was not isolated from all examined samples
P <0.05
Discussion
FGS sometimes presents with severe inflammation of the oral cavity that causes pain and much stress in affected individuals. The aetiology of the disease has been assumed to be multifunctional, and feline immune system responses are probably associated with infectious and non-infectious agents. 4 In this study, we examined, for the first time in Japan, the prevalence of FCV, FHV-1, C felis, M felis and B bronchiseptica in both control cats and cats with FGS.
From the results, FCV was found to be significantly more prevalent in cats with FGS (63%) compared with normal cats (36%). It has been reported elsewhere that cats with FGS are infected with FCV at a relatively higher rate (from 41% up to 96%), and thus FCV infection has been thought to be one of the primary components of FGS.8,18,19 Although 36% of normal cats were infected with FCV in this study, this prevalence was higher than that found in previous studies.11,20 As the prevalence of FCV in cats might differ in various populations, FCV infection is not always essential for FGS. In fact, in the present study, 38% of FGS cats were negative for FCV infection. Recently, a study described that FIV or FeLV antigens were detected in the oral biopsy samples of FGS lesions, although FCV antigens were not detected in the same lesion. 21
Other pathogens besides FCV could play a role in the aetiology of FGS. For FHV-1 infection, we used a previously described conventional PCR assay. 15 In this study, there was no significant difference in the prevalence of FHV-1 between normal (1.4%) and FGS (6.3%) cats. It has been reported that the positive rate of FHV-1 infection in FGS cats is 0–16%.11,18,19 In the present study, the detection rate was almost equivalent in FGS cats. Because the virus is involved in feline upper respiratory tract disease, oropharyngeal swab samples or real-time PCR analysis should have been applied for FHV-1.19,22,23 However, the virus may latently infect ganglion cells of the nervous system, and it is sometimes difficult to detect by PCR and identify as a pathogen. 20
The prevalence of C felis was low in both the control cats (1.4%) and cats with FGS (6.3%) in the present study. Studies in shelters in other countries demonstrated that control cats were infected with the virus at a rate of 0–15%.22,24,25 In these reports, C felis was more often detected from conjunctival or pharyngeal swab samples of cats with upper respiratory infection or conjunctivitis than cats with FGS. Cats with FGS have a higher prevalence of C felis than control cats, but the lower odds ratio indicates a weak association between C felis infection and development of FGS. 19 Therefore, consistent with our results, C felis is not considered the main pathogen associated with FGS, based on previous studies.
In this study, M felis infection was found in 6.9% of normal cats and 3.1% of cats with FGS. Previous reports demonstrated that M felis is detected at a rate of 20–31% in normal cats and 38–47% in cats with FGS.19,3 Although our result is relatively lower than in these other reports, the differences may be the result of analysis of conjunctival or oropharyngeal samples rather than the oral samples, as used in our study.
No cats infected with B bronchiseptica were found in this study. In upper respiratory infections of cats or dogs, the bacterium is sometimes detected from pharyngeal swab samples as the main pathogenic agent and causes severe infectious bronchitis or pneumonia. 26 For FGS, there have been no reports of B bronchiseptica as a primary pathogen. The analysis of oral microflora revealed that B bronchiseptica was not detected from both control and FGS-affected cats. 9 Taken together, these results indicate that the bacterium is not associated with the cause of FGS, although its infection might be identified through examination of oropharyngeal swab samples.
In the present study, bacterial cultures of oral swab samples were examined, and P multocida was most frequently identified in both control and FGS cats. Previous studies demonstrated that the bacterium was often identified from oral swab samples of control cats.10,26 Interestingly, our data showed that the prevalence of P multocida in cats with FGS was significantly lower than in control cats. In addition, E faecalis was identified at a higher rate in FGS cats than in control cats. Intestinal bacteria, including E coli, P mirabilis and Corynebacterium species, were often detected from the FGS cat swab samples. The prevalence of anaerobes was higher in FGS cats than in control groups. These data indicate that the microflora of the oral cavity of FGS cats is disrupted; intestinal bacteria and anaerobes dominate indigenous bacteria such as P multocida.
Although the results of this study provide novel information about the differences in the oral microbial communities of control and FGS cats, the limitations must be considered. First, there was a difference in age distribution between control and FGS cats in the present study. It has been recently reported that the intestinal microbiome of cat undergoes age-dependent changes. 27 It is therefore possible that oral microflora might change with age, regardless of the FGS. However, it is still unclear whether the oral microbiome might change with age. Additional studies are required to compare the oral microbiome in cats across a variety of ages, to determine the impact of the age disparity between control and FGS cats. Additionally, future studies into FGS need to age match controls with FGS cases to remove any potential confounder, because it is assumed that other potential modifiable factors, such as rearing environment, diet or species, could also influence the oral microbiome.28–30
Also, our data did not include bacterial population indicators, such as 16S ribosome RNA gene sequencing. Therefore, it is possible that dominant or easily grown bacteria were preferentially identified. However, our results are supported by the fact that gram-negative bacilli and anaerobes increased in plaques and can be associated with the progression of FGS.9,31
Conclusions
The results of the present study show that the positive rate of FCV was significantly higher in cats with FGS, as found with previous studies. Most importantly, it was demonstrated that the incidence of E faecalis and anaerobic bacteria were more frequently isolated from oral swab samples of cats with FGS than those of control cats. Although this study lacks information about the bacterial population and the changes in oral microflora over time, the present data indicate that the native microflora of the oral cavity of cats with FGS might be disrupted. Future studies to further explore the association between anaerobes, E faecalis and FGS are required, and if confirmed to be correct, may offer additional treatment avenues.
Footnotes
Acknowledgements
The authors would like to thank the efforts of the veterinarians who provided serum and swab samples (Hirano Animal Hospital, Matsuoka Animal Hospital, Yamayoshi Dog and Cat Hospital, Ishizuka Animal Hospital, Sen-nan Animal Hospital, Kuzunoha Animal Hospital, FNP Animal Hospital, Sippo Animal Hospital, Satoh Animal Hospital). The authors also thank Enago (![]() ) for the English-language review.
) for the English-language review.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
