Abstract
Objectives
The aim of this study was to determine the accuracy of a commercial luteinizing hormone (LH) test as an aid in distinguishing between sexually intact and ovariectomized or castrated domestic cats.
Methods
Convenience serum samples collected from sexually intact female and male cats (n = 67) undergoing elective sterilization surgery and archived sera from ovariectomized and castrated cats (n = 54) were tested for LH using a commercial diagnostic assay. Test results were compared with the known reproductive status of the cats. Additionally, sera from sexually intact (n = 54) and ovariectomized (n = 94) queens were collected at specific times of the year to evaluate possible seasonal effects on test results.
Results
Overall test sensitivity was 89.3% (95% confidence interval [CI] 82.3–94.2%), specificity was 92.6% (95% CI 87.1–96.2%) and accuracy was 91.1%. Analysis of results of female cats (n = 216) – sexually intact (n = 87) and ovariectomized (n = 129) – yielded a test sensitivity of 90.8% (95% CI 82.7–96.0%), a specificity of 92.3% (95% CI 86.2–96.2%) and accuracy of 91.7%. Analysis of the results of male cats (n = 53) – sexually intact (n = 19) and neutered (n = 34) – yielded test a sensitivity of 85.3% (95% CI 68.9–95.1%), a specificity of 94.7% (95% CI 74.0–99.9%) and accuracy of 88.7%. The sera of 10 intact queens unexpectedly yielded positive LH results; two of these cats were in estrus, based on visual inspection at the time of ovariohysterectomy. Test accuracy was 94.6% for those 148 samples collected at specific times of the year, with two samples each over three, 3 month periods yielding false-positive results.
Conclusions and relevance
The commercial point-of-care LH test is a useful adjunct to historical and physical examination findings for determination of reproductive status in domestic cats. Repeat testing 24 h later should be considered for those female cats with signs of estrus and initial positive test results.
Introduction
In the absence of medical history or physical evidence of surgical sterilization, such as a tattoo or surgically removed tip of an ear, veterinarians may have difficulty definitively determining the reproductive status – sexually intact or neutered – of adult cats. 1 This is particularly difficult with female cats. Such determination is necessary for the proper diagnosis and treatment of medical conditions and for confirming candidates for surgical sterilization. A commercially available qualitative test for luteinizing hormone (LH) (Witness LH; Zoetis) has been developed for reproductive management in dogs (ie, to determine the appropriate time frame for breeding). 2 Yet, this hormone can also be used as an aid in the determination of reproductive status of both dogs and cats.2,3 The lower limit of LH detection for this test is 1 ng/ml. 2
Sexually intact female animals generally have low serum LH concentrations until estrus, when LH significantly increases.4,5 Ovarian production of estrogen and progesterone negatively affect hypothalamic production of gonadotropin-releasing hormone and pituitary production of LH (Figure 1). In contrast, LH concentrations are increased because of a lack of such negative hormonal feedback; average reported serum LH concentrations are >2.5 ng/ml in ovariohysterectomized female cats.2,4,6,7 Increased LH concentrations have been confirmed in female dogs ovariectomized 5 days to >5 years prior to testing and in female cats ovariectomized 1.6 months prior to testing.3,6 Measurement of LH may help distinguish between sexually intact and ovariectomized female animals because of the presence or absence of this negative feedback mechanism, respectively. Yet, female dogs and cats with clinical signs consistent with estrus at the time of LH testing may yield transient positive LH results because of the LH surge associated with the onset of estrus. 3 Time of testing relative to time of year may also affect the result. Sexually intact female cats, unlike dogs, are seasonally polyestrous; they cycle repeatedly throughout a breeding season, typically February, as the days get longer, through September in the northern hemisphere.8,9 Therefore, testing during the breeding season may increase the opportunity to identify the LH surge because multiple surges may occur. In two previously published studies, researchers reported excellent sensitivity and specificity for using LH concentration as a determinant of female reproductive status of cats.7,10 Differentiation may be important to prevent unnecessary anesthetic and surgical procedures for previously ovariohysterectomized animals and to aid in confirming a diagnosis of ovarian remnant syndrome – defined as the presence of functional ovarian tissue accompanied by signs of estrus following routine ovariohysterectomy.1,11–13

The hypothalamic–pituitary–gonadal axis is depicted for sexually intact and neutered animals. In sexually intact animals, the ovaries and testes predominantly produce estrogen/progesterone and testosterone, respectively, thus inhibiting hypothalamic production of gonadotropin-releasing hormone (GnRH) and pituitary gland production of follicle stimulating hormone (FSH) and luteinizing hormone (LH). With ovariohysterectomy and castration, negative feedback through estrogen/progesterone and testosterone, respectively, is absent, leading to sustained increases of GnRH, FSH and LH
Likewise, sexually intact male animals are expected to have relatively low serum LH concentrations and castrated male animals are expected to have relatively high serum LH concentrations because of a similar feedback mechanism through testosterone associated with the presence or absence of the testes, respectively.5,14 Using LH concentration as an aid to determine reproductive status may be problematic because the authors of one study reported overlapping LH concentrations between sexually intact and castrated male dogs, 5 and the authors of another study reported agreement between test results and reproductive status. 14 To our knowledge, LH testing has not been reported as an aid to distinguish between sexually intact and castrated male cats. LH testing may be useful for male cats with suspected cryptorchidism.
The objective of this study was to contemporaneously determine the accuracy of a commercially available LH test for the identification of reproductive status of female and male domestic cats and to determine the seasonal impact – if any – of the polyestrous reproductive cycle of female cats on test results.
Materials and methods
Samples
Set A
Whole blood of sexually intact female and male cats was collected prospectively during the months of March, April and May at the time of admission for elective sterilization surgery to the Veterinary Community Outreach Program at the University of Florida, College of Veterinary Medicine. Cats originated from north-central Florida animal shelters and rescue organizations, and were deemed good candidates for elective sterilization based on physical examination. Sera were prepared and stored refrigerated at 2–8°C for up to 24 h before testing. In addition, frozen, archived sera of female and male neutered cats were thawed and immediately tested. This portion of the study was approved by the University of Florida Institutional Animal Care and Use Committee (IACUC).
Set B
To evaluate the influence of time of year on LH testing of sexually intact female and ovariectomized cats, sera collected throughout the year were divided into four groups based on month of collection and then analyzed. Groups 1–4 included samples from January–March, April–June, July–September and October–December, respectively. These sera were exclusive to sample set B. Sexually intact female cats originated from the local community (ie, community cats) and animal shelters in east central Alabama and were presented to a university-based spay–neuter program. Frozen, archived sera of neutered female cats were also included in this sample set. This portion of the study was approved by the IACUC at Auburn University.
LH determination
LH concentration was determined using a commercially available, qualitative diagnostic assay (Witness LH; Zoetis) and following the manufacturer’s instructions. 2 Briefly, three drops of serum from each cat were applied to the sample well of this lateral chromatographic test using the supplied pipette. The presence or absence of LH within tested sera was determined by visual interpretation after 20 mins at ambient temperature. LH within sera initially binds to the colloidal gold-labeled anti-dog LH antibodies to form complexes. The immune complexes migrate along the nitrocellulose test strip and accumulate on the test line, resulting in the formation of a pink line. Unbound colloidal gold-labeled anti-dog LH antibodies are captured further down the test strip and subsequently form a pink control line. The formation of a control line for all tests indicates proper function of the test. The presence of a pink line at the test line of equal or greater color intensity than that of the pink line at the control line indicates a positive result. The absence of a pink line at the test line or the presence of a pink line that is of lesser color intensity than that of the control line indicates a negative result.
Testing of sera of sample set A was conducted by one of the authors (BAD) in batch upon the completion of a series of surgical procedures and not contemporaneously with each individual case. Archived samples were analyzed similarly, and individual patient signalment was not directly available during test interpretation. Testing of sera of sample set B was likewise conducted by one of the authors (BG) who was blinded to the reproductive statuses of the cats.
Test performance
Test results from sample sets A and B were analyzed independently and together. Additionally, results obtained for the female cats of sample set B were reviewed by group to determine the influence, if any, of time of year on test results. Test results were compared with the known reproductive status of the cats. Sensitivity (the ability of the test to correctly identify neutered cats with high concentrations of LH among a group of neutered cats) and specificity (the ability of the test to correctly identify sexually intact cats with low concentrations of LH among a group of sexually intact cats) were calculated. Accuracy – the number of cats the test correctly identifies as sexually intact or neutered divided by the number of all cats tested – was determined. Ninety-five percent confidence intervals (CIs) for sensitivity and specificity were calculated using statistical software (MedCalc). Based on determined sensitivity and specificity of the test for all cats and using hypothetical prevalence rates, positive and negative predictive values (PPV and NPV, respectively) were calculated using statistical software (EpiTools; Ausvet).
Results
A total of 121 samples – 67 from sexually intact female and male cats and 54 from neutered cats – comprised sample set A. A total of 148 samples – 94 from sexually intact female cats and 54 from ovariectomized cats – comprised sample set B (Table 1). Combined test results from both sample sets yielded a test sensitivity, specificity and accuracy of 89.3% (95% CI 82.3–94.2%), 92.6% (95% CI 87.1–96.2%) and 91.1%, respectively (Table 2). Analysis of the results of only female cats (n = 216) – sexually intact (n = 129) and neutered (n = 87) – yielded a test sensitivity of 90.8% (95% CI 82.7–96.0%), specificity of 92.3% (95% CI 86.2–96.2%) and accuracy of 91.7%, respectively. Analysis of the results of only male cats (n = 53) – sexually intact (n = 19) and neutered (n = 34) – yielded a test sensitivity of 85.3% (95% CI 68.9–95.1%), a specificity of 94.7% (95% CI 74.0–99.9%) and accuracy of 88.7%.
Numbers of sexually intact and neutered cats for which serum luteinizing hormone testing was performed

Group 1= January–March; group 2 = April–June; group 3 = July–September; group 4 = October–December
Performance data of a commercially available point-of-care luteinizing hormone test compared with known reproductive status with the sera of sexually intact and neutered cats
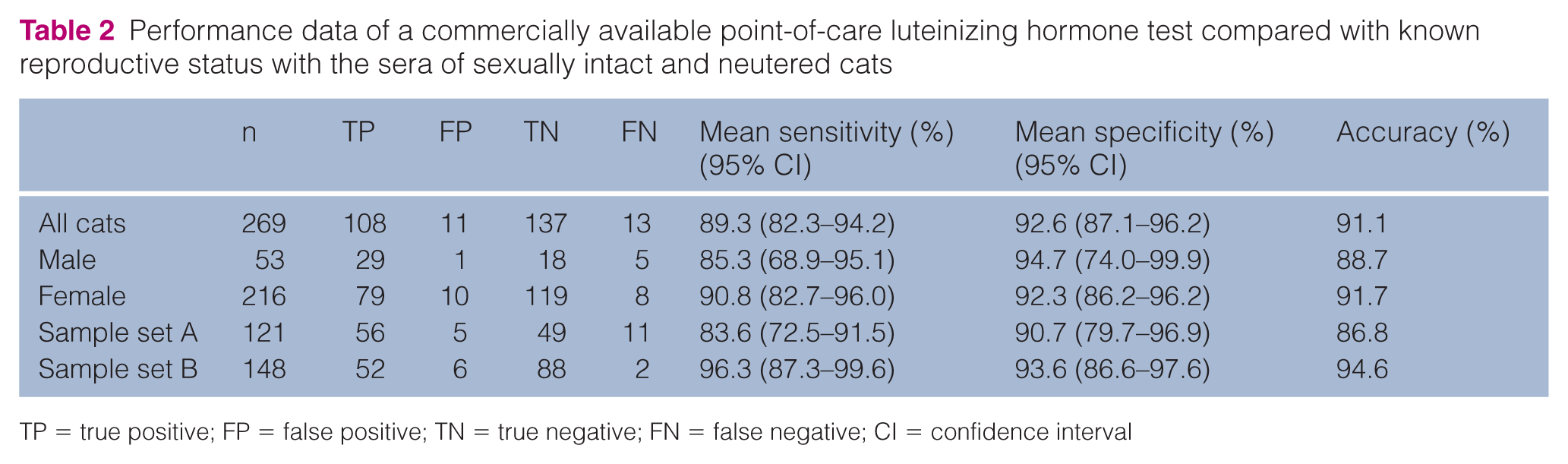
TP = true positive; FP = false positive; TN = true negative; FN = false negative; CI = confidence interval
For sample set A, sensitivity, specificity and accuracy were 83.6% (95% CI 72.5–91.5%), 90.7% (95% CI 79.7–96.9%) and 86.8%, respectively. Unexpectedly, the sera of four intact queens yielded positive LH results. Based on observed behavior at admission and visual inspection of their reproductive tracts at the time of ovariohysterectomy, two of these queens were determined to be in estrus.
For sample set B, sensitivity, specificity and accuracy were 96.3% (95% CI 87.3–99.6%), 93.6% (95% CI 86.6–97.6%) and 94.6%, respectively. The sera of six intact queens unexpectedly yielded positive LH results. Two queens belonged to each of groups 1, 3 and 4; phase of estrous cycle was not recorded.
The PPV and NPV for variable hypothetical prevalence rates of neutered cats are shown in Table 3. In a hypothetical population of cats in which 5% of the cats are neutered, the PPV of the test is 38.8% and the NPV of the test is 99.4%. In a hypothetical population of cats in which 90% of the cats are neutered, the PPV of the test is 99.1% and the NPV of the test is 49.0%.
Calculated positive and negative predictive values for a commercially available point-of-care luteinizing hormone (LH) test in hypothetical populations of cats that are not sexually intact (ie, neutered) with increasing prevalence rates from 5% to 90%*

Calculations based on a sensitivity of 89.3% and a specificity of 92.6%
PPV = positive predictive value; NPV = negative predictive value
Discussion
The LH test evaluated was a useful method of identifying the reproductive status of both female and male domestic cats. Overall test sensitivity at 89.3% is less than that (100%) previously reported;7,10 however, exclusion of male cats from the current analysis improved sensitivity to 90.8%. Overall test specificity at 92.6% is congruent with that (92–94%) previously reported.7,10 In contrast to the previous reports,7,10 the current study included the sera of female cats purposefully collected throughout the year and of sexually intact and castrated male cats.
Unexpectedly, the sera of 10 intact queens yielded positive LH results. Of the four queens that were presented for elective sterilization surgery (set A), the reproductive tracts of two were consistent with estrus (ie, visible ovarian follicles with hyperemic, edematous uterine tissue). Coincidental testing at the time of the LH surge may have yielded positive test results for these two queens. The test manufacturer recommends re-testing 24 h later of those queens for which estrus is suspected and initial positive test results are obtained. 2 Repeat testing of these two queens 24 h later with fresh sera may have yielded negative results, thereby correctly identifying these cats as sexually intact. Assuming this and reclassifying these two cats from ‘false positive’ to ‘true negative’, specificity for distinguishing sexually intact from ovariectomized female cats improved from 92.3% to 93.8% (95% CI 88.2–97.3%) and accuracy improved from 91.7% to 92.6%. Similarly, female cats displaying signs of estrus for which an ovarian remnant is suspected should also be re-tested 24 h later with fresh sera if initial test results are positive. A subsequent negative test result suggests ovarian tissue is present.
The sera of two queens within each of three of the four groups – groups one (January–March), three (July–September) and four (October–December) – unexpectedly yielded positive results. In the southeastern USA, sexually intact female cats are likely to have multiple estrous cycles between January and June, before environmental temperatures are too high such that ovarian function may be negatively affected.8,9 The number of false-positive test results was not higher for groups 1 and 2 (January–June) compared with the number of false-positive test results for groups 3 and 4 (July–December). The sera of all 17 sexually intact female cats of group 2 (April–June) yielded expected negative test results. Therefore, test specificity is not altered by the time of year in female cats of unknown reproductive status.
Time of testing relative to ovariohysterectomy could have affected sensitivity. LH is expected to be high, >1 ng/ml, in ovariectomized animals.2,4,7 Removal of the ovaries leads to increased LH concentration through lack of hormonal negative feedback to the hypothalamus and pituitary gland.4,5 However, the minimum amount of time from ovariohysterectomy to assay-detectable LH concentrations is unclear and likely varies among female cats. For a small number of sexually intact female cats, researchers reported that serum LH concentrations significantly increased by 30 days post-ovariohysterectomy but then decreased to pre-ovariohysterectomy concentrations at 60–120 days post-ovariohysterectomy. 6 In the current study, the duration of time post-ovariohysterectomy to phlebotomy of cats for which archived sera was tested was unknown; the impact of time is thought to be minimal given average reported serum LH concentrations are >2.5 ng/ml in ovariohysterectomized female cats and the lower limit of detection of the LH test is 1 ng/ml.2,6,7 Despite a decrease in LH concentration up to 4 months post-ovariohysterectomy, LH concentrations likely minimally rebound to, or remain at, the detectable limit with a continued lack of negative hormonal feedback. Previously and currently reported sensitivities of 89–100% suggest such occurrence.
Repeated freezing and thawing of archived sera may also have affected our reported test sensitivity. LH may have degraded with each freeze/thaw cycle, and the number of freeze/thaw cycles for these archived sera was unknown. For human females, LH did not significantly degrade in sera subjected to up to 10 freeze/thaw cycles. 15 The authors are not aware of similar studies conducted with feline serum samples.
Although LH concentration was reportedly useful in determining the reproductive status for a small number of sexually intact and castrated male dogs, 14 to our knowledge, assessment of LH concentration for determining reproductive status of male cats has not been previously reported. In the current study, sensitivity was lower and specificity was higher for distinguishing sexually intact from castrated male cats than they were for distinguishing sexually intact from ovariohysterectomized female cats (85.3% vs 90.8% and 94.7% vs 92.3%, respectively) (Table 1). The reasons for this disparity are unclear. Possibly male LH is negatively affected by time of testing in relation to castration. Similar to a suspected ovarian remnant in a female cat showing signs of estrus, LH concentration may be helpful in determining the presence of retained testicular tissue within a male cat exhibiting hormonally mediated behaviors such as urine spraying or inter-cat aggression. 16 Yet, such testing is likely unnecessary because veterinarians can simply examine the cat’s penis for penile spines; their presence with such a male cat suggests a source of testosterone and therefore the probable presence of a testicle.13,17
In contrast, the use of the LH test for the diagnosis of ovarian remnant syndrome (ORS) holds great potential merit because of the difficulty often associated with diagnosis of this syndrome. Indeed, this novel application of the LH test deserves emphasis because of its value as a rapid and non-invasive means of diagnosis of this often frustrating clinical syndrome. Clinical signs of ORS in cats include manifestations of behavioral estrus (eg, vocalization, rubbing, rolling, ‘friendly’ behavior, crouching and/or lordosis and treading in place with their hindlimbs). The onset of these signs may occur as soon as a few days or as late as many years following ovariohysterectomy and although normal inter-estrous intervals of 1–3 weeks may be seen, several months may lapse between periods of behavioral estrus. Many queens display rubbing and rolling behaviors regardless of reproductive status or stage or cycling; therefore, these behaviors are not specific indicators of ORS or ovarian tissue.8,11
Diagnosis of ORS is often challenging, especially when cats are not displaying estrus behaviors at the time of presentation and when intervals between such displays are lengthy. Typically, diagnosis of feline ORS is based on a combination of clinical signs, history, vaginal cytology, sex hormone concentrations, and exploratory laparotomy. In situations in which history and clinical signs are consistent with ORS, performing vaginal cytology concurrent to behavioral signs of estrus may be helpful. Although an increase in cornified vaginal epithelial cells and clearing of normal background mucus are often noted, vaginal changes in estrous queens tend to be subtle. Because behavioral estrus frequently persists beyond hormonal estrus, veterinarians may fail to detect these cytological changes. Additionally, veterinarians may also fail to detect an increase in serum estradiol concentration because elevations in serum estradiol are brief, returning to basal concentrations in as little as 48 h. Also, a single serum estradiol concentration may not be indicative of follicular activity.11,12 Logically, measurement of LH with this rapid point-of-care test may be useful in the diagnosis of ORS. As for a sexually intact female cat, this test should generally remain negative in cats with ORS.
One of the authors (BG) documented three clinical cases of ORS and utilized this point-of-care LH test for each to determine the need for exploratory laparotomy. In brief, three adult queens ranging in age from 2.5–5.5 years with clinical signs consistent with ORS and their sera yielded negative LH test results. Subsequent exploratory laparotomies confirmed the presence of ovarian tissue in each of these cats. Each of these cats had undergone previous ovariohysterectomy at least 2 years prior to presentation, and each had a history of sporadic displays of estrus behavior, including rubbing, rolling, crouching, and/or lordosis and treading. One owner reported that her cat attracted tom cats during this time. Periods of estrous behavior were brief, lasting only a few days in most cases and were separated by long inter-estrus periods ranging from approximately 10–18 months. None of these cats presented during an episode of estrous behavior. Follow-up examinations were performed at 45–60 days and the sera of all three cats yielded positive LH test results. The owners did not report signs of estrus within the following 2 years after which time they were lost to follow-up. These cases illustrate a practical and valuable application of the point-of-care LH test.
The contemporaneous performance data for the test will likely be useful to general and shelter medicine veterinarians presented with cats of unknown reproductive status. Along with history, if available, and physical examination, the result of LH testing may help veterinarians determine reproductive status.
Depending on the population of cats tested – stray/feral or owned – the predictive value of a positive or negative test result will vary (Table 3). For example, the prevalence of sexually intact female cats admitted to a shelter is likely high. The estimated percentage of female cats admitted to a shelter within Alachua County, FL, the community from which the prospectively tested samples (set A) were collected, is 85%; therefore, 15% of female cats are ovariectomized. 18 The probability that a negative LH test result for a cat of this population is predictive of reproductive status – in this case, sexually intact – is likewise high (eg, at a sensitivity of 89.3% and a specificity of 92.6%, NPV = 98.0%). Alternatively, the probability that a positive LH test result for a cat of this population is predictive of reproductive status – in this case, ovariectomized – is low (eg, at a sensitivity of 89.3% and a specificity of 92.6%, PPV = 68.1%). Therefore, a positive test result for a cat in such a population should be interpreted with caution; ideally, LH concentration with a fresh serum sample collected 24 h later should be tested.
The prevalence of sexually intact female cats presented for evaluation at a general practice is likely significantly less than that of those female cats admitted to a shelter; authors of a 2007 survey of US households reported that 20% of owned cats were sexually intact (ie, 80% of owned cats were neutered). 19 Therefore, the probability that a negative LH test result for a cat of this population is predictive of reproductive status – in this case, sexually intact – is low (eg, at a sensitivity of 89.3% and a specificity of 92.6%, NPV = 68.4%). Alternatively, the probability that a positive LH test result for a cat of this population is predictive of reproductive status – in this case, ovariectomized – is high (eg, at a sensitivity of 89.3% and a specificity of 92.6%, PPV = 98.0%). Therefore, a negative test result for a cat of such a population should be interpreted with caution. Re-testing should be considered. Additionally, as for any diagnostic test result, it should be interpreted in association with history, if available, and physical examination findings.
Despite its potential clinical usefulness, the test itself has limitations. Occasionally, test and control lines were faint, making interpretation difficult. Also, serum is the only sample type used for this test; therefore, test users must wait for collected blood to clot and for centrifugation, to facilitate separation of serum from the clot, to end. Users must then wait 20 mins for the test result. This relatively large amount of wait time may not be feasible for some clinical scenarios, such as if a patient is already anesthetized in preparation for surgical sterilization. However, in most situations when the indications for LH testing are identified ahead of time, it offers the advantage of preventing an unnecessary anesthetic and surgical procedure.
Test performance is less than that reported for two laboratory-based ELISAs for measurement of anti-Müllerian hormone (AMH).20,21 Because AMH is only produced by granulosa cells of the ovaries of females and by the Sertoli cells of the testes of males, AMH concentration may be able to distinguish between sexually intact and ovariectomized or castrated cats.20,21 However, the numbers of cats evaluated in each report are less than the numbers of cats that we evaluated in this study, and therefore their performances may change with testing of larger numbers of cats.20,21 Additionally, time to result is significantly increased with either AMH test compared with time to result with the LH test because the former requires veterinarians to submit patient sera to a commercial laboratory.
This study has some limitations. Repeat testing, laboratory quantification of LH concentration, or, if available, testing of purified feline LH, could have confirmed accurate identification of hormonal concentration by the qualitative commercial test at the time of sampling, as well as aided in determination of the true reproductive status of the cats. Additionally, as discussed previously, factors that may have negatively affected sensitivity include the unknown time between phlebotomies and sterilization surgeries, and the unknown number of freeze/thaw cycles and their potential effect on LH stability within the sera.
Conclusions
Veterinarians may have difficulty definitively determining the reproductive status of cats without medical history or physical evidence of surgical sterilization such as a tattoo or surgically removed tip of an ear. This is particularly difficult in female cats with signs of estrus and a history of previous sterilization. Such determination is necessary for the proper diagnosis and treatment of medical conditions, such as ORS, and for confirming candidates for surgical sterilization. The commercially available point-of-care LH test is a useful adjunct to historical and physical examination findings for determination of reproductive status in domestic cats. Repeat testing of those female cats with signs of estrus 24 h later should be considered to exclude coincidental testing at the time of the LH surge. In an attempt to avoid the possible confounder of an LH surge, veterinarians could instead delay testing of suspect female cats until they are not displaying signs of estrus.
Footnotes
Acknowledgements
The authors thank Ms Sylvia Tucker for technical assistance.
Author note
This paper was presented, in part, at the 2016 AAFP conference.
Conflict of interest
MR Krecic is employed by Zoetis, manufacturer of Witness LH. BA DiGangi and B Griffin declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Zoetis supplied the Witness LH tests and, at the time of this study, paid the salary of MR Krecic.
