Abstract
Objectives
Multidrug resistance 1 (MDR1) encodes a protein called P-glycoprotein (P-gp), which serves as an efflux pump membrane protein implicated in intestinal homeostasis and drug resistance. Cyclooxygenase-2 (COX2) is a key enzyme in the synthesis of proinflammatory prostaglandins, tumourigenesis and in mucosal defence. Despite the importance of MDR1 and COX2, changes in their mRNA levels have not been studied in cats with inflammatory bowel disease (IBD) and low-grade alimentary lymphoma (LGAL). The present study aimed to determine the mRNA levels of MDR1 and COX2 in cats with IBD and LGAL, and to evaluate their correlation with clinical signs, histological severity and between genes.
Methods
Cats diagnosed with IBD (n = 20) and LGAL (n = 9) between 2008 and 2015 were included in the current study. Three healthy animals composed the healthy control cats group in which endoscopy was performed immediately before the ovariohysterectomy. All duodenal biopsy samples were obtained by endoscopy. Feline chronic enteropathy activity index was calculated for all cases. IBD histopathology was classified according to severity. MDR1 and COX2 mRNA levels were determined by absolute reverse transcriptase-quantitative real-time PCR.
Results
Statistically significant differences were observed for MDR1 and COX2 mRNA levels between the IBD and LGAL groups. No correlations were observed between molecular gene expression, feline chronic enteropathy activity index and histological grading for IBD, and between MDR1 and COX2 genes. However, a positive statistically significant correlation was observed between MDR1 and COX2 expression in the duodenum of cats.
Conclusions and relevance
MDR1 and COX2 gene expression is increased in cats with LGAL compared with cats with IBD. The control group tended to have lower values than both diseased groups. These results suggest that these genes may be involved in the pathogenesis of IBD or LGAL in cats.
Introduction
Inflammatory bowel disease (IBD) and low-grade alimentary lymphoma (LGAL) are common causes of chronic enteropathies (CEs) in cats.1–6 The aetiology of IBD remains largely unknown, but multifactorial mechanisms are hypothesised to contribute to the pathogenesis.1–3,6 However, LGAL is the most common form of lymphoma in cats and its cause is also unknown.5,7–16 Additionally, progression of IBD to lymphoma has been proposed in cats and humans, but no direct link has been established.9,17
In humans, the multidrug resistance 1 (MDR1) or ABCB1 gene belongs to the family of ABC (ATP-binding cassette) transporters gene.18–20 MDR1 codes for P-glycoprotein (P-gp), an efflux pump membrane protein that actively transports substrates such as bacterial products and drugs from the inside to the outside of cells, is likely implicated in IBD pathogenesis and resistance to treatments.19–22 Previously, it has been described that MDR1a-deficient mice develop colitis and that human patients with IBD show reduced expression of MDR1 in the colon.23,24 Furthermore, elevated P-gp expression levels have been shown in canine IBD and have been associated with poor prognosis. 25 Likewise, MDR1 has been implicated in treatment resistance to various chemotherapy regimens used in veterinary species. 26
Cyclooxygenase 2 (COX2) is an inducible inflammatory regulator isoform by cellular activation, proinflammatory cytokines, growth factors, tumour promoters and prostaglandin mediator in humans and mice.27–30 Moreover, their metabolites have many biological roles, such as prostaglandin E2, which is a mediator of pain, modulator of cytokine production, inducer of regulators of angiogenesis, producer of proinflammatory mediators and promoter of tumourigenesis.31,32 Additionally, overexpression of COX2 may be a consequence of inflammation, leading to increased levels of bcl-2 protein and resistance to apoptosis of cells, and thus enhancing the risk of cancer.33,34 Lately, COX2 has been also considered a constitutive enzyme expressed in the gastrointestinal tract that acts as part of the mucosal defence mediated by prostaglandin and leading to reduced severity of colitis.35,36
An in vitro study has shown that the expression and activity of P-gp can be modulated by COX2. 37 However, this association has not yet been reported in human IBD. 38 Furthermore, a positive correlation between COX2 and the ABC-transporter immunoexpression has been described in different human neoplasms and non-Hodgkin’s lymphoma.39,40
Since an inflammatory response in the mucosa may be affected by higher exposure from a defective intestinal barrier, the aims of the present study were: (1) to determine the mRNA transcription levels of MDR1 and COX2 in cats with IBD and LGAL; and (2) to correlate these values with clinical signs and histological severity, and to evaluate whether there is a correlation between the MDR1 and COX2 genes.
Materials and methods
Study population
Thirty-one cats were included in this retrospective study. The electronic database of the Royal Veterinary College of London and Veterinary Teaching Hospital of the Universitat Autònoma de Barcelona (UAB) was searched between June 2008 and May 2015. Cats with signs of CE and duodenal biopsy samples available in RNAlater (Sigma) were identified. The inclusion criteria were the presence of gastrointestinal signs (>3 weeks duration), complete medical record and not having received any glucocorticosteroids or non-steroidal anti-inflammatory drugs in the previous 3 months. Information obtained included signalment, history, physical examination, clinicopathological testing (haematology, serum biochemistry and total thyroxine [T4]), faecal parasitology and abdominal ultrasonography. Serum cyanocobalamin, folate and specific feline pancreatic lipase immunoreactivity (fPLI) were included if available. Retroviral status of all cats was negative.
Feline CE activity index (FCEAI) was retrospectively calculated for all cats. 2 This scoring is composed of gastrointestinal signs (vomiting, diarrhoea, anorexia, weight loss, lethargy; each sign received 0–3 points), abnormal total protein concentration (yes = 1, no = 0 point), hypophosphataemia (yes = 1, no = 0 point), increased serum alanine aminotransferase and/or alkaline phosphatase activities (yes = 1, no = 0 point) and endoscopic lesions (yes = 1, no = 0 point). A composite score was subsequently calculated yielding values for mild (2–5), moderate (6–11) and severe (⩾12) enteropathy. 41
According to histological diagnosis, the cats were grouped in IBD and LGAL. Biopsies from three healthy control female cats (HCC; mean age 30 months) were taken by endoscopy before ovariohysterectomy (control group). These cats did not present endoscopic abnormalities, gastrointestinal signs or disease.
Owners of the HCC provided signed consent and procedures were approved by the UAB ethical committee (CEAAH 2354).
Collection of endoscopic biopsy specimen
Samples of duodenal mucosa were collected by endoscopy. A minimum of six biopsies from each diseased cat was obtained from the duodenum and fixed in 10% neutral buffered formalin, embedded in paraffin wax, sectioned at 5 µm and stained with haematoxylin and eosin. An additional three biopsies from each cat were stored in RNA later for 24 h and subsequently maintained at −80°C until RNA isolation.
Histopathological classification and immunohistochemistry
For IBD, biopsies were scored according to the criteria of a board-certified pathologist as normal (score 0), mild (1), moderate (2) and severe (3) enteritis based on the architecture features and cellular infiltration of the section. LGAL cases were identified according to the National Cancer Institute working formulation.42,43 When the lymphoma presented 0–5 mitoses per high-power field and a nuclear size <1.5× the size of a red blood cell was classified as LGAL.42,43
CD3 and CD79a immunophenotyping was performed in LGAL and severe IBD cases as previously described. 16
Total RNA extraction and complementary DNA synthesis
Total RNA was individually extracted from duodenum using 500 µl per sample of TRI reagent (Molecular Research Center) and following the manufacturer’s instructions. RNA quantification was carried out with a Nanodrop ND-1000 (Thermo Scientific) and RNA quality checked with the Experion RNA StdSens Analysis Kit (BioRad). RNA integrity values obtained were >8, indicative of excellent RNA integrity and quality. Reverse transcription (RT), to generate cDNA, was performed using 1 µg total RNA, denatured at 70°C for 10 mins with Oligo dT15primer (Promega) and SuperScript III Reverse Transcriptase enzyme (Invitrogen) in the presence of the recombinant ribonuclease inhibitor RNaseOUT (Invitrogen) in a final volume of 20 μl. The reaction was performed at 37°C for 1 h and heat inactivated at 70°C for 15 mins.
Primer design and transcriptional analysis
Primers used for the MDR1 and COX2 gene expression study were designed with Primer3 version 4.0 based on target sequences obtained from the Felis catus database (Table 1). Efficiency of the amplification was determined for each primer pair using serial 10-fold dilutions of pooled cDNA. The efficiency was calculated as E = 10(–1/s) where s is the slope generated from the serial dilutions, when log dilution is plotted against ΔCt (threshold cycle number). 44 The analysis of mRNA levels of target genes, were assessed with absolute RT-quantitative real-time PCR (RT-qPCR).
Sequences and efficiencies of primers used for quantitative real-time PCR analysis in duodenal samples of cats

All reactions were run in the Bio-Rad CFX384 Real-Time PCR Detection System, according to the following protocol: 95°C for 5 mins, 40 cycles at 95ºC for 30 s and 60ºC for 30 s. All samples were run in triplicate.
Determination of plasmid copy number in recombinant Escherichia coli was used for absolute quantification. 45 The pGEM easy vector (Promega) with the amplified product of RT-qPCR was transformed into E coli DH5alpha cells. Bacteria were grown in LB medium overnight and DNA vectors were obtained using Nucleospin Plasmid Quickpure kit (Machery-Nagel), digested with EcoRI to check the insert (Promega) and sequenced with T7 and SP6 primers. The plasmid copy number was determined using the following equation:

Absolute qPCR was carried out under the same conditions using a 107–101 copies/ml dilution of plasmid DNA (pGEM; Promega). Standard curves (Ct-threshold cycle vs log copy number) were constructed for sample copy number determination.
Statistical analysis
Statistical analysis was performed using SPSS statistics software (version 17.0; IBM) adopting a level of significance of P <0.05. Data were determined being non-parametric, therefore the Kruskal–Wallis test was used to compare continuous variables (FCEAI, histopathological score and gene expression) between groups. ANOVA with Tukey’s post-hoc test was used to test differences between IBD and LGAL groups regarding expression of COX2 and MDR1 mRNA. Linear correlations between variables were evaluated using the Pearson correlation coefficient.
Results
A total of 29 cats met the inclusion criteria. According to histological diagnosis, 20 cats were diagnosed with IBD and nine with LGAL. Median age was 119.5 months (range 30–201 months) for the IBD group; for the LGAL group median age was 140 months (range 84–205 months). A statistical difference was not found (P = 0.248). Furthermore, the IBD group presented with a significantly higher body weight (median 4.58 kg, range 3.20–5.44 kg) than the LGAL group (median 3.20 kg, range 2.27–4.25 kg; P = 0.027). No difference was observed regarding body condition score between cats with IBD (median 4) and LGAL (median 3; P = 0.246).
All cats in the IBD group were neutered. There were seven (35%) female and 13 (65%) male cats in the IBD group; the LGAL group was composed of six (67%) female and three (33%) male cats. Breeds represented in the IBD group were domestic shorthair (DSH; n = 11), Siamese (n = 5), domestic longhair (n = 3) and British Shorthair (n = 1) cats. The LGAL group was composed of DSH (n = 7), Birman (n = 1) and Persian (n = 1) cats.
The most common clinical sign in all cats was weight loss (94%), followed by vomiting (81%), lethargy and inappetence (71%), and diarrhoea (68%). Median FCEAI score in the LGAL group (11, range 5–14) was significantly higher than in the IBD group (9, range 5–13; P = 0.030).
Clinicopathological abnormalities detected in this study are summarised in Table 2.
Clinical pathological abnormality in cats with inflammatory bowel disease (IBD) and low-grade alimentary lymphoma (LGAL)

Table includes only parameters that showed alterations
ALT = alanine aminotransferase; fPLI = feline pancreatic lipase immunoreactivity (Spec fPL); – = within reference interval
Concurrent comorbidities were present in some patients with IBD: one cat presented with acute cholangitis, one with chronic cholangitis and one with chronic bronchitis. fPLI serum concentrations were elevated in seven cats. Previous comorbidities under treatment were reported in one cat with hyperthyroidism and one cat with hypercalcaemia. In the LGAL group, comorbidities were also presented: one cat with chronic kidney disease (CKD), one with CKD and hyperthyroidism that was controlled with methimazole treatment, one with hepatic lipidosis, one with increased fPLI and one with hyperthyroidism previously treated with radioiodine therapy.
With respect to the inflammatory cells infiltrating the lamina propria, all cats presented with lymphoplasmacytic inflammation, although three of the cats had concurrent mild neutrophilic infiltration. Twelve cases corresponded to mild, six to moderate and two to severe enteritis. All lymphomas were identified as T-cell immunophenotype. Regarding the control group, all biopsies were scored as normal.
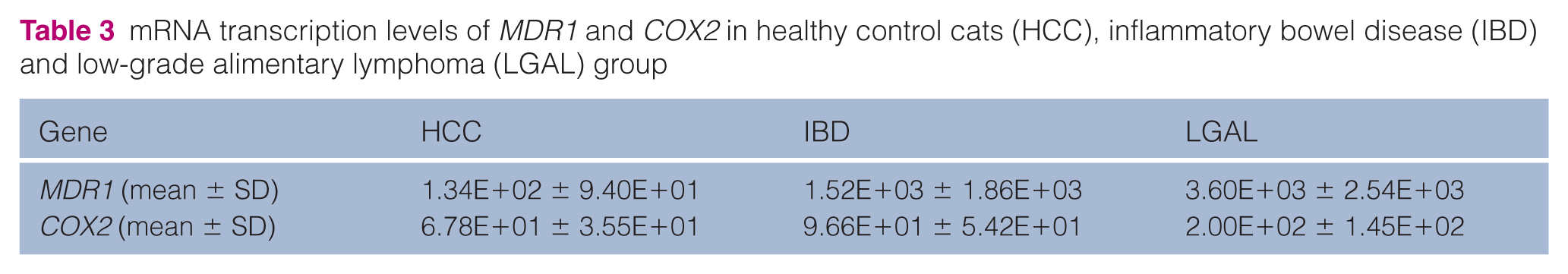
According to the gene expression results, cats with LGAL showed statistically significant higher expression levels than cats with IBD for both MDR1 (P = 0.023) and COX2 (P = 0.034) (Figure 1; Table 3). In addition, the HCC group showed lower expression values than both groups (Table 3), but this group was not included in the statistical analysis owing to the low number of cats.
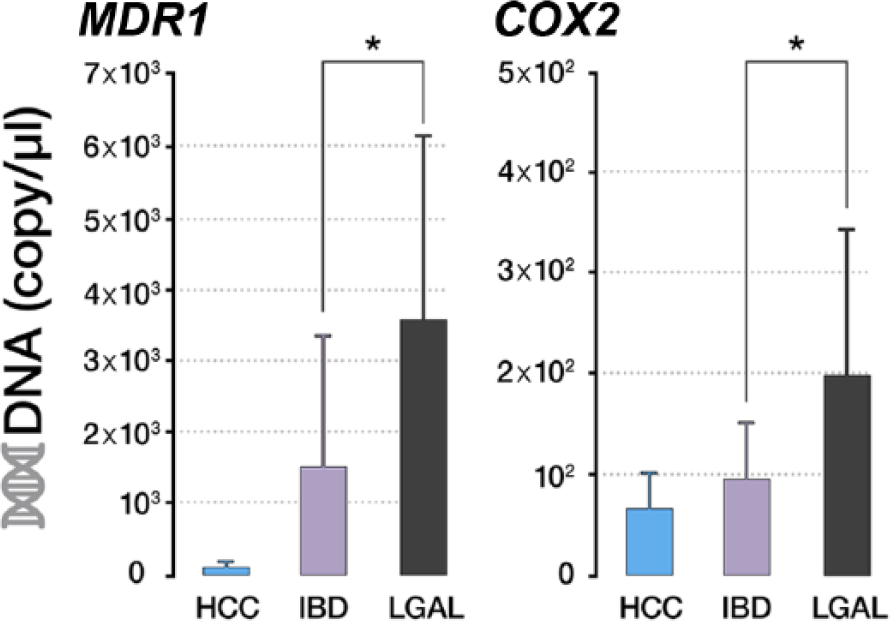
mRNA levels of MDR1 and COX2 genes in duodenal samples of cats. Analysis of mRNA levels was assessed with absolute reverse transcription quantitative real-time PCR. Values represent the means ± SE (n = 3 healthy control cats [HCC]; n = 20 inflammatory bowel disease [IBD]; n = 9 low-grade alimentary lymphoma [LGAL] group). Statistically significant differences between groups are LGAL vs IBD (*P <0.05). HCC group was not included in the statistical analysis
mRNA transcription levels of MDR1 and COX2 in healthy control cats (HCC), inflammatory bowel disease (IBD) and low-grade alimentary lymphoma (LGAL) group
There were no significant correlations detected between FCEAI, histological grade of IBD and either COX2 or MDR1 duodenal gene expression, nor between both genes in the studied groups (P >0.050; r <0.344).
Discussion
The results of the present study confirm previous findings that patients with IBD tend to be younger cats and present with higher body weight than cats with LGAL, although a significant overlap was present.1–6,16,46 Male cats, DSH and Siamese cats were also over-represented in the IBD group, although a predisposition to this disease has been proposed for the latter breed.1–6,16,46 Clinical signs described herein are similar to those previously reported.1–6,16,46,47
Cats with IBD from a previous study presented with similar FCEAI scores to those included in the study herein. 2 To our knowledge, FCEAI has not previously been applied to cats diagnosed with LGAL. A statistically significant difference was found in the FCEAI score between the two diseased groups, likely because LGAL presents with more severe clinical signs and/or chronicity.
Clinicopathological alterations found in this study are similar to previous reports of cats with IBD or LGAL, although none of the cats with LGAL presented with anaemia, which has been reported in other studies.2,3,5,46,47 Furthermore, according to Table 2, hypoproteinaemia, hypoalbuminaemia and hypocobalanaemia were more frequently seen in our LGAL group, similar to previous reports.5,48
Comorbidities were present in several cats in this study. A recent report described that cholangitis is more commonly associated with IBD than triaditis followed by pancreatitis. 47 However, in the present study, fPLI was increased in 50% of cats suffering from IBD vs only two cats with cholangitis confirmed by histology and none of the studied cats presenting with signs of triaditis. Nevertheless, pancreatic or hepatic biopsies were not taken from all patients to ascertain a diagnosis of pancreatitis or cholangitis. 49 Other comorbidities (hepatic lipidosis, CKD, hyperthyroidism, chronic bronchial disease) that were present in these cats have been previously reported in medium-age to elderly cats.47,50–52
Consistent with previous reports, lymphoplasmacytic inflammation was the most common inflammatory pattern defined in IBD and T-cell immunophenotype in LGAL cats.3,6,11,13–16,53
In the present study, HCC showed only mild MDR1 expression. Healthy humans have moderately high MDR1 mRNA expression levels in jejunum and colon. In mice, the highest mRNA levels of MDR1 were found in the normal ileum and colon, and these levels gradually declined proximally towards the jejunum, duodenum and stomach.21,54–56 In humans with IBD, greater and differential MDR1 mRNA levels have been found in Crohn’s disease vs healthy controls, which is similar to our findings. 57 Contrarily, ulcerative colitis has been associated with low expression of the MDR1 gene vs healthy people. 24 In the present study, MDR1 mRNA levels tended to be higher in IBD and LGAL than in HCC, and a statistically significant difference has been observed between the diseased groups. It is not possible to determine if increased MDR1 levels play a role in the aethiopathogenesis of feline IBD or if it is a consequence of the inflammation, as previously reported in mice. 58 In addition, the higher levels of MDR1 gene expression observed in the LGAL vs the IBD group could explain the necessity of a more aggressive therapy in the lymphoma. Future studies to determine whether feline MDR1 gene expression is similar to that found in Crohn’s disease and to determine the contribution of this gene to IBD pathogenesis and the implication with resistance to treatment are needed.20,59
To our knowledge, so far there have been no studies evaluating MDR1 gene expressionin LGAL and IBD in cats; the MDR1 gene has only been studied in a feline lymphoma cell line. 60 In dogs, gastrointestinal lymphoma showed higher mRNA levels of MDR1 in all cases, which is similar to our findings, although there was no mention of histopathological grade and immunophenotype in that study. 61 A recent study reported that canine multicentric lymphomas showed higher mRNA levels of MDR1; however, T-cell type was associated with lower levels in comparison to B-cell lymphomas that showed overexpression. 62 This study included only cats with T-cell immunophenotype; therefore, cats with B-cell lymphoma should be recruited to compare and determine which immunophenotype shows a higher expression. Otherwise, the evidence suggests that MDR1 gene expression in untreated human lymphoma is variable with 10–50% of tumours being positive, whereas 100% of our cases were positive.63,64 Further studies are needed to draw conclusions about MDR1 gene expression in different anatomical locations of feline lymphoma, as well as immunophenotype, and if there is a difference in the patterns of gene expression in treated cats vs untreated cats, as well as its role as a prognostic marker.
Overexpression of COX2 in IBD from gastrointestinal samples of humans and rats, and colonic biopsies of dogs, has been observed vs low levels present in control individuals.34,65 In dogs, mRNA expression was not statistically different between duodenal samples from control and dogs with IBD, but higher COX2 expression was observed in the latter. 66 This may be similar to our findings that cats with IBD tended to present with higher level of COX2 than the HCC group. Untreated human lymphoma may present with high COX2 expression levels and this overexpression may be associated with cell proliferation and angiogenesis.38,67 This may explain how the LGAL group showed higher COX2 expression than cats with IBD. Furthermore, COX2 upregulation in human lymphomas has been associated with the aggressiveness of the tumour, relapse, poor response to therapy and short overall survival.68–71 This aspect was not evaluated in our study because follow-up was not available. Studies with a larger group of cats with IBD and different grades of alimentary lymphoma, and follow-up would be needed to obtain conclusions.
Correlation between COX2 and P-gp immunoexpression has been reported in hepatocellular cancer cells, breast cancer cells and ovarian cancer cells in humans.72–74 One report of human colorectal cancer described correlations between both genes since blocking COX2 downregulated the expression of MDR1. 75 However, no correlation was shown in our study, similar to a previous study in human non-Hodgkin’s lymphoma. 40 Future studies are needed in cats to confirm the correlation of the studied genes and to determine if COX2 inhibitor treatment may be beneficial for LGAL.
The study has several limitations. The diagnosis of LGAL was made based on histopathology and immunohistochemistry features; both combined techniques have a sensitivity of 78% and specificity of 99%. 16 Addition of PCR for antigen receptor rearrangements increases sensitivity to 82.6%, but this test was not performed in the present study; thereby, misdiagnosis is possible. 16 Furthermore, only duodenal biopsies were included in this study, which may have influenced the results of the correlations since both diseases may also affect the jejunum and ileum.3,11,16 However, the main objective of the study was to describe the gene expression of inflammatory and tumour tissues. Another fact is the small number of sick cats likely affecting the power of the statistical analysis. In addition, owing to the small number of HCC, and the reduced number of cats with IBD and LGAL, the results should be considered with caution. A larger number of cats with IBD and follow-up information would be needed to differentiate them into food-responsive and steroid-responsive groups. Furthermore, follow-up would be necessary to determine the role of MDR1 and COX2 in resistance to treatment and prognosis in cancer as well as IBD.
Conclusions
MDR1 and COX2 gene expression is higher in cats with LGAL than in cats with IBD. These results suggest that these genes may be involved in the pathogenesis of IBD or LGAL in cats.
Footnotes
Acknowledgements
The authors thank the Royal Veterinary College of University of London, especially the Clinical Investigation Center (CIC), for all the samples and medical records yielded of cats with IBD and LGAL necessary to perform this study. Fundação para a Ciência e a Tecnologia JC Balasch is thankfully acknowledged for the graphic design of the figures and tables.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
M Teles has a post-doctoral fellowship from Fundação para a Ciência e a Tecnologia (SFRH/BPD/109219/2015) supported by the European Social Fund and national funds from the ‘Ministério da Educação e Ciência (POPH – QREN – Tipologia 4.1)’ of Portugal. J Castro-López received a grant from the Chilean government (Becas CONICYT) for his PhD studies.
