Abstract
Objectives
The aim of the study was to evaluate the analgesic efficacy of gabapentin–buprenorphine in comparison with meloxicam–buprenorphine or buprenorphine alone, and the correlation between two pain-scoring systems in cats.
Methods
Fifty-two adult cats were included in a randomized, controlled, blinded study. Anesthetic protocol included acepromazine–buprenorphine–propofol–isoflurane. The gabapentin–buprenorphine group (GBG, n = 19) received gabapentin capsules (50 mg PO) and buprenorphine (0.02 mg/kg IM). The meloxicam–buprenorphine group (MBG, n = 15) received meloxicam (0.2 mg/kg SC), buprenorphine and placebo capsules (PO). The buprenorphine group (BG, n = 18) received buprenorphine and placebo capsules (PO). Gabapentin (GBG) and placebo (MBG and BG) capsules were administered 12 h and 1 h before surgery. Postoperative pain was evaluated up to 8 h after ovariohysterectomy using a multidimensional composite pain scale (MCPS) and the Glasgow pain scale (rCMPS-F). A dynamic interactive visual analog scale (DIVAS) was used to evaluate sedation. Rescue analgesia included buprenorphine and/or meloxicam if the MCPS ⩾6. A repeated measures linear model was used for statistical analysis (P <0.05). Spearman’s rank correlation between the MCPS and rCMPS-F was evaluated.
Results
The prevalence of rescue analgesia with a MCPS was not different (P = 0.08; GBG, n = 5 [26%]; MBG, n = 2 [13%]; BG, n = 9 [50%]), but it would have been significantly higher in the BG (n = 14 [78%]) than GBG (P = 0.003; n = 5 [26%]) and MBG (P = 0.005; n = 4 [27%]) if intervention was based on the rCMPS-F. DIVAS and MCPS/rCMPS-F scores were not different among treatments. A strong correlation was observed between scoring systems (P <0.0001).
Conclusions and relevance
Analgesia was not significantly different among treatments using an MCPS. Despite a strong correlation between scoring systems, GBG/MBG would have been superior to the BG with the rCMPS-F demonstrating a potential type II error with an MCPS due to small sample size.
Introduction
Gabapentin is a lipophilic structural analogue of the inhibitory neurotransmitter gamma-aminobutyric acid, which has been used for the treatment of seizures and neuropathic pain. Its mechanism of action is unknown, but it involves interaction with voltage-gated calcium channels binding to the alpha2delta subunit.1,2 Systematic reviews and meta-analyses reveal that perioperative administration of gabapentin reduces acute postoperative pain in humans undergoing surgery, including abdominal hysterectomy.3–7 Hence, there is an interest in the use of gabapentin in veterinary medicine.
The analgesic efficacy of gabapentin in cats has only been reported in case series following major orthopedic procedures, 8 wound debridement, 9 and musculoskeletal pain and head trauma. 10 However, multimodal analgesic techniques used in these reports are considered confounding factors and the effects of gabapentin in cats within the perioperative period remain unclear. The drug could be an alternative in the management of acute pain in cats, owing to its high bioavailability (approximately 89%) after oral administration, a potentially favorable characteristic for an analgesic drug. 11 However, it did not produce thermal anti-nociception in laboratory cats. 12
The analgesic efficacy of a drug should be evaluated using valid pain scoring systems. In cats, the Glasgow composite measure pain scale (rCMPS-F) 13 and the UNESP-Botucatu multidimensional composite pain scale (MCPS)14–16 have been recently used for this purpose. However, their correlation has never been reported and it is not known whether study outcome would be the same using the two instruments.
The primary objective of this study was to evaluate the analgesic effects of gabapentin after oral administration in combination with buprenorphine (gabapentin–buprenorphine group [GBG]), and to compare these effects with those of a meloxicam and buprenorphine group (MBG), or buprenorphine alone group (BG) in client-owned cats undergoing ovariohysterectomy. A second objective was to evaluate the correlation between two pain-scoring systems in cats. We hypothesized that: (1) prevalence of rescue analgesia and pain scores would be lower in both the GBG and MBG when compared with the BG; and (2) a strong correlation between the pain scoring systems would exist. This study follows The Consolidated Standards of Reporting Trials (CONSORT guidelines; http://www.consort-statement.org/).
Materials and methods
The study protocol was approved by the animal care committee of the Faculty of Veterinary Medicine, University of Montréal (15-Rech-1794). The study was performed between July and November 2015. Figure 1 describes the timeline of events.

Timeline of events.
Animals
Sixty-nine client-owned domestic cats scheduled for ovariohysterectomy were recruited in a prospective, blinded, randomized, controlled clinical trial after obtaining the owners’ written consent. Animals were housed individually in adjacent cages of a cat ward in the veterinary teaching hospital (Centre Hospitalier Universitaire Vétérinaire) of the Université de Montréal. Food but not water was withheld for approximately 8–12 h before general anesthesia.
Inclusion and exclusion criteria
Individuals were included in the study if they were considered healthy based on medical history, complete physical examination, and hematocrit and total protein testing. Exclusion criteria included aggression, cardiac dysrhythmias on auscultation, pregnancy, lactation, obesity (body condition score >7 on a scale from 1 to 9), 17 anemia (hematocrit ⩽30%) and clinical signs of disease.
Experimental procedure
All cats were premedicated with acepromazine (0.02 mg/kg; Gentès & Bolduc) and buprenorphine (0.02 mg/kg Vetergesic; Champion Alstoe), administered as a single injection into the lumbar epaxial muscles (IM). Approximately 20 mins later, an intravenous catheter was aseptically inserted into the cephalic vein and propofol (Diprivan; AstraZeneca) was administered and titrated to allow endotracheal intubation with a cuffed endotracheal tube. Isoflurane (Isoflurane USP; Fresenius Kabi) in oxygen was administered using a non-rebreathing circuit for maintenance of anesthesia. Cats were positioned in dorsal recumbency on a circulating warm-water blanket. Monitoring was performed using a multi-parametric monitor (LifeWindow 6000V Veterinary Multiparameter Monitor; Digicare Animal Health), which included electrocardiography, capnography, inspired and expired concentrations of isoflurane, esophageal temperature and pulse oximetry. Blood pressure was measured with a Doppler ultrasonic flow detector (Parks Medical Electronics) using an appropriate neonatal cuff size. Balanced crystalloid solution (Lactated Ringer’s Solution; Baxter) was administered at a rate of 5 ml/kg/h intravenously throughout surgery. One veterinarian (BPM) with experience in surgery performed ovariohysterectomy. A 3 cm ventral midline incision was made and a modified three-clamp technique was used. Duration of surgery (first incision until placement of the last suture) and anesthesia (injection of propofol to turning off the vaporizer dial), and time to extubation (turning off the vaporizer dial until extubation) were recorded for each cat. Extubation was performed when palpebral reflexes returned.
Treatment groups
Individual treatment randomization was performed for 69 cats at once, and before the study had begun using a software (www.randomization.com) by two individuals (GMD and JB) who were not involved with treatment administration. Each eligible cat was assigned sequentially a number from 1–69 and this number was given based on the order of arrival. Cats were randomly assigned to receive one of the following three treatments: BG – no other analgesic treatment was administered for surgery; MBG – cats received meloxicam (0.2 mg/kg SC Metacam; Boehringer Ingelheim) immediately after induction of anesthesia. In the GBG, cats were administered gabapentin (50 mg PO). Gabapentin was compounded in gelatin capsules (size 4) in a lactose excipient (146 mg of lactose/capsule) by the institution’s pharmacy. Oral treatments were given approximately 12 h and 1 h before surgery. At these time points, both the MBG and BG received a capsule with lactose (placebo) identical to gabapentin capsules. These treatments were administered by the individual who performed pain assessments. Three milliliters of water were administered orally to each cat after administration of treatments using a syringe.
Data collection
Evaluations were performed before premedication which was approximately 60 mins prior to the induction of anesthesia (time 0, baseline) and at 0.5, 1, 2, 3, 4, 6 and 8 h after the end of surgery by an observer who was unaware of treatment administration. Cats were monitored for adverse effects (eg, ataxia, vomiting, diarrhea, etc) throughout the study period.
Pain and sedation scores
Sedation was evaluated by using a dynamic and interactive visual analog scale (DIVAS). 18 Briefly, the DIVAS is derived by using 100 mm lines. A conscious cat displaying normal behavior with no signs of sedation was marked as ‘0’. A cat in lateral recumbency that would not respond to a handclap was marked as ‘10’ (100 mm). Pain was evaluated using the rCMPS-F 13 and the MCPS in French. 14 Briefly, the rCMPS-F has been developed as a prototype to assess acute pain in cats based on behavioral changes with an intervention level score ⩾4/16. 13 The MCPS is a reliable and responsive instrument to evaluate pain after ovariohysterectomy that involves different domains such as ‘pain expression’ and ‘psychomotor change’. 15 The observer who evaluated pain completed approximately 6 h of video-based training on feline pain assessment using a website (www.animalpain.com.br) before the study had begun.
Rescue analgesia
The need for rescue analgesia was considered the primary outcome of the study and was based on MCPS pain scores. Supplemental analgesia was provided if MCPS scores were ⩾6 and consisted of administration of meloxicam (0.2 mg/kg SC) in the BG and GBG, and administration of buprenorphine (0.02 mg/kg IV) in all groups. The score for rescue analgesia was lower than the original scale in English because blood pressure recordings were not performed, thus lowering the maximum score of the scale. Data collected after the administration of rescue analgesia were not included in the statistical analysis; however, cats continued to be assessed for up to 8 h. Postoperative treatments were administered by investigators who were not involved with pain assessment. The number of cats requiring rescue analgesia and the number of times that rescue analgesia was administered for each cat were recorded. Cats in both the BG and GBG that did not require rescue analgesia during the studying period received a dose of meloxicam immediately after the 8 h time point. A second dose of meloxicam (0.05 mg/kg PO) was administered 24 h after the first dose.
Statistical analysis
Statistical analyses were performed using standard software (SAS version 9.3; SAS Institute). Power analysis was performed before the study. From Benito et al, 16 in which a similar study design was used, the authors assumed that the prevalence of treatment failure would be approximately 70% in the BG, and 20% in both the MBG and GBG. A power analysis with Fisher’s exact test indicated that a minimum sample size of 15 cats per group would be needed to detect a difference 80% of the times at an alpha level of 5%. Data were tested for normality with a Shapiro–Wilk test. Demographic data for each treatment group were compared using one-way ANOVA or χ2 test. Prevalence of rescue analgesia was compared among treatments with the exact χ2 test. A separate subgroup restricted to the cats that received rescue analgesic treatment (response variable ‘rescue time’) was analyzed using a general linear model with treatment as a factor; only the first rescue time was used for this analysis. Mean pain scores and DIVAS sedation of all cats were investigated with repeated-measures linear models with treatment as a between-subject factor and time as a within-subject factor. A priori contrasts were then performed between pairs of means adjusting the level of statistical significance for each comparison with the Benjamini–Hochberg procedure. These analyses were not performed after the 2 h time point because many cats received rescue analgesia by that time point and data were discarded afterwards; group comparisons would have been inappropriate. The Spearman’s rank correlation between the MCPS and rCMPS-F scores was evaluated. The percentage of disagreement for rescue analgesia (%DIRA) where one scale would indicate rescue, but not the other, was calculated between postoperative MCPS and rCMPS-F pain scores. Values of P <0.05 were considered statistically significant.
Results
Fifty-two cats were included in the final analysis. Seventeen cats were excluded after surgery owing to pregnancy (n = 6), signs of upper respiratory tract disease (n = 4), previous ovariohysterectomy (n = 3), aggressiveness (n = 2), anemia (n = 1) and limb deformity (n = 1). Table 1 shows body condition score, body weight, hematocrit, total protein, duration of anesthesia and surgery, and time to extubation. These variables were not different among treatment groups. Mean ± SD dose of gabapentin was 17.3 ± 3.7 mg/kg. Cats remained in hospital for at least 24 h after surgery. None of the cats developed postoperative complications or adverse effects.
Body condition score (BCS), body weight, hematocrit, total protein, anesthesia and surgery times, and time to extubation in cats undergoing ovariohysterectomy

Values are expressed as mean ± SD with the exception of BCS, which is reported as median (range)
GBG = gabapentin–buprenorphine group; MBG = meloxicam–buprenorphine group; BG = buprenorphine group
Rescue analgesia
Sixteen cats required rescue analgesia. Prevalence of rescue analgesia was not different using the MCPS (P = 0.08; GBG, n = 5 [26%]; MBG, n = 2 [13%]; and BG, n = 9 [50%]) (Table 2). Time to first rescue analgesia was not different among treatments (P = 0.77). Two cats (one in the GBG and one in the BG) required a second dose of rescue analgesia.
Number of cats receiving rescue analgesia with the UNESP-Botucatu multidimensional composite pain scale (MCPS) over time (h) following ovariohysterectomy

Table includes the first administration of rescue analgesia. Prevalence of rescue analgesia with the MCPS was not different (P = 0.08; GBG, n = 5 [26%]; MBG, n = 2 [13%]; and BG, n = 9 [50%]) among groups. Differences among treatments for the number of cats requiring first dose of rescue analgesia were not significant (P = 0.08)
GBG = gabapentin–buprenorphine group; MBG = meloxicam–buprenorphine group; BG = buprenorphine group
The prevalence of rescue analgesia would have been significantly different among treatments (P = 0.002) had the intervention been based on the rCMPS-F. In the latter case, prevalence of rescue analgesia would have been higher in the BG (n = 14 [78%]) than the GBG (P = 0.003; n = 5 [26%]) and MBG (P = 0.005; n = 4 [27%]) (Table 3).
Number of cats that would have received rescue analgesia if intervention had been based on the Glasgow feline composite measure pain scale (rCMPS-F) (scores ⩾ 4/16) following ovariohysterectomy

The prevalence of rescue analgesia would have been significantly higher in the BG (n = 14 [78%]) than GBG (P = 0.003; n = 5 [26%]) and MBG (P = 0.005; n = 4 [27%]) if intervention was based on the rCMPS-F
GBG = gabapentin–buprenorphine group; MBG = meloxicam–buprenorphine group; BG = buprenorphine group
Correlation between MCPS and rCMPS-F
The Spearman’s rank correlation indicated a strong association between MCPS and rCMPS-F scores (P <0.0001) (Figure 2). Correlation was strong at baseline, 0.5, 2, 3, 4, 6 (r = 0.7) and at 8 h (r = 0.6), and very strong at 1 h (r = 0.8). The %DIRA between postoperative MCPS and rCMPS-F pain scores for rescue analgesia was 7.8%; the outcome for rescue analgesia could have changed if intervention had been based on the rCMPS-F in 6.4% of cases.

Correlation between the UNESP-Botucatu multidimensional composite pain scale (MCPS) and the Glasgow feline composite measure pain scale (rCMPS-F) in cats following ovariohysterectomy. The Spearman’s rank correlation indicated a strong association between the MCPS and rCMPS-F (P <0.0001) pain scores. Black dots correspond to a number of paired scores using the two scales in 52 cats throughout the study (seven time points with a total of 364 observations)
DIVAS sedation
The DIVAS sedation scores were not different among treatments at any of the time points. Sedation scores were increased in the GBG (P <0.0001), MBG (P <0.0001) and BG (P = 0.0002) at 0.5 h when compared with baseline values (Figure 3).

Mean ± SEM scores for the dynamic and interactive visual analog scale (DIVAS) in cats following ovariohysterectomy. Sedation scores were not different among treatments.
MCPS/ rCMPS-F pain scores
The MCPS and rCMPS-F pain scores were not different among treatments (Figures 4 and 5) at baseline, 0.5, 1 and 2 h. The MCPS pain scores were increased for the GBG and BG treatments at 1 h (P = 0.0001 and P = 0.0005; GBG and BG, respectively) and 2 h (P <0.0001 and P = 0.0007; GBG and BG, respectively) when compared with baseline values. For the MBG treatment, the MCPS pain scores were not increased compared with baseline values at 0.5, 1 and 2 h (Figure 4). The rCMPS-F pain scores were increased for the GBG at 0.5 h (P <0.0001) and 1 h (P = 0.0003) when compared with baseline values. For the MBG and BG treatments, rCMPS-F pain scores after adjustments were not increased compared with baseline values at 0.5, 1 and 2 h (Figure 5).
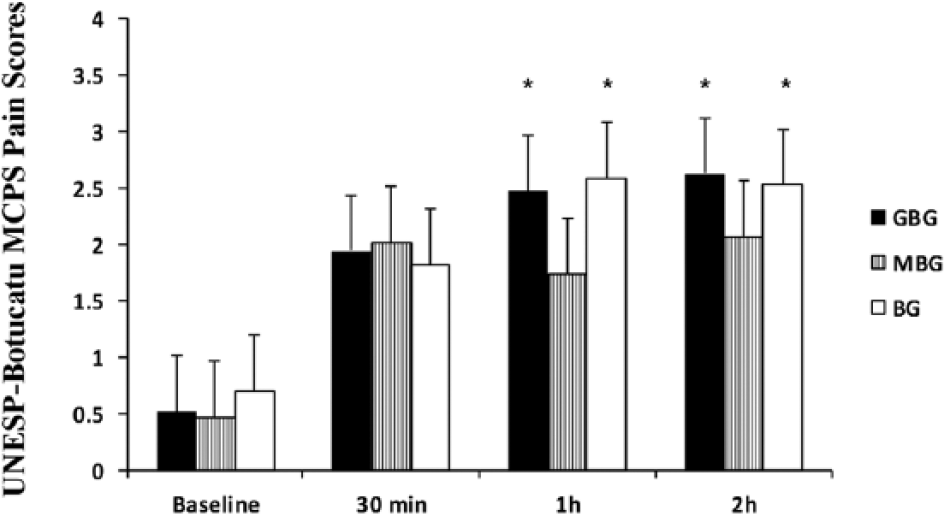
Mean ± SEM scores for the UNESP-Botucatu multidimensional composite pain scale (MCPS) in cats following ovariohysterectomy. Pain scores were not different among treatments.

Mean ± SEM scores for the Glasgow feline composite measure pain scale (rCMPS-F) in cats following ovariohysterectomy. Pain scores were not different among treatments.
Discussion
In this randomized clinical trial, the analgesic effects of gabapentin with buprenorphine were evaluated and compared with those of meloxicam–buprenorphine and buprenorphine alone. The latter two treatments are often used in the management of postoperative pain in cats and there is currently an interest in the use of the combination gabapentin–buprenorphine in feline practice considering that gabapentin is associated with reduced pain scores and postoperative analgesic requirements in humans. Indeed, the drug decreases opioid-induced adverse effects and incidence of chronic postoperative pain.3–7,19 In brief, the results of this study showed that postoperative pain scores and prevalence of rescue analgesia were not significantly different among treatments using the MCPS. However, a type II error may have occurred as a result of small sample size considering that prevalence of rescue analgesia would have been significantly lower in the GBG and MBG than BG using the rCMPS-F. Power calculations expected a prevalence of rescue analgesia of 70% in the BG and not 50%, as observed in this study. Another important finding was that the observed analgesic effect in the GBG was large and in the direction predicted when compared with the BG (two-fold difference between the GBG and BG), suggesting a clinically relevant finding masked by potentially low statistical power. Finally, analgesic effects in the GBG were not different than MBG using either the MCPS or rCMPS-F. Based on these findings, gabapentin could be an alternative for postoperative pain or as part of multimodal analgesic regimens, particularly when non-steroidal anti-inflammatory drugs (NSAIDs) are contraindicated.
Several limitations could explain the discrepancy between the results for the MCPS and rCMPS-F. First, prevalence of rescue analgesia was based on the MCPS, and not the rCMPS-F. A recent systematic review showed that the MCPS is the only pain scale that has been investigated for validity (construct, criterion and content), reliability (internal consistency, intra- and inter-rater, and test re-test) and sensibility (identification of a cut-off point and responsiveness) in cats. 20 For the original rCMPS-F, only construct and content validity were reported but sensitivity was moderate. 13 It is not known whether these factors affected pain scoring and the results. Second, a revised version of the rCMPS-F includes a simple three-point facial scale and a recommended intervention level of ⩾5/20. 21 This new version improved discriminatory ability (lower misclassification rates) to differentiate painful and pain-free cats compared with the previous rCMPS-F. 21 Perhaps the inclusion of facial expressions could have changed pain scores and outcome related to rescue analgesia in the present study. Third, pain scoring was performed using the instruments in two different languages. The MCPS in French has been validated 14 and was preferred for rescue analgesia for the reasons discussed in terms of validity, and as it was in the native language of the observer who assessed pain. Language, gender and cultural differences have been shown to produce intrinsic bias in the assessment of pain in cats. 22 Fourth, pain was assessed by a fourth-year veterinary student. This observer received video-based training on feline pain assessment, but it is possible that the lack of experience with real-time assessment played a role in the results. A previous study showed that veterinary students may provide lower pain scores using the MCPS than individuals with previous experience in pain assessment. Indeed, scores can be significantly different, and depending who assesses pain, the outcome for rescue analgesia might change. 22 In addition, despite a ‘strong’ or ‘very strong’ correlation between the MCPS and rCMPS-F when used by the same observer, the disagreement between the two instruments (%DIRA) was clinically relevant if one considers that for some of these cats, one scale had indicated the administration of supplemental analgesia while the other did not. Further studies using the MCPS and the revised version of the rCMPS-F in the same language, by an observer who is experienced with pain assessment, might provide additional information on these issues. Additionally, a non-inferiority trial comparing the postoperative effects of buprenorphine with gabapentin vs buprenorphine with an NSAID may provide a definitive answer on the efficacy of gabapentin and buprenorphine for treatment of acute pain in cats.
The optimal dose of gabapentin in the treatment of acute pain has not been established. Systematic reviews and meta-analysis showed that single doses from 300–1200 mg are administered 1–2 h before surgery in humans. Few studies reported the administration of multiple doses as described in this study.3–5,7 At this time, it is not clear if multiple preoperative doses are advantageous over a single dose before surgery. 7 In the present clinical study, a dose of 50 mg was chosen based on: (1) clinical experience, expert opinion or consensus in veterinary medicine;23,24 (2) a previous pharmacokinetic study where high bioavailability was reported following oral administration of 10 mg/kg gabapentin in cats; 11 and (3) studies in humans where approximately 15 mg/kg is administered for preoperative pain management (ie, 1200 mg for an individual weighing 80 kg). Dosage regimens in this study seemed to be clinically appropriate; nevertheless, the ideal therapeutic level cannot be confirmed because plasma concentrations of gabapentin were not concomitantly analyzed. Controversies regarding the perioperative analgesic efficacy of gabapentin exist in the human literature. Perioperative administration of gabapentin provided pain relief and decreased opioid requirements after hysterectomy, 7 lumbar spine surgery 6 and total hip arthroplasty. 4 Nevertheless, a recent meta-analysis with trial sequential analyses including 132 randomized clinical trials revealed that firm evidence for use of gabapentin is still lacking. 25
The MCPS and rCMPS-F instruments could not detect significant differences among treatments, likely because cats were removed from statistical analysis following rescue analgesia. This was performed to avoid analysis bias while at the same time introducing selection bias. The approach limited the ability to detect significant differences among treatments using pain scores. However, these scores were increased in the GBG and BG at 1 and 2 h using the MCPS, and in the GBG at 0.5 and 1 h using the rCMPS-F when compared with baseline values. This reflects not only that pain scores are commonly higher after surgery when compared with baseline values, but also that some individuals in these groups could have higher pain scores than in the MBG since the same observations were not recorded in the latter group. Removing cats from statistical analysis after rescue analgesia also limited the ability to evaluate treatment effect from 2–8 h postoperatively and when acute inflammation is present. However, these cats were monitored throughout the studying period and rescue analgesia, the primary outcome of this study, could be provided any time.
There was of particular interest in the evaluation of sedative effects produced by gabapentin since sedation and dizziness have been reported as an adverse effect in humans.3,4,7 One could argue that acepromazine confounded the assessment of sedation; however, the drug is commonly administered as part of premedication for its anxiolytic properties. In addition, acepromazine was administered to all cats enrolled in the study. Therefore, any differences in DIVAS sedation scores among treatments could be attributed to the administration of gabapentin. Sedation scores were significantly increased at 0.5 h when compared with baseline for all treatments. This is not surprising since residual effects of anesthesia are still observed early after extubation. In cats, sedation or other gabapentin-induced adverse effects have not been described.8–11 For healthy cats undergoing ovariohysterectomy, gabapentin did not increase sedation scores or outwardly detectable adverse effects when compared with other treatments.
Conclusions
Postoperative analgesia was not significantly different among treatments using the MCPS. Despite strong correlation between scoring systems, outcome for rescue analgesia may differ when using the MCPS and rCMPS-F. Both the GBG and MBG would have been superior to the BG, with the rCMPS-F demonstrating a potential type II error with the MCPS due to small sample size. Gabapentin in combination with opioids might be an important alternative in the treatment of perioperative pain in cats.
Footnotes
Acknowledgements
The authors acknowledge Dr Geneviève Luca for technical help during the study.
Author note
This paper was presented, in part, at the Association of Veterinary Anaesthetists Autumn Meeting, Prague, Czech Republic, September 2016, and Association of Veterinary Anaesthetists Spring Meeting, Manchester, UK, April 2017.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article. Dr Beatriz Monteiro is a recipient of the Vanier Canada Graduate Scholarship.
