Abstract
Objectives
The aims of this study were to describe the paravertebral brachial plexus (PBP) blockade in cats, and compare the spread of bupivacaine–methylene blue solution (BMS) using two volumes of injection into the PBP.
Methods
Six cadavers (4.4 ± 1.8 kg) randomly received 0.15 ml/kg (LOW) or 0.2 ml/kg (HIGH) per injection of BMS into the PBP. The first injection was made ventral to the transverse process of the sixth cervical vertebra (nerves C6 and C7) at the emergence of nerve roots. A second injection was performed at the edge of the cranial border of the scapula and directed ventrally towards the first rib (C8 and T1). Cadavers were dissected and the presence of BMS in contact with the nerves (success rate) and length of staining were evaluated for each nerve (C6/C7/C8/T1). Length of target nerves in contact with BMS was measured and <15 mm was considered fair, ⩾15 mm sufficient and >40 mm excessive. Complications (aspiration of blood, staining of pleural space and/or spinal cord) were recorded. Paired t-test, Mann-Whitney U-test and χ2 test were used for statistical analysis when appropriate (P <0.05).
Results
Eleven hemilateral brachial plexuses were injected. Aspiration of blood occurred during one injection. Inadvertent pleural or spinal staining was not observed. Success rates (positive nerve staining) in the LOW and HIGH groups, respectively, were as follows: C6: 83% and 80%; C7: 66% and 100%; C8: 66% and 100%; T1: 50% and 60% (P >0.05). Median (range) length of nerve staining was 22 mm (15–39 mm) for LOW and 21 mm (13–35 mm) for HIGH (P >0.05). Excessive nerve staining was not recorded.
Conclusions and relevance
This study describes the PBP blockade in cats with acceptable nerve staining and minimal complications. Success rate and length of nerve staining were not significantly different between the LOW and HIGH groups.
Introduction
Loco-regional anesthetic techniques are used to reduce intraoperative anesthetic requirements and to improve perioperative pain relief as part of a multimodal analgesic approach.1,2
Brachial plexus nerve blockade is often performed in small animal patients undergoing thoracic limb surgery. The axillary brachial plexus (ABP) blockade can be performed either at the level of the scapulo-humeral joint,3,4 or cranial to the acromion. 5 In cats, this technique has been described using electrical nerve stimulation and ultrasonography.2,6 The ‘RUMM’ block has been described in dogs to anesthetize the radial, ulnar, median and musculocutaneous nerves, and is performed at the level of the mid-humerus. 7 A third technique involves needle insertion in proximity with the nerve branches where they emerge from the spinal cord at the level of the intervertebral foramina. This is known as the paravertebral approach for brachial plexus (PBP) blockade. 8 The PBP approach aims to block the emerging cervical (C6, C7, C8) and thoracic (T1) nerve roots. The ABP and RUMM techniques provide anesthesia distal to mid-humerus, whereas the PBP blockade may anesthetize the scapular, humeral, radio-ulnar, carpal and digital areas. The PBP blockade has been described in dogs but not in cats.8–10
The aims of this study were to describe the PBP blockade in cats, and compare the spread of two different volumes of bupivacaine–methylene blue (1:1) solution (BMS) after PBP injections in cat cadavers.
Materials and methods
The cadavers of six cats (4.4 ± 1.8 kg) that had been euthanized for reasons unrelated to the study were included. Cadavers were frozen and thawed overnight before the study. Each cat was positioned in lateral recumbency with the thoracic limb to be injected extended caudally. A foam pad was placed below the neck to facilitate positioning. The skin was clipped and cleaned. An insulated needle (Stimuplex A, 22G, 50 mm; B Braun) was attached to a 3 ml syringe and used for injections. The needle and extension tubing were pre-filled with BMS.
Technique
The transverse process of the sixth cervical vertebra was identified by palpation. The needle was inserted dorsal to the transverse process (2–3 cm from the spinous process) in a ventral direction at a 30–45° angle in sagittal plane, until it contacted the transverse process. This first injection was used to target, and stain, the C6 and C7 nerve roots (Figure 1a). For the second injection, the first rib and mid-point of the cranial border of the scapula were identified by palpation. The needle was inserted at the edge of the cranial border of the scapula and directed ventrally towards the first rib at a 30° angle in sagittal plane and dorsal to the costochondral junction. The second injection was used to target, and stain, the C8 and T1 nerve roots (Figure 1b). Injections were performed after negative aspiration of blood and lack of resistance to injection. One cat received a unilateral injection on the right side as the left side was used for an anatomical study of the PBP (Figure 2).
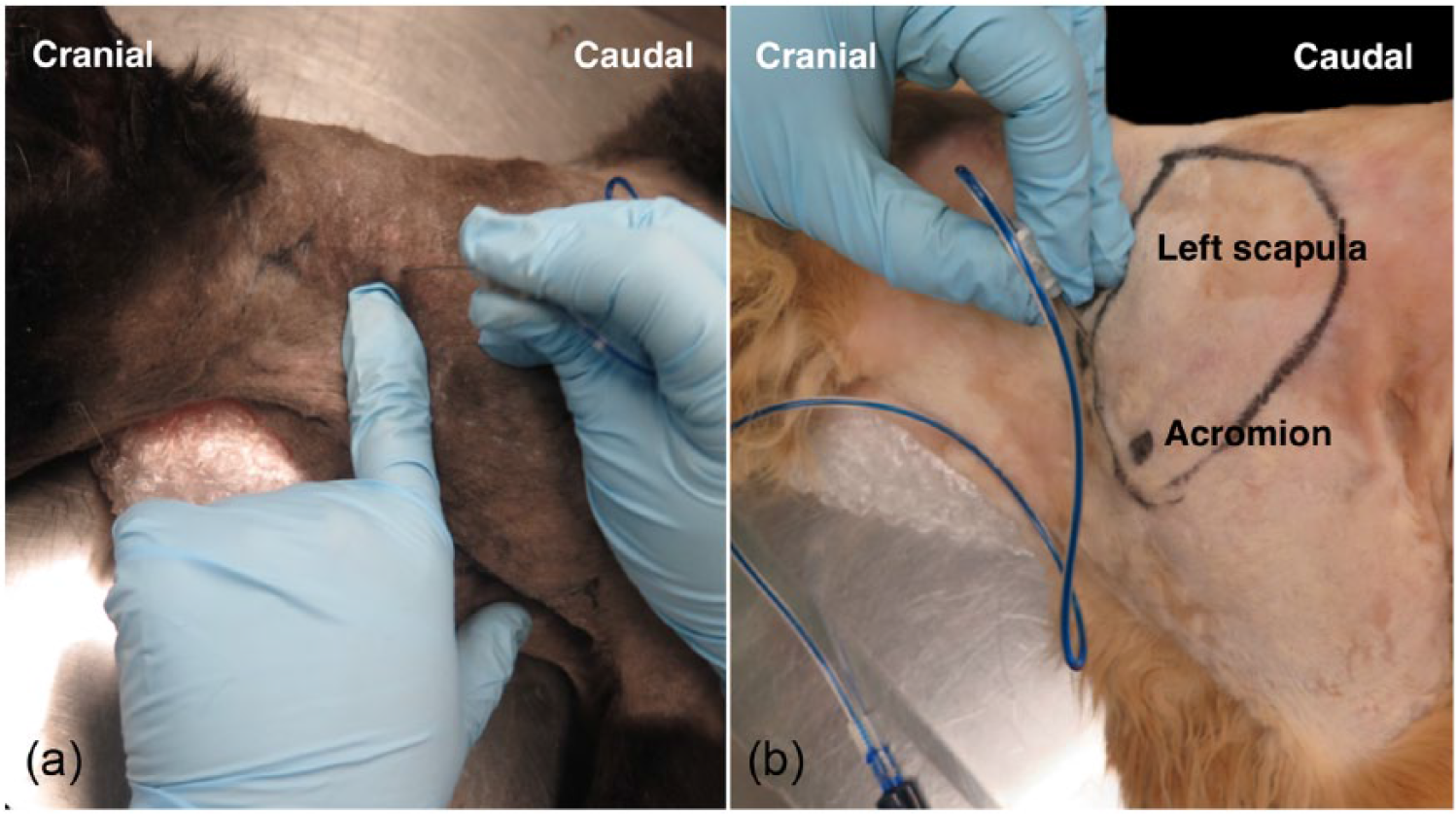
Paravertebral approach to the brachial plexus in a cat. (a) First injection point (C6 and C7 nerve roots). The needle was inserted perpendicular to the skin until it reached the transverse process of the sixth cervical vertebra. (b) Second injection point (C8 and T1 nerve roots). The left thoracic limb was extended caudally and the needle was inserted at the edge of the cranial border of the scapula and directed ventrally towards the first rib

Dissection of the left brachial plexus in a cat. Muscles of the cervical and cranial thoracic areas were removed and the scapula was retracted to allow visualization of C6, C7, C8 and T1 nerve roots and their branches. *Palpable landmarks for injections (transverse process of the sixth cervical vertebra and first rib). A and B = location for first and second injections, respectively; AA = axillary artery; AV = axillary vein; 1 = suprascapular nerve; 2 = subscapular nerve; 3 = axillary nerve; 4 = musculocutaneous nerve; 5 = radial nerve; 6 = median nerve; 7 = ulnar nerve
Assessment
Cadavers were assigned using a random sequence generator (www.random.org) to receive either 0.15 ml/kg (LOW; n = 3) or 0.2 ml/kg (HIGH; n = 3) per injection of BMS. Immediately after injections, the skin was incised to allow visualization and dissection of the acromiotrapezius and spinodeltoid muscles. The rhomboideus and infraspinatus muscles were then incised and the scapula retracted. Nerves of the brachial plexus were dissected close to the intervertebral foramina and first rib. Additionally, the aspiration of blood prior to injection, and staining of visceral pleura, mediastinum and spinal cord were recorded. These were considered to be complications of the technique.
Presence of BMS in contact with the nerves (success rate) was calculated for each nerve (C6, C7, C8 and T1) stained after each block. Length of target nerves in contact with BMS was measured with a ruler and <15 mm was considered fair, ⩾15 mm was considered sufficient and >40 mm was considered excessive.
Statistical analysis
Statistical analysis was performed using a statistical online calculator (Socsci Statistics, www.socscistatistics.com/tests/Default.aspx). Data were tested for normality using the Shapiro–Wilk test. Data are reported as mean ± SD with the exception of length of stained nerves, which is reported as median (range). Groups were compared using a Student’s t-test (parametric data) and Mann-Whitney U-test or the χ2 test (non-parametric data). Significance was defined as P <0.05.
Results
Eleven hemilateral brachial plexuses of six cadavers were injected with BMS. Success rate and length of nerve stained with BMS are presented in Table 1. Overall percentage of successful staining was 85% in the HIGH group and 66% in the LOW group (P = 0.59). The length of nerve staining was sufficient (⩾15 mm) in 15/16 injections in the LOW group and in 14/17 injections in the HIGH group (P = 0.79). Excessive nerve staining (>40 mm) was not observed (Figure 3).
Success rate and length (mm) of nerve staining by the bupivacaine–methylene blue solution using the paravertebral brachial plexus approach

Data are n (%) unless otherwise indicated. Success rate and length of nerve staining were not different between groups. Six cat cadavers (11 hemilateral brachial plexuses) were randomly assigned to receive either LOW (0.15 ml/kg) or HIGH (0.2 ml/kg) volume per injection. Two injections were performed for each block
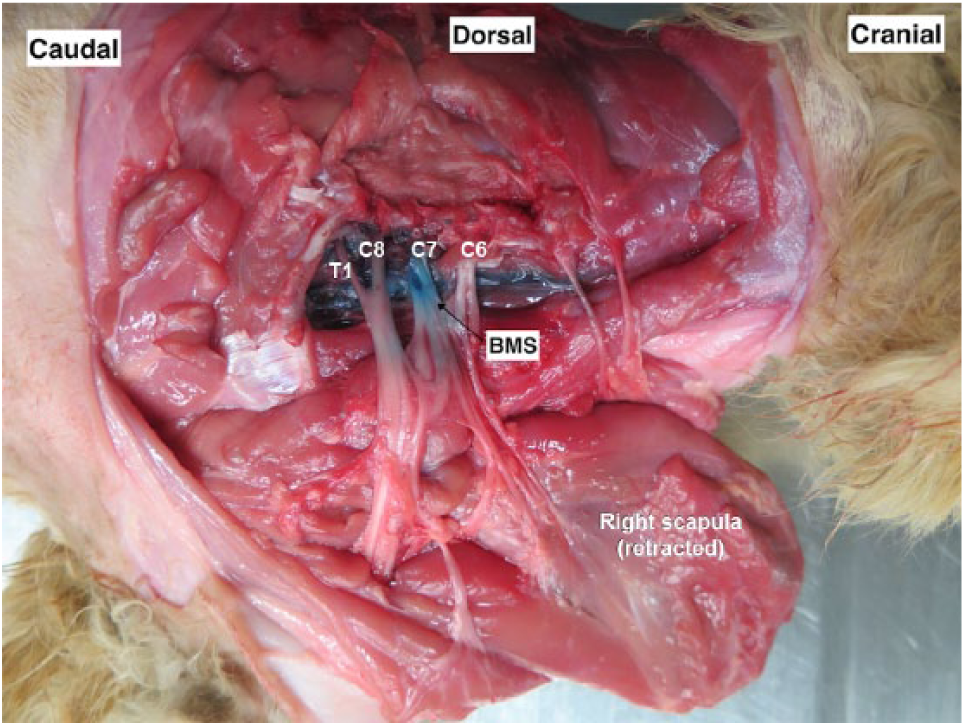
Dissection of the right brachial plexus in a cat after injection of bupivacaine–methylene blue solution (BMS) using the paravertebral brachial plexus approach. The spread of BMS over the C6, C7, C8 and T1 nerve roots was recorded
For one injection site (C6 and C7; HIGH group), aspiration of blood from an unintentional puncture of the jugular vein was noted. The needle was repositioned and injection was performed after negative aspiration of blood. No other complications were observed.
Discussion
In this study using cadavers, the PBP blockade was described in cats using two volumes of BMS. Anatomical studies are useful for identification of landmarks and potential risks, and refinement of local anesthetic techniques before their application in clinical studies.
Aspiration of blood from an unintentional puncture of the jugular vein was recorded once in this study. Negative aspiration of blood before injection is recommended to confirm needle location due to the proximity of the C6 and C7 nerves and the jugular vein. Inadvertent intravenous administration of local anesthetics such as bupivacaine can produce severe signs of cardiotoxicity (ie, hypotension, dysrhythmias, etc) and could be a problem in clinical practice. Puncture of the pleural space/thorax could also be a potential complication with this technique. 11 Indeed, migration of injectate to the pleural space has been reported with the ABP approach; 6 however, staining of the thoracic cavity was not recorded in this study. Accidental intravenous administration of local anesthetic is a concern with this technique.
Phrenic nerve staining was not evaluated in this study; however, its blockade is a potential complication of the brachial plexus technique (both PBP and ABP).9,10 The paravertebral approach inevitably blocks the phrenic nerve as the latter arises from C5 and C6. However, respiratory function due to diaphragmatic hemiparesis was not impaired by unilateral phrenic nerve blockade in dogs. 12
Bupivacaine was mixed with methylene blue solution in an attempt to mimic a clinically relevant solution with similar density and physical properties. One of the limitations of this study is that the distribution of injectate in a clinical patient may be different when compared with cadavers. The actual effects of methylene blue solution in the physical properties and spread characteristics of a local anesthetic are unknown; however, this solution has been used in several similar anatomical studies.4,6,8,13
The study did not identify significant differences between the two volumes of BMS injection; however, a higher overall success rate was obtained in the HIGH group. Body condition score was not systematically evaluated; however, palpation of the vertebral processes seemed more difficult in larger cats possibly due to higher body fat and/or muscle mass in the cervical area. For example, the heaviest cat (7.4 kg) in this study was in the LOW group and had a substantial amount of fat between nerves after dissection. In this cat, BMS did not spread as expected and was deposited medially to the target nerves.
The minimal effective length of nerve in contact with local anesthetic required for a successful blockade is controversial. In previous anatomical studies involving isolated nerves from frogs and canine cadavers,4,14 staining of approximately 15–25 mm along the nerve was considered to be appropriate and likely to produce an effective nerve blockade in vivo.4,14 In this case, both LOW and HIGH volumes would have produced clinically relevant PBP blockade in cats.
More studies are required to determine the safety and efficacy of this loco-regional anesthetic technique (in vivo) in cats.
Conclusions
This study describes the PBP blockade in cats with acceptable nerve staining and minimal complications. Success rate and length of nerve staining were not significantly different between LOW and HIGH groups.
Footnotes
Author note
This paper was presented, in part, at the 2017 Association of Veterinary Anaesthetists’ Spring Meeting, Manchester, UK.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article. Dr Beatriz Monteiro is a recipient of the Vanier Canada Graduate Scholarship.
