Abstract
Objectives
The study aims were to evaluate the feasibility of contrast-enhanced ultrasonography in feline renal transplant recipients in the post-transplantation period and to report findings in a case with presumptive delayed allograft ischemia.
Methods
Cats were imaged postoperatively using contrast harmonic ultrasonography after a bolus injection of a microbubble contrast medium. Time/mean pixel intensity curves were generated for cortical and medullary regions of interest in the renal allograft in each cat. Arrival time, time to peak, wash-in slope, wash-out slope, mean transit time and renal blood flow were calculated for each area.
Results
Within the renal cortices of cats without ureteral obstruction 1 day post-transplantation, arrival time was 2.0–6.3 s, time to peak was 3.6–30.1 s, wash-in rate was 2.45–41.14 mean pixel intensity (MPI)/s, wash-out rate was −2.01 to −0.47 MPI/s and blood flow was 6.1–106.5 MPI/s. Ratio mean transit time was 0.29–1.29. Typical cortical and medullary perfusion patterns were observed in these cats. In one cat with delayed graft ischemia followed by presumptive acute transplant rejection, dynamic and heterogeneous cortical and medullary perfusion was demonstrated. Decreases in cortical blood flow were paralleled by elevated serum creatinine.
Conclusions and relevance
Contrast-enhanced ultrasonography can be used in feline renal transplant recipients and provides both qualitative and quantitative data regarding renal allograft perfusion.
Introduction
Following renal transplantation, azotemia can occur as a result of allograft rejection, acute tubular necrosis or ureteral obstruction among other causes. 1 Protocol renal biopsies and diagnostic imaging are used to differentiate causes of post-transplantation azotemia in people. 2 Post-transplantation biopsies are infrequently performed in cats because of smaller allograft size, cost and long turnaround time; therefore, diagnostic imaging and bloodwork are heavily relied upon.
Contrast-enhanced ultrasonography (CEUS) has been used to evaluate human renal allografts and native kidneys in normal dogs and cats.3–9 The contrast medium consists of gas-filled microbubbles that remain intravascular and detectable for several minutes. 10 CEUS offers superior perfusion detection, as color and power Doppler underestimate small vessels, slow or low-volume flow or flow from unfavorable angles. 11 CEUS allows capillary level perfusion quantification via time-intensity curves, which have been used in people to differentiate normal renal grafts from those with rejection or acute damage.3,4,12,13 Prior to this study, CEUS had not been evaluated in feline renal transplantation. The purpose of this pilot study is to report the preliminary use of CEUS in feline renal transplant recipients in the postoperative period, including recommendations for technique optimization and areas of potential utility for future study.
Materials and methods
Medical records at the University of Georgia were searched for cats that underwent renal transplantation, B-mode ultrasonography and CEUS within 24 h postoperatively. Serum creatinine concentrations were recorded within 2 days preoperatively and 1 day postoperatively (same day of CEUS). For one cat with additional CEUS beyond 1 day postoperatively, serum creatinine within 1 day of each CEUS study was recorded.
Ultrasonography was performed using one of two machines (GE Logiq S8 with 9–11 MHz virtual convex linear transducer and Toshiba SSA-680A with 6C1 1–6 MHz convex transducer). Harmonic imaging software developed by the machine manufacturers was used for CEUS and the mechanical indices were 0.03 (Toshiba) and 0.12 (GE). Each cat received a bolus injection of 0.03 ml/kg of perflutren microsphere suspension (Optison; GE) via cephalic venous catheter, followed by 2–3 ml heparinized saline to flush the catheter. Continuous images were obtained from 0–120 s after injection.
Images were analyzed using image-processing software (OsiriX; Pixmeo). Two regions of interest (ROI) were drawn within the allograft cortex and medulla for each cat at each second. When respiratory or transducer fanning motion was encountered, the ROI was moved to compensate. If there was no renal cortex or medulla on a given image, that time point was excluded from analysis. Mean pixel intensity (MPI) was calculated by the software for each ROI.
For each kidney and ROI, time-intensity curves were created. 14 Peak MPI (MPIpeak), time to peak enhancement from injection (TTP) and arrival time (AT) were gathered from time-intensity curves. 3 After cancelling the pre-arrival period and correcting for background, the following gamma-variate function was fit to each kidney–ROI combination:MPI t = MPIpeak × [(t / TTP)^(β × TTP) × exp{−β × (t − TTP)}], where MPI t is MPI at time t and β (beta) is the slope of the signal intensity rise. 12 β was computed using non-linear least squares via the nls function. 14 Mean transit time (MTT) was the time point that divided the total area under the gamma-variate curve in half. 12 MTT ratio was cortical MTT/medullary MTT. 3 Renal blood flow was MPIpeak × β. 15 Wash-in slopes used the first point after AT where MPI ⩾10% greater than baseline (baseline defined as median MPI before AT) to the first point before TTP where MPI ⩾90% of MPIpeak . 7 Wash-out slopes used the last point after TTP where MPI ⩾90% of MPIpeak to the last point after TTP where MPI ⩾20% greater than baseline. 7 To estimate wash-in and wash-out slopes, linear regressions were fit using the lm function in R. 7
Results
A total of five cats were included (see appendix 1 in the supplementary material). At the time of 24 h postoperative ultrasonographic study, 4/5 cats showed improved serum creatinine levels (see appendix 1 in the supplementary material) and no evidence of transplant complication. The remaining cat (case 5) had a ureteral obstruction noted ultrasonographically and serum creatinine mildly increased compared with the preoperative value. The obstruction was confirmed during revision neoureterocystostomy.
Subjectively, the two-phase pattern of enhancement observed in transplanted kidneys was similar to that previously described: rapid cortical contrast enhancement followed shortly by gradual medullary enhancement. 7 Representative images and time-intensity curves are provided (Figures 1 and 2). Quantitatively, there was considerable variation among transplanted kidneys (Table 1). In case 5, cortical TTP and wash-in rates were prolonged compared with 3/4 and with 4/4 of the remaining cats, respectively, at 24 h post-transplantation.

Contrast-enhanced ultrasonographic images of a representative transplanted kidney at time of peak (a) cortical and (b) medullary intensity, 1 day post-transplantation. Regions of interest are encircled by solid white lines
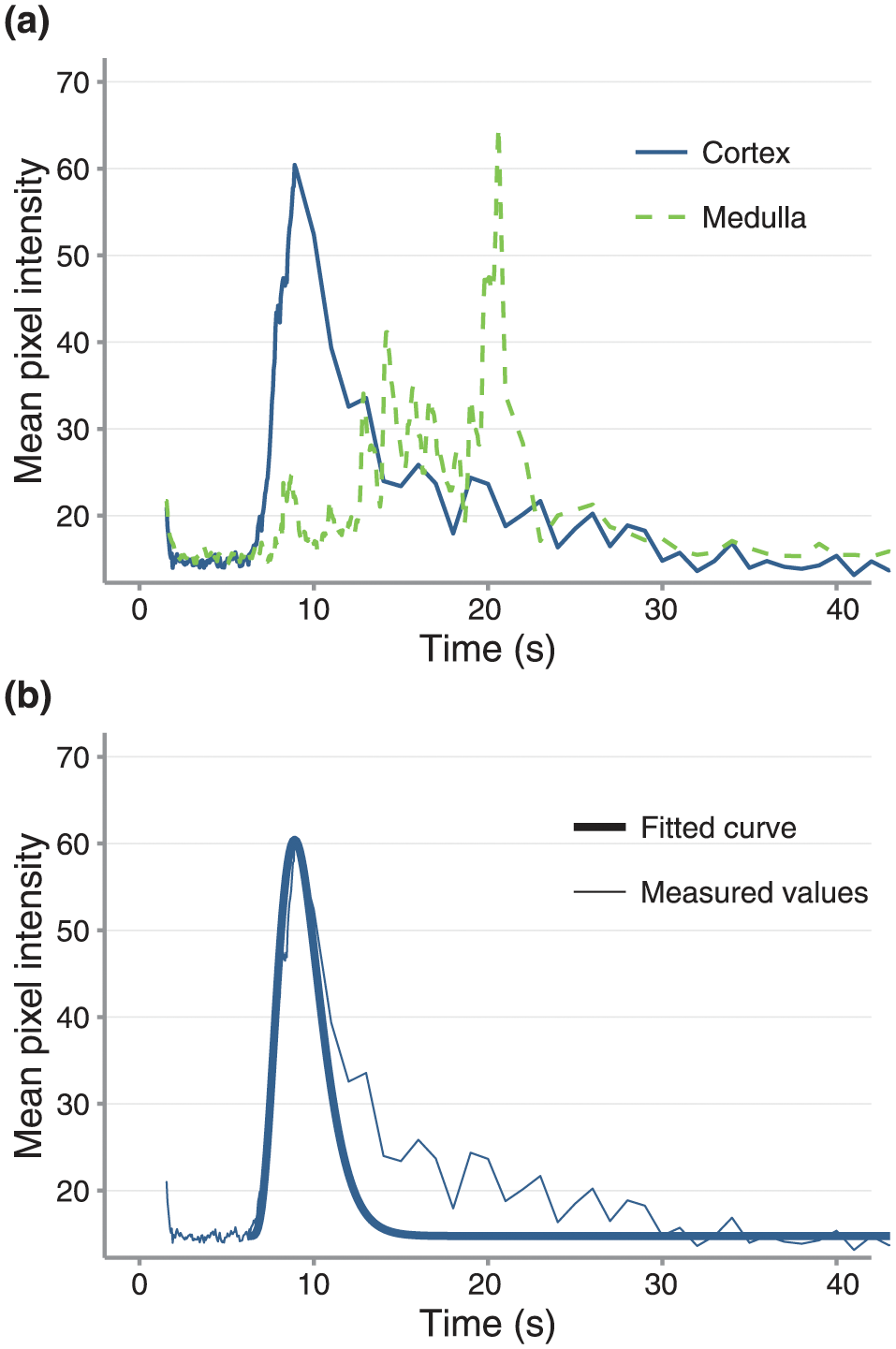
(a) Cortical and medullary time-intensity curves with (b) an overlaid curve fitted to a gamma-variate function for the cortex for the transplanted kidney shown in Figure 1
Results of quantitative contrast-enhanced ultrasonography parameters of renal allografts 1 day post-renal transplantation in clinically improved cats (cases 1–4) and a cat with ureteral obstruction (case 5), as compared with published reference intervals for normal cats

Data are mean ± SD
Leinonen et al (2010) 7
Kinns et al (2010) 6
MPI = mean pixel intensity; MTT = mean transit time; NA = not available
One cat (case 1) had five additional CEUS examinations between 9 and 20 days post-transplantation (Figure 3). Perfusion values obtained for this cat’s studies are provided (Table 2), excluding the second study, which lacked renal enhancement. Lack of perfusion was attributed to renal artery thrombosis, severe vasoconstriction or vascular pedicle torsion. Anticoagulant therapy was initiated. The cat then developed signs consistent with acute rejection, including increased serum creatinine, allograft enlargement and behavioral depression; the presumed acute rejection likely occurred as a result of increased antigenicity of the injured allograft.16,17 Cortical enhancement was highly heterogeneous 10 days post-transplantation, so three separate cortical ROIs were drawn to reflect variability. Concurrent serum creatinine was nearly double the 1 day postoperative value and remained elevated subsequently. Treatment of the acute rejection reaction included ciclosporin (7 mg/kg PO q12h), mycophenolate (10 mg/kg PO q12h) and dexamethasone (0.15 mg/kg IV q12h). The cortical wash-out rate progressively slowed on serial CEUS. At the final study (20 days post-transplantation), the cortical wash-out rate was 4% of its initial value and 2% of the mean reported in normal cats. 7 The ratio MTT was greater than preceding values and all other transplant kidneys (1.7–7.7 times greater), and cortical blood flow fell below the 1 day postoperative range. Forty-six days post-transplantation, the cat developed signs of sepsis and died.

B-mode (left) and contrast-enhanced ultrasonographic images (right) at peak cortical intensity of a transplanted kidney in a cat a series of time points after transplantation. (a) Day 1 post-transplantation: there is uniform cortical contrast enhancement. (b) Day 9: there is little-to-no contrast enhancement, thought to be due to renal artery thrombosis, severe vasoconstriction or torsion of the renal vascular pedicle. (c) Day 10: the cortical enhancement is highly heterogeneous, characterized by multifocal regions that nearly completely lack contrast uptake for the entirety of the study. At this time, the cat was exhibiting signs consistent with an acute rejection episode. (d) Day 12, (e) day 15, and (f) day 20: there is subjective improvement of cortical perfusion compared with prior, characterized by more homogeneous contrast enhancement
Serum creatinine and quantitative contrast-enhanced ultrasonography perfusion values obtained between 1 and 20 days post-renal transplantation in a clinically unstable cat (case 1) *
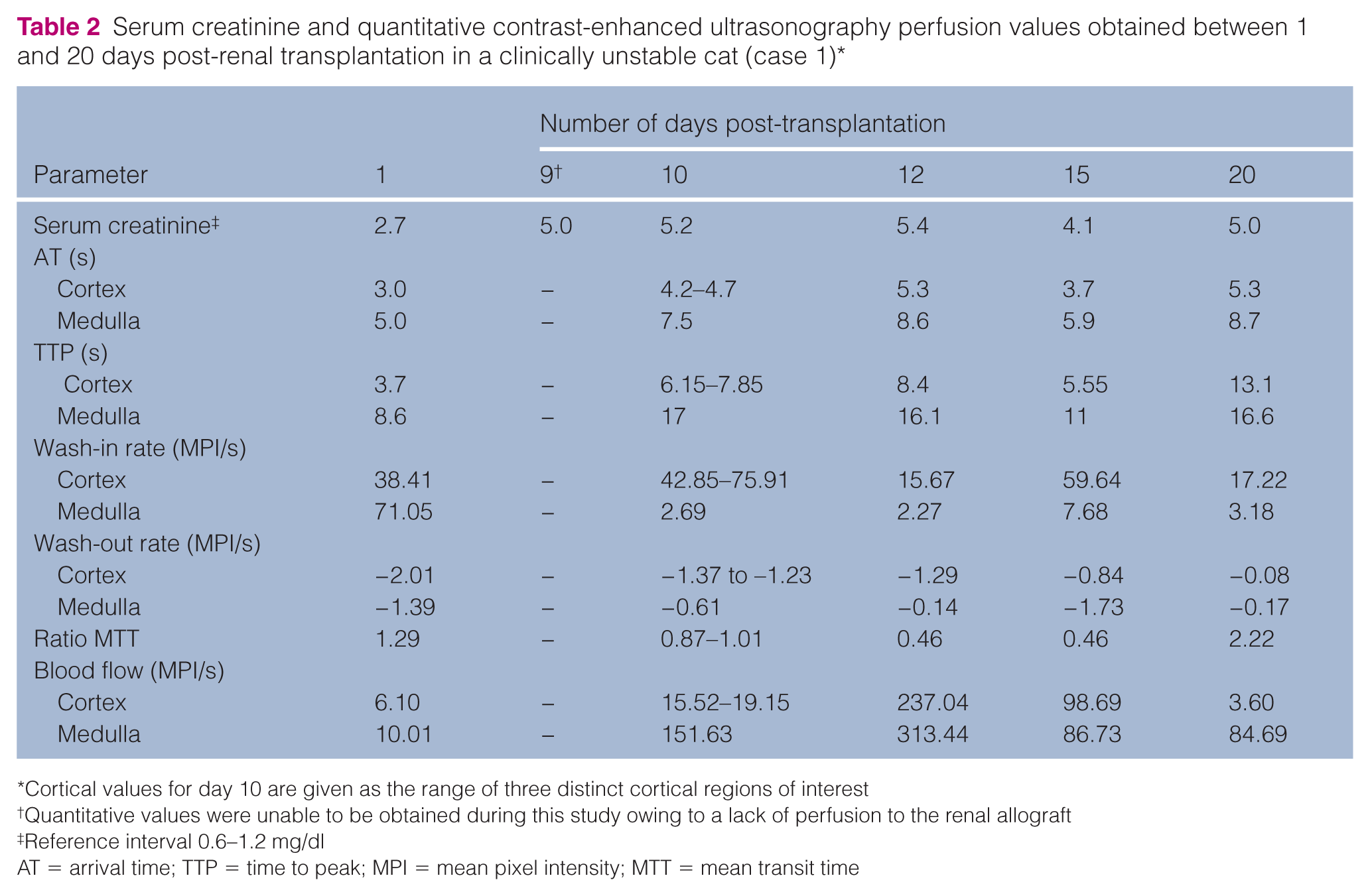
Cortical values for day 10 are given as the range of three distinct cortical regions of interest
Quantitative values were unable to be obtained during this study owing to a lack of perfusion to the renal allograft
Reference interval 0.6–1.2 mg/dl
AT = arrival time; TTP = time to peak; MPI = mean pixel intensity; MTT = mean transit time
Discussion
No complications were noted as a result of contrast medium administration. In the cat with ureteral obstruction (case 5), higher cortical TTP and slower wash-in rates may reflect decreased cortical perfusion, as this cat also had lower allograft blood flow. This is supported by a study demonstrating reduced cortical renal blood flow within 60 mins of ureteral obstruction. 18
When evaluating the cat with ischemic injury and apparent allograft rejection (case 1), dynamic differences in renal perfusion were demonstrated with CEUS that were not apparent on B-mode ultrasonography. Elevated MTT ratio is associated with transplant acute tubular necrosis in people. 3 At 20 days post-transplantation, this cat had a MTT ratio nearly double that of all other CEUS studies in this report. If the association between tubular damage and MTT ratio applies to cats, this could indicate tubular damage in this cat. However, necropsy was declined so definitive conclusions cannot be drawn. Also of note is the progressively reduced cortical wash-out rate and dramatically reduced renal blood flow in the final CEUS studies. Similar to the inverse correlation between renal blood flow and creatinine in human transplant recipients, this cat had persistently elevated creatinine concentrations during this time. 15
Differentiating underlying causes of renal dysfunction is critical when tailoring treatment protocols. In people, early transplant dysfunction is grouped mainly into acute graft rejection or acute tubular necrosis. 19 Acute graft rejection, if detected early, can be reversed with immunosuppressive therapy.20,21 Acute tubular necrosis treatment primarily involves fluid management. 22 Developing a non-invasive method of differentiating causes of feline renal allograft dysfunction, such as CEUS, could facilitate targeted therapy and improve patient outcomes. In support of this goal, additional CEUS parameters shown to be useful for people and heretofore not discussed in veterinary medicine were evaluated in this study. In people, MTT 5 days post-transplantation is inversely related to renal function 30 days post-transplantation, giving prognostic value to CEUS. 3 Antibody-mediated CEUS targeting T lymphocytes detected early renal rejection episodes and demonstrated utility in monitoring response to immunosuppressive therapy, suggesting further promise for CEUS in evaluating renal transplantation rejection. 23
Limitations of this study are primarily due to its retrospective nature. Ciclosporin concentrations can affect allograft perfusion via vasoactive properties; however, insufficient data prevented evaluation of potential ciclosporin influence on CEUS perfusion parameter variability. 24 The authors recommend that transducer fanning during CEUS be avoided, as this leads to gaps in quantitative data collection. Gain should be kept at mid-range to avoid excessive brightness (Figure 3b,c).
Conclusions
This study demonstrates that CEUS is feasible and safe in feline renal allograft recipients and provides qualitative and quantitative perfusion information. CEUS demonstrated dynamic, regional and global changes in renal perfusion in a cat with transplant failure. Prospective and large-scale studies are warranted to determine if CEUS can differentiate between underlying causes of allograft dysfunction or provide prognostic information, as in people.
Supplemental Material
Click here for Supplementary Appendix
Summary of case data for feline renal allograft recipients
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
