Abstract
Objectives
The objective of this study was to compare the sedative effect of butorphanol–dexmedetomidine with buprenorphine–dexmedetomidine following intramuscular (IM) administration in cats.
Methods
Using a prospective, randomised, blinded design, 40 client-owned adult cats were assigned to receive IM dexmedetomidine (10 µg/kg) combined with either butorphanol (0.4 mg/kg) (‘BUT’ group) or buprenorphine (20 µg/kg) (‘BUP’ group). Sedation was scored using a previously published multidimensional composite scale before administration (T0) and 5, 10, 15 and 20 mins afterwards (T5, T10, T15 and T20, respectively). Alfaxalone (1.5 mg/kg) was administered IM at T20 if the cat was not deemed adequately sedated to place an intravenous catheter. Adverse events were recorded. Friedman two-way ANOVA analysed sedation scores within groups. Mann–Whitney Rank Sum test compared sedation scores between groups; Fisher’s exact test analysed the frequency of alfaxalone administration and adverse events. P <0.05 was considered statistically significant.
Results
Sedation scores between groups were similar at baseline, but at T5, T10, T15 and T20 scores were higher in the BUT group (P <0.01). Within both groups, sedation scores changed over time and the highest sedation scores were reached at T10. Requirement for additional sedation was similar between groups: two cats in the BUT group and five cats in the BUP group. One cat and 11 cats vomited (P = 0.002) in the BUT and BUP groups, respectively. No other adverse events were recorded.
Conclusions and relevance
At these doses, IM buprenorphine–dexmedetomidine provides inferior sedation and a higher incidence of vomiting than butorphanol–dexmedetomidine in cats. Butorphanol–dexmedetomidine may be preferred for feline sedation, especially where vomiting is contraindicated.
Introduction
Intravenous (IV) catheter placement in cats can be stressful for the animal and challenging for the operator. Sedation may be required to reduce the animal’s stress, facilitate handling and increase successful IV catheter placement. The intramuscular (IM) route is preferable to other routes (ie, subcutaneous or transmucosal) in terms of ease of injection and a more predictable sedative effect.1,2 When selecting the agents to be used, consideration must be given to both the type of any subsequent procedures that need to be performed, and the need for inclusion of analgesic drugs within the protocol.
Dexmedetomidine, the dextro-optical isomer of racemic medetomidine, has agonist activity at the alpha (α)2-adrenoreceptor and causes sedation by activating the central α2-adrenoreceptors in the locus coeruleus and inhibiting presynaptic noradrenergic activity.3,4 Dexmedetomidine is licensed in many countries for sedation by IM administration in cats, and has been shown to produce similar dose-dependent sedation, analgesia and muscle relaxation to medetomidine. 4 a2-adrenoreceptor agonists are often combined with opioids to exploit their synergistic effects and improve quality of sedation.5,6
In people, butorphanol is a kappa (κ) opioid receptor agonist and a mu (μ) opioid receptor antagonist, 7 while buprenorphine has partial agonist activity at μ opioid receptors. 8 Although this classification has been accepted for animals, including cats, 9 the exact activity of butorphanol and buprenorphine might be different between species.7,10,11 Both butorphanol and buprenorphine are licensed for IM administration in cats. While buprenorphine has been shown to provide superior analgesia than butorphanol in cats undergoing surgical procedures,12,13 there are no studies comparing their sedative effect when combined with dexmedetomidine. 14
The IM administration of butorphanol (0.2 mg/kg) combined with dexmedetomidine (10 µg/kg) provided a superior sedation than dexmedetomidine alone (10 µg/kg) in cats. 6 Cats were successfully sedated when administered IM dexmedetomidine (20 µg/kg) and buprenorphine (10 µg/kg), although minimal or no sedation was produced by buprenorphine alone (10 or 20 µg/kg). 15 In dogs, IM dexmedetomidine (500 µg/m2) and butorphanol (0.2 mg/kg) provided a more reliable sedation for hip radiography than dexmedetomidine and buprenorphine (0.015 mg/kg). 16
In our clinical experience, butorphanol provides a more profound sedation than buprenorphine when administered IM in combination with dexmedetomidine in cats. Therefore, we hypothesised that buprenorphine (20 µg/kg) combined with dexmedetomidine (10 µg/kg) IM provides inferior sedation to butorphanol (0.4 mg/kg) combined with dexmedetomidine (10 µg/kg).
Materials and methods
Ethical approval was given by the University of Nottingham Veterinary School’s Clinical Ethical Review Panel (1225 140901). Using a prospective, blinded and randomised design, client-owned adult cats (>12 months old) requiring sedation or general anaesthesia for non-painful procedures, such as diagnostic procedures, blood sampling, minor wound care and IV treatment administration, were recruited. Cats were excluded if: classification according to the American Society of Anesthesiology (ASA) ⩾4; the animal had cardiac disease precluding sedation with the drugs used; there were signs of hypovolaemia or dehydration; depression or neurological abnormalities, or signs of increased intracranial or intraocular pressure were present; suspicion of a gastrointestinal obstruction existed; and opioid medication or sedative drugs were administered in the past 8 h. Cats had been starved for at least 8 h prior to presentation. Informed owner consent was obtained.
Prior to drug administration, cats were examined by the same observer (RJB). Thoracic auscultation, respiratory rate, heart rate, pulse palpation, mucous membrane colour and capillary refill time were performed, body condition score (BCS) assessed and body mass (kg) recorded. Cats’ temperament was assessed using a score designed for this study (Table 1). Cats were placed in a kennel in the feline ward and allowed to acclimatise for approximately 30 mins.
Simple descriptive scale specifically designed by the authors to assess the temperament of cats prior to intramuscular administration of butorphanol–dexmedetomidine or buprenorphine–dexmedetomidine

Cats were assigned to one of two groups (butorphanol [BUT] or buprenorphine [BUP]) according to a randomisation table (Quickcalcs; Graphpad Software). Cats in the BUT group received 0.4 mg/kg butorphanol (Alvegesic; Dechra) and 10 µg/kg dexmedetomidine (Dexdomitor 0.5 mg/ml; Vetoquinol) IM; cats in the BUP group received 20 µg/kg buprenorphine (Vetergesic 1 ml vials; Ceva Animal Health) and 10 µg/kg dexmedetomidine IM. The two drugs were each drawn into a 1 ml syringe by an anaesthetist not involved in the study and mixed together in a third syringe before dilution with water for injection to a volume of 0.1 ml/kg immediately prior to administration.
Cats were assessed in their kennel prior to injection (T0) by the same observer (RJB), who was unaware of group allocation. Previously published criteria were used to assess sedation quality and were performed in the same order for each cat (see Table 2). 1 Posture was evaluated from outside the kennel door, then the clippers were turned on and held next to the door to judge the response to clipper sounds. Next, the kennel door was opened, and the response to clipping assessed by extending a forelimb at the shoulder and clipping some fur over the cephalic vein. An attempt to place the cat in lateral recumbency within the kennel was made to assess response to restraint.
Multidimensional composite scale used to score sedation in cats after intramuscular administration of butorphanol–dexmedetomidine or buprenorphine–dexmedetomidine 1

The sedative solution was injected in the right or left epaxial muscle by the same operator (RJB) with a nurse restraining the cat. Cats were returned to their kennels and sedation assessed at 5 min intervals up to and including 20 mins post-administration (T5, T10, T15 and T20). Noise was kept to a minimum in the ward and kennels were not covered to allow undisturbed monitoring of sedated cats.
After sedation was scored at T20, the cat was restrained by a nurse and a catheter was placed in the cephalic vein by RJB. If the cat had been aroused during moving from the kennel, it was allowed to relax again. If the cat could not be restrained in sternal recumbency with one forelimb extended from the shoulder and the cephalic vein raised at the elbow, additional sedation was administered using 1.5 mg/kg alfaxalone (Alfaxan; Jurox) IM. During the observation period, any adverse events (eg, vomiting, muscle tremors, agitation, dysphoria) were recorded.
Statistics
Based on the results of a pilot study performed on eight cats, 18 cats per group were necessary to detect a 20% difference in sedation score, with a power of 80%, and an α of 0.05.
Data were tested for normality where appropriate using a Kolmogorov–Smirnov test (SigmaStat 3.5). The Student’s t-test compared age between groups, and a Mann–Whitney rank sum test assessed differences in body mass, BCS, ASA status and temperament. Fisher’s exact test compared sex, breed and incidence of adverse events between groups. Friedman two-way ANOVA evaluated sedation scores within groups and the Mann–Whitney rank sum test compared sedation scores between groups at respective time points. P <0.05 was considered statistically significant. Data are reported as mean ± SD or median (range) as appropriate.
Results
Forty cats were recruited but three were excluded (two in the BUT group, one in the BUP group) owing to misinjection of drugs (n = 1); miscalculation of drugs as incorrect body weight was recorded (n = 1); observation process interrupted by the clinician responsible for the case (n = 1). Data from 37 cats were analysed (18 in the BUT group, 19 in the BUP group). Cats required sedation to undergo the following procedures: ultrasound (n = 11), radiography (n = 11), CT scan (n = 10), blood sampling (n = 2), wound care (n = 2) and chemotherapy administration (n = 1). All required placement of an IV catheter at the end of observation period.
Between groups, body mass, BCS, age, ASA status, temperament, sex and breed were not significantly different (Table 3). Sedation scores were similar at T0, but at T5, T10, T15 and T20 scores were higher (P <0.01) in the BUT group (Figure 1). Within each group, sedation scores changed over time (Figure 1). In both groups maximum sedation was achieved at T10: 7 (3–10) and 5 (0–8) in the BUT and BUP groups, respectively, and sedation scores at T15 and T20 were not statistically different from T10 (Figure 1).
Demographic data of cats in the butorphanol–dexmedetomidine (BUT) and buprenorphine–dexmedetomidine (BUP) groups

FN = female neutered; MN = male neutered; ASA = American Society of Anesthesiologists; DSH = domestic shorthair
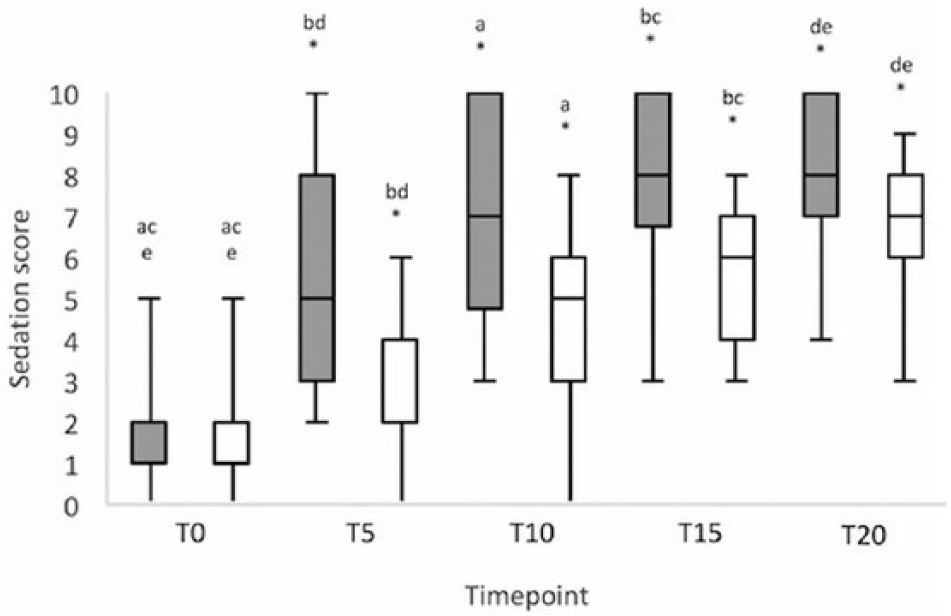
Sedation scores (0 = no sedation; 10 = maximum sedation) obtained prior to (T0) and at 5, 10, 15 and 20 mins (T5–T20) after administering intramuscular butorphanol–dexmedetomidine (shaded bars) or buprenorphine–dexmedetomidine (clear bars) in cats. Boxes represent median and interquartile range; whiskers represent maximum and minimum values. *P <0.01 between groups at same time point. Within same group same letter (a–e) indicates P <0.05 between time points
Twelve cats vomited on at least one occasion during the observation period: one cat (5.6%) in the BUT group at 5 mins and 11 cats (57.9%) in the BUP group (P = 0.002) at 9 (4–15) mins after injection (Figure 2). No other adverse events were recorded. Seven cats required additional sedation for IV catheter placement: two (11.1%) in the BUT group and five (26.3 %) in the BUP group (P = 0.4).

Number of cats that vomited and required additional intramuscular sedation (alfaxalone) for intravenous catheter placement, having received butorphanol–dexmedetomidine (shaded bars) or buprenorphine–dexmedetomidine (clear bars). *P = 0.002
Discussion
The IM administration of buprenorphine or butorphanol combined with dexmedetomidine resulted in sedation of all cats. Nevertheless, the requirement of additional sedation for IV catheter placement was similar. At the doses used, the sedation after buprenorphine and dexmedetomidine was inferior to butorphanol and dexmedetomidine, supporting our hypothesis.
Previous publications demonstrate good sedation following butorphanol–dexmedetomidine administration in cats, while others show a poor sedation after buprenorphine–dexmedetomidine. However, no studies involve a direct comparison, as in the current study. Slingsby et al 14 found no difference in sedation using medetomidine (20 μg/kg) combined with the same doses of butorphanol and buprenorphine as the present study described. This difference may be explained by the use of medetomidine instead of dexmedetomidine, its relative higher dose, by the different score used to assess sedation or by the fact that their sample size was designed to find a difference in postoperative pain rather than sedation scores. Overall sedation scores indicated a greater sedative effect after IM dexmedetomidine (10 μg/kg) and butorphanol (0.1 mg/kg), when compared with dexmedetomidine (10 μg/kg). 6 Nagore et al 17 found the quality of sedation obtained administering IM butorphanol (0.4 mg/kg) and dexmedetomidine (10 μg/kg) was similar to dexmedetomidine (20 μg/kg) and provided better sedation for IV catheter placement. Slingsby et al 15 reported that IM buprenorphine (10 or 20 μg/kg) has a minimal sedative effect in cats up to 6 h post-administration, but was effective if combined with dexmedetomidine (20 μg/kg) within 15 mins. The peak plasma concentration of buprenorphine after IM administration in cats is 3 mins; 18 combining it with dexmedetomidine increases this to 9 mins. 19 In vitro, buprenorphine takes 30 mins to bind the μ opioid receptor, 20 although peak sedative effect of buprenorphine combined with dexmedetomidine is 15 mins. 15 Butorphanol IM reaches peak plasma concentration after 21 mins, 21 yet peak sedation combined with dexmedetomidine has been reported at 15 mins. 17 According to our result the synergistic effect produced combining butorphanol and dexmedetomidine is superior to buprenorphine and dexmedetomidine, confirming previous impressions.6,15,17
The difference in BCS between groups was not statistically significant (P = 0.06), but it is possible that cats in the BUT group had a higher BCS. As actual body weight was used to calculate drug doses, overweight cats would have received more drug per kg of lean body weight, which could have resulted in greater sedation in the BUT group.
Sedation was assessed for 20 mins in every cat, although considering the 30 min receptor binding of buprenorphine in vitro, 20 an extended assessment period could be justified. Previous work has demonstrated little sedative effect of IM buprenorphine alone, 15 and peak sedation at 15 mins after IM administration of butorphanol–dexmedetomidine and buprenorphine–dexmedetomidine.15,17 Furthermore, the duration of dexmedetomidine sedation is dose dependent, 4 and at 40 μg/kg declines after 30 mins, 22 which could lead to a decrease in sedation scores with a longer observation period. According to our results, the maximum sedation recorded was achieved at T10 and maintained until T20 with no difference within groups. Furthermore, 20 mins is a reasonable timeframe in which to wait for a sedation to take effect. Therefore, we believe that a 20 min observation period was adequate, and longer observation would not have affected our results or been clinically useful.
The maximum sedation score was reached 10 mins from administration in both the BUT and BUP groups. Maximum sedation time was 15 mins after IM administration of butorphanol (0.4 mg/kg) and dexmedetomidine (10 μg/kg), 17 and after buprenorphine (10 μg/kg) and dexmedetomidine (20 μg/kg), 15 but sedation was not assessed at earlier time points in these studies.
Onset, peak of effect and quality of sedation using different injection sites in cats has not been investigated. In dogs, injection of dexmedetomidine and hydromorphone into semi-membranous or cervical muscles resulted in a quicker onset compared with the gluteal and lumbar muscles. 23 Furthermore, the administration of acepromazine and morphine into the gluteal muscles gave lower sedation scores at 10 mins after injection than the quadriceps, triceps and cervical muscles. 24 Slingsby et al 15 used the quadriceps muscle, while the lumbar epaxial muscle was used as the site of injection in our study. Nagore et al 17 did not report the site of IM injection. The difference in injection sites might have had an effect on the time to reach the maximum sedation between the reported studies.
When comparing the effect of two drugs (ie, buprenorphine and butorphanol) equipotency should be satisfied. Equipotent doses are yet to be proven; therefore, it is important to select doses used in the clinical setting. Therefore, we utilised the higher end of the reference interval licensed in cats in the UK. The same doses of butorphanol and buprenorphine used here have also been used in previous studies.1,15,17 A dexmedetomidine control group was not included as the aim of the study was to compare in cats the sedative combinations with each other, which has not previously been carried out. Furthermore, earlier work has compared dexmedetomidine alone with butorphanol and buprenorphine combinations.6,15,17
When a cat requires IM sedation, selection of a reliable combination is important to minimise the stress of the animal by avoiding multiple injections and to avoid delaying the procedure. Therefore, our results have clinical value: administration of butorphanol–dexmedetomidine resulted in a more profound sedation than buprenorphine–dexmedetomidine, and, indeed, could be differentiated in most cases by the observer during the study, and so should be preferred for IM sedation for a non-painful procedure. A statistical difference in the number of cats requiring additional sedation for IV catheter placement was not found, possibly because the majority of cats would have tolerated IV catheterisation without sedation. Or, this could reflect a type II statistical error as the study was not designed to find statistically significant differences in additional sedation.
Vomiting is a known side effect of dexmedetomidine administration in cats.17,25,26 Twelve of 37 cats (32.4%) in our study vomited. α2-adrenoreceptor agonists cause vomiting by activation of receptors in the area postrema of the chemoreceptor trigger zone in cats. 27 Opioids have both an antiemetic and an emetic effect,28–30 thought to be associated with their lipophilicity, allowing crossing of the blood–brain barrier and suppressing the medullary vomiting centre located in the solitary nucleus, reticular formation and dorsal motor nucleus of the vagus.30–32 Poorly lipophilic opioids act on the chemoreceptor trigger zone outside the blood–brain barrier causing vomiting.30,33 In the current study 11 cats in the BUP group vomited vs only one cat in the BUT group, which is an important finding when considering sedation or premedication where vomiting is contraindicated. Buprenorphine alone did not cause vomiting in any cats but did not prevent vomiting when combined with dexmedetomidine.1,15,34 Nagore et al 17 found 70% of cats vomited after dexmedetomidine, while only 10% vomited after combining dexmedetomidine with butorphanol. Equally, Papastefanou et al 35 advocated the use of butorphanol, which significantly reduced dexmedetomidine-induced vomiting in cats. The difference in antiemetic activity between buprenorphine and butorphanol could be explained by considering the slow binding characteristics of buprenorphine to the µ opioid receptor in vitro. 20 Further work could assess whether vomiting is reduced by administering buprenorphine before dexmedetomidine, allowing it to exert its effect on the medullary vomiting centre.
The subjectivity of the scoring system used is a limitation of this study. To our knowledge, there is no validated sedation scoring system in cats; therefore, we decided to use one implemented by Santos et al 1 comparing the sedative effect of dexmedetomidine and buprenorphine. A weak point of this system is that unsedated, nervous cats in a crouched position will have a higher baseline sedation score than a relaxed and interactive cat. Nevertheless, this should not have affected our results as the temperament scores at clinical examination were similar between groups as were sedation scores at T0. Another limitation is the necessity of interacting with the cat every 5 mins, which may have roused the patient. However, this was deemed necessary to increase the chances of identifying the time of maximal sedative effect.
Conclusions
At the doses used in the present study, buprenorphine combined with dexmedetomidine administered IM provided inferior sedation than butorphanol combined with dexmedetomidine, with a higher incidence of vomiting.
Footnotes
Acknowledgements
The authors wish to thank the staff at Dick White Referrals who assisted the progress of this study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
