Abstract
Objectives
The aim of this study was to retrospectively describe clinical, radiographic and therapeutic features of feline lungworm infection.
Methods
Medical records of cats with lungworm diagnosis, thoracic radiography and without concurrent diseases between 2013 and 2015 were reviewed. Collection of data included physical examination, haematology, serum biochemistry, therapy with a variety of anthelmintics and outcomes.
Results
Thirty-seven records were recovered and 26 were included in the study. Single infections by Aelurostrongylus abstrusus (n = 15), Troglostrongylus brevior (n = 3) and Capillaria aerophila (n = 1) and coinfections by T brevior/A abstrusus (n = 6) and T brevior/C aerophila (n = 1) were diagnosed. The most common respiratory signs were coughing (n = 12), increased vesicular sounds (n = 10), dyspnoea (n = 9), such as laboured breathing, orthopnoea or open-mouth breathing, and tachypnoea (n = 6). Two cats were subclinically infected. The most common laboratory abnormality was anaemia (n = 7). Radiographic patterns recorded were interstitial (n = 24), bronchial (n = 21), alveolar (n = 10) and vascular (n = 2). Twenty-five cats had a complete recovery within 2–6 weeks of therapy. One kitten died 7 days after the diagnosis.
Conclusions and relevance
Lungworms should always be included in the differential diagnosis in cats living in endemic areas and presenting with respiratory signs and radiographic abnormalities. A copromicroscopic examination should be considered as the first diagnostic step for all cats at risk of lungworm infections. In most cases, timely therapy with a variety of anthelmintics guarantees recovery.
Introduction
Feline lower respiratory tract disorders include inflammatory, infectious, neoplastic and traumatic conditions. The presence of concomitant and predisposing diseases, and non-specific or inconsistent clinical findings make a definitive diagnosis challenging.
The so-called ‘cat lungworm’ Aelurostrongylus abstrusus (Metastrongyloidea, Angiostrongylidae) is the most important nematode affecting the respiratory system of domestic cats (Felis silvestris catus) worldwide. 1 Cats may become infected by ingesting intermediate hosts (ie, snails or slugs), but it is more likely that infection is due to preying on paratenic hosts (eg, birds, frogs, lizards, snakes and rodents).2,3 Capillaria aerophila (Trichocephalida, Capillariidae) is a nematode living in the trachea, bronchi and bronchioles of wildlife and domestic carnivores. 4 Clinical cases of lung capillariosis in cats have been recorded from Italy, Germany, France, Romania, Portugal, Uruguay and Australia.5–10
In the past few years, species of the mollusc-borne nematodes from the Troglostrongylus genus (Metastrongyloidea, Crenosomatidae) have been identified as agents of verminous bronchopneumonia in cats.11,12 These parasites have long been considered to be found in wild felids only,13,14 but recently, Troglostrongylus brevior has been described from domestic cats from Mediterranean countries.11,15–20
Cats infected with lungworms may display the same clinical signs as cats with other respiratory diseases, such as feline bronchial disease/asthma, bacterial, viral and fungal pneumonia, pulmonary oedema, pulmonary contusion and neoplastic disorders.4,21
Adult stages, eggs and larvae of A abstrusus cause coughing, dyspnoea, open-mouth breathing, sneezing and wheezing, although some cats may also be subclinically infected.22–25 The infection is considered occasionally life-threatening, especially in young or immunosuppressed subjects that do not receive prompt therapeutic intervention.4,26,27 Lung capillariosis is usually characterised by a chronic tracheobronchitis, and infected animals may present with chronic dry cough, sneezing, respiratory sounds and wheezing.8,28
Comprehensive clinical information on feline troglostrongylosis is currently lacking. The available information suggest that this parasite severely affects kittens and young cats, which often show severe dyspnoea, coughing and nasal discharge.10,11,16–18,29 Infection in kittens may be fatal despite prompt anthelmintic treatment. Adult animals are usually subclinically infected.1,11,16,29
There is significant merit in expanding knowledge of clinical aspects of feline respiratory parasitoses, as lungworms have recently gained the scientific interest of researchers and practitioners due to the clinical severity, the frequent presence of mixed infection, the challenges posed in their diagnosis and treatment, and their apparent spread throughout many European countries.
The present article describes the clinicopathological, radiographic and therapeutic features of 26 cases of single and mixed feline lungworm infections diagnosed in 2013–2015 in endemic areas of Italy.
Material and methods
Animals
Between 2013 and 2015, 37 cats were diagnosed with lungworms at the Veterinary Teaching Hospital of the Faculty of Veterinary Medicine – University of Teramo, Italy. Of these animals, 26 met the inclusion criteria of the study: (i) positivity for lungworms at the copromicroscopic examination, (ii) a complete radiographic analysis of the thorax, (iii) no evidence of other concurrent disorders or diseases and (iv) a complete post-treatment follow-up.
Of the 11 excluded cats, nine with single infection (four by A abstrusus, one by T brevior and two by C aerophila) and two mixed infections (one by A abstrusus and T brevior, and one by A abstrusus and C aerophila) were excluded because of an incomplete radiographic analysis or lack of follow-up, while two further A abstrusus-infected animals were excluded because of concurrent diseases (ie, diabetes mellitus, hypertrophic cardiomyopathy).
Clinical examination and radiographic procedures
At referral, available historical data were collected for each of the 26 cats. All animals were subjected to a physical examination and to two or three radiographic views of the thorax. Haematology, serum biochemistry and serology for feline immunodeficiency virus antibody and feline leukaemia virus antigen were also obtained.
Radiographs were reviewed by an ECVDI diplomate (MV) and findings were classified as previously described for respiratory diseases of cats. 6 Bronchial signs were defined as mild (first generation of bronchi visible), moderate (second generation visible) and severe (third generation visible). Alveolar patterns were defined as mild (isolated fluffy infiltrates), moderate (well defined with air bronchograms) and severe (lobar sign). Reticular and nodular interstitial patterns were recorded and interstitial changes were defined as mild (interstitial framework visible but could be bronchial pattern), moderate (interstitial framework can be distinguished from bronchial) and severe (undisputed reticular interstitial pattern).
An enlargement of pulmonary arteries, veins, or both, was recorded as vascular pattern. 30
Oropharyngeal and conjunctival swabs were collected from six cats with upper respiratory signs (ie, nasal and/or ocular discharge, sneezing) for real-time (RT) PCR for feline herpesvirus-1 (FHV-1) and Chlamydophila felis and RT nested PCR for feline calicivirus (FCV).
Parasitological procedures
Faecal samples from each cat were subjected to a copromicroscopic analysis with a classical floatation with zinc sulfate and with the Baermann–Wetzel method. 31
First-stage larvae (L1s) of metastrongyloids found at the Baermann’s examination were identified based on key morphological and morphometric characteristics. 1 The identity of the larvae was confirmed using molecular assays specific for the ribosomal DNA of A abstrusus 32 and T brevior. 16 Eggs of C aerophila found on faecal floatation were identified according to size, morphological and microscopic features, 33 and their identity genetically confirmed using a test specific for the mitochondrial DNA of the nematode. 34
Specific anthelmintic therapy and follow-up
All cats included in the retrospective study received anthelmintic treatment and were rechecked every 2 weeks. Physical examinations, a complete radiographic study of the thorax, copromicroscopic and molecular tests were carried out for each animal until clinical recovery, negative parasitic tests or death.
Results
Signalment and history
All animals (13 males and 13 females) were domestic cats, aged from 2 months to 11 years (median 24.9 months). All cats lived outdoors or were allowed to roam. Fifteen (cats 1, 2, 5, 7–11, 14, 17 and 19–23) were stray animals, five (cats 12, 13, 16, 24 and 25) came from different feline shelters or colonies from central Italy and six (cats 3, 4, 6, 15, 18 and 26) were privately owned cats living indoors with outdoor access.
Details on signalment, history and presenting complaints are reported in Table 1.
Details of parasitic infections, signalment, history, presenting complaint(s) and physical examination findings of the 26 cases

M = male; F = female; Mn = male neutered; A abstrusus = Aelurostrongylus abstrusus; T brevior = Troglostrongylus brevior; C aerophila = Capillaria aerophila
Free access to external environment
Clinical examination and diagnostic procedures
At clinical examination, 24 cats showed respiratory distress, while two did not show any clinical signs. The most frequent signs observed were: coughing (n = 12), breathing difficulties (n = 9), tachypnoea (n = 6), abdominal breathing (n = 5), ocular and/or nasal discharge (n = 5) and sneezing (n = 4). Lung auscultation revealed increased vesicular breath sounds (n = 10) and adventitious sounds, such as wheezing (n = 2) and crackles (n = 1). Other signs documented were anorexia, hyporexia, lethargy, weight loss and pyrexia (Tables 1 and 2).
Percentages of clinical signs in the 26 cats diagnosed with single or mixed lungworm infection
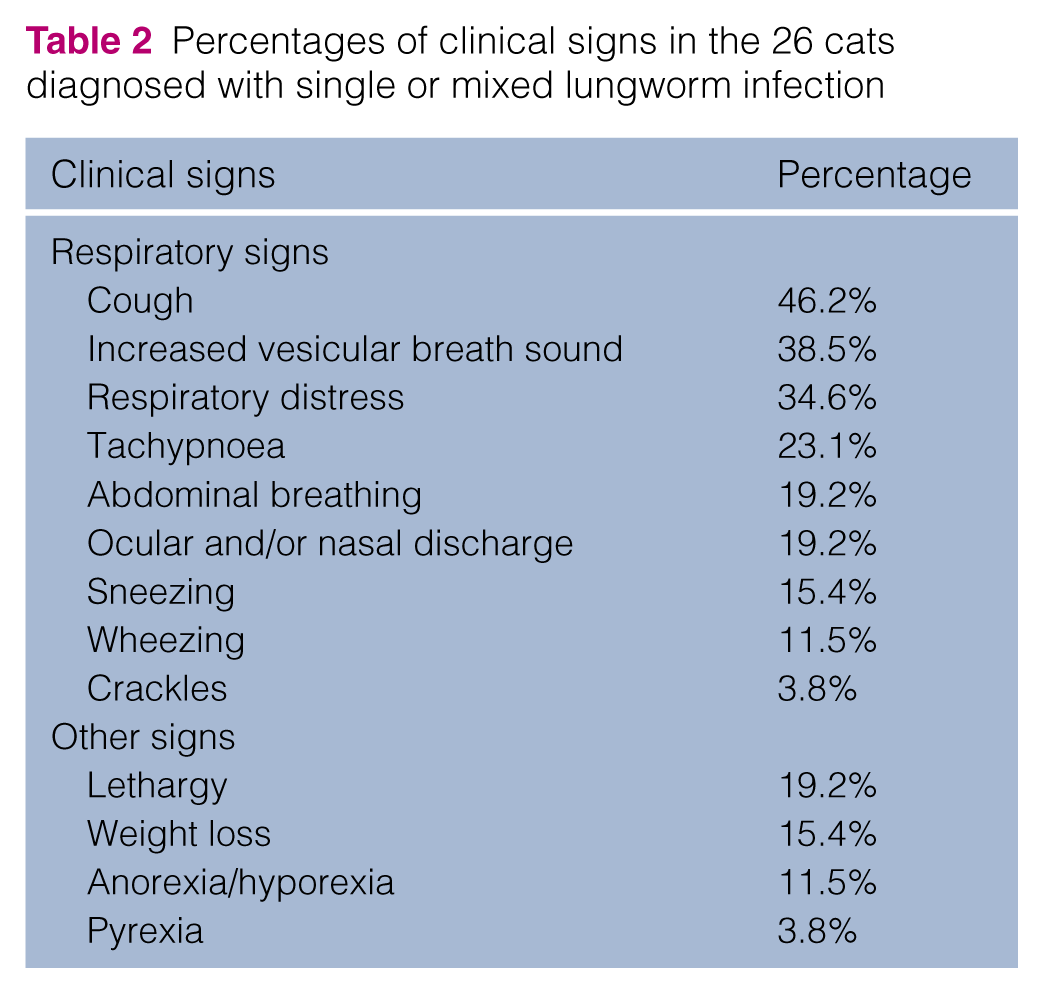
Haematological results for each cat are detailed in Table 3. The most common haematological abnormality was mild normocytic, normochromic anaemia (n = 7). Three cats had neutrophilia and only one cat had eosinophilic leukocytosis.
Haematological, serum biochemical and serological data of the 26 cats at referral

N = value in reference interval; Hct = haematocrit; WCC = white blood cell count; AST = aspartate aminotranferase; ALT = alanine aminotransferase; CK = creatinine kinase; FeLV = feline leukaemia virus; FIV = feline immunodeficiency virus
Bands were not noted
Regarding biochemical analysis (Table 3), two cats showed mild increased aspartate aminotransferase (AST) and/or alanine aminotransferase (ALT) levels.
Radiographic patterns for all cats are summarised in Table 4. The most common abnormalities were interstitial (n = 24; reticular in 19, nodular in five) and bronchial (n = 21). Alveolar changes were observed in 10 animals. Two cats had caudal lobar pulmonary artery dilatation (Figures 1 and 2).
Thoracic radiographic features and follow-up of cats studied 6
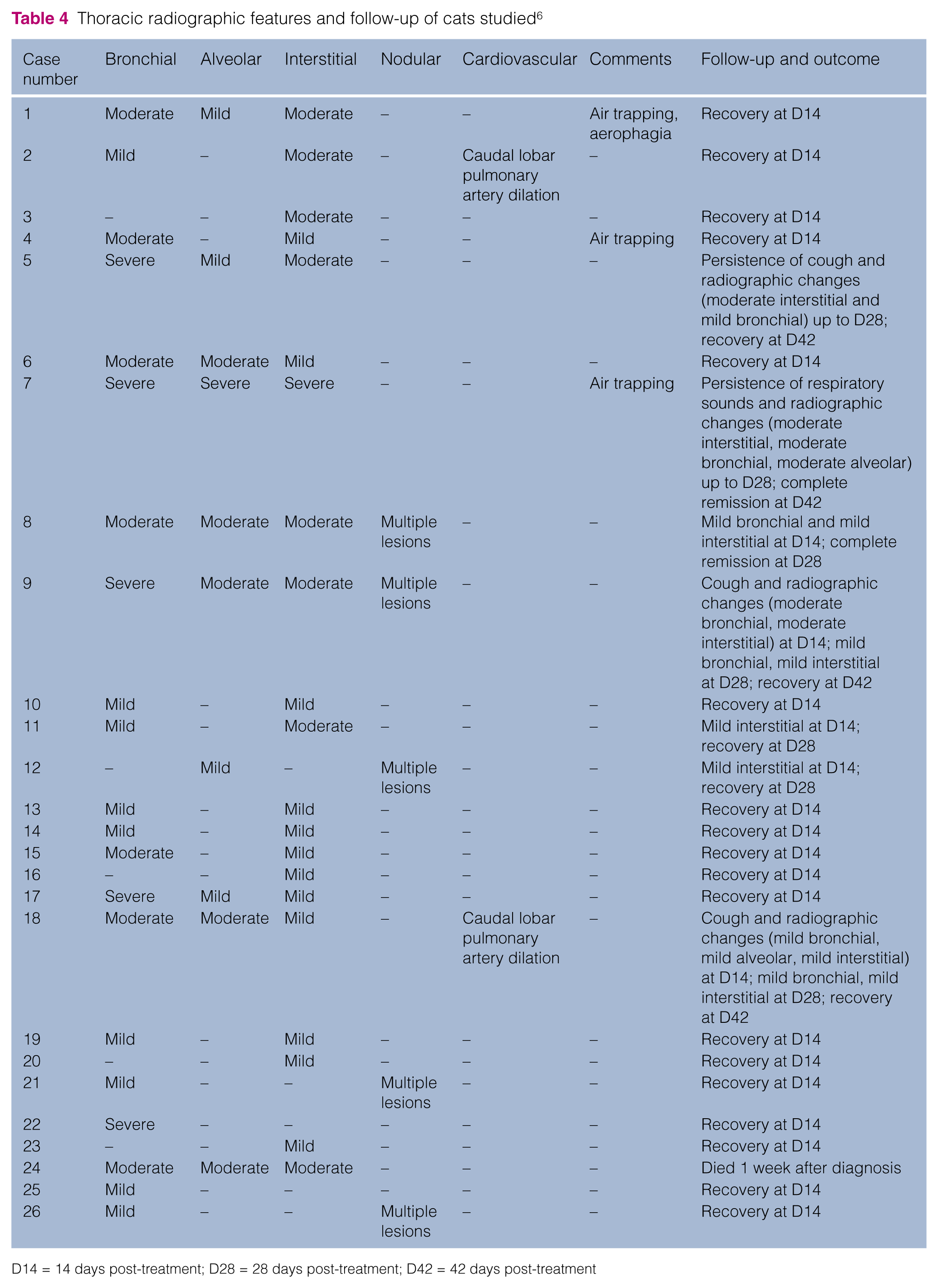
D14 = 14 days post-treatment; D28 = 28 days post-treatment; D42 = 42 days post-treatment
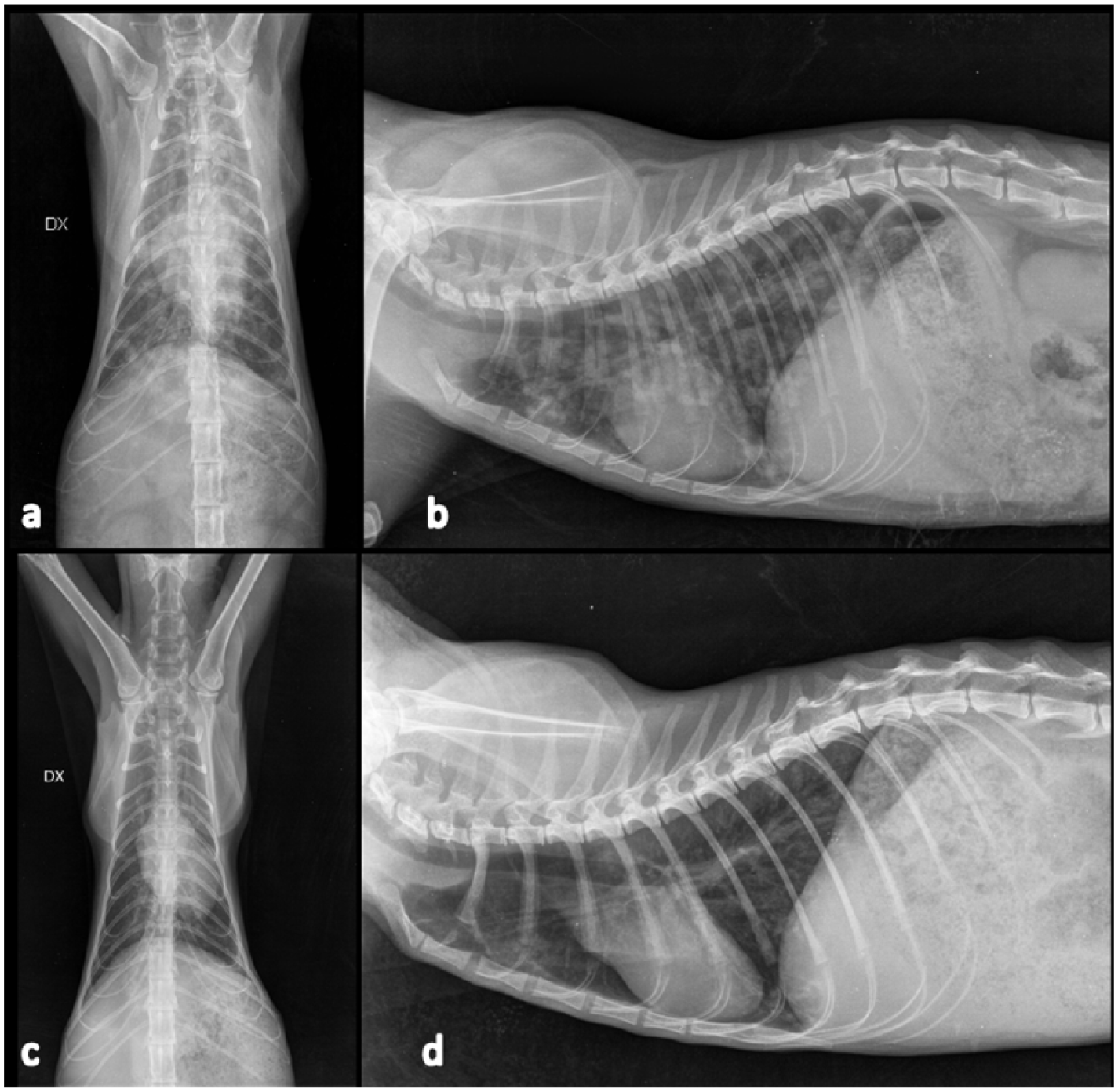
Radiographic studies of a 7-year-old domestic cat infected by Aelurostrongylus abstrusus (cat 8) at referral (a,b) and 2 weeks (c,d) after therapy with moxidectin. Note the presence, at referral (a,b), of a moderate bronchial pattern, a well-defined alveolar pattern in the cranial lobes with small air bronchograms, and multiple nodular lesions through the lung fields. At day 14 (c,d), the cat showed the resolution of nodular lesions and a mild bronchointerstitial pattern

Radiographic studies of a 4-month-old kitten with mixed infection by Aelurostrongylus abstrusus and Troglostrongylus brevior (cat 18), at days 0 (a,b) and 28 (c,d). At referral (a,b), the cat showed a moderate bronchial pattern, alveolar pattern with air bronchograms in the right caudal lung lobe, and a mild reticular interstitial pattern. Enlargement of caudal lobar pulmonary vessels are visible on ventrodorsal projection. At day 28 (c,d), the cat showed only a mild bronchointerstitial pattern
All cats tested for FHV-1, FCV and C felis scored negative at PCR.
Faecal and biomolecular examinations
A single infection by A abstrusus was diagnosed in 15 cats. Ten cats had T brevior either alone (n = 3) or as coinfection with A abstrusus (n = 6) or C aerophila (n = 1). One cat had a single infection by C aerophila (Table 1). The molecular tests confirmed the microscopic identification of parasite elements in all cats.
Therapy and outcomes
Anthelmintic therapy and treatment outcomes are summarised in Table 5. Twenty-five cats made a full recovery (ie, resolution of clinical, radiographic and parasitological negativisation), while a 3-month-old kitten died after an acute onset of dyspnoea.
Aetiological agents, therapeutic protocols, follow-up and outcome

D14 = 14 days post-treatment; D28 = 28 days post-treatment; D42 = 42 days post-treatment; XR = radiographic; Aa = Aelurostrongylus abstrusus; Tb = Troglostrongylus brevior; Ca = Capillaria aerophila; FBZ = febendazole; MOX = moxidectin; EMO = emodepside; MO = mylbemicin oxime; EPN = eprinomectin
Two positive for A abstrusus and T brevior and one positive for A abstrusus.
One positive for A abstrusus and T brevior and one positive for A abstrusus.
One died 1 week after therapy and still positive for T brevior
Oral fenbendazole (Panacur; MSD Animal Health) administered at 50 mg/kg q24h for three consecutive days led to clinical, radiographic and infection resolution in 3/3 A abstrusus-positive cats, with two showing improved body condition score 2 weeks post-treatment.
Twelve A abstrusus-positive cats received one (n = 4), two (n = 7) or three administrations (n = 1) of imidacloprid 10%/moxidectin 1% (Advocate; Bayer Animal Health) spot-on before the infection resolution. Clinical recovery was observed 2 weeks after the first dose in 11/12 cats, while in one cat clinical signs disappeared after 4 weeks. Resolution of radiographic signs was observed 2 weeks after the first dose in the majority of cats (7/12), while in others recovery from radiographic signs were observed at the second (3/12) or third (2/12) follow-up performed, respectively, 4 or 6 weeks after the first administration of moxidectin.
In 2/2 cats with troglostrongylosis one administration of Advocate was also able to determine infection resolution, while one cat showed clinical and radiographic signs up to 4 weeks after therapy.
Three cats, co-infected by A abstrusus and T brevior, also received two (n = 1) or three administrations (n = 2) of Advocate before a clinical, radiographic and infection resolution.
Two cats received a single spot-on solution containing emodepside 2.1%/praziquantel 8.6% (Profender; Bayer Animal Health), which was effective in treating T brevior in mixed infections either with A abstrusus or C aerophila, and in assuring a full recovery.
A single administration of milbemycin oxime (2 mg/kg body weight [BW]) and praziquantel (5 mg/kg BW) in tablets (Milbemax; Novartis Animal Health) was able to assure a complete recovery in two cats infected by T brevior, either alone or in mixed infection with A abstrusus. Conversely, a 3-month-old cat with a mixed infection (T brevior and A abstrusus) died suddenly from acute respiratory failure 1 week after therapy.
One cat, affected by lung capillariosis, was successfully treated with a single administration of a spot-on solution containing a combination of fipronil 8.3%, (S)-methoprene 10%, eprinomectin 0.4% and praziquantel 8.3%, (Broadline; Merial).
Discussion
Epidemiological data
All infected cats lived outdoors or were allowed to roam, thus confirming the correlation between the occurrence of lungworm infections and lifestyle of animals. 35 In general, young animals, with a less efficient immune system, and adult and old cats, with better hunting skills and a cumulative greater risk of exposure, may be infected by lungworms.25,35,36 The majority (65.4%; 17/26) of animals in this study were less than 1 year old and 84.6% of cats (22/26) aged not more than 4 years. The recent records of T brevior in domestic cats have shown that this parasite mostly occurs in kittens rather than adults.10,11,16–18,29 Accordingly, in the present study all cats infected by T brevior (10/26) were less than 4 months old.
Clinical findings
In general, clinical presentation and severity of signs depend on lungworm species, parasitic burden and host age.22,37,38 For instance, the large size of adult T brevior and C aerophila greatly impact on their anatomical localisation through trachea, bronchi and bronchioles which are more easily blocked by worms, mainly in young cats,11,16 while A abstrusus live in the lung parenchyma with mature worms found at the terminal parts of the bronchioles, and it is assumed that clinical signs are primarily due to egg production and L1 migration within the respiratory tract. 39
Despite these apparent differences, clinical signs in cats affected by different species of lungworms (either in single or mixed infection) often overlap,11,16 as observed in this study (Tables 1 and 2), thus confirming the lack of specificity of clinical findings in cat lungworm infections. In agreement with these results, the most common clinical signs described for feline aelurostrongylosis are coughing and dyspnoea.25,27,36,40 In four cats affected by aelurostrongylosis, described in the present study, weight loss was recorded as observed in experimentally infected cats. 41 Analogously, signs of troglostrongylosis recorded here are consistent, with the most common being dyspnoea, tachypnoea, severe coughing and nasal discharge.11,16,17
Although nasal discharge and sneezing are in most cases signs of upper airways disease, in this study they were recorded in six patients, as previously observed in cats with infectious pneumonia 42 or aelurostrongylosis. 36 This is probably due to the presence of concurrent and/or secondary infection or airway material into the nasopharynx. 43
A comparative analysis cannot be undertaken here for C aerophila, as the nematode was recorded in only two cats showing coughing and respiratory distress, and one of them was also infected by T brevior.
Clinicopathological findings
The most common finding was a mild normocytic, normochromic anaemia, also observed in natural and experimental A abstrusus infections,41,44 probably due to the chronic inflammatory stimulus triggered by lungworms. Anaemia of inflammatory diseases is characteristically non-regenerative 45 or, less commonly, mildly regenerative. 46 However, it is worth noting that the presence of immature red blood cells observed in a cat, and already described in A abstrusus-infected cats, 47 could be related to hypoxia and not necessarily with a regenerative response to anaemia.
In general, respiratory disorders and endoparasitism in cats can stimulate increased circulating eosinophils48,49 and, accordingly, eosinophilia was observed in cases of natural or experimental aelurostrongylosis.27,41,49,50 However, in the present study eosinophilia was detected in only one cat, while two animals displayed eosinopenia. Cats experimentally infected by A abstrusus displayed eosinophilia in 2 or 4 weeks that persisted for 6 months.41,50 Furthermore, it seems that the number of circulating eosinophils is affected by the activity of lungworm infection within the host. 49 Thus, detection of peripheral eosinophilia could be affected by the time of the diagnosis, and subsequently the eosinophilic count cannot be a valuable diagnostic tool for lungworm infections.
Increased liver enzyme was unexpected, and this finding in feline lower respiratory tract disorders is anecdotal. 21 The hepatic involvement could be expected in the cat presenting hyporexia and weight loss (cat 3), but on the other hand is unexplained for the other cat (cat 13). Although speculative in this case, it should be taken into account that disorders of other organs secondarily affect the liver, and several factors, such as hypoxia, toxins and inflammatory mediators, may contribute to hepatic clinicopathological abnormalities. 51
Hyperglobulinaemia and increased serum proteins, not observed here, can be found in feline bronchial disease, fungal pneumonia, chronic foreign body or aspiration pneumonia or bronchiectasis due to chronic antigenic stimulation.43,52,53 However, increased serum protein is not a common finding in lungworm infection and therefore is not useful in identifying cats infected by lungworms.41,47,54
Radiographic examination
The radiographs showed interstitial and bronchial patterns in 92.3% and 80.8% of cases, respectively. This is consistent with radiographic abnormalities commonly observed in cats affected by aelurostrongylosis.27,36,40,44,55–57
As observed in past and recent studies,26,36 the clinical severity of lungworm infections may only partially correspond to the severity of radiographic signs (Tables 1 and 4). In particular, radiographic changes, such as bronchial thickening and a poorly defined nodular pattern throughout the lung fields, may be evident before the onset of clinical signs,36,39 and a lack of radiographic abnormalities is rarely reported in cats with infectious pneumonia, despite the presence of clinical signs.42,58,59
On the other hand, A abstrusus-positive cats with an alveolar pattern usually show overt clinical signs, 36 and all but one cat, with alveolar involvement, needed multiple treatments and/or showed long-standing signs. Furthermore, a 3-month-old cat, diagnosed with T brevior with a moderate alveolar pattern, died 1 week after treatment.
Despite being aspecific, the presence of radiographic changes, including vascular pattern, suggest a possible lungworm infection in the differential diagnosis in cats, either with respiratory signs and/or apparent healthy, exposed to risk factors.
Anthelmintic treatments and outcome
The present data provide further information on the efficacy of some anthelmintics in the treatment of felid lungworm infections.
The efficacy of fenbendazole for treating aelurostrongylosis, as described in other studies,5,60–62 is confirmed here. However, its use in oral formulations may be impaired by difficulties of the administration to indocile or feral animals and the need for at least a 3 day course.61,62
It was previously shown that spot-on moxidectin guarantees a successful elimination of larval shedding in cats with natural aelurostrongylosis. 61 This molecule was confirmed here to be efficacious in assuring a full recovery from single or mixed lungworm infections, after single or repeated administrations. The number of administrations required may be related to the severity of pulmonary involvement, and/or to other concurrent infections (eg, T brevior). Although the administration of spot-on moxidectin appeared unsuccessful in treating troglostrongylosis in a kitten, 11 in this study a single administration of this compound assured a complete recovery in two kittens. Therefore, the previous failure of moxidectin in treating T brevior 11 could be related to a delay of parasiticide treatment in relation to the severity of the clinical signs. Further studies are warranted to investigate how the severity of lung damage, the time of moxidectin administration and the presence of mixed infections may indeed influence the efficacy of this molecule in treating lungworm infections. Thus, moxidectin is a good option for treating A abstrusus and T brevior in single or multiple administrations, depending upon the severity of radiographic lesions and/or concurrent presence of various lungworms.
Emodepside has shown high therapeutic performances in treating aelurostrongylosis in both natural and experimental conditions.62,63 In this study, one administration of emodepside was efficacious to interrupt larval shedding and to lead to complete clinical and radiological remission in two kittens with mixed lungworms infection (A abstrusus and T brevior; T brevior and C aerophila). Therefore, it could be argued that this molecule could have the potential to treat T brevior and C aerophila infections as well (Table 5) and, thus, further studies on a large scale are warranted.
Information available on the efficacy of milbemycin oxime in treating feline lungworms is poor. A single administration of this compound here was effective in stopping larval shedding and ensuring clinical recovery of two kittens with single (T brevior) and mixed (A abstrusus and T brevior) infections. On the other hand, the death of a kitten infected by A abstrusus and T brevior calls for further studies aiming at evaluating the efficacy of milbemycin in lungworm infections and the possible factors able to influence its efficacy in different clinical conditions.
Different compounds have shown efficacy against lung capillariasis in cats.64–66 In the present study, a single administration of eprinomectin was able to guarantee a full recovery in a cat, as previously described. 66
Conclusions
Feline lungworm infections are frequently associated with respiratory signs, sometimes severe, especially in young subjects. A complete history should be collected to investigate the exposure to risk factors (eg, outdoor habitat, strong predatory instinct). Findings from history and clinical signs are non-specific, and a lack of them does not preclude a differential diagnosis of lungworm infection. Thoracic radiographs are advisable in cats with lungworm infection regardless of the presence of clinical evidence. Hence, copromicroscopic examinations should be considered as a first step in the diagnostic work-up for feline respiratory airway diseases and, especially in endemic areas, they could also be a useful screening tool in apparently healthy cats exposed to risk factors. This would allow a prompt anthelmintic therapy that, in most cases, guarantees a complete clinical and parasitological recovery. To date, few products are licensed against A abstrusus infection. However, a variety of compounds, such as moxidectin, emodepside, milbemycin oxime and eprinomectin showed promise against T brevior and C aerophila, and further large field studies are warranted, if one considers the current spread of lungworms, either in single and coinfections.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The authors have performed this study on the basis of their freedom of research.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
