Abstract
Objectives
Dermatophytosis, commonly known as ringworm, is a superficial fungal skin disease and zoonosis. Pythium oligandrum is a micromycete with mycoparasitic properties that is used in agriculture to control fungal infections on plants. Formulations containing P oligandrum were also developed for the treatment of dermatophytoses, but only a small number of case studies have been published. In order to document the process in simplified conditions in vitro, we investigated the effectiveness of P oligandrum against three pathogenic dermatophytes common in domestic animals.
Methods
Cultures of the pathogens grown on nutrient media and experimentally infected cat hair were treated with P oligandrum preparations in therapeutic concentration and the changes were documented by microscopic videos and scanning electron microscopy.
Results
There was strong mycoparasitic activity of P oligandrum against Microsporum canis, Microsporum gypseum and Trichophyton mentagrophytes.
Conclusions and relevance
P oligandrum was demonstrated to be effective against three common causes of dermatophytosis in vitro.
Introduction
Dermatophytosis is a superficial fungal disease of skin and hair. 1 In domestic animals, especially in pets, it is a significant issue as it is a highly infectious and contagious zoonosis. The most commonly isolated pathogen from cats is Microsporum canis, however Microsporum gypseum and Trichophyton species have been also isolated. 1 Although this is a self-limiting condition, treatment is recommended to shorten the course of the disease and to minimise spread to other animals and people. Systemic antifungal drugs are used to eradicate the pathogenic microorganisms from the hair follicle and sporicidal topical therapy is used to treat the haircoat. Topical therapy helps to prevent shedding of viable infective spores to the environment, which might complicate treatment monitoring, and potentially serve as a source of infection for susceptible hosts. The most widely used topical antifungal agents include antifungal azole-based shampoos, lime sulfur and enilconazole. In some situations, topical therapy is used as the sole treatment of choice when systemic therapy is cost prohibitive and/or is not approved for use in some species (eg, horses and cattle). Topical therapy needs to be repeatedly applied until the infection is eliminated, making efficacy, safety and ease of application important issues.
Pythium oligandrum is a soil micromycete with mycoparasitic properties that has been successfully used in agriculture to control fungal infections on plants. It is naturally widespread worldwide. Fungi susceptible to the treatment with P oligandrum in plant protection include Fusarium species, Pythium spinosum, Pythium nunn, Pythium ultimum and Pythium irregulare. 2 One comparative study on sugar beet culture showed that P oligandrum was more effective than a standard chemical antifungal agent. 3 The antifungal efficacy of this microorganism is directly related to its growth needs. P oligandrum forms classical, invasive, very fast growing hyphae, spherical oogonia with small spikes 21–31 μm in size and produces oligandrin, a complex of various enzymes and ferments, which facilitate its penetration into the body of the target fungus. 4 Genes for these enzymes have not been identified in the pathogenic Pythium species, 5 parasitising on animals and plants. Another specific biological characteristic of Pythium species is the production of sporangia that release zoospores. Zoospores of P oligandrum attack the hyphae of target fungi. 6 In essence, this organism obtains necessary growth nutrients only by consuming target fungi. When there are no more nutrients left it vanishes from the area. 7 There are no reports of negative effects of P oligandrum on any organisms expect the target pathogenic fungi. There are clinical reports of P oligandrum use in the treatment of dermatophytosis in people and animals.8–11 Given that dermatophytosis is a self-limiting disease, it is tempting to hypothesise that the activity of P oligandrum in this clinical setting might be supported by the host cell-mediated immunity that might also contribute to the elimination of the infection. Although there are two P oligandrum commercial formulations available, to our knowledge, mycoparasitism of P oligandrum against these dermatophytes has not yet been documented. The aim of this study was to visualise in vitro the antifungal effects of P oligandrum against the mycelial form of three common dermatophyte pathogens: M canis, M gypseum and T mentagrophytes.
Materials and methods
Testing of two P oligandrum formulations
Two commercially available formulations of P oligandrum were tested (Dermasin and Ecosin; Bio Agens Research and Development). Dermasin is formulated as an oil suspension; Ecosin is supplied as an effervescent tablet for dilution in water. Products were used as recommended by the manufacturer. Ecosin and Dermasin were used for gross observations of effects on Petri dish growth. Ecosin was used for electron microscopy observations. Dermasin was used for filming the P oligandrum effects on infected hairs.
Confirmation of P oligandrum identity by Sanger sequencing
To confirm the identity of P oligandrum in the formulations used in our study, we isolated total DNA from the P oligandrum products and performed direct Sanger sequencing of the evolutionary conserved 18S rDNA genes. The analysis revealed the correctness of the active agent in the pharmacological formulations used in our experiments (Figure S1).
Cultures of pathogenic fungi in vitro
Cultures from collection strains of M canis, M gypseum and T mentagrophytes were used. The fungi were grown both on Sabouraud’s dextrose agar and Dermatophyte Test Medium (OXOID CZ; Thermo Fisher Scientific) and incubated at room temperature (25–29°C). Photographic documentation of the mycoparasitism started on cultures 24 h postinoculation and took 5 days. Microscopic documentation was performed on well-grown colonies 14–19 days postinoculation. In the cat hair study, cultures were used after 7 days of growth. Grown pathogenic fungi were identified by microscopic examination performed by a professional mycologist (DK).
Macro- and microphotography and electron microscopy
Changes in gross colony morphology were documented using the macro mode of a Nikon Coolpix P510 camera.
A Nikon optical microscope with an attached Canon EOS 1100D camera and an Olympus BX50 optical polarising microscope with the DP70 digital camera were used to document microscopic changes. To visualise the changes on fungal culture plates, an Olympus SZX16 optical binocular microscope equipped with the Canon EOS digital camera was used. Video documentation of macro- and microscopic changes was carried out using sequence scanning and QuickPHOTO MICRO 3.0 software.
The electron microscopy was performed using a Tescan VEGA3 XMU scanning electron microscope (SEM); the optimum magnification for documentary photography was 5000–10,000×. Photos of the gilded samples were taken in the secondary electrons mode.
Experimental studies
Macroscopic colony growth changes
After allowing inoculated fungal culture plates to incubate for 48 h, 5 ml Ecosin were added to the surface of the plates. The plates were observed for 5 days and changes in macroscopic morphology were documented. Experiments were repeated in duplicate. Untreated plates served as controls.
Microscopic changes in colony growth
After allowing fungal culture plates to incubate until mature colonies were present (14–19 days), the effect of P oligandrum against the thick-grown mass of the pathogens was evaluated. Ecosin water solution (5 ml) or Dermasin oil (1 droplet) were applied directly to the colony and the disappearance of the hyphae was documented using continuous photography of randomly selected sites in the colonies. Based upon pilot data, the optimal time to make initial observations was 4–6 h after addition of P oligandrum for Microsporum species and 3–4 h for T mentagrophytes.
Cat hair test
Sterile cat hair was added to the surface of 8 day-old fungal culture plates and incubated for 10 days at room temperature (25–29°C). When hairs were visibly covered with dermatophyte growth, a sample of cat hair and fungal growth was mounted on a glass microscope slide in mineral oil. After microscopically confirming the presence of fungal growth on hairs, three droplets of Dermasin were added to the mineral oil mount. Photomicrographs were taken hourly on day 1 and then once daily on days 2, 3, 4, 6, 7 and 8 post-treatment along with microscopic video documentation. The cat hair test was repeated with Ecosin with the exception that hair samples were treated for 2, 5 and 24 h before SEM – this timing, which roughly corresponded to the start, maximum and end of the reaction between P oligandrum and the fungi observed in macroscopic experiments, was selected in order to maximally visualise the main steps of the reaction in detail.
Results
In our initial experiment, we compared the macroscopic development of the colonies of pathogenic fungi on nutrient media growing spontaneously and in the presence of aqueous P oligandrum suspension. All of the pathogens were visibly affected by P oligandrum, but growth was observed on the edge of the colonies that were not in direct contact with the liquid (Figure 1).
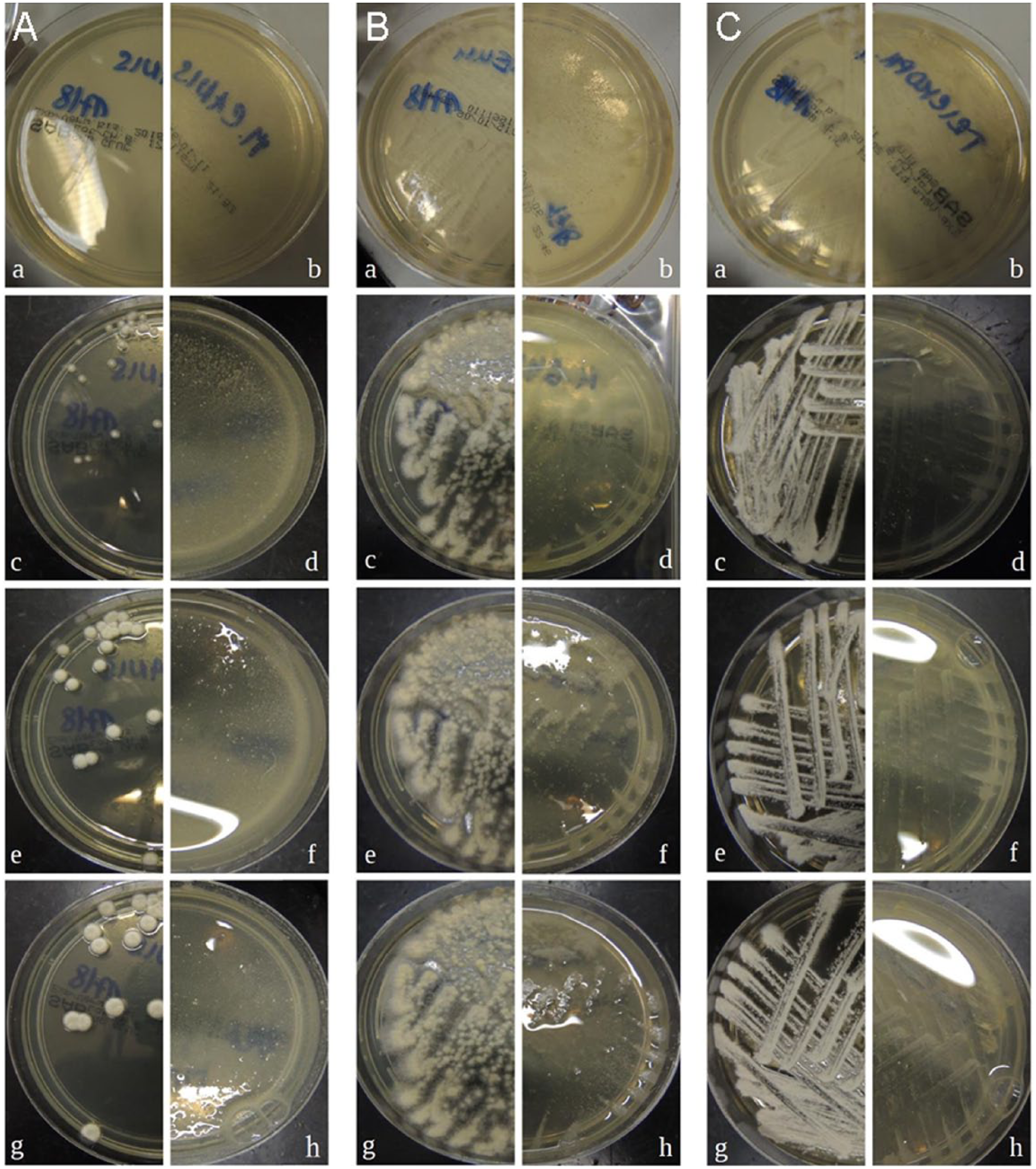
Growth of (A) Microsporum canis, (B) Microsporum gypseum and (C) Trichophyton mentagrophytes on nutrient media. The images on the left of each column (a,c,e,g) document the growth of the intact mycoparasite culture; the corresponding images on the right (b,d,f,h) show the culture treated with Pythium oligandrum. Photographs (a,b) 48 h after inoculation, (c,d) 72 h, (e,f) 96 h and (g,h) after 120 h
Subsequently, we aimed to visualise the P oligandrum antifungal activity in vitro by a microscopic documentation of mature fungi colonies after the addition of aqueous or oil suspension of P oligandrum. Photo microscopy and video microscopy revealed that hyphal growth was affected within a few hours of contact with the biological agent (supplementary material Figures S2 and S3 and Videos S4–S9 show the quickly disappearing hyphae of all of the pathogenic fungi). Macroscopic changes were most remarkable and rapid with T mentagrophytes and slowest with M gypseum, according to the time of observation. We also performed long-term observations, which revealed that a regrowth of the hyphae in the cleared-up areas started slowly about 72 h after exposure to P oligandrum. The findings were similar with both P oligandrum formulations. Moreover, we were able to see that P oligandrum actively searches for its ‘prey’. Figure 2 was observed 7 days after Ecosin droplets were applied to the edge of a Petri dish a few mm away from the M canis culture. It showed that the P oligandrum hyphae grew and attacked the hyphae of M canis and intensified their growth in the direction towards the colonies of pathogenic fungus.

Growth of the thick invasive hyphae of Pythium oligandrum from the bottom of the picture toward the hyphae of the Microsporum canis culture (above, much thinner; blue arrows) on a nutrient medium. Note the swollen hyphae of the M canis being attacked by P oligandrum (centre of the picture; red arrows). Scale bar = 0.5 mm
Additionally, a cat hair test, in which sterile cat hair samples were infected by M canis, M gypseum and T mentagrophytes, revealed that hyphae, which covered a hair, were completely gone within several hours of P oligandrum treatment. In the case of M canis and T mentagrophytes (Figure 3, and supplementary material Videos S10 and S11), the main part of the process took 3–6 h, while the disappearance of M gypseum hyphae and especially of its macroconidia took significantly longer. We documented these effects through video microscopy of a single cat hair treated with an oil suspension of P oligandrum. The SEM images document a randomly selected hair taken from a hair sample that underwent treatment with the aqueous P oligandrum suspension. Figure 4 shows a hair before treatment and other hairs after 5 and 24 h of the treatment. Note the damaged structure of the hair – parts of the keratin forming the naturally scaled structure of hair were consumed by the dermatophyte.

(a) A cat hair strongly invaded by Trichophyton mentagrophytes and (b) the same hair after 6 h of Pythium oligandrum treatment. Also see Video S11 in the supplementary material
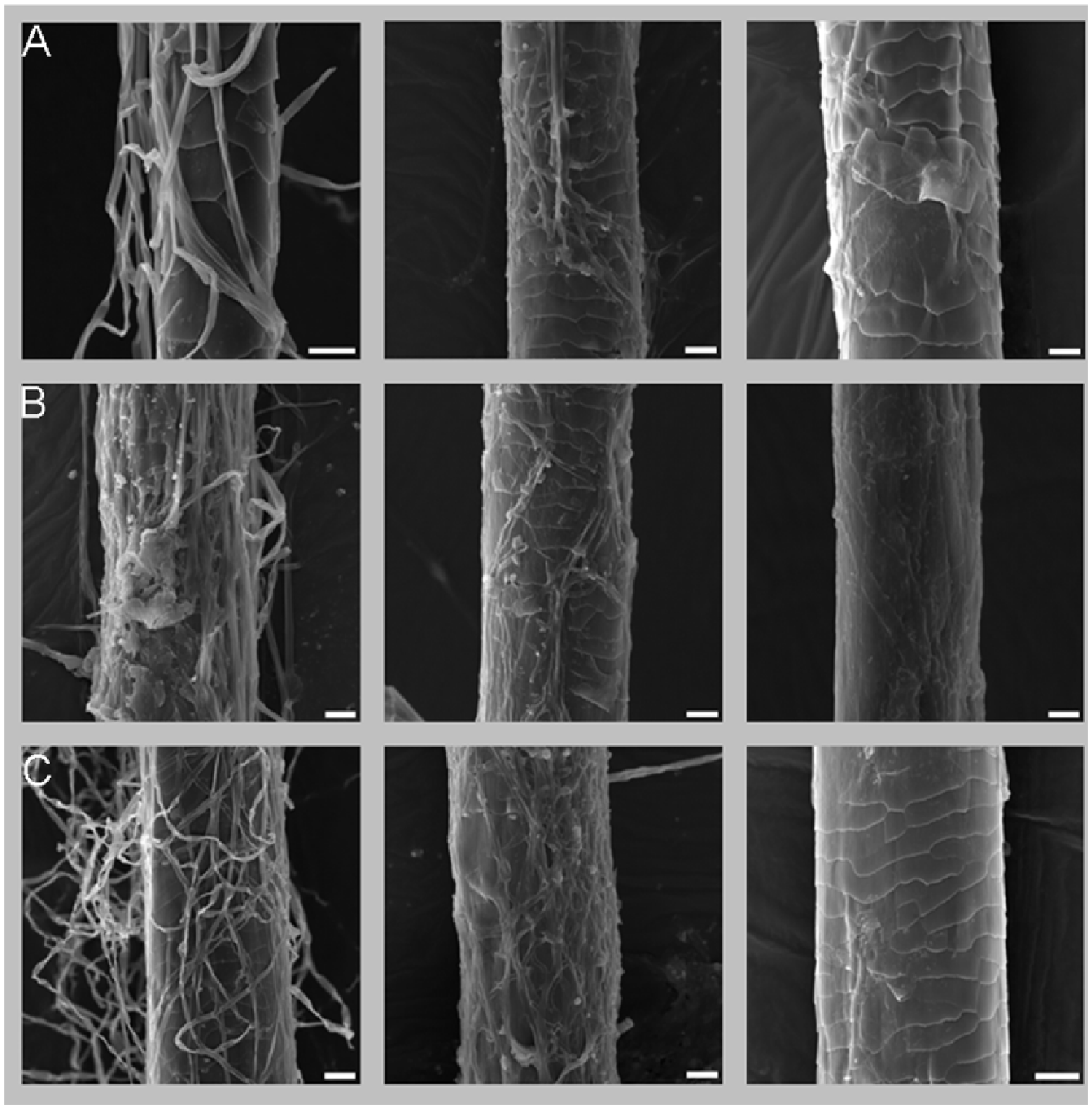
Scanning electron microscopy images of cat hair infested with Microsporum canis (row A), Microsporum gypseum (row B) and Trichophyton mentagrophytes (row C). The images in the left column show a hair before treatment, those in the centre show a hair after 5 h in a Pythium oligandrum solution and figures in the right column depict a hair after 24 h in a P oligandrum solution. Scale bar = 10 μm in each image. Note that the hyphae are severely damaged after 5 h of P oligandrum action and that the hair treated for 24 h is practically without hyphae, and its topical structure is also damaged – parts of the keratin forming the naturally scaled structure of the hair were consumed by the dermatophyte
Discussion
In this observational study, we documented that P oligandrum has in vitro antifungal efficacy against M canis, M gypseum and T mentagrophytes. Macroscopically, fungal colonies in contact with this biological antifungal agent showed a rapid loss of hyphae, presumably due to direct elimination by P oligandrum. At this time it is unknown whether these effects would mirror the naturally occurring infections, similar to what has been reported in agricultural applications on plants. 12
There is a consistent need for the continued development of drugs to treat dermatophytosis. Topical therapy is important for the elimination of the infection from the haircoat, but many animal owners refuse to use products that they perceive to be messy or odorous. The development of resistance is also a concern. 13 The use of alternative therapies, such as an agent with naturally competitive relationship is an interesting option. The natural biological antifungal properties of P oligandrum demonstrated in vitro suggest that this may offer an alternative topical antifungal treatment for dermatophytosis. To support this hypothesis, we have shown the antifungal activity of P oligandrum, demonstrated on experimentally infected cat hair, which can be considered as a simplified model for the treatment option of an animal with superficial dermatomycosis. We also observed the loss of natural scales on the hair surface at the sites that were massively infested by the pathogenic hyphae. This observation suggests that the dermatophytes can attack the whole haircoat, and that topical antifungal therapy should never be underestimated.
To our knowledge, there are no reports of harm to any organism or environment, except of the target fungi by P oligandrum, although it has been used in agriculture to a large extent for more than two decades. The publications about use of P oligandrum for treatment of dermatophytoses in vertebrates also do not report any unusual effects,8–10,14 with the exception of several observations of transitory local alopecia of the dermatophytosis affected sites, 11 which was probably an effect of the dermatophytosis.
In our study, P oligandrum microscopic studies were similar to those carried out on plants in agriculture. 15 These studies describe P oligandrum as an aggressive mycoparasite that attacks the hyphae of pathological fungi, including Pythium species.3,6 The observations shown in our study confirm that P oligandrum attacks the hyphae of M canis, M gypseum and T mentagrophytes in a similar fashion. Moreover, in our study, the elimination of pathogens by P oligandrum was video documented as a dynamic process. The experiment with the cat hair being a real substrate for the tested pathogens gave similar results as the observation carried out on cultures. The regrowth of the pathogenic dermatophyte after approximately 72 h after the treatment suggests that one bolus of P oligandrum does not destroy the pathogen totally and needs to be used repeatedly. A field study carried out among veterinarians using Ecosin for treatment of cats and dogs implies that application over two consequent days followed by a 2 day break between the individual applications might be ideal. 14 This is consistent with our findings, where regrowth of hyphae of dermatophytes was observed. 12 Further research is needed to confirm the correct treatment protocol for use in practice.
Conclusions
Our results showed that P oligandrum is an aggressive mycoparasite of M canis, T mentagrophytes and M gypseum. The mycoparasitism was proven on cultures of the pathogens and with comparable results on a cat hair sample, which served as a simplified model for animal treatment. These findings are consistent with previously published studies in plants and case reports in veterinary medicine. It is necessary to emphasise that the concentration of the pathogens in our experiments was much higher than would be expected in an infected animal. Nevertheless, even in this artificial setting with pathogen overdose, the P oligandrum formulations proved their effectiveness. We believe that our findings will be useful for further in vitro studies and in vivo treatment studies in animals, and possibly prospectively, in people.
Supplemental Material
Click here for Supplementary Figure 1
Sanger sequencing of the P oligandrum used in the study. Evolutionarily conserved 18S rDNA genes were sequenced to prove the identity of the effective agent
Supplemental Material
Click here for Supplementary Figure 2
Fungal culture of M canis (a) a moment before the application of a P oligandrum preparation and (b) 30 mins after the P oligandrum application. The P oligandrum preparation was applied in the region visible in the far upper left edge of the picture. See also Video S5
Supplemental Material
Click here for Supplementary Figure 3
Fungal growth of M gypseum before the treatment (a) and 30 mins after the application of a P oligandrum preparation (b). Note the decrease in hyphae density in the central part of (b). See also Video S6.
Footnotes
Acknowledgements
We wish to acknowledge Professor Dr Karen A Moriello DVM, DACVD, for her kind consultation during our work. We wish to acknowledge Ing Eva Žallmannová PhD, for the editing of our manuscript.
Supplementary material
The following files are available: Figure S1: Sanger sequencing of the P oligandrum used in the study. Evolutionarily conserved 18S rDNA genes were sequenced to prove the identity of the effective agent.
Figure S2: Fungal culture of M canis (a) a moment before the application of a P oligandrum preparation and (b) 30 mins after the P oligandrum application. The P oligandrum preparation was applied in the region visible in the far upper left edge of the picture. See also Video S5.
Figure S3: Fungal growth of M gypseum before the treatment (a) and 30 mins after the application of a P oligandrum preparation (b). Note the decrease in hyphae density in the central part of (b). See also Video S6.
Video S4: Hyphae of M canis (white) disappearing within several minutes. The scanning began just 6 h after the addition of aqueous suspension of P oligandrum to the culture.
Video S5: Hyphae of M canis (white) disappearing within 30 mins while treated with an oil suspension of P oligandrum.
Video S6: Hyphae of M gypseum (white) disappearing within 30 mins. The scanning began 3 h after the addition of the aqueous suspension of P oligandrum.
Video S7: Hyphae of M gypseum (white on pink Sabouraud nutrient medium) disappearing within 65 mins of the treatment with an oil suspension of P oligandrum. Note that the oval-shaped spores of the pathogen are also eliminated by P oligandrum.
Video S8: Hyphae of T mentagrophytes (white) disappearing within several minutes. The scanning began 3 h after the addition of aqueous suspension of P oligandrum.
Video S9: Hyphae of T mentagrophytes (white) disappearing during the treatment with oil suspension of P oligandrum.
Video S10: Cat hair overgrown with M canis treated with oil suspension of P oligandrum. The documented part of the treatment process took 6 h.
Video S11: Cat hair overgrown with T mentagrophytes treated with oil suspension of P oligandrum. It took 6 h until the hair was visually clear of hyphae.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The Institute of Geology, project RVO 67985831, is acknowledged for granting the use of the scanning electron microscope.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
