Abstract
Objectives
Cardiorespiratory parameters and anaesthesia quality in cats anaesthetised with either intramuscular (IM) alfaxalone or ketamine both combined with dexmedetomidine and butorphanol for castration were evaluated.
Methods
Thirty-two client-owned cats were randomly assigned to receive either alfaxalone (A; 3 mg/kg IM) or ketamine (K; 5 mg/kg IM), combined with dexmedetomidine (10 μg/kg) and butorphanol (0.2 mg/kg). Heart rate (HR), respiratory rate (RR) and rectal temperature (T°) were recorded prior to drug administration. Pulse rate (PR) and RR were recorded 10 (T10) and 15 (T15) mins after injection (T0). Cardiorespiratory values (PR, RR, SPO2, blood pressure, PE′CO2) were recorded every 5 mins for the duration of the procedure. Pain at injection, intubation and recovery were evaluated with simple descriptive scores. Feasibility of anaesthesia was evaluated by the number of top-ups of anaesthetic needed. Cat attitude, ability to walk and presence of ataxia were assessed several times after extubation (Texmin) and the time between injection and extubation recorded. Pain was assessed at Tex120 and Tex240 with the 4Avet-pain score.
Results
The RR was significantly lower in group K at T10 (RRK = 28 ±13.35 breaths per minute [brpm], RRA= 43.24 ±7.04 brpm) and T15 (RRK = 28 ±11.53 brpm vs RRA = 43 ±12.18 brpm). Time to extubation was significantly longer in group A (TA = 62 ±14.6 mins, TK = 45.13 ± 7.38 mins). Cats in group K needed more top-ups, were more ataxic at Tex120, had a worse recovery score at Tex60 and were less willing to walk at Tex30.
Conclusions and relevance
Cats receiving alfaxalone had a longer but better quality recovery. Cardiorespiratory parameters were stable and within clinically acceptable values following IM injection of either alfaxalone or ketamine in healthy cats. Intramuscular alfaxalone is a suitable alternative to ketamine for short procedures requiring anaesthesia when used in combination with dexmedetomidine and butorphanol.
Introduction
Cat castration is a common procedure carried out in general practice and requires general anaesthesia. It is often carried out on young healthy cats that may resent physical restraint and intravenous catheter placement. Ketamine combinations are widely used for anaesthesia in cats. 1 It produces a cataleptoid state involving unconsciousness and somatic analgesia but not muscular relaxation. 2 Therefore, ketamine is commonly administered in combination with an α2-agonist and an opioid in order to improve muscle relaxation compared with when ketamine is administered alone.3–9 Ketamine, α2-agonist and opioid combinations appear to be practical for surgical procedures of short duration and provide adequate maintenance of ventilation.1,10 Recently, there has been international discussion about the non-medical use of ketamine. It has raised some discussion about the availability of ketamine in the veterinary market in the future. This uncertainty has led veterinarians to seek suitable alternative intramuscular (IM) anaesthetic agents for cats and dogs.
Alfaxalone is an anaesthetic induction agent widely used in feline anaesthesia.11–17 The European licensing of alfaxalone is for intravenous (IV) injection only; however, the Specific Product Characteristics have permitted IM injection of alfaxalone in Australia, New Zealand and South Africa for a decade. To our knowledge, only a few studies have been published reporting IM administration of alfaxalone in cats, and these have reported very few side effects.15,18–21 A single report describes the combination of alfaxalone, dexmedetomidine and butorphanol given IM in a clinical setting. 21 Cats recovering from a ketamine-based anaesthesia protocol have also been described as hypersensitive to touch and noise, making the recovery unsettled.3,5,22,23 In addition, IM injections of ketamine are reported as painful, probably owing to the acid pH of the solution. 24 Given the aversive reactions to ketamine, alfaxalone IM may well provide a better recovery quality and a less aversive response to IM injection than ketamine. 22 Therefore, the study aim was to investigate the use of intramuscularly administered alfaxalone as a general anaesthetic agent vs ketamine in cats hospitalised for castration. The objectives of the study were to: (i) establish whether IM administration of alfaxalone, when combined with dexmedetomidine and butorphanol, was suitable to induce and maintain anaesthesia for the duration of a cat castration, possibly followed by an identification procedure such as an ear tattoo or subcutaneous microchip placement; (ii) compare cardiorespiratory and recovery quality data between this protocol and one using ketamine combined with dexmedetomidine and butorphanol; (iii) determine whether alfaxalone given IM has fewer side effects and if the duration of general anaesthesia is well matched to the duration of the procedure.
Materials and methods
Animals
Thirty-two male cats presented for castration (16 in each group), weighing 3.62 ± 0.75 kg with a mean age of 10.34 ± 5.4 months, were recruited to this study after obtaining informed owner consent and approval from the Association of Veterinary Anaesthetists (AVA) ethical committee (2015-002). The primary measure of interest was the quality of recovery. A sample size calculation was undertaken, based on previous observations. For a power of 80% at an alpha of 0.05, 24 cats were needed. This study was performed in a set time period and therefore we decided to recruit as many cats as possible.
Study design
This was a prospective, randomised, clinical study involving two parallel groups. In association with dexmedetomidine and butorphanol IM, group A received alfaxalone IM, and group K received ketamine IM. All cases were enrolled over a 3 week period during the winter of 2015 at ONIRIS (Nantes-Atlantic National College of Veterinary Medicine, Food Science and Engineering) animal reproduction service. All the procedures (surgery, tattoo, microchip injection) were performed by final-year students supervised by an intern.
Anaesthesia
All cats were starved overnight. A complete physical examination was performed and American Society of Anesthesiologists’ status I was confirmed in all animals. At this physical examination heart rate (HR) in beats per minute (bpm), respiratory rate (RR) in breaths per minute (brpm) and rectal temperature were recorded. Cats were weighed and randomly (simple block randomised by drawing of lots) allocated to one of the two groups. Both groups were administered drugs IM in the lumbar muscle with a 23 G × 5/8” needle; 0.01 mg/kg dexmedetomidine (Dexdomitor; Elanco) and 0.2 mg/kg butorphanol (Torbugesic; Zoetis) was mixed with either 3 mg/kg alfaxalone (Alfaxan; Dechra) or 5 mg/kg ketamine (Imalgène 1000; Mérial). Reactions to needle insertion and drug injection were scored separately with a descriptive score (see Appendix 1 in Supplementary material) 25 by the same operator, who was not blinded. Following drug administration cats were kept in a holding box and observed continuously. Ten (T10) and 15 (T15) mins after the injection, quality of sedation (see Appendix 2 in Supplementary material) 18 was scored by a blinded observer and HR and RR were recorded (by auscultation and observation, respectively). An IV catheter was placed in the cephalic vein at T10. A SpO2 probe (TidalGuard HD; Midmark) was placed on the tongue at T15. Endotracheal intubation with a cuffed endotracheal tube (3 or 3.5 mm ID) was then attempted following spraying the arytenoid cartilages with 0.1 ml 2% lidocaine solution (Xylovet; Ceva). Signs of inadequate anaesthesia at this time point (eg, chewing, moving)triggered the administration of an IV top-up of either ketamine or alfaxalone, depending on group allocation. A top-up syringe was prepared for each cat by a separate investigator, who was aware of group allocation. The top-up comprised either ketamine or alfaxalone appropriately diluted to give 0.2 ml per top-up per cat and provided either 0.3 mg/kg of alfaxalone or 0.5 mg/kg of ketamine. The number of top-ups was recorded, as well as the time between the IM injection and the intubation. The quality of intubation was assessed by the blinded operator using a descriptive scale (see Appendix 3 in Supplementary material). 15
Procedure
Following endotracheal intubation, cats were placed in dorsal recumbency and connected to a Bain breathing circuit providing 1 l/min of 100% oxygen. An oscillometric cuff of the appropriate size (40–60% of the limb circumference) was placed over the brachial artery for indirect measurement (NIBP) of systolic (SAP), mean (MAP) and diastolic arterial blood pressure (DAP) (petMAP Graphic; Ramsey Medical). This blood pressure measurement has previously been evaluated and shown good agreement with direct blood pressure measurement. 26 Cats were connected to a side-stream capnograph (V9004; Surgivet) with a low dead space endotracheal tube connector, including a gas sampling port, and SpO2 was also monitored. Once all the monitoring devices were in place, cats were aseptically prepared in order to be castrated. Before starting surgery, meloxicam (0.3 mg/kg) was administered subcutaneously and lidocaine 2% (Xylovet; Ceva) 1 mg/kg was injected subcutaneously in two sites at the level of each incision on the left and right side of the scrotum.
From the time of intubation until the end of the procedure, cats were monitored every 5 mins by checking and recording pulse rate (PR) from pulse oximeter, RR, NIBP, end-tidal carbon dioxide concentration (PE’CO2), SpO2 and depth of anaesthesia (by checking jaw tone, eye position and palpebral reflex). Anticipated noxious stimuli (lidocaine injections, skin incisions, tractions on the testicle, spermatic cord canal and artery ligations, testicle removal) were recorded. After castration, an ear tattoo for identification was undertaken in some cats (n = 10). Occurrence of reactions (movement, eg, lifting of the head, flexion/ extension of the neck, or chewing) during identification procedure was recorded. Cats were monitored until palpebral and swallowing reflexes returned. The time between the IM injection, the start of surgery and extubation were recorded, as well as surgical duration. Signs of inadequate anaesthesia such as limb or body movements, and sudden increases in RR, HR and NIBP were used to determine the need for a top-up injection, in which case a 0.2 ml top-up of either alfaxalone or ketamine was administered IV.
After extubation, HR, RR and rectal temperature were recorded once. Cats were maintained on a heat mattress and observed continuously until they raised their heads and their temperature remained above 37°C. The time to keep their heads raised and maintain sternal recumbency were recorded. Sedation score (see Appendix 2 in Supplementary material), recovery quality (see Appendix 4 in Supplementary material), 18 ability to walk, presence of ataxia, attitude and position were recorded 15, 30, 45, 60, 120 and 240 mins after extubation (Texmin). 15 Pain was assessed at Tex120 and Tex240 using a composite pain score 4AVet (see Appendix 5 in Supplementary material). If the pain score was >6, the cat was given buprenorphine 0.02 mg/kg subcutaneously (SC). Cats were discharged 6 h after extubation and were given buprenorphine 0.02 mg/kg SC just before leaving, whether they received rescue analgesia or not.
Statistical analysis
Baseline physiological parameters (HR, PR, RR, NIBP, T°) were normally distributed and values measured at the initial clinical examination were compared between groups using an unpaired t-test. The normality of physiological data measured after the IM injection, evaluated using a Shapiro–Wilk test, was found not to be normally distributed. Therefore, these data were analysed using a mixed-model analysis with Bonferroni post-hoc correction with P values <0.05. Drugs used and time points were entered as fixed variables and the animal itself as the random effect. All the non-parametric (categorical) data were analysed with a Kruskal–Wallis rank sum test. An unpaired t-test was performed to compare the number of top-ups needed per cat, as well as the number of cats needing a top-up.
Results
There was no difference in age, weight or breed between the two groups of cats. The cats from groups A and K had a mean ± SD age of 10 ± 4 months and 10 ± 6 months, respectively, a mean ± SD body weight of 3.51 ± 0.56 kg and 3.73 ± 0.92, respectively, and were mainly of European shorthair breed apart from one Maine Coon and one Persian cat in group K.
Injection, insertion of needle, sedation and intubation score are presented in Table 1. The injection score was significantly higher than the needle insertion score.
Median (range) insertion of the needle score, injection of the drug score and intubation score in cats from groups A and K
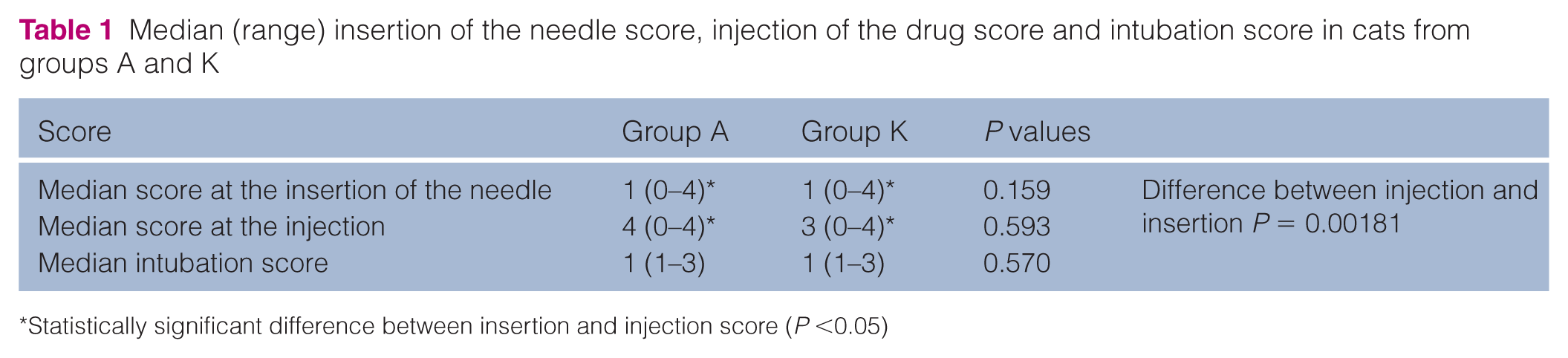
Statistically significant difference between insertion and injection score (P <0.05)
The number of cats needing a top-up of anaesthetic was significantly higher (P = 0.006) in group K (10/16 vs 4/16 in group A), as well as the number of top-ups needed per cat (P = 0.03) (mean ± SD 0.25 ± 0.45 group A vs 2 ± 2.24 in group K).
Cardiovascular variables were not different between groups (Table 2). The PR and MAP between intubation and extubation in both groups showed a significant time effect. RR was significantly lower at 10 and 15 mins after the injection in group K from baseline or compared with group A. A time effect was also found in both groups. The rectal temperature in recovery was significantly decreased compared with baseline in both groups (Table 2).
Baseline, intraoperative and postextubation values for 32 cats undergoing castration under alfaxalone (A) or ketamine (K) anaesthesia

Baseline values presented as mean ± SD for pulse rate (PR; beats per minute), respiratory rate (RR; breaths per minute) and body temperature (T◦). Physiological values are given for 10 and 15 mins after injection (T10 and T15). Intraoperative (from 20–55 mins) and postextubation values presented as median (range) for PR, RR, systolic arterial blood pressure (SAP; mmHg), mean arterial blood pressure (MAP; mmHg), diastolic arterial blood pressure (DAP; mmHg), peripheral arterial haemoglobin oxygen saturation (SpO2, %) and end expiratory pressure in carbon dioxide (PE′CO2; mmHg)
Statistically significant difference between group (P <0.05)
Statistically significant difference over the time (P <0.05)
NA = not applicable
One cat from group A presented signs of cyanosis 5 mins after the IM injection, before endotracheal intubation. It was believed to be apnoeic, but after intubation the cat began breathing spontaneously without need for manual ventilation. This cat was a Persian, and it was hypothesised that this cyanotic episode could be explained by its airway conformation. However, as this event happened very quickly, it is difficult to make a firm conclusion. For these reasons we decided to exclude this cat.
Surgery time was not different between groups (mean ± SD 27±4.69 mins in group A vs 27.27±2.49 in group K), but the time between intubation and extubation was significantly longer (P = 0.00046) in cats from group A (mean ± SD 62.06 ± 14.67 mins vs 45.13±7.38 in group K). Both groups had an equivalent number of cats that received an ear tattoo (seven [group A] and three [group K] cats)
During recovery, most cats from both groups were calm until 240 mins after extubation. One cat from group K showed signs of excitement during the first hours following extubation. All cats were in sternal recumbency between 15 and 30 mins after extubation. Cats from group A were significantly more willing to walk 30 mins after extubation (9/16 vs 2/16; P = 0.010) and less ataxic 120 mins after extubation (P = 0.003). Cats from groups A had a significantly better quality of recovery compared with cats from group K, 60 mins after the extubation (Table 3). Three cats (one from group A and two from group K) needed rescue analgesia, two cats at 120 mins after extubation and one at 240 mins after extubation (Table 3).
Median (range) recovery, sedation and pain score in cats from group A and group K at different time points after extubation

Statistically significant difference between group (P <0.05)
NA = not applicable
Discussion
The present study compares the use of ketamine and alfaxalone combined with dexmedetomidine and butorphanol when given IM to cats undergoing castration. Both combinations allowed full general anaesthesia and endotracheal intubation, with only minor side effects observed in both groups of cats.
Cats from both groups had a high injection score, indicating pain on injection, with no significant difference in scores between groups. This has been previously described for IM injection of both alfaxalone and ketamine.18,24 IM injection of ketamine has been reported to feel like a sting, although there are no published papers investigating this. 24 It has been suggested that pain on injection with alfaxalone is due to the large volume.18,21 To try to differentiate discomfort from needle insertion and product injection, each step was scored separately. For both groups, the solution injected triggered more severe reactions from the cats than the needle through the skin and muscles. It was not possible to determine for alfaxalone if cats reacted to the irritant property of the solution or to the volume. Owing to the difficulty in restraining non-sedated cats, the injection of the drugs occurred immediately after the insertion of the needle. Therefore, discrimination between both scores might be difficult. Taking into account the fact that the operator marking theses scores was not blinded makes this result debatable and difficult to interpret.
Duration of surgery was the same in both groups, but the time between intubation and extubation was significantly longer for cats in group A. Cats from group K required a higher rate of anaesthetic IV redosing, suggesting the dose of ketamine in our study was inadequate for castration by veterinary students in a teaching hospital setting. To the best of our knowledge, there were no equivalent studies reported in the literature at the time of undertaking this study. Therefore, the drug doses were selected on the basis of the clinical experience of the study’s designer. It is therefore possible that the alfaxalone and ketamine doses used were not equipotent. Most of the top-ups were needed at 30 mins after initial IM injection, corresponding to the start of the surgical procedure. Data collection may have delayed the start of surgery compared with a clinical private practice setting, putting the surgical incision during a declining phase of anaesthesia with ketamine. It is possible the ketamine dose used in this study might have a shorter duration of action than the alfaxalone dose used. This could explain why >50% of group K cats needed additional ketamine compared with previous studies. This finding could also be caused by the different mode of action of alfaxalone and the ketamine. Ketamine is a dissociative agent which does not provide muscle relaxation, whereas alfaxalone, a gamma-aminobutryic acid A receptor agonist, provides good muscle relaxation. Owing to the constellation of clinical signs, including muscle relaxation, swallowing and the presence of a palpebral reflex, that was used to judge the requirement for top-ups, this could have biased the cats in ketamine group to require more top-ups than the cats in the alfaxalone group, irrespective of relative depth of anaesthesia. It is also possible that the dose of dexmedetomidine in this study was not adequate to provide long enough muscle relaxation in group K and using a higher dose of dexmedetomidine would have obviated the need for further top-ups in the ketamine group.
A previous study using alfaxalone described an unpredictable time of recovery, as well as very light plane of anaesthesia with 5 mg/kg of alfaxalone associated with 10 μg/kg dexmedetomidine with or without 0.1 mg/kg hydromorphone. 18 Other studies using a different dose of alfaxalone IV or IM showed that anaesthesia time was dose relative.12,19 Our study confirmed that surgical depth of anaesthesia could be attained with this IM alfaxalone-based association with the chosen doses.
RR in both groups decreased significantly compared with baseline. Decreased RR and apnoea have been previously described in cats that received a combination of medetomidine and ketamine.5–7 Furthermore, cats from group K showed a lower RR 10 and 15 mins after the IM injection. A time effect with a decrease of RR was also observed from both groups. Earlier studies suggest that alfaxalone has a dose-dependent effect on respiratory depression.12,13,17 However, SpO2 was within the normal range in both groups and the RR remained equivalent and within normal limits during the anaesthesia time before intubation without oxygen supplementation and after intubation when given 1 l/min oxygen. It is therefore possible that the significant difference in RR before intubation might have been an incidental finding.
Recovery quality from alfaxalone has been previously assessed when given IV and IM, with controversial results.12,14,15,18,19,22,27 The recovery was found to be associated with episodes of paddling and trembling. 14 However the quality was described as either good to excellent,12,15,21 or bad quality associated with excitement.14,18,19
After extubation, cats from the group A seemed to have a better-quality recovery than cats that received ketamine, with no alfaxalone cats showing excitement, as has been previously described for IM injection.18,19 A better recovery quality after alfaxalone compared with ketamine has also been shown after both drugs were given IV. 22 After extubation, cats from group A also seemed more willing to walk and were less ataxic, which might be explained by the dissociative effect of ketamine.
During the recovery time, three cats (one from group A and two from group K) needed rescue analgesia. Castration and tattoo were performed by final-year students, making the time of this procedure significantly longer compared with when performed by an experienced veterinary surgeon. Extra rescue analgesia may not be required in a practice setting. The use of perioperative meloxicam could bias the difference between groups; however, the aim of this study was not to assess the analgesia effect of the protocols used.
Conclusions
Both ketamine and alfaxalone combined with dexmedetomidine and butorphanol provided adequate anaesthesia for castration in cats with minimal effects on cardiorespiratory variables for a private practice setting. The combination alfaxalone–dexmedetomidine–butorphanol when given IM seems a suitable anaesthetic protocol for minor surgical procedures lasting <1 h. However, pain on injection of alfaxalone is concerning. Further research is necessary to evaluate the reason and reduce the pain during injection of the drug. 28
Footnotes
Supplementary material
Injection reaction score, sedation score criteria, intubation score criteria, recovery score criteria and 4AVet pain scale.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
