Abstract
Objectives
The objective of this study was to determine if modification of inspired oxygen concentration or positive end-expiratory pressure (PEEP) would alter bronchoalveolar lavage (BAL)-induced changes in pulmonary mechanics or atelectasis, as measured using ventilator-acquired pulmonary mechanics and thoracic CT.
Methods
Six experimentally asthmatic cats underwent anesthesia and non-bronchoscopic BAL, each under four randomized treatment conditions: 100% oxygen, zero PEEP; 30% oxygen, zero PEEP; 100% oxygen, PEEP 2 cmH2O; and 30% oxygen, PEEP 2 cmH2O. Pulse oximetry was used to estimate oxygen saturation (SpO2). Ventilator-acquired pulmonary mechanics and thoracic CT scans were collected prior to BAL and at 1, 5 and 15 mins post-BAL.
Results
While receiving 100% oxygen, no cat had SpO2 <91%. Some cats receiving 30% oxygen had decreased saturation immediately post-BAL (mean ± SD 70.8 ± 31%), but 6/8 of these had SpO2 >90% by 1 min later. There was a significant increase in airway resistance and a decrease in lung compliance following BAL, but there was no significant difference between treatment groups. Cats receiving no PEEP and 30% oxygen conserved better aeration of the lung parenchyma in BAL-sampled areas than those receiving no PEEP and 100% oxygen.
Conclusions and relevance
Alterations in pulmonary mechanics or atelectasis may not be reflected by SpO2 following BAL. The use of 30% inspired oxygen concentration failed to show any significant improvement in pulmonary mechanics but did diminish atelectasis. In some cats, it was also associated with desaturation of hemoglobin. The use of PEEP in this study did not show any effect on our outcome parameters. Further studies using higher PEEP (5–10 cmH2O) and intermediate inspired oxygen concentration (40–60%) are warranted to determine if they would confer clinical benefit in cats undergoing diagnostic BAL.
Introduction
Respiratory diseases represent some of the most common and serious conditions in pet cats. Cats can develop asthma, chronic bronchitis, pneumonia, interstitial lung disease and neoplasia, which impact on both the quality and quantity of life.1–7 Physical examination, blood laboratory values and thoracic radiography are helpful in excluding non-respiratory disorders (eg, heart disease), but they do not provide a ‘cellular’ diagnosis. Indeed, thoracic radiographs are unable to differentiate bacterial pneumonia, some fungal pneumonia, certain types of interstitial lung disease and neoplasia. 8
Collection of samples from the lung by instilling and aspirating saline into and out of the airways of anesthetized cats (ie, bronchoalveolar lavage [BAL]) is considered a more definitive test, allowing microscopic evaluation and culture.9,10 Unfortunately, the procedure dilutes pulmonary surfactant and makes an animal more susceptible to alveolar collapse. This, coupled with retention of saline in alveoli, leads to impaired gas exchange and hypoxemia.11,12 In addition, BAL is performed under anesthesia in veterinary patients, and most anesthetic protocols further compromise gas exchange, clearance of secretions and cardiovascular function. Veterinary patients are typically anesthetized with 100% oxygen, leading to loss of the so-called nitrogen skeleton and making alveolar collapse more likely.13–15 Not surprisingly, BAL and anesthesia may be associated with morbidity and mortality in cats with concurrent respiratory disease.11–15
The objective of this study was to determine if modification of inspired oxygen concentration or positive end-expiratory pressure (PEEP) would alter BAL-induced changes in pulmonary mechanics or atelectasis as measured using ventilator-acquired pulmonary mechanics and thoracic CT. It was hypothesized that the use of PEEP at 2 cmH2O or an inspired oxygen concentration of 30% would improve pulmonary mechanics and decrease evidence of atelectasis compared with no PEEP or 100% O2 in cats undergoing BAL. As feline asthmatics represent a common population receiving diagnostic BAL and as they are at high risk for small airway collapse, experimentally asthmatic cats were used in this study.
Materials and methods
Cats and treatment groups
Six cats with experimentally induced asthma were used in the present investigation. Of the six cats, five were male and one was female. The mean age was 1.8 years; mean body weight was 4.5 kg. Each cat was bred from a high-responder asthmatic cat colony and was screened prior to study for pre-existing sensitization to Bermuda grass allergen (BGA) by intradermal testing and BAL fluid analysis. Induction of asthma was performed using a previously described protocol, with >17% eosinophils required for study enrollment.10,16 After sensitization, the cats continued to receive aerosolized BGA challenges (500 μg/ml) weekly to maintain an asthmatic phenotype. Allergen challenges were also administered 24 h prior to each trial and data collection. This study was approved by the University of Missouri Animal Care and Use Committee (number 6912).
Each cat was given four treatments in random order: (1) 100% oxygen, zero PEEP (ZEEP100); (2) 30% oxygen, zero PEEP (ZEEP30); (3) 100% oxygen, PEEP 2 cmH2O (PEEP100); and (4) 30% oxygen, PEEP 2 cmH2O (PEEP30). There was a minimum of a 2 week washout period between treatments.
Anesthesia and mechanical ventilation
Cats were sedated with 30 mg intravenous (IV) ketamine to facilitate IV catheter placement. Anesthesia was induced with propofol (6 mg/kg IV) and maintained via continuous rate infusion of propofol (0.3 mg/kg/min). If the depth of anesthesia appeared inadequate as judged by animal movement, the presence of palpebral reflex or spontaneous respiration, propofol boluses of 0.5–1.0 mg/kg IV were administered. Orotracheal intubation was performed using standardized cuffed endotracheal tubes (internal diameter 4 mm, length 14 cm).
Following intubation, mechanical ventilation was initiated using a pneumatically driven critical care ventilator (Engstrom Carestation; GE Healthcare) under the assigned treatment conditions. The cats were ventilated in neonatal mode using volume-controlled ventilation. Respiratory rate was set at 10 breaths per minute with an inspiratory to expiratory ratio of approximately 1:3. Tidal volume (TV) was initially set at 10 ml/kg and adjusted at 2.5 ml/kg increments without exceeding 15 ml/kg to ensure an end-tidal partial pressure of carbon dioxide (PCO2) of 35–40 mmHg (Cardell 9405 monitor; Midmark Animal Health). Heart rate and pulse oximetry were continuously recorded throughout the anesthesia. Body temperature and end-tidal PCO2 were recorded prior to and at the conclusion of data collection.
Ventilator-acquired pulmonary mechanics and pulse oximetry
Expiratory and inspiratory breath hold maneuvers were performed at each time point on the ventilator to acquire pulmonary mechanics. The ventilator continuously (breath-by-breath) calculated and recorded airway resistance (Raw) and respiratory system compliance. Compliance was calculated by the ventilator using the following formula: Compliance = TVexpiratory/(plateau pressure [Pplat] – extrinsic PEEP [PEEPe] – intrinsic PEEP [PEEPi]). Data were captured manually and electronically using ventilator software (EView software; GE Healthcare).
Pulse oximetry was used to record oxygen saturation (SpO2) throughout the entire procedure (Cardell 9405 monitor; Midmark Animal Health). The nadir SpO2 was recorded for each cat immediately following BAL. Baseline variables were recorded after 5 mins of ventilation, and additional data was collected at 1 (T1), 5 (T5) and 15 (T15) mins after BAL.
Lavage collection
Following baseline data collection, the endotracheal tube was disconnected from the ventilator and BAL was performed using a non-bronchoscopic technique. Briefly, an 8 Fr red rubber tube was gently advanced down the endotracheal tube until resistance was felt. A 20 ml aliquot of sterile saline was instilled and aspirated. The volume aspirated was recorded. The endotracheal tube was reconnected to the ventilator under the assigned treatment conditions.
CT
Thoracic CT scans were performed at baseline and at T1, T5 and T15. CT scans were obtained using a 64-detector row CT scanner (Aquilion 64; Toshiba America Medical Systems) during inspiratory and expiratory breath holds. Contiguous transverse images were acquired with 0.5 mm collimation, matrix of 512 × 512, pitch of 1.0, small focal spot size, 10–12 cm field of view, 120 kVp and 250 mAs. Images were reconstructed with a high-resolution algorithm and 1 mm-thick sections (no interval). Images were transferred to an Empiric PACS system (Encompass; Fujifilm Medical Systems USA), viewed using an OsiriX workstation (v. 3.9.4, 32 bit; www.osirix-viewer.com) using a lung window (window width 1600; window level −550). A scoring system (0–4) was also used to represent the proportion of each lung lobe occupied by saline: 0 = <5%, 1 = 5–25%, 2 = 25–50%, 3 = 50–75%, 4 = >75%. These scores were assigned to each lung lobe at 1, 5 and 15 mins post-BAL. Regions of interest (ROI) were drawn in the lung parenchyma most affected by the BAL and in the least severely affected area on the same transverse image for each time point post-BAL (Figure 1). Lung attenuation (in Hounsfield units) was measured in the most and least severe ROI.

CT images were acquired from six cats under four randomized treatment conditions at baseline and 1 (T1), 5 (T5) and 15 (T15) mins after bronchoalveolar lavage (BAL). A transverse slice at the level of the caudal and accessory lung lobes was selected for each cat. Two different regions of interest (ROI) were selected from each cross-sectional image post-BAL: the first representing the area of lung least affected, the second representing the area of lung most affected. Representative transverse slices from one cat during the 100% oxygen, zero positive end-expiratory pressure trial at: (a) baseline, (b) T1, (c) T5 and (d) T15. The green outlines in each image represent the least affected ROI (on the left side of each image) and the most severely affected ROI (on the right side of each image)
Statistical analysis
Statistical analysis was performed using commercially available software (Stata; StataCorp). Data were summarized as mean ± SD. Normality was visually inspected using histogram plots of the data, and with the exception of SpO2, the other parameters appeared normally distributed. ANOVA was used to evaluate treatment, time, time by treatment and repeated measures on time. Treatment differences in nadir SpO2 were compared using a Mann–Whitney U-test. A Tukey multiple comparisons procedure was used post-hoc to determine individual differences when appropriate. Total lung lobe scores were analyzed with one-way ANOVA. A P value <0.05 was considered significant.
Results
BAL retrieval
There was no significant difference in BAL fluid retrieval between individual cats or treatment groups. Overall, the mean recovery of saline was 47 ± 12%.
SpO2
SpO2 data for all treatment groups at baseline, T1, T5 and T15 are displayed in Figure 2. At baseline, SpO2 was ⩾94% in all cats for all treatments. Overall, cats treated with 30% oxygen had a significantly lower SpO2 than those treated with 100% oxygen. It is important to note that the observed nadir in SpO2 never decreased below 91% in any cat treated with 100% oxygen. In contrast, four cats each in the ZEEP30 and PEEP30 groups had SpO2 measurements below 90% immediately after BAL. The SpO2 increased to 90% or greater in 6/8 of these cats by the time of the 1 min reading; in the remaining two cats it was >90% by T15. SpO2 nadirs were significantly lower for all cats receiving 30% oxygen (70.8 ± 31.0%) compared with all cats receiving 100% oxygen (97.5 ± 3.2%).
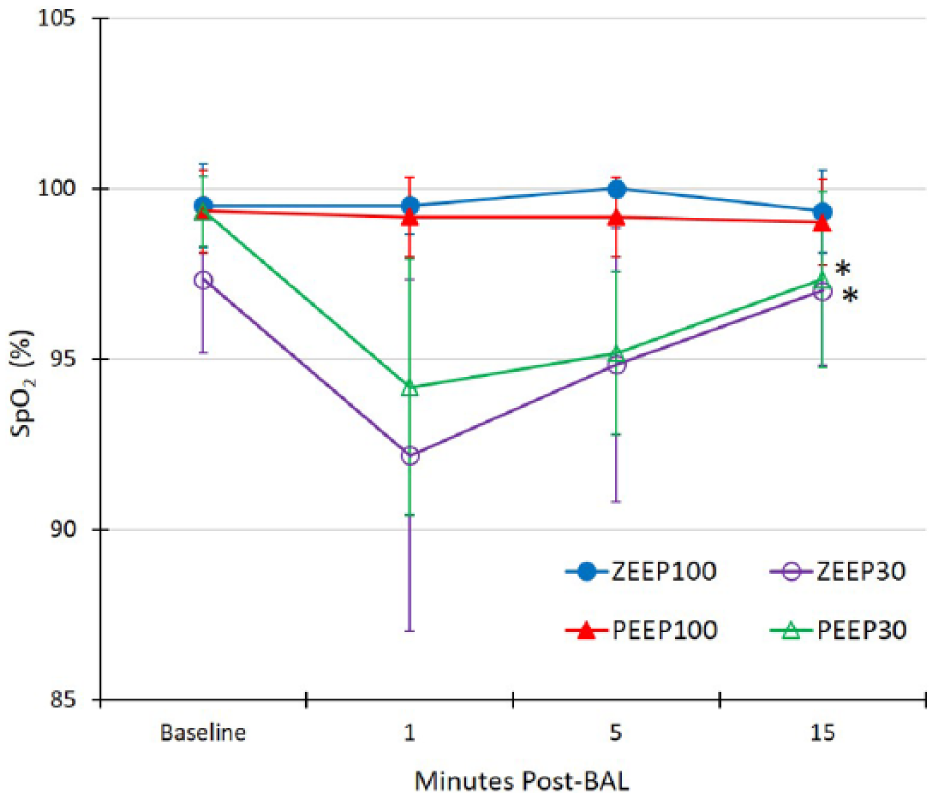
Six experimentally asthmatic cats were randomized to receive each of the following treatments with a 2 week washout in between: 100% oxygen, zero positive end-expiratory pressure (PEEP) (ZEEP100), 30% oxygen, zero PEEP (ZEEP30), 100% oxygen, PEEP 2 cmH2O (PEEP100) and 30% oxygen, PEEP 2 cmH2O (PEEP30). The SpO2 was recorded at each time point. The SpO2 for each time point is given as mean ± SD.
Pulmonary mechanics
Raw was significantly higher after BAL for all treatment groups combined (Figure 3a). Baseline Raw for all groups was 55.2 ± 7.8 cmH2O/l/s. After BAL, Raw increased to 107.4 ± 41.4 cmH2O/l/s. There was no significant difference between treatment groups, nor was there a difference in Raw between 1, 5 or 15 mins for any treatment.

Ventilator-acquired pulmonary mechanics were performed prior (*) to and 1, 5 and 15 mins after bronchoalveolar lavage (BAL) in six anesthetized experimentally asthmatic cats randomized to the aforementioned treatment groups (see Figure 2). (a) The airway resistance (Raw) for each time point is given in this figure as mean ± SD. Post-BAL the mean Raw for all treatment groups was significantly higher vs baseline (*). (b) The compliance for each time point is given in this figure as mean ± SD.
Respiratory system compliance was significantly lower after BAL for all treatment groups combined (Figure 3b). Compliance for all groups was 6.3 ± 1.3 ml/cmH2O pre-BAL and 3.5 ± 1.0 ml/cmH2O post-BAL. There was no significant difference in compliance between treatment groups or between the 1, 5 or 15 min time points.
CT
The BAL resulted in increased lung attenuation manifested by ground glass opacification to complete opacification of affected lung segments or lobes. Affected lung segments or lobes were decreased in volume secondary to atelectasis leading to rightward (20/24) and leftward (4/24) mediastinal shifts. As the TV was kept constant before and after BAL, non-affected lung lobes or segments became relatively hyperinflated, increasing in volume and decreasing in lung attenuation. With respect to distribution of saline in lungs post-BAL, as reflected by the scoring system, there was no significant difference between treatment groups or times. However, more saline was distributed in the right middle, right caudal and accessory lobes compared with the other lobes (mean ± SD, cranial segment of the left cranial lobe 0.5 ± 0.9; caudal segment of the left cranial lobe 0.8 ± 1.2; left caudal 1.4 ± 1.8; right cranial 0.4 ± 0.7; right middle 2.3 ± 1.6; right caudal 2.6 ± 1.9; accessory 2.3 ± 1.8).
Mean attenuation of severely affected ROI is shown in Figure 4. The mean attenuation of the severely affected ROI for all treatment groups was significantly higher than the least affected ROI following BAL. There was no change in attenuation in the severely affected ROI over time (1, 5 and 15 mins) in any treatment group. While treated with ZEEP30, attenuation of pulmonary parenchyma was significantly less than for all other treatment groups. In addition, the attenuation of the ZEEP100 group was significantly higher than for all other treatment groups.
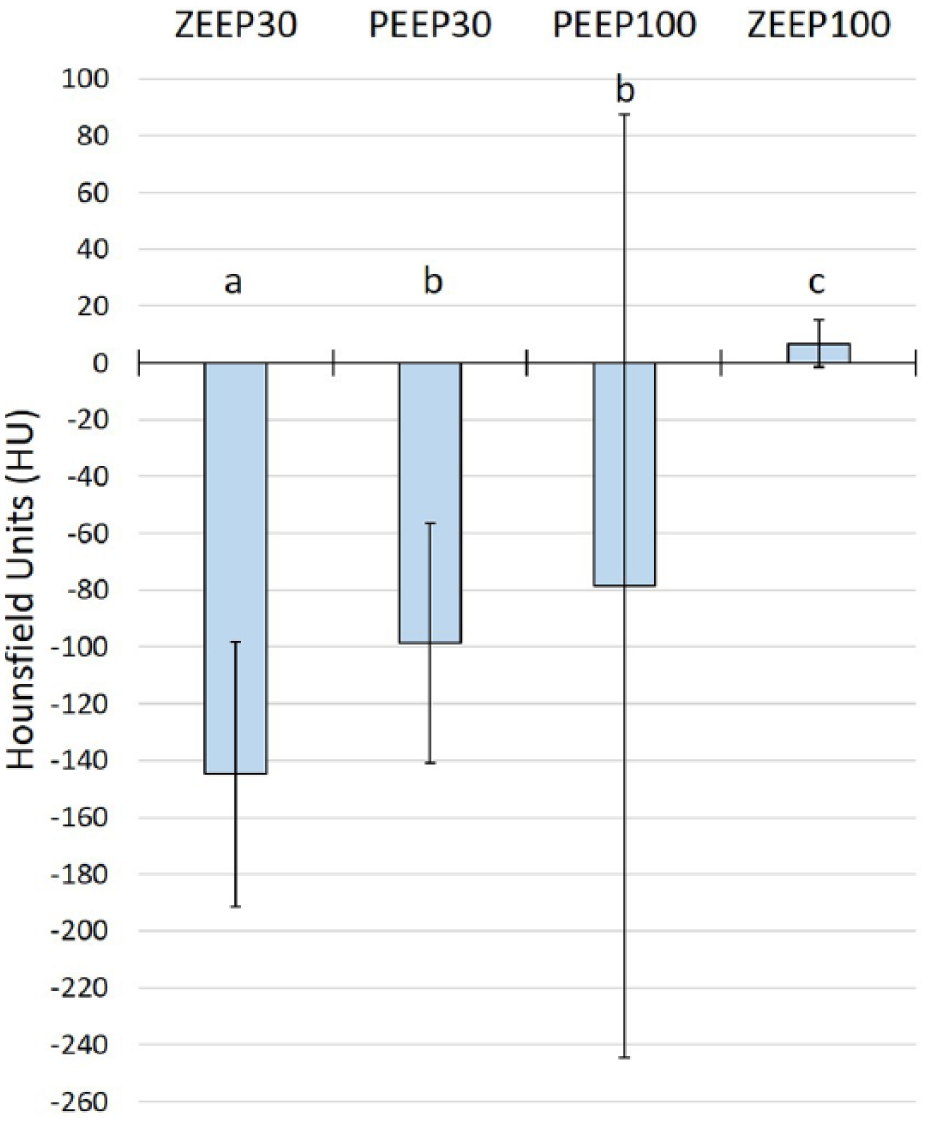
Most and least affected regions of interest (ROI) were chosen from CT scans acquired 1, 5 and 15 mins after bronchoalveolar lavage (BAL) in six anesthetized experimentally asthmatic cats randomized to the aforementioned treatment groups (see Figure 2). Lung attenuation (in Hounsfield units) was measured in each ROI. The mean attenuation post-BAL is given in this figure as mean ± SD, with each letter (a, b and c) indicating a significant difference (P <0.05) from each of the other letters. For all time points, attenuation in the severely affected ROI was significantly higher compared with the least affected ROI (–900 ± 25 HU)
Discussion
The purpose of the present study was to determine if altering inspired oxygen concentration or end-expiratory pressure could modify the adverse effects of anesthesia and BAL on oxygenation, pulmonary mechanics and pulmonary aeration. As expected, BAL negatively affected Raw and compliance, and it increased atelectasis. Modification of PEEP or inspired oxygen concentration did not significantly alter the effects of BAL on pulmonary mechanics. While 30% inspired oxygen concentration decreased the negative effects of BAL on pulmonary atelectasis compared with 100% inspired oxygen concentration, the lower oxygen concentration was associated with further desaturation of hemoglobin. The treatments did not affect the sampling of BAL fluid in terms of percentage retrieved or distribution within the lungs. This study was performed in experimentally asthmatic cats, a population in which BAL is frequently performed and which are prone to small airway collapse post-BAL. Although cats with other respiratory disorders may have different physiologic and anatomic responses to BAL, the results of this study still have important clinical implications.
General anesthesia leads to pulmonary atelectasis by three main mechanisms: (1) decreased functional residual capacity (FRC) due to increased pleural pressure and loss of muscle tone; (2) gas absorption behind collapsed or occluded airways due to decreased FRC and easily diffusible gas; (3) loss of functional surfactant due to lack of spontaneous deep breaths.13,14,17,18 Loss of epithelial integrity and damage to type I and II alveolar cells impairs fluid transport and can result in alveolar space edema. Effects of atelectasis include decrease in pulmonary compliance, impaired oxygenation, lung injury and increased pulmonary vascular resistance. 14
One primary strategy for preventing and reversing atelectasis under anesthesia is the application of PEEP. PEEP prevents the decreased FRC normally seen with anesthesia and minimizes alveolar collapse by applying adequate pressure to keep airways open. 15 The second main strategy for addressing anesthesia-induced atelectasis is use of a lower inspired oxygen concentration, which causes less absorption atelectasis and may also be less cytotoxic. 15
Our hypothesis was that the use of PEEP would improve pulmonary mechanics, as has been shown in anesthetized dogs. 19 As PEEP prevents the loss of FRC associated with anesthesia, it concurrently prevents associated increases in Raw and decreases in compliance. 18 In the current study, we did not see any effect of PEEP on pulmonary mechanics. It is possible that higher PEEP (eg, 5–10 cmH2O) would have elicited an effect. It is also possible that the lack of statistical difference was due to the small sample size. In this study, no alveolar recruitment maneuvers (RMs) were performed. When complete airway collapse has already occurred, PEEP alone may not reverse it; an RM may be performed to restore vital capacity. The addition of PEEP following an RM has been shown to prolong significantly the effects of the RM, even while receiving 100% oxygen.13,15,19,20
Not surprisingly, cats receiving 30% oxygen had a lower nadir SpO2 than those receiving 100% oxygen. Other studies assessing the effect of oxygen concentration under anesthesia have shown that administration of 30–40% oxygen was often associated with lower oxygenation (PaO2 or SpO2) which was short-lived; however, they also documented reduction of atelectasis.21–23 Moreover, a prior study in cats showed impairment of gas exchange with use of 100% oxygen, as evidenced by a lower ratio between PaO2 and FiO2 (PaO2/FiO2), likely due to increased atelectasis. 24 Given the risks for desaturation with 30% inspired oxygen concentrations, future research should address whether intermediate inspired oxygen concentration (eg, 40–60%) might provide the advantage of decreased atelectasis and also prevent desaturation of hemoglobin.
As hypothesized, post-BAL, cats in the ZEEP30 group had less attenuation than both groups receiving 100% oxygen, showing some benefit to a lower inspired oxygen concentrations, consistent with multiple previous studies in animals and people.21–24 The PEEP30 group also had less attenuation than the ZEEP100 group, although it is unclear whether this improvement was due to the lower inspired oxygen, the application of PEEP or a combination of both.
Both PEEP and a lower inspired oxygen concentration were employed in this study in an effort to prevent and treat alveolar collapse under anesthesia. However, it is worth mentioning that neither approach is without possible harmful effects. PEEP may lead to adverse cardiovascular effects and can increase intragastric and intra-abdominal pressure, making regurgitation or vomiting more likely in the perioperative period.13,15 As discussed above, the use of a low oxygen concentration comes with increased risk of hypoxemia.13,15 Equally important to consider is the fact that, in this study, SpO2 did not always reflect pulmonary function or anatomic changes (Figure 5). As such, it is important that the risks of any anesthetic protocol be assessed on a case-by-case basis.
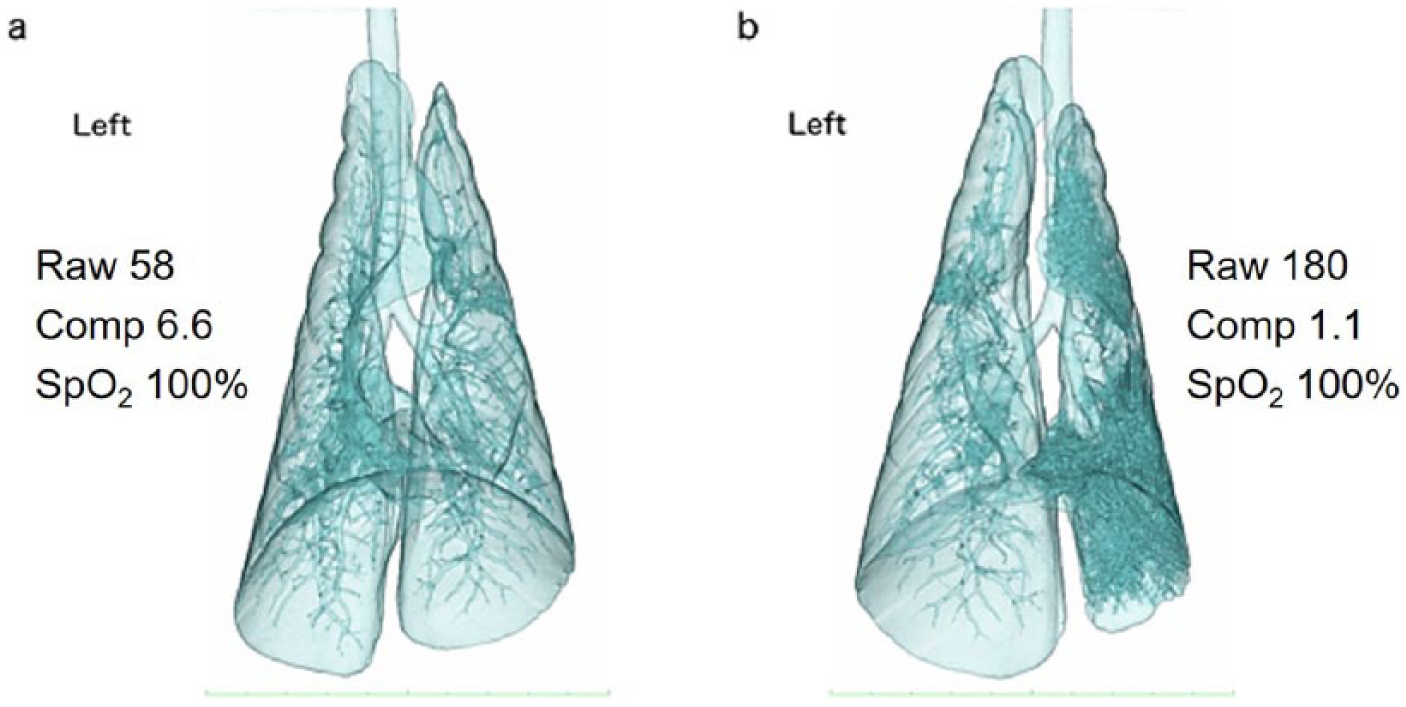
CT images were acquired from six cats under four randomized treatment conditions at baseline and 1, 5 and 15 mins post-bronchoalveolar lavage (BAL). Three-dimensional (3D) reconstructions were created to subjectively assess the atelectasis of the lungs. Both of these 3D reconstructions are from one cat during the ZEEP100 trial: (a) pre-BAL and (b) 1 min post-BAL. There is a noticeable change in lung volume and attenuation following BAL. The Raw and compliance (Comp) that were collected in tandem with the CT are also shown. At the time both of these scans were performed, the cat’s SpO2 was 100%
This study had several limitations. First, the small sample size may have been underpowered for some assessed outcomes. Second, because cats were all ventilated with set TVs, total lung volumes as measured by CT reconstruction could not be used as a measurable outcome parameter. Third, cats did not undergo monitoring for arterial blood pressure or arterial blood gas (ABG) analysis. Monitoring blood pressure may be important for determining the safety of PEEP. ABG analysis would be helpful to better assess patient oxygenation, especially given that pulse oximetry may not be entirely accurate in cats. Fourth, cats were not paralyzed with neuromuscular blockers. Although they were maintained with adequate IV anesthesia and the ventilator was monitored for patient triggering, the possibility of some spontaneous breathing cannot be ruled out. Paralytics are rarely used in feline clinical practice and our study was designed to mimic conditions associated with BAL collection in pet cats. It should be noted that per clinical guidelines in human medicine, maintaining respiratory muscle tone is an important strategy to preventing anesthesia-induced atelectasis. 13 It is important to also note that the application of PEEP or a specific FiO2 cannot be used during bronchoscopy-guided BAL, although these methods could be employed before and after the procedure.
Conclusions
The use of 30% inspired oxygen concentration failed to show any significant improvement in pulmonary mechanics but did diminish atelectasis. Unfortunately, the lower oxygen concentration was also associated with further desaturation of hemoglobin in some cats. The modest levels of PEEP employed were without effect on any of our outcome parameters. Although this study was performed in experimentally asthmatic cats, cats with other respiratory disorders may have similar physiologic and anatomic responses to BAL. Alterations in pulmonary mechanics or atelectasis may not be reflected by SpO2 following BAL. Further studies are warranted to define interventions that will decrease the negative effects of diagnostic BAL and anesthesia.
Footnotes
Acknowledgements
We would like to thank Dr Marie Kerl and Dr Megan Grobman for their support in this project; Jan Kunkel for her assistance with CT; and Dr Patrick Pithua for his help with statistical analysis.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding for this study was provided by discretionary funds by the investigators.
