Abstract
Objectives
This study compared methods of mite retrieval from community cats in the Ohio River Valley region of the USA and determined incidence of parasitic mites in this region.
Methods
In total, 493 community cats were humanely trapped and anesthetized for a trap–neuter–return program. Cats received a dermatologic examination, ear swabs, superficial skin scraping, flea combing, acetate tape preparation and feces collection. All samples were examined microscopically. Large volumes of hair and scale from flea combing were dissolved in 10% potassium hydroxide and centrifuged with Sheather’s solution. Fecal samples were mixed with Sheather’s solution, filtered and centrifuged.
Results
Ear swabs were significantly (P <0.05) better than other methods for finding chigger mites and Otodectes cynotis, and skin scraping was significantly better than ear swabs for finding Cheyletiella species. Only cats with O cynotis had clinical lesions. Mites remained identifiable for 6 months at room temperature. Mite incidence rates were as follows: Notoedres cati (1/493 cats), 0.002 (95% confidence interval [CI] 0–0.006); Lynxacarus radovskyi (2/493 cats), 0.004 (95% CI 0–0.01); Demodex gatoi (5/493 cats), 0.01 (95% CI 0.001–0.019); chigger mites (10/493 cats), 0.02 (95% CI 0.008–0.033); Cheyletiella species (12/493 cats), 0.024 (95% CI 0.011–0.038); and O cynotis (124/493 cats), 0.252 (95% CI 0.213–0.29).
Conclusions and relevance
Ear swabs are recommended when O cynotis or chigger mites are suspected. Skin scraping is more likely to yield positive results than ear swabs, but was not significantly better than acetate tape preparations, flea combing or fecal flotation for finding Cheyletiella species. Mites can remain identifiable for prolonged periods at room temperature. With the exception of O cynotis, the incidence of feline parasitic mites in the Ohio River Valley region of the USA is low; however, D gatoi and L radovskyi were present in the area and should be considered in cats with dermatologic disease attributable to them. In this population of community cats, asymptomatic carriage of mites was common.
Introduction
Domestic cats are affected by various ectoparasites including Cheyletiella species, Notoedres cati, Demodex gatoi, Otodectes cynotis and fleas that can cause significant disease. Fleas are by far the most important ectoparasite of domestic cats worldwide and a great deal is known about how to identify fleas and evidence of flea infestations. There is comparatively little information regarding best practices for cutaneous parasitic mite retrieval and incidence rates in cats. Mite infestation is often suspected or listed as a differential diagnosis in cats with dermatologic disease. Clinical signs are variable and can include pruritus, hair barbering, hypotrichosis, alopecia, excoriations, erythema, otic discharge and pruritus, scaling, crusting, miliary dermatitis and indolent lip ulcers, any of which may vary in intensity and distribution. 1 Asymptomatic carriage is also possible. Owing to this variability in presentation, as well as hypersensitivity responses to very small numbers of mites and the fastidious grooming behaviors of cats, documenting the presence of parasites on examination is frequently elusive.1–3 Clinicians and owners often need to complete parasite treatment trials in order to rule out infestation, which may be time consuming or expensive.1–3 This may be particularly difficult in the case of D gatoi because treatment usually requires 6 weekly lime sulfur dips.1–3 The pertinent scientific literature has shown that contagious parasitic mites can be identified on fecal analysis, and many textbooks and veterinarians recommend examining fecal samples from cats that are suspected of being parasitized.1–8 Silbermayer et al have suggested that fecal analysis may be preferable to skin scraping and acetate tape preparations for finding D gatoi; however, this case report was limited to two cats. 6 Despite these recommendations and findings, to our knowledge there have not been any large-scale studies conducted that have used fecal examination in addition to traditional surface sampling methods (ear swabs, skin scraping, flea combing and acetate tape preparation) as a means of parasitic mite retrieval in the domestic cat.
Owing to the difficulty of finding feline parasitic mites, in particular D gatoi, the incidence of this mite is unknown for most areas of North America. D gatoi is known to occur in the Gulf Coast states (Texas, Louisiana, Mississippi, Alabama and Florida), although it is unlikely that D gatoi is truly limited to these geographical regions. The Ohio River Valley region of the USA is a large area that encompasses parts of Illinois, Indiana, Ohio, Pennsylvania, West Virginia, Virginia, Kentucky and Tennessee, and lies north of the Gulf Coast states. Following discussion with local veterinarians and a few reports of D gatoi outside of the Gulf Coast region, it was hypothesized that D gatoi extends into this area of the USA, affecting cats native to the region. This study sampled community cats from Kentucky and southern Indiana, two states central to the Ohio River Valley region. Information regarding contagious parasite distribution range and prevalence is of use to feline practitioners and clients to determine appropriate differential diagnoses for patients that present with dermatologic signs that may be attributable to parasitic mites.
The American Society for the Prevention of Cruelty to Animals uses the term ‘community cats’ to encompass any unowned cat, including feral cats, lost or abandoned cats and cats that are fed or intermittently cared for by one or more residents in a community. Community cats may act as or be perceived to act as a source of parasitic mites for owned cats. It is expected that community cats would be at a higher risk for parasitism than owned cats and would provide a good population for retrieval of parasitic mites in order to accomplish the goals of this study, including a comparison of mite retrieval methods in cats, as well as determination of the incidence of contagious parasitic mites.
Materials and methods
Animals
Over the course of 14 months (11 collection days) from February 2014 to April 2015, a total of 493 community cats were sampled from Kentucky and southern Indiana after being humanely trapped by members of the community for a trap–neuter–return (TNR) program operating in Louisville, Kentucky. Cats were sampled during all months except May, July and August. Upon admission, cats were assigned an identification number based on order of admission and were housed overnight in individual traps draped with sheets to prevent contact with adjacent cats in a warehouse where surgery was performed the following day. Cats were anesthetized with 0.1 ml/2.27 kg body weight combined intramuscular injection of tiletamine 50 mg/ml, zolazepam 50 mg/ml, ketamine 80 mg/ml and xylazine 20 mg/ml. Isoflurane was available and used as necessary to maintain an adequate plane of anesthesia in each cat. Anesthesia was monitored by trained veterinary technologists and surgeries were performed by licensed veterinarians.
After surgery, cats had a dermatologic examination performed by the primary investigator and samples were collected as follows: both ears were swabbed with separate cotton-tipped applicators moistened with mineral oil; a flea combing of the head, neck, dorsum and lateral trunk using a total of 20 strokes per cat was performed; acetate tape preparations of any scale and lesional sites when present, in addition to difficult-to-groom locations intrascapularly and on top of the head; a superficial skin scraping with a #10 blade scalpel intrascapularly, on the top of the head, as well as at any lesional sites; and fecal collection by gloved digital rectal examination. The left ear tip of each cat was removed as a means of permanently identifying them as altered; vaccines for feline viral rhinotracheitis, calici, panleukopenia and rabies, and subcutaneous fluids, were administered. The cats were wiped with dilute chlorhexidine solution, ears cleaned (EpiOtic Advanced; Virbac) and ivermectin (Ivomec; Merial) applied aurally, and a combination imidacloprid and pyriproxyfen product (Advantage II; Bayer) applied topically on the dorsum. Cats were monitored during recovery, and individually housed and fed overnight at the same facility then released back to their trapping locations the following day. To ensure that cats were never sampled more than once during the study period (eg, if a cat had been trapped twice in the 14 month period) any cats already missing the left ear tip were not examined or sampled.
This study adhered to the principles of good clinical practice as set by the US Department of Agriculture and was accepted by the primary investigator’s animal care committee as an acceptable use of animal models for evaluation. Informed consent was obtained from the executive director of the TNR organization for use of cats in their care.
Sample analysis
Owing to the number of samples collected during each TNR event, it was not feasible to examine most samples on the day of collection. Manigot found that mites remained identifiable for 21 days in mineral oil at room temperature. 9 Throughout this study, positive skin scrapes, ear swabs, fecal flotations and processed flea combings were kept at room temperature and periodically checked over 6 months to see how long mites would remain identifiable.
Samples were mounted onto slides and stored as follows: ear swabs were rolled on microscope slides with the use of additional mineral oil as necessary and a coverslip was sealed in place with quick-dry clear nail varnish applied around the edges as a means of temporary fixation; collections of hair and scale from flea combing were either applied to a microscope slide with mineral oil and a coverslip secured as described if a small amount of sample was collected (98/493 samples) or sealed in plastic Ziploc bags and refrigerated if a large amount was collected (395/493 samples); acetate tape preparations were applied to microscope slides with gentle digital pressure; skin scrapings were placed in mineral oil on microscope slides and coverslips secured as described; and an approximately 1 cm3 sample of feces was placed in a sealed plastic cup and refrigerated.
Large-volume flea comb samples and fecal samples were processed within 24 h of collection. Hair and scale from flea combing was dissolved in 1 ml 10% potassium hydroxide (KOH) in a 12 ml conical centrifuge tube in a warm water bath for 30 mins at which point a commercial Sheather’s solution (sugar dissolved in water to a specific gravity of 1.27) was added to a reverse meniscus and the solution centrifuged at 1500 rpm (444 xg) for 10 mins in a swing bucket centrifuge with a coverslip on the surface. The coverslips were allowed to sit on the tubes for an additional 10 mins after centrifugation before being removed and placed on a microscope slide. The edges were sealed with clear nail varnish. Fecal samples were broken down and mixed with 10 ml of Sheather’s solution using a tongue depressor and then poured through a stainless steel mesh tea strainer and funneled into 12 ml conical centrifuge tubes. Sheather’s solution was added to a reverse meniscus and the solution centrifuged at 1500 rpm (444 xg) for 5 mins with a coverslip on the surface. The coverslips were allowed to sit on the tubes for an additional 10 mins after centrifugation before being removed, placed on a microscope slide and sealed with clear nail varnish.
All acetate tape preparations were examined by the primary investigator within 24 h of collection. All other samples were examined within 1 week of collection. All samples were examined microscopically with a low-power objective lens (× 100 total magnification) with the light condenser turned down, with higher power (× 400 total magnification) used for mite identification as necessary. All parasitic mites (adult and juvenile forms) were recorded. Any mites that were not clearly identifiable were sent to a board-certified veterinary parasitologist for examination (M Dryden).
Statistics
All analyses were performed using SAS V 9.3. Incidence, percentage and exact 95% binomial confidence intervals (CIs) were calculated for each mite and method. Pairwise comparisons of mite counts between methods for each mite type was performed using repeated measures ANOVA with Tukey’s test. In addition, incidences were compared between each method, and fecal incidence by McNemar’s tests. Incidence, percentage and exact 95% binomial CIs were also calculated for each method that found any mite. A logistic regression was fitted by a generalized estimating equation approach to test for differences in incidence between methods. A binomial distribution with a logit link function was used. An independent correlation structure was used to describe correlation between cats. Multiple comparisons were corrected for by Tukey’s test.
Results
In total, 141/493 cats had at least one sample that contained contagious parasitic mites, including N cati (n = 1), Lynxacarus radovskyi (n = 2), D gatoi (n = 5), chigger mites (n = 10), Cheyletiella species (n = 12) and O cynotis (n = 124). Thirteen cats had more than one type of contagious mite found on sampling. The non-contagious mite D cati was found in samples from three cats, and free-living, non-parasitic mites (eg, grain mites, dust mites) were found in samples from 24 cats (Figure 1). In total, 15/24 samples positive for free-living mites were fecal flotations.

Free-living, non-parasitic mite found on fecal flotation in a community cat
N cati was identified on fecal flotation from one individual. This cat also had a chigger mite and O cynotis identified on fecal flotation (Table 1, cat number 87), and no clinical signs of dermatitis. The incidence of N cati was 0.002 (95% CI 0–0.006).
Number(s) of contagious mites (excluding Otodectes cynotis)* found by collection method

Cats are listed in the order in which they were examined. Cats were given identification (ID) numbers upon admission
Data available upon request
FC = flea comb; SS = skin scrape; T = acetate tape preparation; Fe = fecal; E = ear swab; AU = both ears; AD = right ear; AS = left ear
L radovskyi was identified on fecal flotation from two individuals (Figure 2). Neither cat had clinical lesions. The incidence of L radovskyi was 0.004 (95% CI 0–0.01).
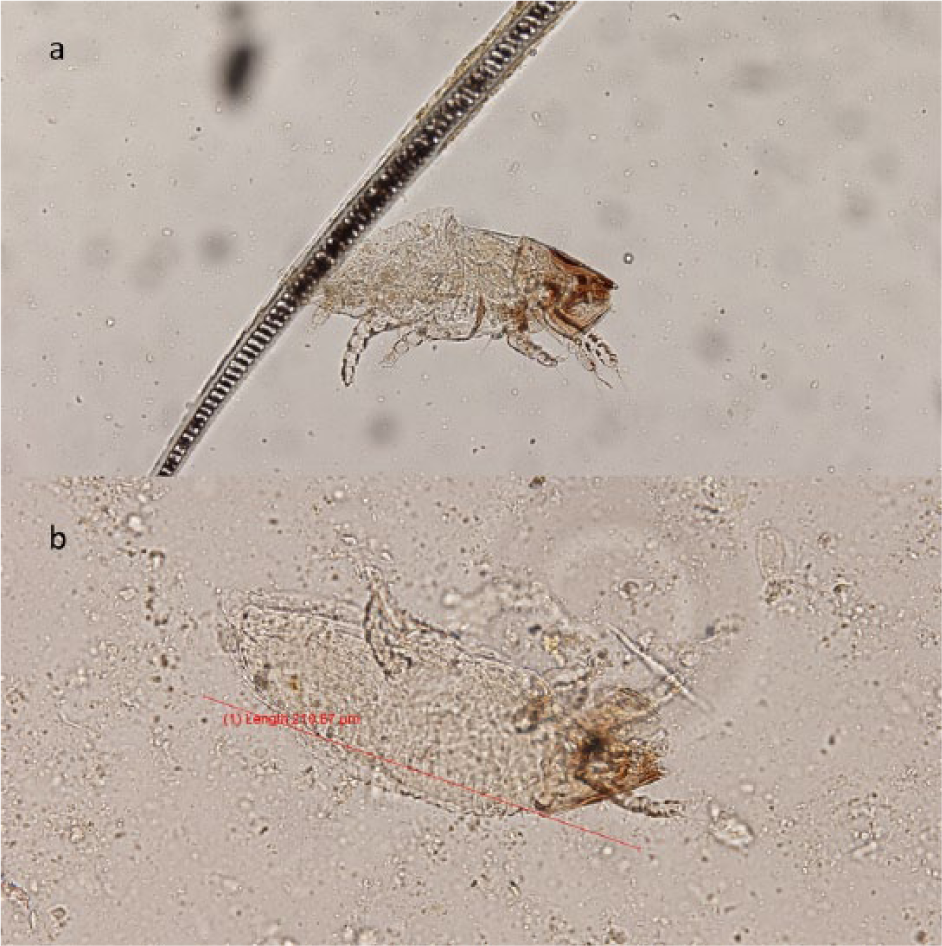
(a) Lynxacarus radovskyi found on fecal flotation in a community cat; (b) length of L radovskyi shown (210.87 μm)
D gatoi was found in fecal flotations, flea combs and skin scraping in five individuals (Figure 3). Skin scraping always produced equal or greater numbers of mites than fecal flotation (Table 1). There was no statistical difference between methods of mite retrieval for D gatoi. Four of the cats with D gatoi had no clinical lesions. One cat had crusting on the head, neck and right pinna; however, O cynotis was also identified in ear swabs from this individual (Table 1, cat number 81). The incidence of D gatoi was 0.01 (95% CI 0.001–0.019).
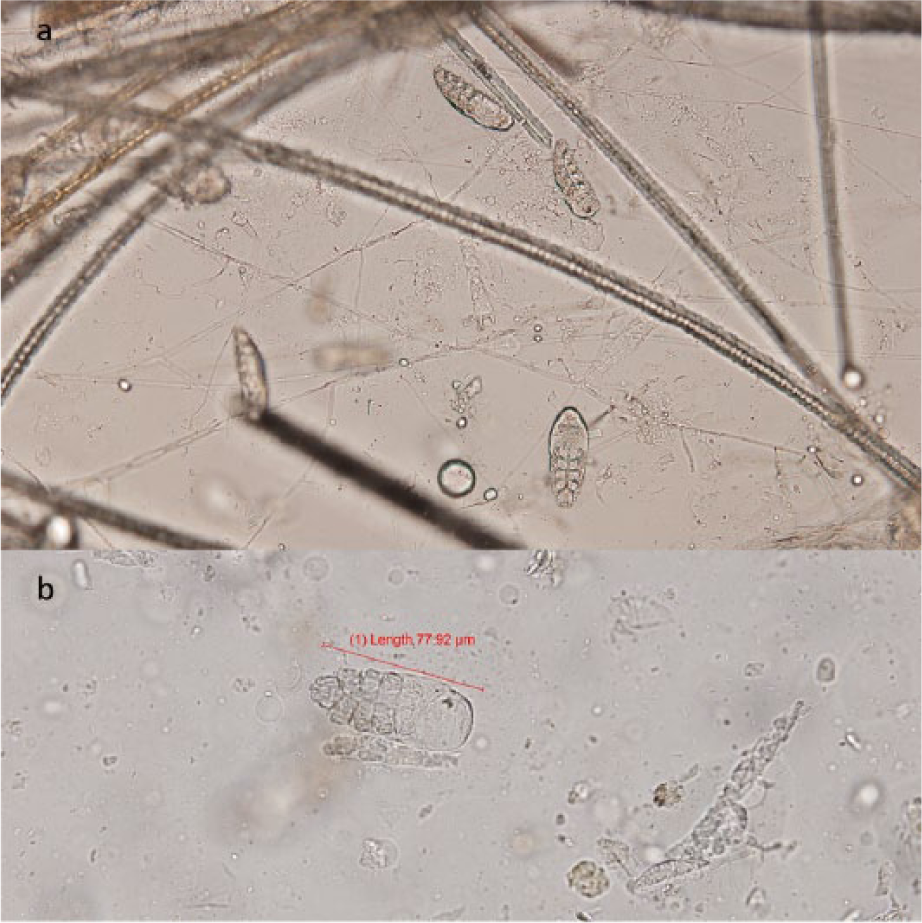
(a) Multiple Demodex gatoi from skin scraping; (b) D gatoi found on fecal flotation (length 77.92 μm)
Chigger mites were identified in ear swabs, fecal flotations and flea combs from 10 cats (Figure 4). Ear swabs were significantly better than all other methods for finding chiggers (P <0.05). None of the cats with chigger mites had clinical lesions. The incidence of chigger mites was 0.02 (95% CI 0.008–0.033).
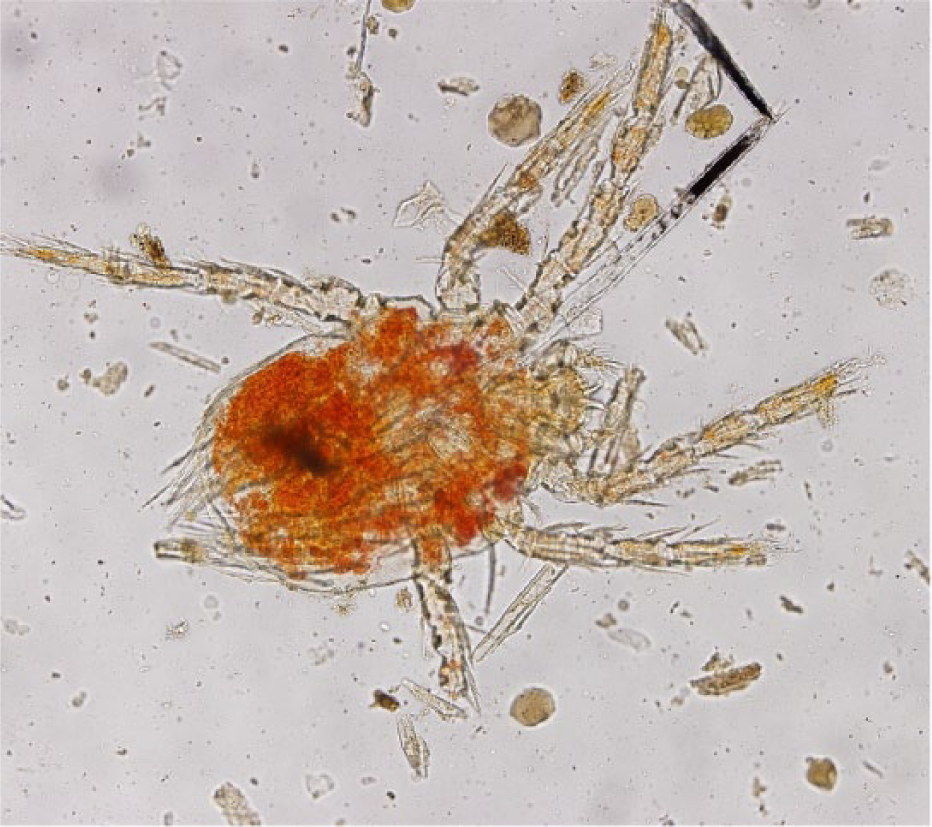
Chigger mite found on fecal flotation from a community cat
Cheyletiella species were found in 12 cats by all sampling techniques except ear swabs, and skin scraping was significantly better (P <0.05) than ear swabs for finding the mite. Four individuals had clinical lesions and all of these had O cynotis identified concurrently (Table 1). Lesions consisted of pinnal thickening, crusting and alopecia, as well as copious dark-to-purulent otic discharge. Cheyletiella species incidence was 0.024 (95% CI 0.011–0.038).
O cynotis was the most commonly identified parasitic mite; it was found in 124 cats by all methods tested. Ear swabs were significantly better than any other method for finding O cynotis (P <0.05). In total, 22/124 cats with O cynotis had clinical signs of otitis externa, including otic discharge, crusting in or around the pinnae, excoriations and alopecia of the pinnae or adjacent skin, and aural hematoma. The incidence of O cynotis was 0.252 (95% CI 0.213-0.290).
D gatoi, O cynotis and chigger mites from skin scrapings, fecal flotations, processed flea combing and ear swabs from multiple cats were kept at room temperature for 6 months during the study period. All mites were still identifiable at the end of the 6 month period.
Discussion
Despite the significant potential for dermatologic disease, there is little information regarding the best practices for retrieval of parasitic mites in domestic cats. In part, this is suspected to be secondary to the fastidious grooming behaviors of cats, in addition to small numbers of mites causing clinical signs in some cats, making it difficult for clinicians to diagnose parasitism without a response to treatment trial.
N cati and L radovskyi were identified, respectively, in one and two cats on fecal analysis but no other samples in this study. The literature states that both of these mites are usually found in abundance on skin scraping or hair examination in cats affected by them.1,10,11 To our knowledge, this is the first report of L radovskyi found on fecal flotation, and the first report of this mite in the study region. L radovskyi is a hair-clasping astigmatid mite of cats that has previously been identified in Texas, Florida and Hawaii in the USA.11,12 None of the cats from which these samples were collected had clinical signs of dermatitis. Cats may groom other cats and kittens that live in their colonies, whether indoor or outdoor, and in the process ingest mites originating from those other cats. Also, cats are carnivorous hunters and if mites are found in fecal samples of cats permitted to hunt, it is possible that those mites came from prey species such as a rabbit in the case of N cati. 1 In this instance, it is unknown if N cati and L radovskyi were causing a pruritic hypersensitivity accounting for single mites found on fecal flotation or if the mites belonged to another affected individual living in close contact or potentially a prey species that was eaten.
In addition to L radovskyi, this is also the first report of D gatoi from Kentucky and southern Indiana, two states central to the Ohio River Valley region of the USA. D gatoi differs from other species of Demodex mites in that it is transmissible between cats via casual contact.2,3,13 Clinical signs vary from asymptomatic carriers of large numbers of mites to severe pruritus with barbered hair, alopecia and occasional miliary dermatitis or indolent lip ulcers due to an apparently small number of mites.1–3 These findings suggest that some cats may develop a hypersensitivity to D gatoi that results in clinical signs. Because D gatoi inhabits the superficial keratin layers of the epidermis, it is relatively easy for cats to remove the mite through grooming. It would be expected that if a cat was infested by D gatoi the mites could be found on fecal analysis after ingestion.1–3,13 Silbermayer et al found higher numbers of D gatoi in fecal samples than skin scraping or cellophane tape preparations from two co-habitating cats parasitized by D gatoi, one with associated pruritus and one without. 6 In contrast, in this study D gatoi was identified in five individuals and the mite was found more frequently and in higher numbers on skin scraping than fecal flotation, and it is unknown whether affected cats were pruritic but only one had clinical lesions that were most likely attributable to concurrent O cynotis. Owners collected fecal samples after treatment with ivermectin in the study by Silbermayer et al, 6 which may have caused mite death and easier removal by grooming contributing to more mites on fecal analysis than in the current study where cats were not treated prior to sample collection. In addition, although both studies used similar flotation solutions and techniques, the sedimentation/flotation technique used by Silbermayer et al was not described in detail and may have differed slightly from the technique used in the current study resulting in possible differences in mite numbers. 6
This study found that ear swabs were significantly more likely to find chigger mites and O cynotis than any other method of sampling. This is not surprising, as the parasitic larvae of chigger mites are found in and around cats’ ears, and the preferred location for O cynotis is the external ear canal and adjacent skin of their hosts.1,12,14 In contrast, the surface-dwelling Cheyletiella species were found in all sampling methods except ear swabs; however, skin scraping was the only method found to be significantly better than ear swabs for the detection of these mites.
Fecal flotation was the most common method by which non-parasitic, free-living mites (grain mites, dust mites) were found (see Figure 1). If a clinician is not confident in the identification of mites, finding free-living mites may lead to an inappropriate diagnosis of mite infestation in patients. Samples should always be examined by a trained individual, and if the identity of a mite is in question, the advice of a specialist should be sought.
Of note in this study is that only cats with O cynotis had clinical signs detected on dermatologic examination. Degree of pruritus was not known for the cats that were sampled, but none displayed clinically apparent lesions due to infestation by mites other than O cynotis. It is possible that these cats were pruritic and if they were more closely monitored this sign would be apparent. Also, cats were examined after having been altered, so female cats had their abdomens shaved and cleaned, which may have removed evidence of barbered hair or crusting on the ventral abdomen.
Seasonality did not appear to affect the number of mites detected (see Table 1); however, cats were not sampled during the months of May, July or August over the study period, so it is possible that during these months a seasonal effect was missed.
In order of increasing frequency, the percentage of cats with samples containing contagious parasitic mites in this study was 0.2% N cati, 0.4% L radovskyi, 1.0% D gatoi, 2.0% chigger mites, 2.4% Cheyletiella species and 25.0% O cynotis. The incidence of O cynotis is comparable with that found by Akucewich et al in Florida (37%) and Thomas et al in Oklahoma (19%).15,16 These previous studies did not include incidences of other mites.
Similar to what was found by Manigot, mites kept in mineral oil preparations can remain identifiable for prolonged periods at room temperature. 9 In addition to samples kept in mineral oil (ear swabs and skin scrapings), this study found that mites in samples from centrifugation with Sheather’s solution (fecal flotation and flea combing with KOH preparation) also remained identifiable for prolonged periods. The significance of this finding is that examination of samples collected from cats does not necessarily need to be performed immediately, provided that a coverslip is secured in place and the samples are kept at room temperature. If clinicians do not have the time to examine skin scrapings fully or other samples in detail at the time of collection they can store them as described in order to evaluate them at a later time. In this study, the use of readily available quick-dry clear nail varnish was used to secure coverslips to slides prior to microscopic examination. The exception to this is acetate tape preparations, which tend to dry out after a short period of time resulting in mite dessication.
Conclusions
If O cynotis or chigger mites are suspected in a cat, ear swabs are the best method for detection of those mites. If Cheyletiella species are suspected, skin scraping is more effective than ear swabs for detection. For all other mites and methods no statistical differences between skin scraping, acetate tape preparation, flea combing, fecal flotation or ear swabs could be determined. With the exception of O cynotis, the incidence of parasitic mites in community cats of the Ohio River Valley region of the USA is low. Clinicians in this region need to be aware that both L radovskyi and D gatoi may be found on their feline patients and they should be considered as differential diagnoses in cats with dermatitis or pruritus that may be attributable to them.
Footnotes
Acknowledgements
We would like to thank Jackie Wells for her help collecting and processing samples, Deborah Keys for her assistance with the statistical analysis, and Alley Cat Advocates for allowing the use of cats under their care for the purposes of this study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received partial funding from the American College of Veterinary Dermatology research grant. Costs not covered by the grant were self-funded.
