Abstract
Objectives
The objective was to determine if decreased platelet function could be detected after treatment with aspirin and/or clopidogrel in healthy cats using three point-of-care platelet function tests that evaluate platelet function by different methods: Multiplate (by impedance), Platelet Function Analyzer 100 (by mechanical aperture closure) and Plateletworks (by platelet counting).
Methods
Thirty-six healthy cats were randomly assigned to receive one of three oral treatments over an 8 day period: (1) aspirin 5 mg q72h; (2) aspirin 20.25 mg q72h; or (3) clopidogrel 18.75 mg q24h. Cats treated with 5 and 20.25 mg aspirin also received clopidogrel on days 4–8. Platelet aggregation in response to adenosine diphosphate and collagen ± arachidonic acid was assessed on days 1 (baseline), 4 and 8. Aspirin and clopidogrel metabolites were measured by high-performance liquid chromatography. Platelet function in response to treatment was analyzed by ANCOVA, linear regression and Spearman correlation.
Results
The only solitary aspirin effect was detected using Plateletworks with collagen in cats treated with 20.25 mg. The only effect detected by Multiplate was using arachidonic acid in cats treated with both aspirin 20.25 mg and clopidogrel. All clopidogrel treatment effects were detected by Platelet Function Analyzer 100, Plateletworks (adenosine diphosphate) and Plateletworks (collagen). Drug metabolites were present in all cats, but concentrations were minimally correlated to platelet function test results.
Conclusions and relevance
Platelet Function Analyzer 100 and Plateletworks using adenosine diphosphate ± collagen agonists may be used to detect decreased platelet function in response to clopidogrel treatment. Either aspirin is not as effective an antiplatelet drug as clopidogrel, or the tests used were not optimal to measure aspirin effect. Cats with heart disease are commonly prescribed antiplatelet drugs to decrease the risk of aortic thromboembolism. Platelet Function Analyzer 100 and Plateletworks may be useful for confirming clopidogrel treatment in these cats.
Introduction
Aortic thromboembolism (ATE) is a complication of heart disease in cats, and is associated with a guarded outcome and high rate of euthanasia.1–5 Finding left atrial enlargement or spontaneous echo contrast in cats with heart disease may prompt antiplatelet therapy. 4 The drugs most commonly used are aspirin and clopidogrel, but the evidence that such therapy decreases platelet function, reduces the risk for ATE or improves long-term prognosis for all cats with heart disease, is based on limited evidence.1,3,4 It has recently been reported that clopidogrel, 18.75 mg once daily, is more effective than aspirin, 81 mg every 3 days, in preventing recurrent ATE, 6 but recurrence of ATE may be seen with both drugs, and the benefits of aspirin and clopidogrel and optimal dosing regimens in preventing a first ATE event are not known.4,6
Monitoring the hemostatic effect of antiplatelet drugs was historically not routinely performed in human or veterinary patients because optical aggregometry using platelet-rich plasma and template bleeding time are too cumbersome and operator-dependent for clinical application.7–9 Several user-friendly, minimally operator-dependent, point-of-care tests for monitoring antiplatelet therapy have become available.7,8 These tests are being evaluated in humans to allow individual dose adjustment and identification of individuals resistant to therapy.7,8
A long-term objective of our laboratory is point-of-care monitoring of antiplatelet therapy of cats at risk for ATE in an effort to optimize therapy. We previously reported institutional reference intervals for three whole blood point-of-care platelet function tests that measure platelet function by different methods: Multiplate (Diapharma), by impedance aggregometry; Platelet Function Analyzer 100 (PFA-100; Siemens), by mechanical aperture closure that evaluates platelet activation under conditions of high shear stress; and Plateletworks (Helena), by platelet counting. 10 The objective of the present study was to determine if treatment of healthy cats with aspirin, clopidogrel or a combination of the two drugs causes changes in platelet function measurable with these tests. A secondary objective was to determine if aspirin and clopidogrel plasma drug metabolite concentrations were correlated with these changes. Documenting utility of these tests in healthy cats would form the foundation for their use in cats with heart diseases.
Materials and methods
Animals
Animal use was approved by the University of Guelph Animal Care Committee in accordance with the standards set by the Canadian Council on Animal Care and the Ontario Animals for Research Act (1980). Thirty-six facility-owned, non-pedigreed cats were used. Cats were selected so that age (mean 6 years, range 3–12 years) approximated the demographic mean age at which cats are diagnosed with hypertrophic cardiomyopathy.6,11–15 Health was confirmed by history, physical examination, complete blood count, serum biochemical profile, feline leukemia virus/feline immunodeficiency virus testing and activated clotting time as previously described. 10
Treatment groups
Cats were randomly assigned to one of three treatment groups, with males and females randomized separately to ensure equal sex distribution within each group, using a computerized method (Microsoft Excel 2007). All treatment groups were similar with respect to mean weight and age, as well as age range and male:female ratio (see Table 1 in the supplementary material). Each treatment group contained 12 cats that received one of three treatments during an 8 day period beginning on day 1 after baseline platelet function testing earlier the same day: group 1, aspirin (Aspirin [Bayer], compounded in the Pharmacy, Ontario Veterinary College) 5 mg orally every 3 days (aspirin-5); group 2, aspirin 20.25 mg (1/4 of an 81 mg tablet) orally every 3 days (aspirin-20); group 3, clopidogrel (Plavix; Sanofi-Aventis) 18.75 mg (1/4 of a 75 mg tablet) orally daily. Additionally, group 1 and group 2 cats also received dual-agent therapy with the addition of clopidogrel, 18.75 mg orally daily, on days 4–8. On day 1, group 1 and group 2 aspirin treatments, and group 3 clopidogrel treatments, were given between 11:00 and 15:00 after baseline blood samples had been obtained. On day 4, group 1 and group 2 aspirin treatments were given between 07:00 and 08:00 prior to blood sampling, while clopidogrel treatments were given between 11:00 and 15:00 after blood sampling. All other aspirin and clopidogrel treatments were given between 07:00 and 08:00.
Blood collection and platelet function testing
On days 1 (baseline), 4 and 8, cats were sedated with ketamine (2 mg/kg) and butorphanol (0.2 mg/kg), given intravenously via a saphenous vein, for jugular venepuncture with a 21 G butterfly needle, as previously described. 10 For each test day the order in which cats were tested was randomly determined. Blood was collected into vacuum tubes in the following sequence: 10 1 ml was drawn first as a ‘hemostasis discard’ into a heparin tube (BD Vacutainer 367884) and used for biochemistry profile and drug metabolite assays; 4.0–4.5 ml was collected into a citrate tube (for Multiplate and PFA-100); and 1 ml was collected into an EDTA tube (for complete blood count and Plateletworks). A 6 ml syringe was used to collect 2–3 ml for transfer into Plateletworks agonist tubes (which do not have a vacuum), 10 and any residual blood was used to measure activated clotting time using MAX-ACT tubes. 10 A maximum of 9.5 ml blood was collected on each sampling day.
Platelet function testing was performed with Multiplate, PFA-100 and Plateletworks using the agonists adenosine diphosphate (ADP), collagen (COL) ± arachidonic acid (AA), as previously reported. 10 Citrated samples were kept under continuous rocking agitation (Coulter blood mixer) for 10–15 mins, at which point an aliquot was removed for Multiplate analysis. The Multiplate analyzer performs assays in duplicate, so a single analysis was performed. After initiating Multiplate analysis, PFA-100 analysis of the citrated samples was started. Duplicate (and triplicate) analyses by PFA-100 were performed sequentially rather than at the same time. While Multiplate and PFA-100 analyses were being performed, the EDTA-anticoagulated and Plateletworks ADP and COL samples were transported to the Animal Health Laboratory, University of Guelph. The samples were gently manually rocked during transport.
Platelet counts were obtained from the Plateletworks ADP, Plateletworks COL and EDTA-anticoagulated samples, in that order, by impedance (Abaxis HM-5 analyzer). After completion of impedance-based platelet counts, platelet counts were obtained in the same order by optical methods (ADVIA 2120i analyzer). 10 All impedance platelet counts were obtained in duplicate; single values were obtained for optical platelet counts. 10 All platelet counts were obtained within 10–30 mins of blood collection. Blood smears were made of all EDTA-anticoagulated samples and the feather edge examined for platelet clumping at 10–40× magnification, which was graded as 1 (no or few small aggregates), 2 (several medium-size aggregates) or 3 (numerous large aggregates).
Drug metabolite measurements
Heparinized blood samples were centrifuged for 5000 × g for 8 mins at 4 ºC within 10 mins of blood collection and plasma stored at −80 ºC in 1.2 ml cryogenic vials (Corning). At the completion of the study, frozen samples were shipped to the Clinical Pharmacology Laboratory, North Carolina State University College of Veterinary Medicine, for drug metabolite analysis using high-performance liquid chromatography (HPLC) methods developed in their laboratory (see the materials and methods in the supplementary material). For aspirin-treated cats, the metabolite salicylate was measured. For clopidogrel-treated cats, the inactive carboxylic acid metabolite (SR 26334) was measured. 16
Statistical analysis
Data were analyzed using SAS version 9.1. Normality of data distribution was determined by the Shapiro–Wilk test. As data distribution was not normal for at least one of the parameters being examined, Spearman correlation coefficients were used as follows: (1) to evaluate for effects of body weight or body condition scores on drug metabolite concentrations and platelet function tests results; and (2) to evaluate for effects of drug metabolite concentrations on platelet function test results. Data were logarithmically transformed to improve fit for parametric tests analysis within the SAS mixed procedure model. Specifically: (1) data were analyzed by ANCOVA, using treatment day within each group as the independent variable, platelet function test result as the dependent variable, and age, sex, platelet count and hematocrit as confounding variables; (2) differences in drug metabolite concentrations between groups and days were analyzed by t-test and ANOVA; and (3) linear regression models were used to examine effects of combined salicylate concentrations from groups 1 and 2. Level of significance was set at P <0.05.
Results
Sedation and blood sample collection was uncomplicated for all cats. The combined range of day 1, day 4 and day 8 optical platelet counts in EDTA-anticoagulated samples of all groups was 173–819 × 109/l (reference interval [RI] 93–514 × 109/l), with a mean of 324 × 109/l and median of 340 × 109/l. Two values were excluded from this range: 75 × 109/l on day 4 in group 1, and 142 × 109/l on day 4 in group 3. These values were considered falsely low, which was attributed to likely sampling artifact as the first smear displayed grade 3 clumping and the second smear grade 2–3 clumping. Four other blood smears with grade 3 clumping were associated with platelet counts >200 × 109/l. Two other samples had platelet counts <200 × 109/l, one of which (173 × 109/l) was associated with grade 2 clumping and the other (180 × 109/l) with grade 1 clumping. All other samples had platelet counts >200 × 109/l and grade 1–2 clumping. For each platelet function test and respective agonist, the median, interquartile range and range of values for each treatment group and day are shown in Figures 1–8. Analysis by ANCOVA did not identify any influence of age, sex, platelet count in EDTA-anticoagulated samples (all values included) or hematocrit on treatment effects.

Box and whisker plots of Multiplate (arachidonic acid agonist) arbitrary aggregation units (AU*min) for the three treatment groups on days 1 (baseline), 4 and 8, depicting median, interquartile range and range of values for each group. ∆ Significantly different from baseline (P <0.001). Dashed lines: upper (1290 AU*min) and lower (590 AU*min) limits of reference interval 10

Box and whisker plots of Multiplate (adenosine diphoshpate agonist) arbitrary aggregation units (AU*min) for the three treatment groups on days 1 (baseline), 4 and 8, depicting median, interquartile range and range of values for each group. Dashed lines: upper (1760 AU*min) and lower (110 AU*min) limits of reference interval 10
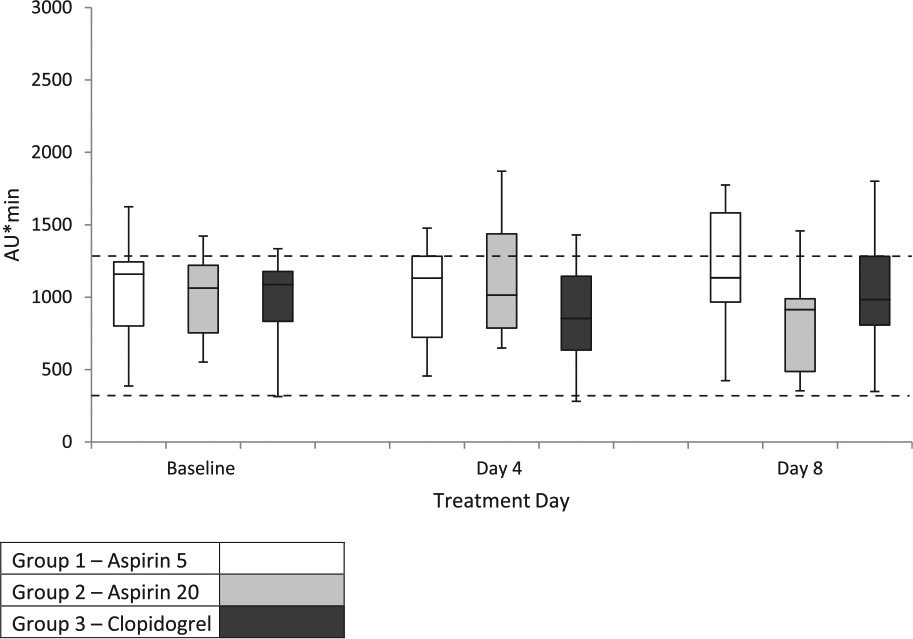
Box and whisker plots of Multiplate (collagen agonist) arbitrary aggregation units (AU*min) for the three treatment groups on days 1 (baseline), 4 and 8, depicting median, interquartile range and range of values for each group. Dashed lines: upper (1290 AU*min) and lower (320 AU*min) limits of reference interval 10

Box and whisker plots of Platelet Function Analyzer 100 (adenosine diphosphate/collagen cartridges) closure times for the three treatment groups on days 1 (baseline), 4 and 8, depicting median, interquartile range and range of values for each group. ∆ Significantly different from baseline (P <0.005 for all comparisons). Dashed lines: upper (89 s) and lower (46 s) limits of reference interval 10

Box and whisker plots of Plateletworks (adenosine diphosphate agonist) percentage aggregation determined by impedance platelet counts for the three treatment groups on days 1 (baseline), 4 and 8, depicting median, interquartile range and range of values for each group. ∆ Significantly different from baseline (P <0.05 for all comparisons). Dashed lines: upper (92%) and lower (18%) limits of reference interval 10

Box and whisker plots of Plateletworks (adenosine diphosphate agonist) percentage aggregation determined by optical platelet counts for the three treatment groups on days 1 (baseline), 4 and 8, depicting median, interquartile range and range of values for each group. ∆ Significantly different from baseline (P <0.001 for all comparisons). Dashed lines: upper (98%) and lower (25%) limits of reference interval 10
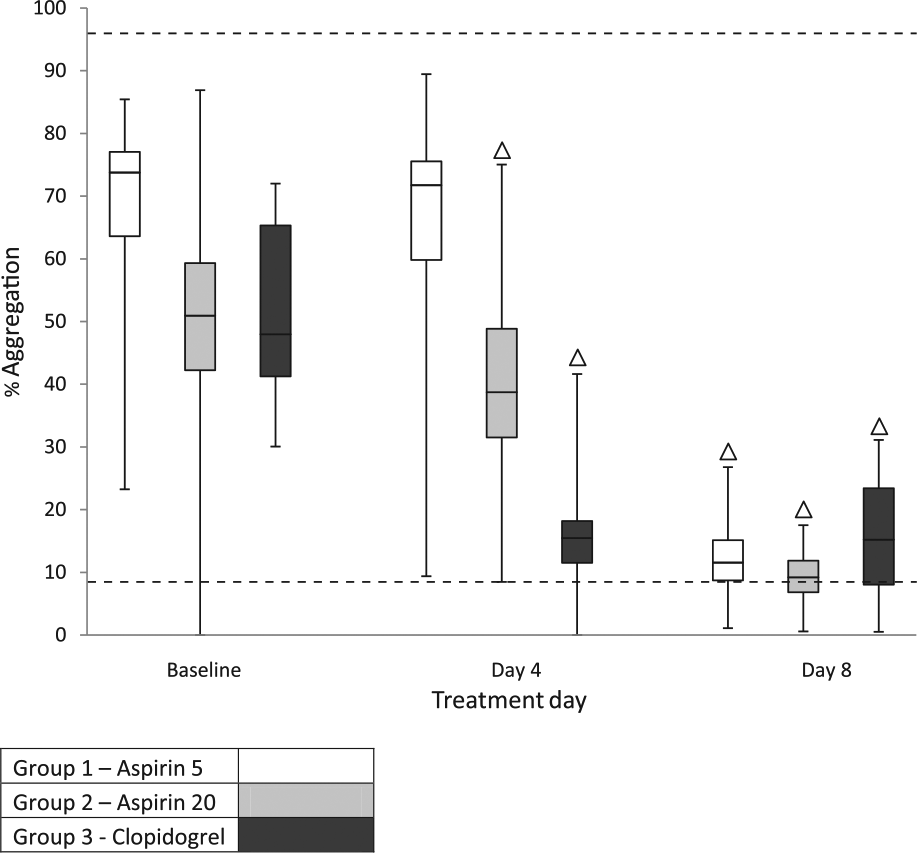
Box and whisker plots of Plateletworks (collagen agonist) percentage aggregation determined by impedance platelet counts for the three treatment groups on days 1 (baseline), 4 and 8, depicting median, interquartile range and range of values for each group. ∆ Significantly different from baseline (P <0.05 for all comparisons). Dashed lines: upper (96%) and lower (9%) limits of reference interval 10

Box and whisker plots of Plateletworks (collagen agonist) percentage aggregation determined by optical platelet counts for the three treatment groups on days 1 (baseline), 4 and 8, depicting median, interquartile range and range of values for each group. ∆ Significantly different from baseline (P <0.001 for all comparisons). Dashed lines: upper (100%) and lower (14%) limits of reference interval 10
For Multiplate, there was a significant decrease in platelet function compared with baseline only when using AA as an agonist in group 2, on day 8, when clopidogrel was administered in addition to aspirin-20 (Figures 1–3). Neither aspirin nor clopidogrel as single agents resulted in decreased platelet function.
For PFA-100, there were significant decreases in platelet function compared with baseline when clopidogrel was used, either alone or in conjunction with aspirin-5 and aspirin-20 (Figure 4). However, although median values were significantly different with clopidogrel therapy, changes in closure time for individual cats varied widely, ranging from no apparent change in some cats to up to two- and three-fold elevations in others (Figure 4). On day 8, closure times were greater than the upper limit of the RI in seven cats in group 1, four cats in group 2 and seven cats in group 3. There were no significant changes in platelet function when aspirin at either dose was used alone. There was no significant difference in platelet function after cats in group 3 had received 8 vs 4 days of therapy with clopidogrel.
For Plateletworks, significant decreases in percentage aggregation, using either ADP or COL agonists, occurred when clopidogrel was used alone or in conjunction with aspirin-5 or aspirin-20, using either impedance or optical platelet counts (Figures 5–8). Percentage aggregation decreased for all individual cats treated with clopidogrel. In group 1 there was a significant increase in platelet function on day 4 using ADP agonist with impedance platelet counts (P = 0.008) but not optical platelet counts (P = 0.144). In group 2, there was a significant decrease in platelet function on day 4 using COL agonist with impedance platelet counts (P = 0.014) but not optical platelet counts (P = 0.986). There were no significant differences in platelet function after cats in group 3 had received 8 vs 4 days of therapy with clopidogrel.
Metabolites of aspirin and clopidogrel were detected in all cats (see Table 2 in the supplementary material). Salicylate concentrations on day 8 were significantly lower than on day 4 in group 1, while the difference approached significance in group 2. Salicylate concentrations were moderately correlated on day 8 with Plateletworks (COL, impedance platelet counts) in group 1 (rho = 0.60, P = 0.04), and with Plateletworks (ADP, impedance platelet counts) in group 2 (rho = 0.58, P = 0.05). Using linear regression, salicylate concentrations weakly predicted PFA-100 results (R2 = 0.02, P = 0.02) and Plateletworks results using both ADP and COL agonists, with both impedance and optical platelet counts (R2 = 0.25–0.29, P = 0.001).
There were no differences in SR 26334 concentrations between day 4 and day 8 in group 3 or between groups on day 8. SR 26334 concentrations were significantly correlated with Plateletworks (ADP) using impedance platelet counts on day 4 (rho = 0.60, P = 0.04) in group 3 and with Multiplate (ADP) on day 8 in group 2 (rho = 0.74, P = 0.02).
Discussion
The objective of this study was to determine if changes in platelet function in healthy cats altered by antiplatelet drugs commonly prescribed to cats at risk for ATE could be detected by three user-friendly point-of-care platelet function tests. The results indicate that: (1) the drugs were absorbed and did decrease platelet function; (2) that PFA-100 and Plateletworks can be used to document clopidogrel effects; (3) that clopidogrel had a stronger antiplatelet effect than aspirin with the tests used; and (4) that there may be a weak dose–response relationship for aspirin with PFA-100, and for aspirin and clopidogrel with Plateletworks.
For Multiplate, the only significant change in platelet function compared with baseline was noted when aspirin-20 was administered in conjunction with clopidogrel on day 8, using AA as the agonist. This overall poor detection of platelet dysfunction by Multiplate was unexpected as platelet aggregometry is historically considered the reference method of platelet function testing, and whole blood aggregometry using a different instrument has been used to document clopidogrel, but not aspirin, effects.17,18 Such results could signify a lack of antiplatelet effects by the drugs; however, given detectable changes in platelet function by other tests in this study, this is unlikely. The use of Multiplate in cats to monitor drug effects has only been reported in one other study. 19 In that study, eptifibatide dramatically decreased platelet function in response to ADP, but abciximab did not. One difference between that study and the present study is that hirudin was used as anticoagulant instead of citrate. Citrate was used in our study to minimize sample volume (as Multiplate and PFA-100 could thus use the same sample), and comparison across tests as all three assess their respective agonists in citrate-based solutions. However, citrate is not an ideal anticoagulant for Multiplate in dogs, 20 and, despite the previous study reporting robust responses to AA, ADP and COL, and generation of RIs for Multiplate in cats, 10 it is likely that citrate is not the optimal anticoagulant for monitoring aspirin and clopidogrel therapy in cats. Other variables to be evaluated and optimized in future studies of Multiplate in cats include sample handling, platelet agonist type and concentration, and assay duration.
The purpose for using different agonists in platelet function studies is to reflect the multiple pathways in which platelets may be activated and that antiplatelet drugs may target. All three agonists used in the present study are effective in inducing platelet aggregation in cats, but AA may be the most sensitive to aspirin inhibition.10,18,21–26 For this reason, AA was included in Multiplate testing, as this could be accomplished using the same sample as for ADP and COL testing. As previously noted, the lack of response to aspirin may reflect poor optimizing of Multiplate (including choice of anticoagulant) or weak aspirin effect. The observation that an aspirin effect was only detected by Multiplate with the higher dose of aspirin in conjunction with clopidogrel using AA may indicate that AA is more sensitive to inhibition and that combined aspirin and clopidogrel therapy had combined antiplatelet effects that just achieved the threshold of Multiplate detection.
Because cats were given a fixed clopidogrel dose of 18.75 mg, regardless of body weight (which is standard clinical practice), 6 clopidogrel metabolite concentrations were examined by correlation with body weight and platelet function test results in an effort to detect dose–response relationships. (An assay for active metabolites was not available at the time the study was performed.) Assuming that SR 26334 concentrations are positively correlated with active metabolites, it is interesting that a correlation between SR 26334 concentrations and Multiplate results were also only seen with combined aspirin-20 and clopidogrel on day 8, where combined antiplatelet effects may have allowed this relationship to be seen.
PFA-100 (ADP/COL cartridges) has previously been reported in normal cats and cats with heart disease.10,27 In neither study were cats treated with antiplatelet drugs. In the present study, clopidogrel caused a significant increase in closure times, indicating that this test may be suitable for monitoring therapy. No significant difference was noted when cats had received clopidogrel for 4 vs 8 days. These findings are consistent with the previous study reporting that clopidogrel at a dose of 18.75 mg once daily is effective as a platelet inhibitor where maximal effect may be achieved by 4 days of therapy. 17 Although clopidogrel significantly increased median closure time, some values in all treatment groups remained within the RIs and clopidogrel effect was not detectable in all cats. It is not known if this is owing to individual cat resistance to clopidogrel, wide confidence intervals associated with the test or lack of sensitivity of the test for clopidogrel effect in some cats. In people, it is well documented that the PFA-100 ADP/COL cartridge does not detect clopidogrel effect in all individuals. This is, at least in part, owing to activation of platelets via the P2Y1 receptor, which has a major role in platelet activation by COL at the high shear rates found in this assay. 28 This receptor is not blocked by clopidogrel, which is selective for the P2Y12 receptor. 28
A difference in platelet function was not detected by PFA-100 when ASA at either dose was used alone. This is consistent with some previous studies where variable inhibition by aspirin to responses to ADP and COL have been seen.18,22–25 This is attributed to ADP and COL having some platelet activation pathways independent of thromboxane A2, varying doses of aspirin and varying strengths of the agonists. The PFA-100 cartridge that contains adrenaline (epinephrine) and COL may be better suited to document the effects of ASA, although in one study aspirin did not inhibit adrenaline-induced aggregation. 22 The additional use of the adrenaline/COL cartridge was precluded by ethical guidelines for the volume of blood that could be acquired from each cat at each time point, and by time constraints for being able to test all samples within a prescribed time frame.
The use of Plateletworks (ADP) has previously been shown to identify clopidogrel effect in healthy cats. 29 Results of the present study were consistent with the findings of a previous report, which used the same dose of clopidogrel. There was less overlap in ranges than with PFA-100, and in all cases individual response to clopidogrel was apparently detected. (For individual cats with low or low–normal baseline values it is possible the change in percentage aggregation could be due to analytic variation.) 10 In the present study, COL agonist was also evaluated. Results with ADP and COL were similar with respect to clopidogrel, except that the overlap of ranges with COL was greater, and using COL instead of, or in addition to ADP, does not appear to offer any advantage for monitoring clopidogrel therapy. Plateletworks did not detect an effect of aspirin-5 but did detect an effect of aspirin-20, using impedance platelet counts. This may be owing to the higher dose of aspirin. However, the large overlap in ranges indicates that in most cases an individual cat’s response would not be detectable. As with PFA-100, the limitations of Plateletworks to detect aspirin effects may be partly due to the use of ADP and COL agonists. An AA agonist tube is available for Plateletworks but was not used because of the aforementioned blood volume and time-to-analysis constraints.
The previous Plateletworks study evaluated sample holding time prior to analysis, concluding that ADP-induced aggregation was minimally affected until 30 mins after blood collection. 29 In contrast, the Plateletworks manufacturer recommends that human samples be analyzed within 10 mins. In the current study, the ADP tubes were analyzed first, followed by the COL tubes. This resulted in the COL tubes being analyzed within 20–30 mins, and the effect of this delay on COL-induced aggregation is not known.
The previous Plateletworks study measuring clopidogrel effect used impedance platelet counts, for which the assay is validated. 29 In the previous report from our laboratory, it was concluded that Plateletworks could also be used with optical platelet counts. 10 The present study further demonstrates that decreased platelet function in healthy cats administered clopidogrel alone or in conjunction with aspirin can also be detected with optical platelet counts using the ADVIA 2120i analyzer. Although most Plateletworks findings were similar when percentage aggregation was calculated using the two platelet counting methods, two results were different. First, on day 4, group 1 (aspirin-5) impedance-based percentage aggregation actually increased, while optically based percentage aggregation did not. This may be due to random error associated with wide confidence intervals, or reflect that optically based methods may be more accurate than impedance-based methods for feline platelet counting. 30 Second, when assessed by impedance, a significant decrease in platelet function was noted with Plateletworks (COL) when cats were administered aspirin-20. However, the trend with the optical values was similar and would likely have become significant with a larger number of cats; and, as previously noted, the magnitude of the decrease in platelet function was not great enough to make Plateletworks (COL) useful for monitoring aspirin therapy.
As previously noted, because the drugs were being given orally, drug metabolite concentrations were measured primarily to confirm drug absorption. This proved to be particularly important with aspirin, as minimal antiplatelet effects were seen. The aspirin-20 treatment resulted in an average aspirin dose of 3 mg/kg and day 4 salicylate concentrations of 14.51 ± 6.65 µg/ml, while the aspirin-5 treatment resulted in an average aspirin dose of 0.9 mg/kg and significantly lower day 4 salicylate concentrations of 4.02 ± 1.05 µg/ml (see Table 2 in the supplementary material). These findings are consistent with previous pharmacokinetic reports of aspirin in cats.24,25,31 The lowest aspirin dose for which salicylate concentrations have been previously reported is 10 mg/kg, resulting in peak salicylate concentrations ranging from 30–42 µg/ml after the first dose to 60–100 µg/ml after daily repeated treatments.25,31 Salicylate concentrations on day 4 in the present study were obtained 3–6 h after aspirin administration, at which time they would be slightly reduced from peak concentrations.25,31
Drug metabolite concentrations were also examined in an effort to identify dose–response relationships to platelet function test results, especially as fixed doses were given. It is important to note in this context that the most active drug against platelet function is aspirin, not salicylate. Plasma salicylate concentrations result from both acetylation of cyclooxygenase and hydrolysis of aspirin by the liver and other tissues. This occurs rapidly such that aspirin is often not detectable at the time of peak salicylate concentrations. 25 Nonetheless, plasma salicylate concentrations are dependent on aspirin concentrations; therefore, a dose–response relationship to platelet function testing may still exist. In a previous study, aspirin doses of 10 mg/kg, resulting in peak salicylate concentrations of 30–42 µg/ml, caused reduced aggregation in response to COL in 2/10 cats, while doses of 25 mg/kg, resulting in peak salicylate concentrations of 50–70 µg/ml, caused reduced aggregation in response to COL in 4/10 cats. 25 Neither dose affected aggregation in response to ADP or thrombin. In an earlier study, a dose of 25 mg/kg resulted in much higher peak salicylate concentrations of 250–300 µg/ml, but an assay other than HPLC was probably used and no platelet function tests were performed. 31 However, when salicylate concentrations of 200–300 µg/ml were achieved with a much higher single dose of aspirin (approximately 160 mg/kg), ADP-induced platelet aggregation was markedly decreased, suggesting a dose effect. 24 In the present study, weak, but significant, aspirin dose effects were noted with PFA-100 and Plateletworks. The effect on PFA-100 was much weaker than on Plateletworks, likely reflecting the lower sensitivity of PFA-100. The antiplatelet doses recommended for cats include doses ranging from 5 mg/cat to >40 mg/kg every 3 days, as well as doses between these two values given every 2 days to twice a week.3,6,18,22,23 Depending on the platelet function test used, there is some evidence that higher doses do cause greater platelet dysfunction; however, in the single, retrospective, study comparing aspirin doses in cats with heart disease, there was no difference between survival or recurrence of ATE between low and high doses. 3
Similarly to aspirin, clopidogrel plasma concentrations were inferred through the measurement of the inactive carboxylic acid metabolite SR 26334. 16 Although PFA-100 and Plateletworks showed decreased platelet function in response to clopidogrel, this was not reflected by correlation between magnitude of change and SR 26334 concentrations, except with Plateletworks (ADP) using impedance platelet counts on day 4 in group 3, and with Multiplate (ADP) on day 8 in group 2. This may be because SR 26334 is not a good surrogate with respect to clopidogrel’s antiplatelet properties. It is also possible that the clopidogrel dose was sufficiently high such that all cats achieved near maximum possible platelet inhibition, regardless of individual variability in absorption and metabolism. Previous studies of clopidogrel in cats have not measured plasma SR 26334 concentrations.
In the present study, clopidogrel resulted in more effective platelet inhibition than two ‘low-dose’ aspirin treatments. This may indicate that clopidogrel is a more effective antiplatelet drug than aspirin. It is also possible, however, that evaluation of aspirin was not ‘equivalent’ to that of clopidogrel, as platelet function tests using AA and adrenaline were not used. However, ultimately, the value of a platelet function test has to be proven by correlation to clinical outcome, and the results of a trial comparing clopidogrel (18.75 mg q24h) and aspirin (81 mg q72h) for the prevention of recurrent ATE are relevant in this context, as recurrence is significantly lower and survival significantly higher in the clopidogrel group. 6
Because aspirin and clopidogrel inhibit different platelet activation pathways, combination therapy may be used in an effort to increase effect. 4 An additive or synergistic effect was not documented in the present study. This may either be because no such effect was present in vivo or that the detectable antiplatelet effect in the tests used was already at maximum from clopidogrel.
A limitation of the study is that echocardiograms were not performed to rule out subclinical cardiac disorders. Cardiomyopathy did not affect PFA-100 results in one study, 28 but it is not known if platelet response to aspirin or clopidogrel is different in cats with cardiomyopathy compared with normal cats. Results of Multiplate or Plateletworks in cats with cardiomyopathy have not been reported; however, cats with mild cardiomyopathy are unlikely to have platelet dysfunction. 32 The prevalence of moderate-to-severe subclinical cardiomyopathy was reported to be <4% in one study and similarly low in another.33,34 Based on these reports, there was a risk of including, at most, two cats with moderate-to-severe cardiomyopathy, which would probably have had minimal effect on results.
Another limitation is that the sequence of filling of blood tubes was not randomized, which may have introduced bias in the test results. Given the numerous potential sources of preanalytic variation with platelet function testing, minimizing preanalytic variation by strict procedure and having potential biases was considered preferable to attempting to control for such variation by randomization. The sequence of collection into discard tube, citrate tube and then EDTA tube was based on the manufacturer’s recommendation when using Vacutainer brand tubes with butterfly needles. As the Plateletworks tubes do not contain a vacuum, once vacuum-tube draws were completed, the needle adapter was rapidly removed and a syringe used for collection; this latter method was considered preferable to performing a second venepuncture.
A further limitation is that, despite minimally traumatic venepuncture and obtaining a ‘discard’ prior to collecting the sample into EDTA in an effort to minimize platelet clumping during blood collection, some clumping occurred. In our previous report, marked platelet clumping on blood smear evaluation was only present in 4/55 samples, and only one of these samples had a platelet count below the RI. 10 The results of the current study are similar, with marked platelet clumping in 4/108 samples, of which one platelet count was below the RI. In neither study did baseline count affect platelet function test results. Platelet clumping could affect platelet function test results both by lowering platelet count and possibly by reflecting that activation of non-aggregated platelets has occurred. Platelet clumping is potentially of greatest concern with Plateletworks, which relies on platelet counting. Specifically, falsely low baseline platelet counts would underestimate percentage aggregation. However, Plateletworks is relatively ‘resistant’ to this effect. If one examines, for example, the effect of lowering the EDTA platelet count progressively from 500 × 109/l to 100 × 109/l, with a postagonist platelet count of 10 × 109/l, percentage aggregation only drops from 98% to 90%. The lower the baseline EDTA platelet count, the greater is the effect of it being falsely low. In this study, platelet counts were sufficiently high, and agonist-induced platelet aggregation sufficiently strong (especially with ADP), to minimize the effects of overall minimal–mild platelet clumping. Also, the higher the postagonist platelet count (reflecting platelet inhibition in Plateletworks), the greater is the effect of a falsely low baseline platelet count. It is therefore possible that the decrease in percentage aggregation seen after antiplatelet therapy would be somewhat enhanced by platelet clumping in baseline samples. However, this would affect both aspirin and clopidogrel, so does not detract from the observation that Plateletworks (ADP or COL) is superior for detecting clopidogrel effect and/or that clopidogrel is a more potent platelet inhibitor. It is likely that some platelet clumping occurs below the level of detection by optical microscopy as used herein, and this may contribute to the relatively wide confidence intervals of Plateletworks and the other two tests. 10 The effects of platelet clumping on results in other studies of these tests in cats were not reported. However, in a previous report, most platelet clumping was associated with normal platelet counts, consistent with the observation in this report that a numerical effect on platelet function tests was not observed. 35
A final limitation is in regard to sample handling. Samples were continuously rocked until analysis. This was undertaken for logistical reasons to standardize sample handling and permit testing of multiple cats in one day, as well as comparison of results to our previous study. Because samples were being analyzed promptly, we considered gentle continuous rocking to, potentially, cause less platelet activation than re-suspension after a brief rest period, and this was consistent with results of pilot studies using the PFA-100, where continuous agitation resulted in fewer flow obstructions (data not shown). Multiplate analysis was performed sooner than the manufacturer’s recommendation for people of waiting 30 mins, although earlier analysis is valid.19,36 The manufacturers’ recommendations for sample handling for the three tests are for people, and optimal sample handling for cats is not known.
Conclusions
Platelet inhibition by clopidogrel was not detected by Multiplate using citrate anticoagulant and ADP, COL and AA at the specified concentrations; was detected in some cats by PFA-100 using ADP/COL cartridges; and was potentially detected in all cats by Plateletworks using ADP or COL tubes. None of the tests detected platelet inhibition by aspirin. For the methods used in this study, Plateletworks using ADP or COL tubes may be recommended to confirm clopidogrel administration in cats.
Footnotes
Acknowledgements
We would like to thank, from the University of Guelph, Dr William Sears, Department of Population Medicine, and Gabrielle Monteith, Department of Clinical Studies, for assistance with study design and statistical analysis; and the veterinary and laboratory technicians, Health Sciences Centre, Department of Clinical Studies, and the Clinical Pathology Laboratory, Animal Health Laboratory, for assistance with blood collection and platelet function testing. We would also like to thank Delta Dise, Clinical Pharmacology Laboratory, North Carolina State University, for performing drug metabolite assays as well as Dr Howard Dobson, Christina de Rivera, Jennifer Brandow, Melissa Brooks and the staff at Vivocore Inc for management and care of the cats.
Supplementary material
The following files are available: Supplementary Table 1: Age, sex and weight of cats Supplementary materials and methods: Methods for drug metabolite analysis using high-performance liquid chromatography developed at the Clinical Pharmacology Laboratory, North Carolina State University College of Veterinary Medicine Supplementary Table 2: Aspirin metabolite (salicylate) and clopidogrel metabolite (SR 26334) concentrations determined by high-performance liquid chromatography
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by a grant from OVC Pet Trust.
