Abstract
Objectives
The aim of this study was to compare the sensitivity of acetate tape impression with skin squeezing and superficial skin scraping methods for the diagnosis of burrowing mites (Notoedres cati) in cats.
Methods
Samples were collected from 50 cats showing signs of notoedric mange. The most affected region was selected for sampling using acetate tape impression with skin squeezing, and superficial skin scraping.
Results
No significant difference was observed in the number of mites found by both methods, regardless of the stage of development of the mite. The tests showed a high correlation to the total number of mites (r = 0.928). However, in two animals it was possible to confirm the presence of N cati only by the acetate tape impression test.
Conclusions and relevance
We conclude that the acetate tape impression test is a good method for confirming the presence of N cati in cats. In our experience, this technique is as sensitive as superficial skin scraping, even in animals presenting with a low infestation. Moreover, the technique is less traumatic and allows the collection of clinical specimens in more sensitive locations, such as the eyelids, lips and paws.
Introduction
Notoedric mange is a skin disease caused by the obligate ectoparasite Notoedres cati. Notoedres mites are smaller than Sarcoptes species, demonstrating ‘thumb print’-like dorsal striations, shorter limb stalks and a dorsal anus. 1 These mites, unlike canine Sarcoptes species mites, are found in large numbers in cats and are easily diagnosed. 2 Although notoedric mange is considered a rare disease, 3 it is common in Brazil, Greece and Iran.4–6 It is highly contagious through direct skin contact,1,7 and has been reported in a range of mammals,8,9 including mostly wild and domestic felids.10–12
Notoedres cati mites excavate burrows into stratum corneum of the epidermis, where eggs are deposited by females. These burrows damage keratinocytes, leading to cytokine release (especially interleukin-1), causing cutaneous inflammation, 13 acanthosis and intense pruritus. The most common clinical sign is hyperkeratosis with gray crusts and scale associated with intense itching of the pinnae, face and head. 13 In chronic cases self-mutilation occurs leading to excoriation, scaling and lichenification, which can deform the face of the animal and cause anorexia, emaciation and, in rare cases, death.10,14
The diagnosis of notoedric mange is performed based on the host that is involved, clinical signs and parasitological examination. 1 The last of these aims to investigate the presence of mites in their adult or immature form. Superficial skin scrapings are the most commonly used laboratory diagnostic method, owing to their effectiveness, low cost and the location of the mites (ie, stratum corneum of the epidermis). 7 However, this methodology is traumatic and can be painful, requiring physical restraint in uncooperative or aggressive animals. 15 Another diagnostic technique, such as the use of acetate tape impression (tape strip test), has been used for the diagnosis of fungi, bacteria and non-burrowing mites such as Cheyletiella blakei. In this technique a strip of transparent adhesive tape is pressed onto the suspected area of the skin to capture some specimens of mites and yeast. 7 The acetate tape impression test with skin squeezing has also been proved to be effective for the diagnosis of Demodex canis and Sarcoptes scabiei in dogs.16,17 However, there are still no studies proving the effectiveness of acetate tape impression test for the diagnosis of N cati in cats.
Therefore, the aim of this study was to compare the accuracy of the acetate tape impression test with skin squeezing with superficial skin scraping for the diagnosis of notoedric mange in cats.
Materials and methods
This study followed the rules of experimentation of the Brazilian College of Experimentation (COBEA) and was approved by Ceará State University’s ethics committee on the use of animals (3383876/2014). Fifty crossbreed cats, both male and female, aged between 3 months and 4 years, presenting with clinical signs of notoedric mange, were included in this study. The animals were subjected to physical and dermatologic examination, and the data were catalogued in a clinical and epidemiological form. Lesions were classified according to the following score system for severity of notoedric lesions: 18 0 = healthy skin demonstrating no lesions, no alopecia, no scratching; 1 = mild skin lesions, mild alopecia, occasional scratching or lesions locally limited to the ears; 2 = moderate skin lesions, moderate alopecia, intensive scratching, wounds secondary to scratching; 3 = severe skin lesions, lesions affecting more than head region, severe alopecia, thick/crusty and scabby appearance of the skin, intensive scratching, inducing to self-trauma injury (Figure 1).

Dermatologic lesions caused by notoedric mange. (a) Score 1: alopecia, lichenification and erythema localized to the ear region; (b) score 2: moderate alopecia and presence of scratching wounds localized to the ear region; (c) score 3: alopecia associated with crusty appearance of the skin of the ear and face of the cat
The most affected region was selected for sampling using acetate tape impression with skin squeezing, and superficial skin scraping from the same lesion, on adjacent sites. For the acetate tape impression, a 4 cm piece of transparent tape was placed on the selected lesion and then the skin was squeezed. The tape strip was removed and placed directly on a microscope slide. For the skin scrapings, the hair of the cats was removed over the sampling areas, and skin scrapings were made with a 2 cm scalpel blade. The material collected was transferred to a microscope slide and immediately clarified in 10% potassium hydroxide (KOH). Both slides were analyzed on a light microscope at × 10 magnification for quantification of eggs, larvae, and nymphs or adult forms of N cati. All samples were evaluated within 8 h of collection. All cats were treated with two doses of ivermectin 200 µg/kg, subcutaneously, every 10 days.
Statistical analyses were performed using Graphpad Prism 6. Results were compared using a paired t-test; significance was set at P <0.05. The Pearson correlation coefficient test was used to determine the correlation between the two tests. Bland–Altman analysis was performed to evaluate the agreement between both measurements, which was based on the average of the differences (bias) in the total number of mites found in each test.
Results
On clinical examination it was observed that 44% (22/50) of the cats presented with score 1, 32% (16/50) score 2 and 24% (12/50) score 3. The parasites were classified based on morphologic and morphometric characteristics according to Scott et al. 1 All life stages of notoedric mites were observed in the analyzed samples; however, in two animals it was possible to verify the presence of N cati only by the acetate tape impression test (see Table 1 in the supplementary material). The diagnosis was carried out more easily with the tape impression, as fewer artifacts such as scabs, debris and blood cells were observed (Figure 2). Furthermore, the identification of the adult form by tape impression was easier as most of them were still alive and moving during the evaluation. All cats were successfully treated with ivermectin.
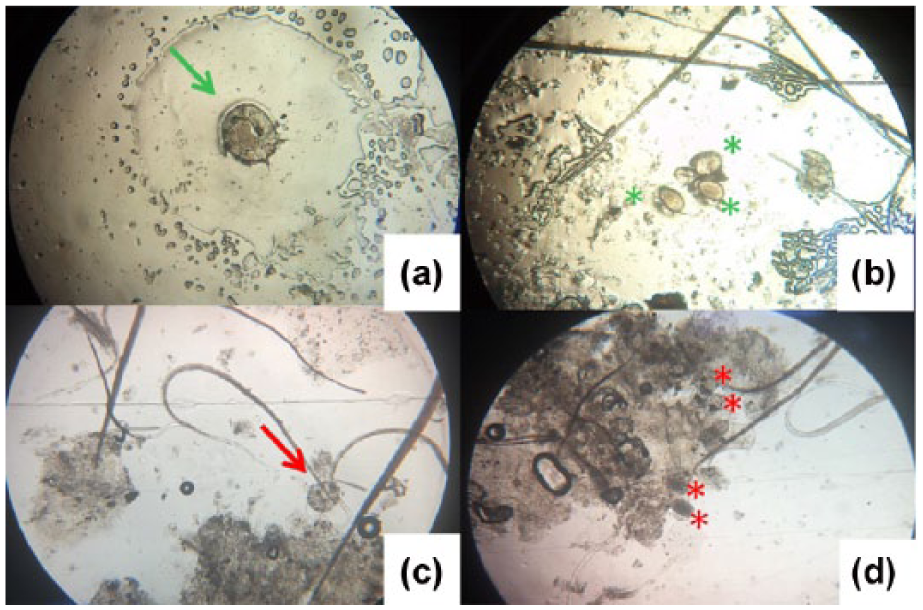
Representative images of Notoedres cati mites on parasitologic test. (a) Adult form (green arrow) and (b) eggs (green asterisks) of N cati in samples obtained by acetate tape impression. (c) Nymph (red arrow) and (d) eggs (red asterisks) of N cati in samples obtained by superficial skin scraping. Note the presence of scabs and cellular debris in the superficial skin scraping sample
Parasitologic evaluation showed no significant difference between the methods, regardless of the development phase of the mite. We observed an average (mean ± SD) of 4.18 ± 9.99 eggs, 10.38 ± 15.47 larvae, 14.7 ± 27.1 nymph/adults and a total of 29.04 ± 49.2 mites by the superficial skin scraping method; and an average (mean ± SD) of 2.30 ± 4.27 eggs, 9.36 ± 15.95 larvae, 13.60 ± 29.17 nymph/adults and a total of 25.02 ± 48.2 mites by the acetate tape impression method (Figure 3a–d). Furthermore, the tests showed a high correlation to the total of mites (r = 0.928).

Distribution of notoedric mites in samples obtained by acetate tape impression with skin squeezing and by superficial skin scraping: (a) eggs, (b) larvae, (c) nymph/adult mites and (d) total number of mites. Results of (e) Bland–Altman analysis and (f) average number of mites observed according to the clinical score for cats infested with notoedric mange. HPF = high-power field
Bland–Altman analysis was performed to compare both diagnostic tests (Figure 3e) and showed a bias of 4.02 ± 18.01 (95% confidence interval −32.3 to 40.3). The mean ± SD number of mites observed by the acetate tape impression was 5.7 ± 2.9, 19.3 ± 9.2 and 83.6 ± 64 mites in cats presenting the clinical scores of 1, 2 and 3, respectively (Figure 3f).
Discussion
The current gold standard test for diagnosis of notoedric mange is the superficial skin scraping, as it is both efficient and reproducible. 7 However, this method is not only traumatic, but also challenging in uncooperative or aggressive animals, occasionally requiring chemical restraint. 15 Herein, we show that the acetate tape impression method is as sensitive as the skin scraping method for the investigation of N cati. Few studies have evaluated diagnostic techniques for notoedric mange. 10 As far as we are aware, no study has compared acetate tape impression and skin squeezing with superficial skin scraping, underlining the importance of the results of this study.
The distribution and characteristics of the lesions, such as crusting at the edge of the pinnae, which rapidly spreads to the face, eyelids and neck, 13 and intense pruritus presented by the cats, were highly suggestive of notoedric mange. This facilitated the selection of the animals in this study.
No significant difference was observed in the number of mites found by both methods, regardless of the stage of development of the mite. In addition, the results of Bland–Altman analysis showed a very low discrepancy between the results of total mite count of both tests. Therefore, in our hands, the acetate tape impression test proved to be as sensitive as superficial skin scraping.
Even though the tape strip test has not been recommended for diagnosing notoedric mange, this test proved to be effective even in low levels of infestation. Moreover, in this study, the conventionally used superficial skin scraping test showed false-negative results in two cats with a low level of infestation. We speculate that visualization of the mites is more difficult with the skin scraping method, for two reasons: (1) the adult mites no longer move as 10% KOH is lethal to them, and (2) there is a higher amount of cellular debris and blood present. The latter has been previously reported in dogs.16,17 We used 10% KOH as the clearing agent, which digests the keratin preserving the mites. Despite this, we observed more debris in the skin scraping method, likely because it is more invasive.
The tape impression method proved as efficient and effective as the skin scraping test, with the advantage of being less traumatic and painful for the cat. Moreover, skin scrapings performed with a scalpel blade increase the risk of injury to both the cat and the user. It has become increasingly important that veterinarians avoid situations where blood-borne pathogens can be transmitted to people. In addition, using a new blade for each cat is expensive and there is risk of transmitting diseases (eg, Bartonella species, Rickettsia species, feline leukemia virus, feline immunodeficiency virus) when the blade is re-used. 7
Conclusions
In our center, the acetate tape impression test is a good method for diagnosing N cati infestation in cats. We were even able to diagnose notoedric mange in animals presenting with low infestation. Moreover, it is less traumatic and facilitates reproducible collection of clinical specimens in more sensitive locations, such as the eyelids, around the lips and the paws.
Footnotes
Supplementary material
Table to show the number of eggs, larvae and nymph/adults observed on skin scraping and tape strip samples.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
KOS was supported by grants from Scientific Initiation of State University of Ceará (IC-UECE).
