Abstract
Practical relevance:
Respiratory parasites infecting domestic cats are attracting increased attention in feline clinical practice. In addition to the most commonly recognised ‘cat lungworm’ Aelurostrongylus abstrusus, Troglostrongylus brevior and Capillaria aerophila are now considered important pathogens of the respiratory tract of cats.
Global importance:
These parasites are being increasingly diagnosed in several regions of the world and a continuous update on epidemiological changes and advances in diagnosis and control is of practical importance.
Aims:
This article reviews current knowledge of lungworms affecting cats, with a special focus on recent insights into diagnosis and management of the diseases they cause. The article also explores some potential new avenues for control of feline parasitic respiratory diseases, and highlights some key areas requiring further research.
Lungworms of cats
Traditionally, feline lungworms have been reported to be Aelurostrongylus abstrusus (Nematoda, Metastrongyloidea, Angiostrongylidae) and Capillaria aerophila (syn Eucoleus aerophilus) (Nematoda, Trichocephalida, Trichuridae). 1 Of these, A abstrusus (ie, the ‘cat lungworm’) has long been regarded as the most significant respiratory parasite of domestic cats (Felis silvestris catus) in terms of worldwide geographic distribution and clinical importance.1,2 The adult stages of A abstrusus (length ~5–12 mm, width ~54–80 µm) reside in nodules within the bronchioles, alveolar ducts and alveoli of infected hosts.
Conversely, C aerophila has been little recognised as a significant pathogen until recently. 2 This nematode infects the lungs of domestic and wild carnivores and, sometimes, people. 2 The adult stages (length ~20–30 mm) are slender (width ~60–180 µm), whitish and filamentous, and live beneath the epithelium of the trachea and bronchi. 3 This nematode has a direct life cycle (Figure 1). After mating, the females release eggs that are coughed, ingested and passed in the faeces of the infected animal. In the environment, they reach infectivity in 1–2 months. 4 Earthworms have been hypothesised to act as facultative intermediate or paratenic hosts, but this biological feature has not been conclusively established.3,4 Animals become infected by ingesting larvated eggs present in the environment or, potentially, infected earthworms.

The adult stages of Capillaria aerophila live under the epithelium of the trachea and bronchi. After mating, the females release eggs that are coughed, ingested and passed in the faeces of the infected animal. In the environment, they mature and become infective. Earthworms have been hypothesised to act as facultative intermediate or paratenic hosts. Animals become infected by ingesting environmental larvated eggs or, potentially, infected earthworms

It is now known that A abstrusus and C aerophila are not the only lungworms to infect domestic cats. The genus Troglostrongylus (Nematoda, Metastrongyloidea, Crenosomatidae), which includes four species of lungworms, mostly reported from wildlife (eg, leopard, African wildcat), 5 has also now been identified in domestic cats. Troglostrongylus brevior, first described in Felis silvestris lybica, the African wildcat, and in Felis chaus, the Jungle cat, 6 has recently been reported.2,5 Adult worms of T brevior vary from ~5–13 mm in length and ~294–430 µm in width, and inhabit the bronchi and bronchioles of infected hosts.

T brevior and A abstrusus have a similar biological cycle (Figure 2). Adult females lay eggs that develop within the lungs. First stage larvae (L1s) reach the pharynx via the respiratory escalator, then are swallowed and pass into the environment in cat faeces.2,4 –7 L1s continue their life cycle in intermediate hosts (slugs and snails), in which they reach the third, infective, larval stage (L3) after a variable period of time dependent on the invertebrate species and environmental temperature.4,6 –9 Animals become infected by ingesting the intermediate host containing L3s or, most often, paratenic hosts including rodents, frogs, lizards, snakes and birds (Figure 2).4,6,7 Recent evidence suggests that T brevior might also be directly transmitted from an infected queen to her litter. 10
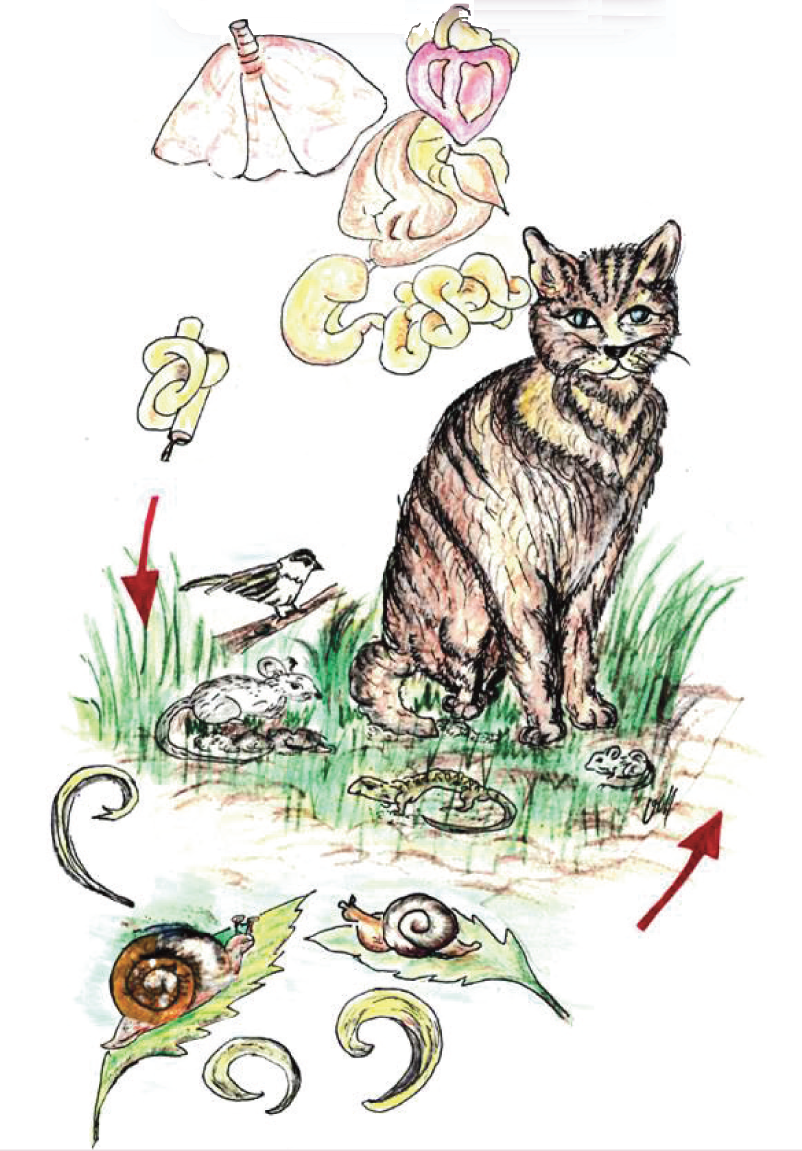
The adult stages of these nematodes live in the respiratory system of the definitive host: bronchioles, alveolar ducts and alveoli (Aelurostrongylus abstrusus) or bronchi and bronchioles (Troglostrongylus brevior). The females release eggs that develop within the parenchyma and, after hatching, first stage larvae (L1s) pass up the respiratory escalator, are swallowed and excreted via the faeces, and reach the environment. L1s continue their development in intermediate hosts (slugs and snails), in which they reach the third larval stage (L3), which is infective for cats. Cats become infected by ingesting the mollusc or, more frequently, paratenic hosts (rodents, frogs, lizards, snakes, birds). Thereafter, L3s migrate to the lungs via the haemolymphatic vessels and evolve to adulthood in the respiratory system
In the past few years, several studies have shed light on new epidemiological scenarios, and provided a better understanding of practical aspects of feline aelurostrongylosis (eg, diagnosis and therapy) and intriguing novel data on ‘new’ and/or neglected lungworms affecting domestic cats. The present article provides an update on current knowledge and discusses priorities for the practitioner.
New paradigms for a changing epidemiology
It is speculated that the geographic range of lungworms infecting cats is possibly expanding. Global warming, movements of animals from endemic to free regions, changing vector phenology, and modification in wildlife habitat and populations may be involved. 11 Alternatively, heightened awareness may have led to an increase in documented clinical diagnoses of previously unknown or underestimated nematodes.
A abstrusus
A abstrusus is distributed worldwide and may infect all cats regardless of their habitat, lifestyle, sex and breed. It is present in nearly every European country, and has been described in Australia and the Americas, as well as in some reports from Asia and Africa.2,11 Owned animals and cats living indoors or with few chances to access the environment are at less risk of A abstrusus infection. However, infection should not be ruled out in household cats. Young animals, with a less efficient immune system, and adult and old cats, with better hunting ability and a cumulative greater risk of exposure, may be infected by A abstrusus.12 –15
C aerophila
C aerophila has long been found in wildlife. In Europe it has occasionally been reported in companion animals, while for many years up to 5% of feral cats in Australia have consistently been found to be infected with C aerophila.2,16 Over the past couple of decades there has been an increase in the number of clinical descriptions, and epidemiological studies have documented the occurrence of C aerophila in cats from different countries,3,17,18 and in humans as well. 19 In some European countries it is likely that wildlife may influence the dispersion of lung capillariosis in cats, as suggested by recent genetic studies that have indicated that C aerophila populations co-infect pets and wildlife in the same areas. 20
T brevior
Nematodes of the genus Troglostrongylus have historically been regarded as parasites of wild felids2,5 but infections in domestic cats are being increasingly reported, mostly in young animals. The first report of troglostrongylosis in a domestic host was from the last century when a feral cat was found to harbour T brevior. 21 Since 2010, however, T brevior has been described in domestic cats from several European islands (Ibiza, Sicily, Sardinia, Crete),22 –25 and in central and southern Apennine regions of Italy.10,26 –28
This increased detection rate in domestic cats has raised questions regarding the origin and actual distribution of T brevior in felid populations. 2 It is not known whether: (i) T brevior infects F s catus more frequently than previously thought and that historically it has been mistaken for A abstrusus; (ii) recent reports reflect increased infection in domestic felids only in particular areas; and/or (iii) some epidemiological drivers have nurtured a spillover of lungworms from wildlife to F s catus. 2
Reduction of woodland habitat drives wild animals into new hospitable environments, such as suburbs and cities, in search of anthropogenic food sources. These movements increase the contact between wildlife and pets and contribute to the spread of parasites harboured by wildlife. Wild animals such as the European wildcat (Felis silvestris silvestris), in which T brevior has been found, play a role as spreaders of the parasite in those areas where wildcats and domestic cats live in sympatry.5,29,30 Indeed, T brevior has been mostly described thus far in cats living in areas of Italy and Greece where wildcat populations are present.2,5,30,31 Additionally, in domestic cats, T brevior is often found in association with A abstrusus.22,25 –27
A recent retrospective study demonstrated negligible occurrence of Troglostrongylus species in domestic cats living in sub-Apennine territories of central Italy during the years 2002–2013. 31 The high presence of T brevior in wildcats originating from the same geographic areas, 30 along with other findings,29,32 ultimately confirm the role of the European wildcat as a natural host of T brevior. Other data collected in 2014/2015 have shown that the prevalence of monospecific infections by T brevior or A abstrusus in domestic cat populations living in regions of the sub-Apennine territories of central Italy is similar. 33 These recent studies suggest that in the past few years some epidemiological and biological drivers might have nurtured bridging infections by T brevior between wildlife and F s catus, and that the nematode is now expanding its host range to include domestic hosts.
This assumption is corroborated by recent findings pertaining to the closely related metastrongyloid Angiostrongylus vasorum, which affects canids in Europe. The high occurrence of this mollusc-borne nematode in foxes living in Italy, Denmark and the UK and the dense fox population in these same areas have been incriminated in the transmission of this parasite to the domestic dog.11,34 –37 Studies have demonstrated the existence of shared populations of A vasorum among dogs, foxes and wolves in endemic regions.37 –39
Possible influence of global warming
Another possible driver influencing current changes in feline lungworm epidemiology is global warming. Temperature, moisture and water availability affect the development and survival of molluscs. The development of nematode larvae in snails and slugs is also known to be influenced by temperature changes. For instance, the higher the average temperature, the higher the rate of larval development of A abstrusus in Helix aspersa (Figure 3), the Mediterranean edible snail. 40 H aspersa, one of the most widely distributed land snails in the world, 41 has been deliberately or accidentally imported into different regions and is widely regarded as a pest outside its native Mediterranean range. 42 This mollusc is one of the most important intermediate hosts of A abstrusus and T brevior, and it is plausible that it has contributed (and is contributing) to the expansion of feline metastrongyloids in European countries. 43

The Mediterranean land snail Helix aspersa may be contributing to the expansion of feline metastrongyloids

Open-mouth breathing in a cat infected with Aelurostrongylus abstrusus
Changes in geographic distribution influenced by global warming have been predicted for A vasorum via a climatic-based model. 44 No similar information is available for A abstrusus or T brevior; however, with similar biological cycles, the same factors involved in the expansion of canine angiostrongylosis would likely also affect feline lungworms. Further studies in this field are warranted.


Diagnostic features
Respiratory parasitoses of cats are often challenging to diagnose clinically because other conditions present with similar signs. Differential diagnoses include bacterial infections, other parasitic infections such as pulmonary toxoplasmosis, respiratory mycoses, feline bronchial disease/asthma, airway foreign bodies and pulmonary tumours.11,16 Signs caused by A abstrusus can be identical to those associated with feline bronchial disease/ asthma. 16 Furthermore, treatment for bronchial disease/asthma with corticosteroids and bronchodilators may produce a clinical recovery in cats with respiratory parasitoses; thus veterinarians have no reason to suspect a misdiagnosis.3,16 There is no evidence at the moment, but it is likely that similar confusion might arise in cats infected with T brevior.
Features of radiographic examinations are not pathognomonic for aelurostrongylosis.57,58 The most common findings in cats infected by A abstrusus and T brevior are an alveolar pattern followed by bronchial and interstitial patterns, bronchial wall thickening and increased interstitial opacity (Figure 5), depending on the worm burden and the chronicity of infection.

(a) Right lateral and (b) ventrodorsal thoracic radiographs of a 2-year-old cat with Aelurostrongylus abstrusus infection. A complex pulmonary pattern is visible throughout the lung fields, characterised by bronchial and alveolar changes and a mild reticular interstitial pattern. (c) Right lateral and (d) ventrodorsal thoracic radiographs of a 3-month-old kitten infected by Troglostrongylus brevior. An interstitial pattern (c,d) is present throughout the lungs fields and there is enlargement of the caudal lobar pulmonary artery (d). Courtesy of Dr Paolo Emidio Crisi, Teaching Veterinary Hospital, University of Teramo, Italy
Assays for the serological diagnosis of feline lungworms are still awaited. A recently developed immunofluoresence antibody test (IFAT) able to detect antibodies against A abstrusus in sera from both experimentally and naturally infected cats showed promising sensitivity and specificity. 59 However, currently, definitive ante-mortem diagnosis of respiratory parasitosis can be achieved only with copromicroscopic or airway wash examinations that reveal L1s of A abstrusus and T brevior, and eggs of C aerophila.
Direct faecal smears and classical sedimentation and flotation methods are unreliable for diagnosing metastrongyloid infections, because of the unpredictable presence of L1s, inadequate sample size, low sensitivity and larval osmotic damage (Figure 6) due to the high specific gravity concentrated solutions.11,49,60 The gold standard faecal test to diagnose feline aelurostrongylosis and troglostrongylosis is the Baermann migration method. However, it requires 12–48 h before larvae can be found and specific skills in discriminating between L1s.2,11,61 In addition, repeated examinations are necessary to avoid false negative samples due to prepatent infections or intermittent eliminating and non-eliminating shedding cycles.11,43

Dehydrated larva of Troglostrongylus brevior observed on flotation with zinc sulphate solution
An accurate morphological and morphometric appraisal of L1s is necessary to achieve a species-specific diagnosis but microscopic differentiation between A abstrusus and T brevior (Figure 7) is often difficult due to overlapping morphological features and individual variations in some characteristics.2,5,6,62 The lengths of A abstrusus and T brevior L1s are ~360–415 μm and ~300–357 μm, respectively.2,4,5,23,43,60,63 However, A abstrusus L1s as short as ~300 μm have been reported in cases confirmed with histological 64 and genetic characterisation. 22 Thus, L1s of the two species should also be differentiated based on head and tail features.
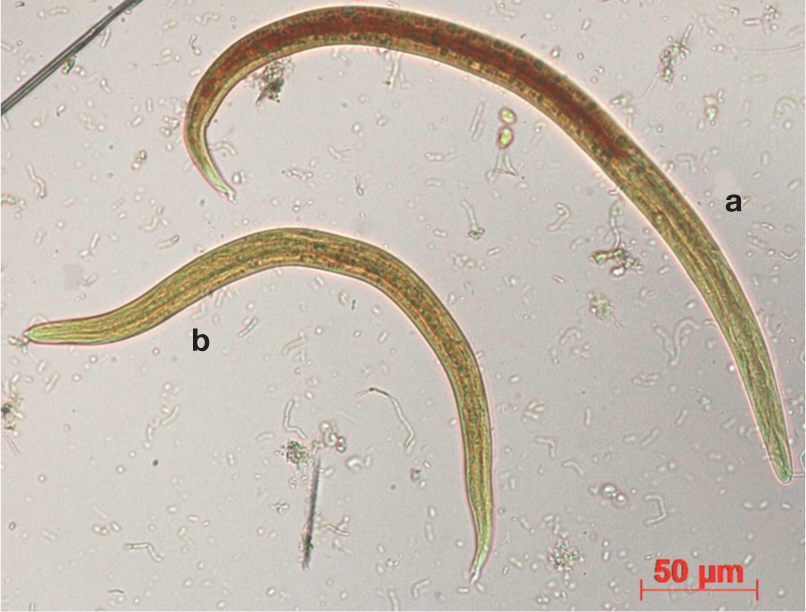
First stage larvae of (a) Aelurostrongylus abstrusus and (b) Troglostrongylus brevior
The head of A abstrusus L1s is rounded and has a terminal oral opening, while the tail is kinked (S-shaped) with distinct knob-like or small finger-like projections at the tip of cuticular spines (Figure 8).5,7,43 L1s of T brevior have a pointed anterior extremity and a subterminal (dorsal) oral opening, while the tail gradually tapers towards the extremity, and possesses a deep dorsal incisure and a shallower ventral incisure near the tip (Figure 8). 5
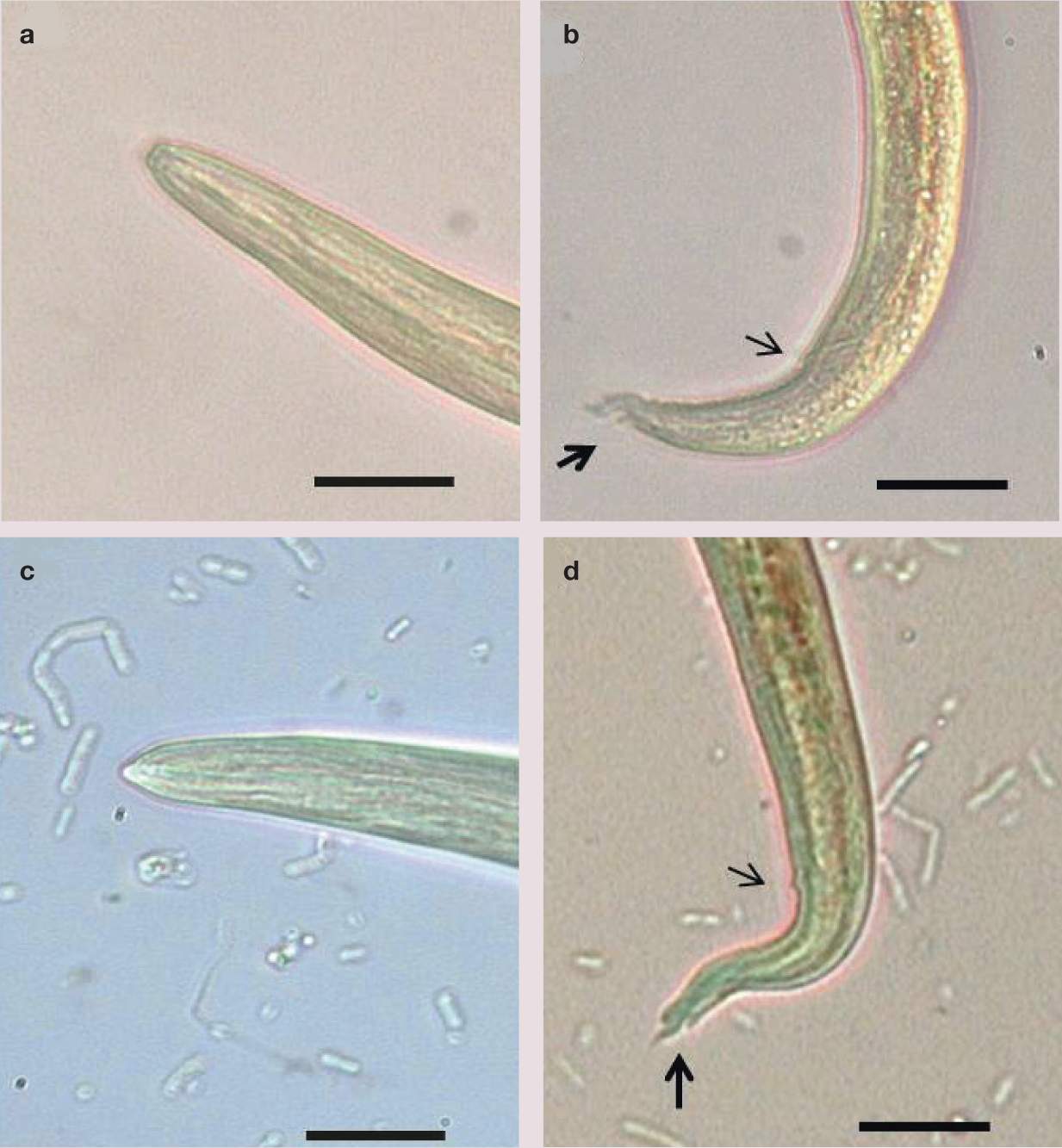
(a,b) First stage larva of Aelurostrongylus abstrusus: (a) magnification of the anterior end, showing a rounded extremity and a terminal oral opening; (b) magnification of the tail, which is kinked, with distinct knob-like or small finger-like projections at the tip of cuticular spines. Note the dorsal incisure (bold arrow) and the anal opening (light arrow). (c,d) First stage larva of Troglostrongylus brevior: (c) magnification of the anterior end, showing a pointed head and a subterminal oral opening; (d) magnification of the tail, showing the presence of a deeper dorsal incisure (bold arrow) and a shallower ventral incisure. Note the anal opening (light arrow). Scale bar = 20 μm
L1s of A abstrusus (and presumably of T brevior, although no evidence is published) can be detected in other samples such as transtracheal aspirates, tracheal swabs, bronchoalveolar lavage fluid and pleural effusions. However, sampling from the respiratory tract requires sedation or anaesthesia and these procedures may fail to detect larvae due to absence of significant pulmonary tissue involvement, limited numbers of L1s, prepatent infection or poor sample recovery. 11

Lung capillariosis is diagnosed by identification of typical eggs on conventional copromicroscopy. Eggs of C aerophila (Figure 9) have a net-like outer shell, measure ~60–65 x ~25–40 μm, are barrel shaped, and have asymmetrical bipolar plugs with no thickening at the base. 65 These ova should be differentiated from those of feline whipworms or other capillariids. There is a paucity of data on feline Trichuris species, as they have been recorded extremely rarely and only in limited geographical areas.4,11 Their eggs reportedly have symmetrical and ringed plugs and differ in size compared with those of C aerophila. Faecal examination may be complicated by the fact that cats may shed trichurid eggs laid by other capillariids or by parasites of ingested prey. 11
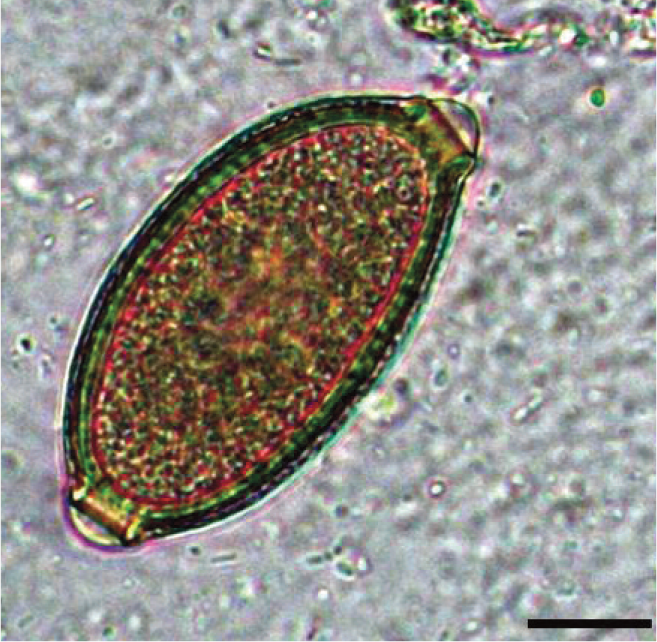
Egg of Capillaria aerophila, showing the typical barrel shape, asymmetry of polar plugs and absence of ring thickening at the base of the plug. Scale bar = 10 μm
Differential characteristics of the most important metastrongyloid L1s and trichurid eggs that can be found in cat faecal samples are described in Table 1.
Differential features of metastrongyloid larvae and trichurid eggs that can be found in feline faecal samples

A minimum length of ~300 μm has been reported in cases confirmed by histological 64 and genetic characterisation 22
To date, described in domestic cats in limited areas: Sri Lanka, Majorca, Hawaii,67 –69 Sicily, 70 Sardinia 71 and northern Spain 63
Parasite with a non-respiratory localisation
Larvae found in bronchi or trachea measured 300–320 µm in length: 6 growth during larval passage in faeces has been hypothesised as a cause of the size variation5,72
Slight individual variation in shape of the caudal end has been reported 5
New diagnostic methods
Innovative assays have been validated for the genetic identification of A abstrusus, T brevior and C aerophila in different biological samples. A duplex PCR based on markers within the ribosomal (rDNA) internal transcribed spacer 2 region (ITS2) was developed to discriminate between faecal L1s of A abstrusus and T brevior in a single cat with a mixed infection. 73 The two metastrongyloids may also be identified by species-specific nested PCRs based on differential markers within the ITS2 region; high sensitivity and specificity was verified on large subsets of samples.26,27 Very recently, a triplex semi-nested PCR has been validated for the simultaneous discrimination of A abstrusus, T brevior and Angiostrongylus chabaudi; 74 the last is a very rare cardiopulmonary nematode of wild felids that has been recorded in a mixed infection with lungworms in a domestic cat. 75
All of these recently developed molecular assays have proved powerful for the diagnosis of feline aelurostrongylosis and troglostrongylosis, overcoming the constraints of classical diagnosis. The PCR assay specific for A abstrusus was validated on a panel of faecal (ie, faeces, flotation supernatant, Baermann sediment) and pharyngeal swab samples from infected cats. It showed a specificity of 100% and a sensitivity of ~97%, and was able to reveal infected cats that were copromicroscopically negative. 76 Similarly, a species-specific PCR proved effective in identifying T brevior in faeces and pharyngeal swabs, even in cats co-infected with A abstrusus or C aerophila.27,28 These findings have demonstrated that a pharyngeal swab is the most suitable sample for the molecular diagnosis of A abstrusus and T brevior with respect to both experimental sensitivity and practicality (avoiding the difficulties of adequate faecal collection in the field, long parasite prepatent and migration periods, laboriousness of DNA extraction from faeces, and presence of PCR inhibitors in faecal samples).26,76 The assays have proved successful in field studies investigating the occurrence of A abstrusus and T brevior in cat populations13,31 and the efficacy of parasiticides in clinical settings.27,54

A similar diagnostic assay specific for a partial region of the mitochondrial (mtDNA) gene encoding for the cytochrome oxidase subunit 1 (COX-1) of C aerophila has been validated. 17 This method displayed a specificity of 100% and sensitivity of 97–100% using faecal samples from cats (and dogs) positive or negative for C aerophila and other endoparasites, including A abstrusus. 17 A similar genetic approach has proved invaluable in assessing the distribution of C aerophila haplotypes in different host species and geographic regions, enabling a better understanding of the current epidemiology of lung capillariosis in Europe. 20

Current and future priorities
Lungworms are gaining more attention in veterinary medicine. More frequent identification of feline lungworm disease could be due to increased prevalence of infections, expanding geographic range, increased awareness, or a combination of these factors. However, it is likely that in certain areas (eg, Europe) lungworms will be increasingly recognised as having a clinical role in respiratory disease of cats.
Practitioner vigilance
From a practical perspective, lungworms infecting cats can be problematic to identify and diagnose, even in the presence of clinical signs. Indeed, any respiratory sign in an at-risk cat (eg, free-roaming animals, kittens) should always ring an alarm bell for clinicians working in endemic areas or with travelling pets. Any suspected case should undergo appropriate diagnostic work-up and, especially in endemic areas, even apparently healthy cats should be screened regularly for lungworms. A routine check for metastrongyloid infection is of importance also for any cat in an endemic region that requires general anaesthesia, given the potential fatal risks associated with lungworm infections. 53
Practitioners should be highly conscious of the importance of lungworms, especially A abstrusus, and must be vigilant for these infections, both in cats living in endemic areas and cats living in territories considered free of infection. These nematodes may be easily introduced into free regions, and thus they should not be overlooked in the presence of compatible clinical signs or ruled out in apparently healthy cats. Veterinarians should start routinely applying the Baermann migration technique (currently infrequently utilised in practice) and perform a careful appraisal of trichurid eggs in cat faeces. 11 Whipworm and capillariid infections in cats are often confused by practitioners, and that it is likely that most capillariid infections are misdiagnosed as ‘whipworm’ infections.2,65
Current armoury
The above advice assumes particular importance in the light of the fact that, in most cases, lungworm treatment protocols are straightforward and effective, especially against A abstrusus and C aerophila. To summarise the treatment data discussed on pages 14 and 15:
Spot-on moxidectin/imidacloprid was effective in stopping larval and egg shedding, respectively, by A abstrusus and C aerophila, and in curing clinical signs in naturally infected cats.82,90
Spot-on emodepside,81,83 selamectin 85 and eprinomectin86,87 similarly showed high efficacy in the treatment of aelurostrongylosis in natural and/or experimental conditions.
Clinical reports in single animals have documented promising activity of moxidectin 54 and emodepside 27 in curing T brevior infection.
Spot-on eprinomectin proved to be efficacious in stopping larval shedding in cats with natural T brevior infection, 87 as well as against both developing larvae and adults of T brevior under experimental conditions. 88
Faecal egg shedding of C aerophila can be reduced or eliminated following a single treatment with eprinomectin.91,92 Also, cats treated with eprinomectin harboured significantly fewer C aerophila lungworms compared with untreated cats. 92
Oral milbemycin oxime has been effective in treating single cases of A abstrusus or T brevior infection. 26
Topical emodepside was effective in treating a case of lung capillariosis, 27 suggesting that a formal evaluation of this formulation for treating C aerophila is worthwhile.
Further field studies involving larger numbers of cases are necessary to evaluate the efficacy of milbemycin oxyme against A abstrusus, T brevior and C aerophila; of selamectin against T brevior and C aerophila; and of moxidectin and emodepside against T brevior. Such studies should take into account the observation that anthelmintic efficacy in treating T brevior seems greatly dependent on the age of the cat, parasite burden, timing and number of administrations, presence and severity of lung damage, and/or host immunity.23,26,27,54
New avenues for control
The potential efficacy of parasiticides in the prevention of metastrongyloid infections opens new avenues for the control of cat lungworms. As a key example, spot-on moxidectin has proved effective in preventing (and treating) infection caused by the closely related parasite A vasorum. 93 It has also been shown that dogs in areas endemic for A vasorum treated every 3 months with milbemycin oxime are half as likely to be infected as those that are not treated, 94 and that monthly treatments with this molecule have the potential to prevent the establishment of A vasorum in dogs. 95 Given that these anthelmintics and others (eg, emodepside, selamectin) have shown efficacy in treating lungworms, further studies are warranted to investigate this management option. However, identifying a risk season for seasonal chemoprevention of lungworms is difficult. Recommendations for chemoprevention would have to consider each local epidemiological situation and take into account the fact that paratenic hosts of A abstrusus and T brevior may be present throughout the year.
The activity of eprinomectin in the chemoprevention of aelurostrongylosis when administered against prepatent developmental stages in experimental conditions is truly promising from a clinical perspective. Administration of this parasiticide 86 is able to limit the progressive pulmonary changes usually occurring during the course of infection.45,96 Field studies are, however, required to support this efficacy, and to evaluate the activity of other anthelmintics in the prevention of cat lungworm infections.
Moxidectin needs to be considered for its important pharmacokinetic properties. This drug remains at detectable levels for weeks after treatment, 97 and it has been demonstrated that consistent administrations of topical spot-on formulations containing moxidectin induce elevated and sustained steady-state plasma concentrations. 98 Thus, repeated administration of moxidectin has the potential to protect animals prospectively against parasites. For instance, the steady-state in cats is reached after three to five treatments, and this protected against infection by hookworms and Dirofilaria immitis.99,100 Studies evaluating the efficacy of moxidectin steady-state in protecting from subsequent infection by lungworms would be very interesting in terms of offering further approaches in the management of these infections.
‘Novel’ lungworms
From an epidemiological perspective, if the current trends in the apparent expansion of feline lungworms continue, we will face an increasing number of clinical cases in a variety of geographic areas. Additionally, other little-known lungworms could gain more importance. For instance, the metastrongyloid Troglostrongylus subcrenatus has so far only been recorded in two cats, one in Malawi, 66 the other in Sicily. 23 Oslerus rostratus has previously only been described in cats in limited areas including Sri Lanka, Majorca and Hawaii; however, more recently, there have been reports in two single cats from Sicily and Sardinia.5,67 –71 Further diagnostic and clinical problems will arise if more cats infected by these two lungworms are identified in wider geographic regions, because their L1s are similar to those of other metastrongyloids and there are no known therapeutic options. 2 This is especially relevant given that these ‘novel’ lungworms may be described in mixed infections, making a definitive diagnosis at the species level truly complicated.
Summary
Continuous awareness of lungworms affecting cats is crucial to further elucidate their geographic distribution and document possible expansion, to refine diagnostic techniques and to improve the current methods available for treatment and prevention of respiratory parasitoses.
Key Points
Feline respiratory parasites –Aelurostrongylus abstrusus, Troglostrongylus brevior and Capillaria aerophila – are gaining increased attention in feline practice due to their worldwide distribution and clinical importance.
Recently, new insights into clinical and laboratory diagnosis, and management and treatment of these nematodes have been gained.
Continuous attention to respiratory parasitoses affecting cats is crucial to further elucidate their geographic distribution, to assess new diagnostic methods and to improve treatment and prevention approaches.
Feline practitioners are encouraged to include feline lungworms in the differential diagnosis of feline respiratory diseases and to apply appropriate diagnostic, therapeutic and preventive measures.
Footnotes
Acknowledgements
The authors thank all of the following friends and colleagues for their valuable participation in studies on feline respiratory nematodes: Fabrizia Veronesi, Andrea Boari, Antonio Frangipane di Regalbono, Anastasia Diakou, Paolo Emidio Crisi, Emanuela Di Giulio, Silvana Meloni, Raffaella Iorio, Barbara Paoletti, Roberto Bartolini, Giulia Morganti and Giuseppe Castagna. The authors are grateful to Matilde Vicovaro for the drawings in Figures 1 and ![]() .
.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Conflict of interest
ADC holds a post-doc fellowship on parasites other than feline lungworms and she has participated in the present review in the framework of her freedom of scientific research.
