Abstract
Objectives
The aim of the study was to determine the clinical benefit and adverse event profile of toceranib phosphate in the treatment of feline oral squamous cell carcinoma (FOSCC).
Methods
Data obtained from the medical records of cats with oral squamous cell carcinoma diagnosed between 2010 and 2014 treated with toceranib phosphate were compared with medical record data from cats that did not receive toceranib, cytotoxic chemotherapy or radiation, to determine the response to toceranib treatment and adverse event profile of toceranib in cats. Concurrent use of non-steroidal anti-inflammatory drugs (NSAIDs) was allowed.
Results
Forty-six cats with FOSCC were included; 23 received treatment with toceranib (group 1) and 23 did not (group 2). The overall biological response rate in group 1 was 56.5%. Median survival time of toceranib-treated cats was significantly longer at 123 days compared with 45 days in cats not treated with toceranib (P = 0.01). Cats achieving stable disease or better on toceranib therapy had significantly longer progression-free survival (P <0.0001) and median survival (P = 0.0042) times than those with progressive disease on toceranib. Administration of NSAIDs was also associated with significantly improved survival time (P = 0.0038) among all cats. Anorexia was common but may reflect the underlying disease in these patients. Toceranib was well tolerated in cats, with the most common side effect being mild gastrointestinal toxicity.
Conclusions and relevance
Toceranib was well tolerated in cats with oral squamous cell carcinoma and may lead to improved survival times, especially when combined with NSAIDs. NSAID administration was also associated with improved survival times, and the relative benefit of toceranib and NSAIDs is difficult to determine from this retrospective study. Despite improvement in survival times, long-term survival in this patient population remained poor. As toceranib was well tolerated and may improve survival time, prospective evaluation of toceranib alone is warranted to assess response as a single agent and as part of multimodal therapy in an effort to achieve a more durable response in FOSCC.
Introduction
Feline oral squamous cell carcinoma (FOSCC) is the most common oral neoplasm in cats, representing >60% of oral tumors in cats. 1 Many treatments have been described, but, because of rapid local recurrence or progression, survival times have been poor. Owing to the advanced stage of disease in most cats at the time of diagnosis, surgical resection is rarely possible. 2 Even when possible, mandibulectomy is associated with high morbidity and local recurrence is seen in >50% of cats. 3 Conventional radiation therapy has shown little benefit as a sole treatment modality, with a median survival time (MST) of 1.5–5.5 months.4–8 Conventional chemotherapy has also not been beneficial for the treatment of FOSCC.7–11 Two studies of accelerated radiation protocols, with and without concurrent carboplatin chemotherapy, have shown modest improvement in survival of 163–174 days with a 29–30% 1 year survival.12,13 While the 1 year survival was improved relative to other studies, the long-term survival was still poor and 19% of cats developed regional or distant metastasis, suggesting the need for systemic treatment for FOSCC. 12
Toceranib phosphate (Palladia; Zoetis) is a small-molecule inhibitor that blocks several receptor tyrosine kinases on the cell surface, including the split-kinase family member kit, vascular endothelial growth factor (VEGF), platelet-derived growth factor and FMS tyrosine kinase 3. 14 Toceranib acts as a competitive inhibitor of adenosine triphosphate and prevents receptor phosphorylation and activation of downstream signaling pathways. 14 Toceranib’s mechanism of action in solid tumors has not yet been fully established but may be due direct antitumor effects, inhibition of angiogenesis or both. 15
FOSCC behaves very similarly to human head and neck squamous cell carcinoma (HNSCC) and FOSCC is considered a model for HNSCC. Both FOSCC and HNSCC are locally aggressive neoplasms with most patients succumbing to local disease rather than distant metastasis. 16 The molecular drivers of human and feline tumors also appear to be similar. 16 Both HNSCC and FOSCC demonstrate overexpression of epidermal growth factor receptor (EGFR).17,18 Increased angiogenesis is demonstrated in both HNSCC and FOSCC with increased VEGF in HNSCC and high microvascular density in FOSCC.16,19,20 One study also demonstrated increased c-kit expression in HNSCC relative to normal tissues. 21 While c-kit expression has not been evaluated in cats, given the similarities between HNSCC and FOSCC c-kit overexpression is plausible in FOSCC. While no studies with toceranib have been performed in cats, preliminary evaluation of toceranib phosphate in dogs with carcinomas showed an overall biological response rate of 71% in dogs with a variety of carcinomas. 15 Of dogs with head and neck carcinoma, 87.5% experienced a clinical benefit, with 75.0% of dogs having a measurable response. 15 Toceranib phosphate targets EGFR and is also antiangiogenic. These facts, combined with the observed biological benefit in dogs with measurable carcinomas to toceranib therapy, suggest toceranib may be an effective treatment option for FOSCC.
No prior studies have evaluated the use of toceranib in cats. Our hypothesis is that treatment of FOSCC with toceranib will be well tolerated and result in improved survival times relative to cats not treated with toceranib.
Materials and methods
Cases were selected by reviewing the medical records of all cats presented to The Animal Medical Center (AMC) Oncology Department between 2010 and 2014 for a biopsy or cytology indicating a diagnosis of FOSCC. Additional cases were identified by the review of AMC biopsy submissions during the same time period. Cases were eligible for inclusion if they met the following criteria: cytological or histological diagnosis of FOSCC and no prior chemotherapy, radiation therapy or surgery other than incisional biopsy for a diagnosis. Cats were assigned to group 1 if they were prescribed toceranib and had one or more follow-up evaluations while on the drug. Cats were assigned to group 2 if they were not treated with toceranib, chemotherapy or radiation at any time from diagnosis of FOSCC to death. Concurrent use of non-steroidal anti-inflammatory drugs (NSAIDs) was allowed in both groups. The signalment (breed, age, gender), date of diagnosis, toceranib dose/schedule, adverse reactions, response assessment, concurrent medications and date of death were recorded for all cats. This information was obtained from the medical records or from telephone conversations with the referring veterinarian or owner. If owners were unable to recall the exact date of death the first, 15th or last day of the month was used if the owner reported the cat died at the beginning, middle or end of the month, respectively. If owners were unable to recall when in the month the cat died, the first of the month was used to avoid overestimating survival times. Cats were excluded from survival analysis if the month of death was not known.
Response assessment in these patients was often subjective, as measurement of oral tumors was difficult in awake patients and cats were not routinely sedated for oral examination or advanced imaging. Although the assessment of stable and progressive disease was subjective in many cases, all complete and partial responses were documented based on RECIST (response evaluation criteria in solid tumors) in dogs. 22 A complete response was recorded when no visible disease was present and partial response when >30% decrease in the longest measured dimension size was noted. Stable disease was recorded when <30% decrease, <20% increase or a subjective assessment of stable disease was noted for at least 45 days on toceranib. Progressive disease was recorded if >20% increase in the longest dimension was present or a subjective assessment of progressive disease was noted on physical examination for patients without tumor measurements. Progression-free survival (PFS) was recorded as the time from when toceranib therapy commenced until the date progressive disease was noted. Overall survival was recorded as the date from the time of diagnosis until the time of death. The response rate to toceranib was reported as the overall biological response rate, which includes patients with stable disease, partial response and complete response. Adverse events were graded based on the Veterinary Cooperative Oncology Group’s (VCOG) common terminology criteria for adverse events version 1.1 and, with the exception of chronic kidney disease (CKD), were recorded as the highest grade experienced by an individual patient at any point in the study. 23 CKD was noted as an adverse event only if there was a change in grade from baseline values during therapy.
Descriptive statistics for continuous data used in formal hypothesis testing was tested for normality by assessment of error residuals and determined to be normal and analyzed by ANOVA, while categorical variables were analyzed by a χ2 analysis. To assess survival time, risk factors for mortality were compared using the Kaplan–Meier product-limit estimates All analyses were conducted using a commercial statistical program (SAS 9.4; SAS Institute) and a type one error probability of 0.05 was utilized as an indication of statistical significance.
Results
Fifty-six cats with FOSCC were identified that had not had curative intent surgery or radiation therapy, of which 46 cases had sufficient information for inclusion in this study. There were 23 cats assigned to group 1 (toceranib treated) and 23 cats assigned to group 2 (no toceranib treatment). Cats excluded from the study included five cats from group 2 for lack of follow-up dates for both PFS and overall survival and five cats from group 1 as they had no follow-up office visits at the AMC after being started on toceranib. For the five cats excluded from group 1 owing to lack of follow-up, 2/5 owners elected not to treat their cats and did not administer the prescribed toceranib; 1/5 cats had only one dose of toceranib given after which the owner elected to discontinue therapy owing to physical difficulty administering the toceranib tablets; and 2/5 cats were prescribed 2 weeks’ worth of toceranib but did not return to AMC and were subsequently seen at alternate hospitals with insufficient information in the medical record to determine response, adverse events or duration of toceranib therapy. The patient signalment data are summarized in Table 1. There was no significant difference between group 1 and group 2 in patient gender, breed, or tumor size. Among all cats, half had anorexia as a presenting complaint; there was no significant difference in the number of anorexic cats or severity of anorexia between group 1 and group 2. Cats in group 1 were significantly more likely to have mandibular tumors than those in group 2 (P = 0.03). No cats had confirmed lymph node metastasis though only one cat had lymph node cytology performed. Of all the cats, 26 had pretreatment thoracic imaging and 2/26 had abnormalities noted (one cat in group 1 with a mediastinal mass suspected to be a mediastinal cyst, and one cat in group 2 with a solitary pulmonary mass with a cytological diagnosis of carcinoma). No cats were treated with radiation therapy at any point from the diagnosis of FOSCC to death.
Summary of signalment variables in all cats
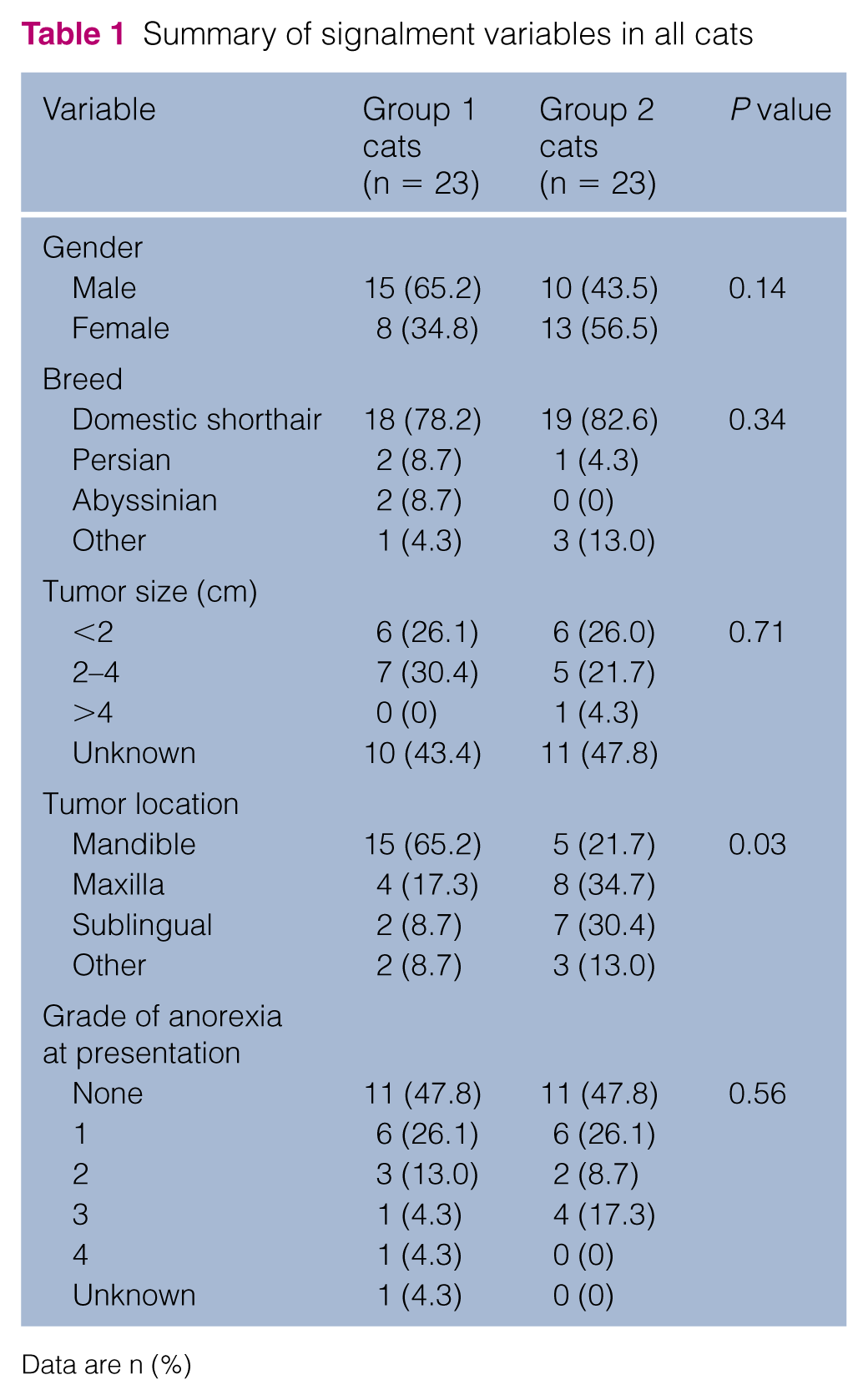
Data are n (%)
Within group 1, one cat achieved a complete response, two had partial responses, 10 had stable disease and 10 had progressive disease for an overall biological response rate to therapy with toceranib ± NSAIDs of 56.5%. All three cats that achieved complete and partial responses were treated with both toceranib and NSAIDs. Of the five cats treated with toceranib alone, 3/5 had stable disease. Only one cat had additional therapy after progression on toceranib. This cat was given carboplatin with no response noted. Two cats in group 1 were lost to follow-up after progression and were censored from the survival analysis at day 35 and day 48. Of the remaining 21 cats in group 1, none were alive at the time of data analysis. Twenty cats died or were euthanized owing to local progression of their FOSCC. The remaining cat died after developing a hyperbilirubinemia of unknown cause. Within group 2, biological response rate could not be evaluated as only 4/23 cats had an assessment of tumor response between diagnosis and death. Of the four cats with evaluation of tumor response, none had complete or partial responses. Two cats treated with NSAIDs had stable disease for 51 and 99 days, respectively. Two cats not treated with NSAIDs had progressive disease noted at 2 and 42 days following diagnosis, respectively. In group 2, 22/23 cats died or were euthanized owing to local progression of their FOSCC. One cat died of an aortic thromboembolism; progressive FOSCC was noted at the time of death.
MST in group 1 was significantly longer at 123 days (range 34–419 days) compared with 45 days (range 1–303 days) in group 2 (P = 0.01), as illustrated in Figure 1. The median PFS in cats achieving stable disease or better on toceranib was 112 days (range 48–263 days). PFS could not be evaluated in group 2 because few cats in group 2 had recheck examinations between the time of diagnosis and death. Cats achieving stable disease or better on toceranib had significantly longer PFS (112 vs 29 days; P <0.0001) and overall survival (201.5 vs 73 days; P = 0.0042) than those that did not respond to toceranib, as illustrated in Figures 2 and 3. The Kaplan–Meier 6 month survival for group 1 was 33.3% and 1 year survival 16.6%. No cats in group 1 were alive at 2 years. The 6 month survival for group 2 was 17.3% and 1 year survival 0%. Within group 1, anorexia at diagnosis was associated with a shorter overall survival time. Anorexia at diagnosis was not a significant prognostic factor within group 2. There was no significant difference in survival time in either group based on tumor location, tumor size at diagnosis or toceranib dose. Other factors with the potential to impact treatment response are summarized in Table 2 for group 1 and in Table 3 for group 2.
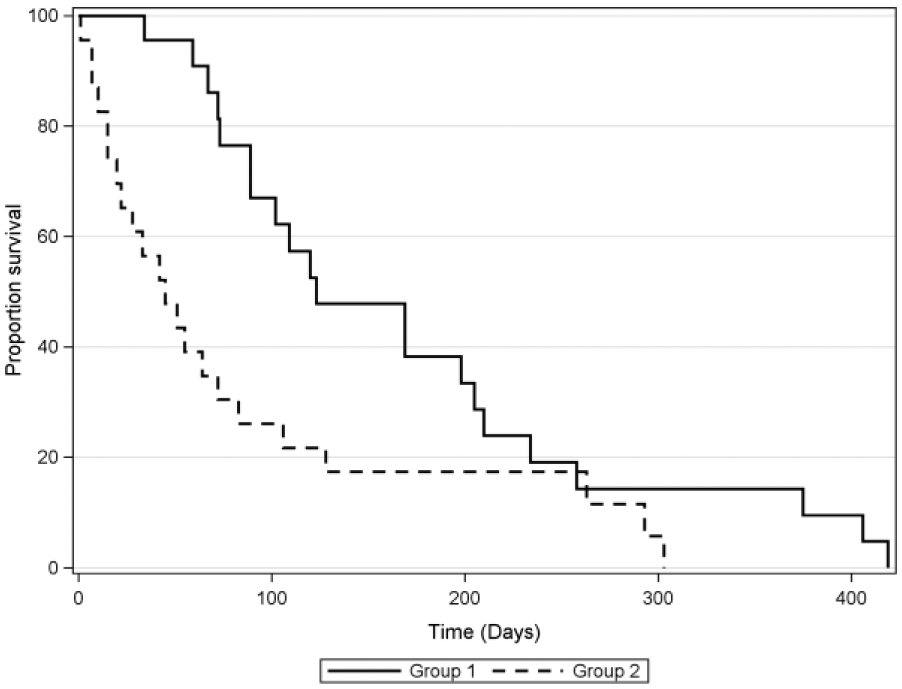
Kaplan–Meier survival curves for cats with feline oral squamous cell carcinoma. Cats in group 1 were treated with toceranib while cats in group 2 did not receive toceranib. Group 1 had a statistically significant longer survival than group 2 (P = 0.01)

Kaplan–Meier progression-free survival (PFS) curves for group 1 divided by response to toceranib. Cats achieving stable disease or better had a statistically significant longer PFS compared with cats with progressive disease (PD) (P <0.0001)

Kaplan–Meier survival curves for group 1 divided by response to toceranib. Cats achieving stable disease or better had statistically significant longer survival compared with cats with progressive disease (PD) on toceranib therapy (P = 0.0042)
Median progression-free survival (PFS), overall survival (OS) and P values associated with prognostic factors evaluated in group 1 only

NSAID = non-steroidal anti-inflammatory drug; SD = stable disease; PR = partial response; CR = complete response; PD = progressive disease
Median overall survival (OS) and P values associated with prognostic factors evaluated in group 2 only
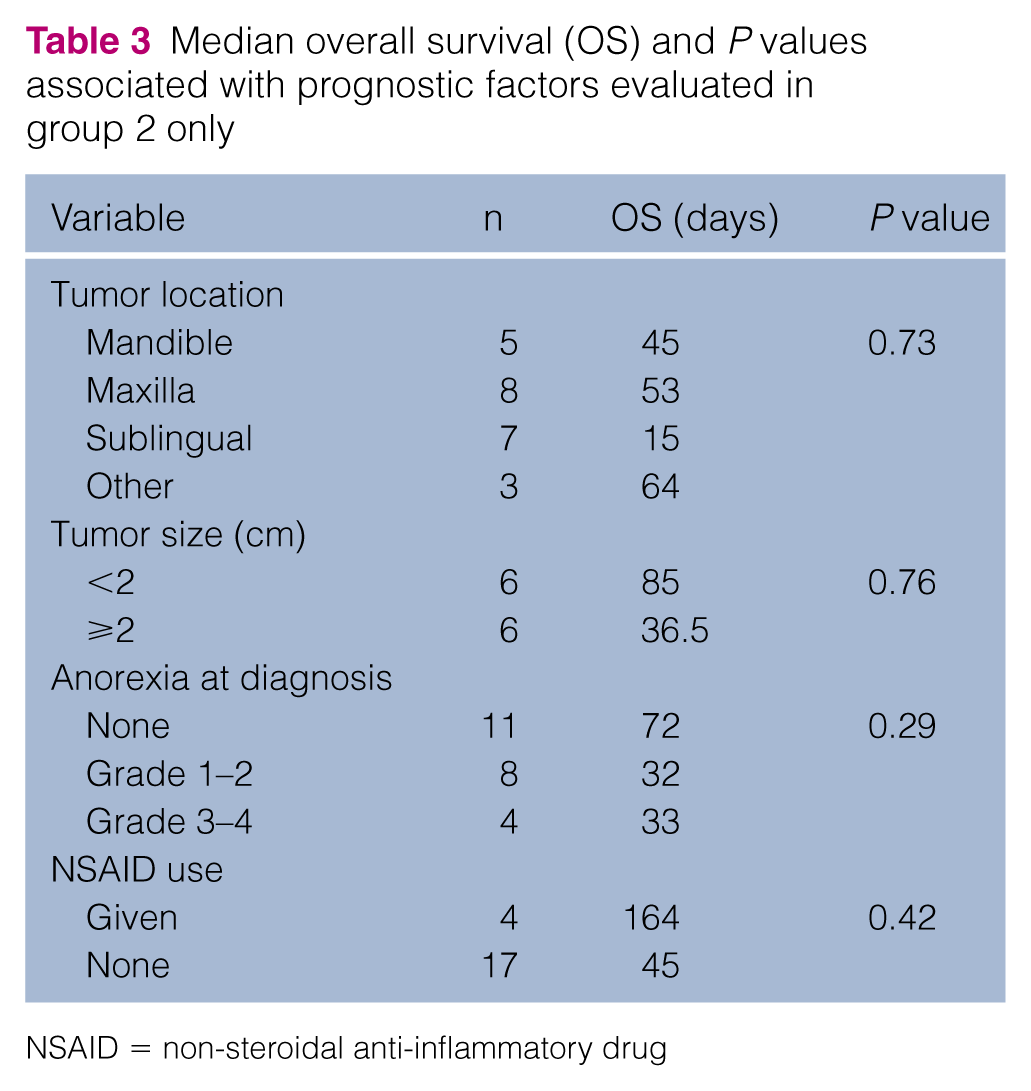
NSAID = non-steroidal anti-inflammatory drug
The median starting toceranib dose in group 1 was 2.52 mg/kg (range 2.0–3.33 mg/kg) with 10 cats treated every other day, 12 cats treated 3 days per week and one cat treated 2 days per week. Three cats underwent dose escalations of toceranib and three cats dose reductions during the study (one each for weight loss, gastrointestinal toxicity and neutropenia). Adverse events were only evaluable in group 1 as few cats in group 2 had recheck examinations between the time of diagnosis and death. Adverse events for group 1 are summarized in Table 4. The most common adverse event was anorexia, with 69.5% of cats experiencing anorexia at one or more time points during the study. No cats required unscheduled office visits or hospitalization for gastrointestinal toxicity. Two cats had short (<7 day) treatment delays due to vomiting or diarrhea, and the toceranib dose was subsequently reduced in one of these cats, while the other was able to continue on toceranib without a dose reduction or recurrence of clinical signs. Two cats developed afebrile neutropenia; VCOG grades I and II. One cat had a short treatment delay and subsequent dose reduction. Toceranib was continued in the second cat and the neutropenia spontaneously resolved. One cat developed a grade 3 hyperbilirubinemia and alkaline phosphatase (ALP) elevation of unknown cause (total bilirubin 2.3 mg/dl, ALP 75 U/l) 28 days after starting toceranib and died at home the day after this was noted. Two cats developed alanine transaminase (ALT) elevations with no clinical signs of hepatic dysfunction. One cat developed a grade I ALT elevation after 12 days on toceranib (ALT 193 U/l) and was treated with Denamarin (Nutramax Laboratories) along with toceranib and the ALT elevation resolved. A second cat developed a grade IV ALT elevation after 30 days on toceranib (ALT 212 U/l). This cat did not receive any treatment for the elevated ALT, and the ALT elevation persisted for the duration of the time the cat was treated with toceranib and resolved when toceranib was discontinued owing to disease progression. Progressive azotemia was noted in five cats, two of which had pre-existing azotemia. All five cats that developed progressive azotemia were treated with both toceranib and NSAIDs. Details of group 1 cats with azotemia are summarized in Table 5. Urinalyses were evaluated in 12/23 cats on toceranib, and none had clinically significant proteinuria (urine protein: creatinine >0.4) while on toceranib. Lameness, which has been reported in dogs receiving toceranib, was not reported in any of the cats.
Adverse events, group 1 only

CKD = chronic kidney disease; ALT = alanine transaminase; ALP = alkaline phosphatase
Renal toxicity, group 1 only

Twenty-two group 1 and 2 cats received an NSAID. Dose and schedule are detailed in Table 6. Among all cats, use of NSAIDs was associated with a significantly longer survival time of 169 vs 55 days, as can be seen in Figure 4 (P = 0.0038). In group 1, 18/23 cats were treated with piroxicam or meloxicam (Metacam; Boehringer Ingelheim) compared with 4/21 cats (two cats unknown) in group 2. In group 1, there was a trend towards a longer survival in cats treated with an NSAID (169 vs 81 days; P = 0.052). Of the five cats in group 1 not given NSAIDs, four had CKD and the remaining cat had concurrent gastrointestinal small cell lymphoma treated with prednisolone, precluding use of an NSAID in these five cats. In group 2, cats treated with NSAIDs had a numerically superior survival than cats not treated with NSAIDs (164 vs 45 days); however, the number of cats treated with NSAIDs was low and this was not statistically significant (P = 0.42).
Non-steroidal anti-inflammatory drug (NSAID) administration in all cats

EOD = every other day

Kaplan–Meier survival curve for all cats divided by use of non-steroidal anti-inflammatory drugs (NSAIDs). Cats given NSAIDs had significantly longer overall survival compared with those not treated with NSAIDs (P = 0.0038)
Discussion
To our knowledge, this is the first study reporting the use of toceranib in cats for any tumor type. In this population of cats with FOSCC, treatment with toceranib resulted in survival times >2.5 times longer than cats not given toceranib. However, despite the improvements seen with toceranib the long-term survival in this patient population remained poor. Only three cats (all treated with toceranib and NSAIDs) remained alive at 1 year following diagnosis and no cats were alive at 2 years. While the survival improvement with toceranib was statistically significant, few cats achieved objective or durable responses. The clinical benefit and low toxicity observed supports further study of toceranib for cats with FOSCC; however, the small number of objective and durable responses suggests toceranib may be best used as part of multimodal therapy.
The adverse events seen in toceranib-treated cats were similar to those reported in dogs, though no evidence of proteinuria or lameness was noted in any of the treated cats. Anorexia was a common finding in toceranib treated cats; however, the high incidence of anorexia prior to treatment suggests that many of these patients experienced anorexia due to their FOSCC as opposed to adverse events from toceranib. Development of vomiting and diarrhea is likely a better assessment of toceranib-induced gastrointestinal toxicity in these patients, and anorexia alone may not warrant discontinuing therapy. Instead, enteral nutritional support with feeding tubes should be considered in anorexic cats if owners wish to continue therapy. Five toceranib-treated cats developed progressive azotemia during the study. All five cats were also administered piroxicam, a drug known to be nephrotoxic, and thus the azotemia could be either from piroxicam, toceranib or the combination. Our data suggest that toceranib may have the potential to be hepatotoxic in cats. Two cats developed ALT elevations; one grade I and one grade IV. The difference in grade of the ALT elevation despite similar numeric elevation in both cats is due to a change in the established reference interval for ALT during the course of the study. Hyperbilirubemia, mild serum ALP elevation and death were seen in one cat. While the cause was unknown, this cat had experienced prolonged, severe anorexia and hepatic lipidosis is considered a plausible explanation for the hyperbilirubinemia and mild serum ALP elevation in this cat, though toxicity from toceranib is also possible. Liver enzyme elevation has previously been noted in dogs treated with toceranib and future studies of toceranib in cats should include close monitoring for hepatotoxicity. 24
Any study, especially a retrospective one, has inherent bias. Cats were not assigned randomly to group 1 and group 2, and follow-up evaluation and use of concurrent medications was not standardized. Use of NSAIDs is the factor considered most likely to affect treatment outcome as NSAIDs may have antineoplastic effects against FOSCC. 25 The results of this study show cats prescribed NSAIDs had significantly longer survival times than those not given NSAIDs. However, caution must be used when interpreting the significance of NSAIDs in this study. The frequency of NSAID use was not equal between group 1 and group 2 (see Table 6), with cats in group 1 significantly more likely to be prescribed NSAIDs than cats in group 2 (P <0.0001). This suggests that owners or veterinarians choosing to treat with toceranib were more likely to treat with NSAIDs as well. The uneven distribution of NSAID administration between group 1 and group 2 means that caution must be used when interpreting the significance of NSAID use across the population as a whole. All five cats in group 1 not prescribed NSAIDs had concurrent disease (four with CKD and one treated with corticosteroids for small cell lymphoma). Because of the retrospective design, we cannot determine if treatment with NSAIDs led to improved survival time in group 2 or if the concurrent diseases present in cats from group 1 not given NSAIDs resulted in shorter survival times. Thus, the patient population not given NSAIDs may be biased towards those cats likely to have shorter survival times. Additionally, cats were included in the NSAID-treated group if NSAIDs were prescribed at any time, regardless of the duration. The short duration of NSAID administration in several cats with prolonged survival times may have biased the results in favor of NSAIDs.
The impact of euthanasia on survival time is a limitation of many retrospective veterinary studies, including this one. Owners who elected to pursue treatment with toceranib for FOSCC may have been less likely to euthanize their cats in the face of progressive disease prolonging survival time in the absence of a treatment effect. PFS has less potential for bias; however, this could not be compared between groups 1 and 2 as few cats in group 2 returned to the hospital, preventing calculation of PFS. However, there are two observations that argue against bias caused by the timing of owners’ decision to euthanize. First, the MST for group 2 is similar to historic reports for cats with untreated FOSCC, suggesting NSAIDs alone did not introduce bias and supports the use of this group of cats as a contemporaneous control population. Second, within group 1, all owners elected to treat their cat; thus, we can assume, all group 1 cat owners were equally committed to an effort to prolong life, making the decision to euthanize more consistent within group 1. Response to toceranib remained strongly prognostic for both PFS and MST within group 1, suggesting that improved survival with toceranib is not due to differences in the timing of euthanasia.
Another potential confounding factor is that the dose and frequency of toceranib was not standardized between cats, and seven cats were treated with doses below the 2.4 mg/kg dose established in dogs as the lowest dose expected to consistently reach blood levels of >40 ng/ml required for inhibition of target receptors.15,24 Despite the lack of standardization of toceranib dose, our study showed no significant difference in MST between cats treated with a toceranib doses greater than the population median of 2.52 mg/kg vs those treated with less than the median dose. This suggests that the pharmacokinetics and minimum efficacious dose may be different in cats than in dogs and warrants further investigation in future studies.
To date, the best long-term survival for FOSCC has been achieved with accelerated radiation therapy, with a 29–30% 1 year survival rate.12,13 Recently, a pilot study combining neoadjuvant therapy with thalidomide, bleomycin and piroxicam followed by surgery, accelerated radiation therapy and adjuvant chemotherapy with thalidomide, bleomycin and piroxicam resulted in long-term survival in 3/6 cats treated. 26 While the number of cats treated was small, those results were promising and also support the use of antiangiogenic agents, such as toceranib, as part of multimodal therapy. In the USA, use of thalidomide is tightly regulated owing to its potential to cause birth defects and the drug is not available for use in veterinary medicine. Toceranib may represent a good choice for combination with accelerated radiation protocols, owing to its antiangiogenic effects and single-agent efficacy in cats.
Conclusions
While long-term survival was not achieved in this study population, a clinical benefit was seen in >56% of the cats treated with toceranib. NSAIDs also were associated with improved survival times, and the relative benefit of toceranib and NSAIDs is difficult to determine from this retrospective study. Further investigation of toceranib alone is warranted to better define single-agent efficacy, and its use as part of a multi-agent treatment protocol may lead to improved responses with acceptable toxicity. The response seen to toceranib in this study makes it suitable for incorporation into a multi-agent protocol for prospective evaluation in an effort to achieve a more durable response in FOSCC.
Footnotes
Acknowledgements
The authors thank Kenneth E Lamb, PhD, of Lamb Statistical Consulting and Medical Editing in West Saint Paul, MN, USA, for providing statistical analysis of the data in this paper.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
