Abstract
Objectives
The aims of the study were to determine the heart rate, rhythm, number of ventricular premature complexes (VPCs) and atrial premature complexes (APCs) in unsedated cats with asymptomatic hypertrophic cardiomyopathy (HCM), using 24 h ambulatory electrocardiogram (ECG) (Holter) in the home environment and to compare the results with those from healthy control cats.
Methods
Fifteen privately owned cats with asymptomatic HCM entered the study. Data from 23 healthy cats, previously published by our group, were used as the control. Clinical examination, measurement of blood pressure, echocardiographic examination, Holter recordings and biochemical analyses were performed in all cats. Holter recordings were obtained in the home environment.
Results
Three-lead ECGs of good quality were obtained from 15 cats with HCM. The median heart rate was 145 (interquartile range [IQR] 137–184) beats per minute for the cats with HCM. All cats with HCM presented with normal sinus rhythm; 60% of cats had intermittent sinus arrhythmia. Median number of VPCs was 3 (IQR 1–17). Three cats had APCs and three cats had both escape beats and intermittent sinus arrhythmia.
Conclusions and relevance
Asymptomatic cats with HCM had few VPCs and APCs on 24 h ambulatory ECG in the home environment. Intermittent sinus arrhythmia was observed in 60% of cats with HCM. The present study shows that the heart rate and the number of VPCs and APCs in cats with asymptomatic HCM are similar to that found in healthy control cats.
Introduction
Hypertrophic cardiomyopathy (HCM) is the most commonly diagnosed heart disease in cats and might lead to clinical signs of congestive heart failure, thromboembolic disease and sudden death.1,2 However, some cats with asymptomatic HCM may survive for a long time and die of a non-cardiac cause.3–5 HCM is defined as a hypertrophied, non-dilated left ventricle (LV) in the absence of any other systemic or cardiac disease that might produce a similar degree of hypertrophy. 6 Cats with HCM may exhibit a variable pattern of ventricular thickening ranging from concentric to focal hypertrophy, which can lead to ischaemia. Myocardial ischaemia is believed to contribute to the production of replacement fibrosis, which contributes to diastolic dysfunction. Histological findings in cats with hypertrophy include myofibre disarray, narrowing of intramural coronary arteries and replacement or interstitial fibrosis.7,8 The replacement fibrosis in the heart muscle of HCM cats may contribute to arrhythmias.
A recently published study of cats with asymptomatic HCM using ambulatory electrocardiogram (ECG) in the home environment showed that cats with asymptomatic HCM had more frequent and complex ventricular and supraventricular arrhythmias than normal cats. The overall heart rate in cats with asymptomatic HCM was similar to normal cats in that study. 9 Sudden death in humans with HCM is thought to be a direct consequence of ventricular tachyarrhythmia. 10 A study using ambulatory Holter in human patients with HCM, where 48% of the patients were classified as New York Heart Association Functional Classification I (cardiac disease but no symptoms and no limitation in ordinary physical activity), showed a high prevalence of ventricular premature complexes (VPCs; 88%), as well as a moderate prevalence of supraventricular tachycardia (37%) and ventricular tachycardia (31%). 11 Ambulatory ECG is a more sensitive method for detection of arrhythmia than a standard ECG recording performed in the clinic.12,13
The aim of the present study was to evaluate the heart rate, rhythm, number of VPCs and atrial premature complexes (APCs) in unsedated cats with asymptomatic HCM using 24 h ambulatory ECG in the home environment and to compare these with data obtained from healthy control cats previously published by our group.
Materials and methods
Recruitment and ethical considerations
Cat owners were informed of the study by advertisements placed in the journal of the Swedish Cat Club (SVERAK), on website home pages of cat breed associations and by providing information at the Veterinary Clinic Djurdoktorn. The study was approved by the Uppsala ethical committee for animal research (C325/5, C327/8, C309/5), by the Swedish Animal Welfare Agency (2005-2383) and by the Swedish Board of Agriculture (31-14010). All cat owners gave their informed written consent for their cats to participate.
Study design
The study was prospective. Healthy cats and cats with HCM were clinically examined, blood pressure was measured and echocardiography performed. 14 Blood samples were obtained for analysis of serum creatinine, alanine aminotransferase (ALT), blood glucose (IDEXX VetTest Chemistry Analyzer 8008; IDEXX) and total thyroxine (T4) (Immulite 2000; Siemens Healthcare Diagnostics).
Inclusion criteria
Inclusion criteria for cats with HCM were as follows: (1) echocardiographic measurements of the LV end-diastolic wall (LVWd) and/or the interventricular septal end-diastolic wall (IVSd) >6 mm; (2) normal respiratory rate and respiratory pattern; (3) systolic blood pressure (SBP) ranging from 100 to 170 mmHg; (4) unremarkable clinical examination (dental disease without clinical significance was allowed); (5) serum creatinine, ALT, blood glucose and total T4 within the reference intervals (RIs). Inclusion criteria for the healthy cats were as follows: (1) absence of left atrial dilation or LV hypertrophy on echocardiographic examination (ie, left atrium to aortic diameter ratio [LA/Ao] <1.5 and LVWd and IVSd ⩽5 mm; (2) no evidence of other structural heart disease; (3) unremarkable clinical examination, including absence of murmurs, arrhythmias or gallop rhythms on auscultation; (4) serum creatinine, total T4 and blood glucose within the normal RIs; (5) SBP <175 mm Hg; (6) presence of normal sinus rhythm on in-clinic Holter evaluation. 14
Exclusion criteria
Exclusion criteria for all cats were as follows: (1) clinical signs of heart failure; (2) structural heart disease other than HCM; (3) systemic or topical medical therapy; (4) concomitant acute or chronic diseases.
Blood pressure
Blood pressure was measured by Doppler sphygmomanometry (The Cat Doppler; Thames Medical) on a front leg, according to the guidelines of the American College of Veterinary Internatl Medicine (ACVIM) Consensus Statement for the identification of hypertension in cats. 15
Echocardiography
Two-dimensional (2D) and M-mode echocardiographic (Philips EnVisor HD and Philips HD 11 XE; Philips Medical Systems) images were obtained using a 12 MHz probe placed on the right precordium with the unsedated cat positioned in right lateral recumbency. Colour flow Doppler was used to visualise any turbulent flow across the mitral valve and/or in the LV outflow tract. The echocardiograms were recorded and analysed according to the recommendations of the American Society of Echocardiography and the Echocardiography Committee of the speciality of Cardiology, ACVIM.16,17 The LVWd and the IVSd were measured in M-mode, and in case of focal hypertrophy measurement was made in 2D. LA was measured with the 2D method for measuring LA and aorta using the right parasternal short-axis 2D view.18,19
Ambulatory ECG recording device (Holter device)
A digital ambulatory ECG recording device (Holter [HR-100; Welch Allyn]) was used for the ECG recordings. The sampling frequency was 200 Hz (samples per second), and effective analogue/digital bit resolution was 0.5 µV. The Holter device weighed 100 g and was carried by the cats in a garment with a pocket. Seven electrodes were fixed to a shaved area on the ventral thorax of the cat, as described previously. 14 The Holter device provided a three-lead ECG. No sedation was used. After the Holter device was fitted, a walking test was performed for all cats before they were allowed to roam freely in the indoor environment at home. During the ambulatory ECG recordings, owners were asked to fill in a questionnaire concerning whether or not the cats were stressed by the Holter garment and the recorder.
Analyses and interpretation of the ambulatory ECG recordings were made semi-manually (PCH 200 Holter Analysis System; Welch Allyn) according to previously published data. 20 The type and the timing of all the beats were manually identified by two of the authors (SH and AT) as the software analysis system often incorrectly annotates normal complexes as being abnormal. The complete ambulatory ECG recordings were manually inspected. The software programme was used for the calculation of the heart rate and by adding missing beats the heart rate was recalculated manually. Diagnostic strips with a duration of 7 s were inspected manually to certify that all complexes were recognised by the computer, and, if not, the missing complexes were added manually both when calculating the heart rate and when diagnosing APCs and VPCs. VPCs occurring during the first hour of ambulatory ECG recording were excluded, to avoid the influence of stress, that might interfere and possibly increase the number of VPCs. The heart rate was calculated in the analysis system semi-manually every 30 mins. During the manual examination of the recording, both the lowest and the highest heart rate of the individual cats were documented. Sinus arrhythmia was diagnosed when there were alternating periods of slower and more rapid heart rates with a variation of 0.1 s or more in successive P-P intervals. 20
Statistical analysis
Data for the HCM cats were investigated for normal distribution using the Ryan–Joiner test. Blood pressure was normally distributed, and mean values and SD are presented. For variables that were not normally distributed, the median value and interquartile range (IQR) are shown. Comparisons between cats with HCM and healthy controls14 were made using the Kruskal–Wallis test (for age, heart rate, APCs and VPCs) and the χ 2 test (for presence of sinus arrhythmia and sinus rhythm). The level of statistical significance was set at P <0.05. A commercial statistical package was used (Minitab 16; Minitab).
Results
Cats
Fifteen privately owned cats aged 1.4–11.2 years (median 4.5 years, IQR 1.7–6.8 years), weighing 3.5–8.4 kg (median 5.3 kg, IQR 4.0–6.8 kg) with HCM were included in the study. Eight different breeds were represented: Norwegian Forest Cat (n = 4), Cornish Rex (n = 4), domestic shorthairs (n = 2), Maine Coon (n = 1), British Shorthair (n = 1), Persian (n = 1), mixed breed (n = 1) and European Shorthair (n = 1). Ten cats were male (all neutered) and five were female (all neutered).
Thirteen of the cats were indoor cats and two were outdoor cats. On physical examination, 12/15 had grade 3/6 murmur and 1/15 had grade 4/6 murmur. Two of the cats had a gallop rhythm and no murmur. No arrhythmia was auscultated.
Twenty-three healthy control cats (10 males, 13 females), aged 1–15 years (mean ± SD 5.5 ± 4.1 years), weighting 3.0–7.4 kg (mean ± SD 4.3 ± 1.1 years) were included. Ten different breeds were represented. 14
Echocardiography
The medians of 2D guided M-mode measurements of LVWd and IVSd in the HCM cats were 6.5 mm (IQR 6.0–8.6 mm, range 5.1–8.6 mm) and 6.5 mm (IQR 5.8–7.1; range 5.3–7.1), respectively. The median LA/Ao measured on 2D echocardiography was 1.13 mm (IQR 1.07–1.29, range 0.9–1.8). LA enlargement was documented in 2/15 cats with HCM.
The healthy control cats were echocardiographically normal. 14
Holter recordings
Three-lead ECGs of good quality were obtained in the 15 cats with HCM with ambulatory ECG recording. The mean duration of the ambulatory ECG recording was 23 h, 11 mins (range 22–24 h). After a short adjustment period, all cats accepted the garment with the ambulatory ECG recording device. Owners of 14/15 cats reported that the cats adjusted to both the garment and Holter device during the recording.
Normal sinus rhythm was present in all the cats with HCM. Sinus arrhythmia occurred intermittently in 9/15 (60%) HCM cats (Figure 1).

Holter recordings. Sinus arrhythmia was present in many cats with hypertrophic cardiomyopathy. The figure shows periods of slower and faster heart rates (HR) with lengthening and shortening of the P-P intervals
All of the healthy control cats had a normal sinus rhythm. In 18/23 cats (78%) intermittent sinus arrhythmia occurred. 14
Heart rate
In the cats with HCM the median maximum heart rate was 245 beats per minute (bpm) (IQR 205–273 bpm) and the median minimum heart rate was 117 bpm (IQR 105–137 bpm) with an overall median heart rate of 145 bpm (IQR 137–184 bpm). Heart rate decreased after a short period of 4 h adjustment time (Figure 2).
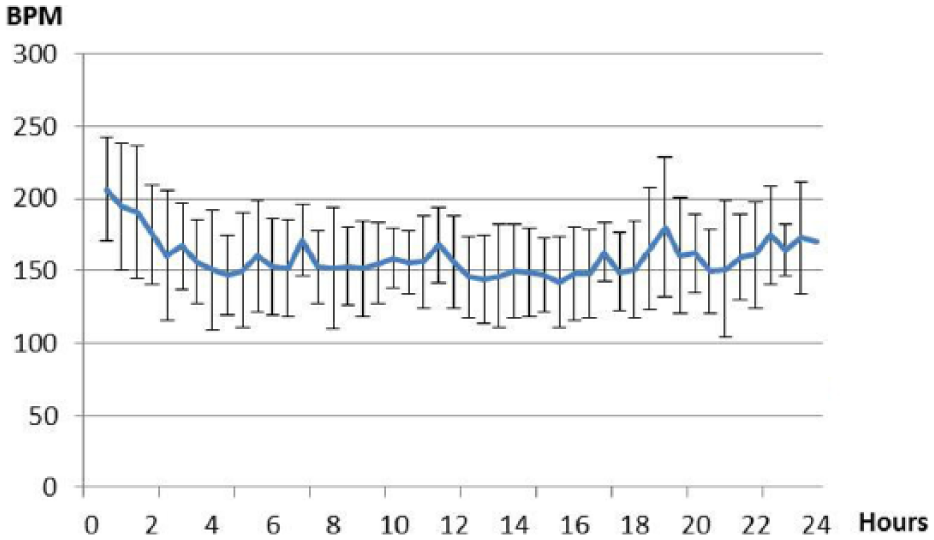
Heart rate over time. Mean heart rate with SD in all cats with hypertrophic cardiomyopathy every 30 mins from the start of the ambulatory electrocardiogram recording at the clinic until the Holter device is taken off the next day. BPM = beats per minute
The healthy control cats had a mean maximum heart rate of 235 ± 25 bpm, a mean minimum heart rate of 131 ± 23 bpm and a mean heart rate of 168 ± 25 bpm. 14
VPCs and APCs
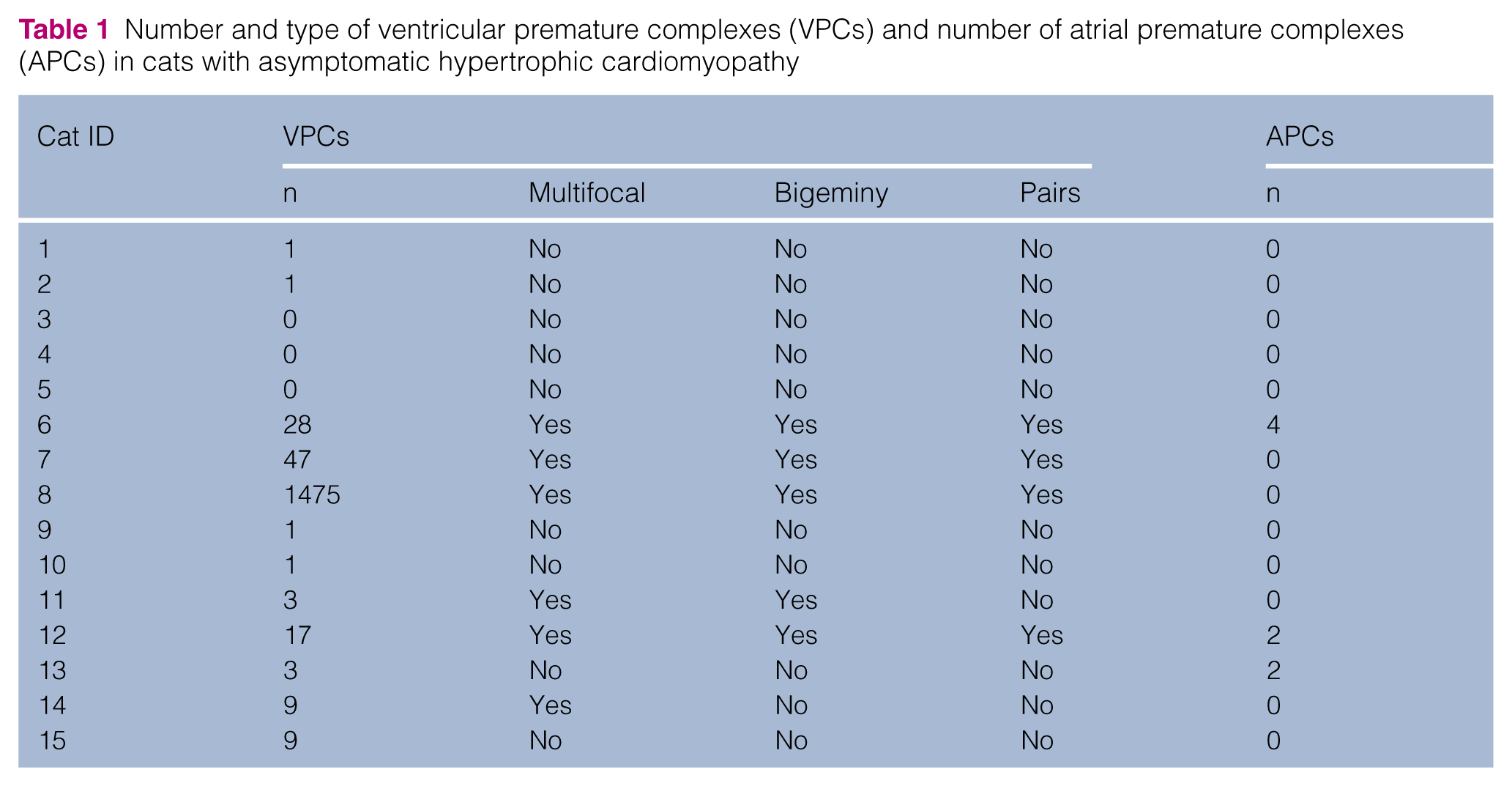
VPCs were present in 12/15 (80%) of the cats with HCM (Table 1; Figure 3). The number of VPCs ranged from zero to 1475 complexes per 24 h period, with a median of three (IQR 1–17). Bigeminal VPCs were documented in five cats, of which four also had VPCs in pairs. The cat that had the maximum number of VPCs showed pairs and bigeminy and multifocal VPCs, and the LV wall was 6.9 mm.
Number and type of ventricular premature complexes (VPCs) and number of atrial premature complexes (APCs) in cats with asymptomatic hypertrophic cardiomyopathy

Holter recordings in a cat with asymptomatic hypertrophic cardiomyopathy. Ventricular premature complexes occur earlier than normal. They are not associated with a P-wave and are broader than the normal complexes if they originate in the ventricles. HR = heart rate
Two of the cats with HCM had two APCs and one cat had four APCs.
In the healthy control cats VPCs (range 0–119 complexes per 24 h period; median 2) occurred in 17/23 (74%). A single APC was found in one cat. 14
Escape beats
Escape beats (range 2–12 beats per 24 h) were documented in 3/15 (20%) cats with HCM on the ambulatory ECG recording (Figure 4). All of these cats had intermittent sinus arrhythmia. All escape beats occurred between 10:00

Holter recordings in a cat with asymptomatic hypertrophic cardiomyopathy. Escape beats occur after a longer pause than normal. They are not associated with a P-wave and are broader than the normal complexes if they originate in the ventricles
Blood pressure
Mean blood pressure was 140 ± 13 mmHg for the cats with HCM.
Comparison with data from the control group
The age of cats with HCM and of control cats did not differ significantly (P = 1.0). Heart rate, number of VPCs, APCs or presence of respiratory sinus arrhythmia did not differ significantly between the groups. All cats had a normal sinus rhythm.
Discussion
The present study shows that the heart rate of cats with asymptomatic HCM was within the RI during most of the recording time, and few arrhythmias appeared during 24 h ambulatory ECG recording in the home environment. The results were similar to those obtained in the healthy control cats and to other studies of healthy cats.14,21,22 Another study reported that asymptomatic cats with HCM had more frequent and severe arrhythmias than the cats in the present study. 9 In the present study, 80% of the cats had VPCs compared with 100% in the previous study. These differences might be due to different cat populations and also to how well the individual cats accepted the Holter device, as stress might give rise to extra VPCs. In the previous study 61% of the owners reported that their cats’ daily activities were moderately to severely affected by the Holter monitor. 9 This is a higher percentage than obtained in our study, where 7% of the owners reported that cats did not get adjusted to the Holter device.
Heart rate in the present study decreased after a short adjustment time for all cats with HCM. This might be caused by increased sympathetic tone before adjusting to the Holter device, in agreement with a previous study. 9 The maximum (245 bpm), minimum (117 bpm) and median (145 bpm) heart rate of the cats with HCM in our study were similar to those obtained from healthy cats and to heart rates in healthy cats studied with ambulatory ECG recordings in other studies.9,14,21,22
All of the cats in our study had asymptomatic HCM and 13/15 had no LA enlargement. Although VPCs were present in 80% of cats with HCM in our study, the total number of VPCs was low, which is in accordance with a previous study. 9 One of the cats with HCM in the present study showed many VPCs during the 24 h recording with multifocal origin. A retrospective study reported that 96% (102/106) of cats presenting with ventricular tachyarrhythmias in a hospital setting had an abnormal echocardiogram, 23 which suggests that VPCs in cats often coexist with structural heart disease. Another retrospective study showed that 57% of cats with HCM had ECG changes, and arrhythmias were documented in approximately 7%, mainly by episodic VPCs. 2 The ambulatory ECG in the present study showed that asymptomatic cats with HCM had few VPCs in a 24 h period, similar to the control group and other studies of healthy cats.9,14,21,22 The reason for the relatively few VPCs found in cats with HCM in the present study might be the asymptomatic presentation and that the median LV chamber wall was only 6.5 mm. Arrhythmias might arise from the myocardium after an ischaemic event and subsequent development of replacement fibrosis. 24 It may be speculated that in mild forms of asymptomatic HCM, as in our study, fibrosis might not be present.
The numbers of APCs per 24 h period for the cats with HCM were similar to the controls and other studies of healthy cats.9,14,21 The finding differs from one study of cats with asymptomatic cardiomyopathy where 88% (15/17) had supraventricular arrhythmias compared with 20% (3/15) in our study. 9
Atrial arrhythmias are sometimes difficult to interpret on ECG because their configuration is similar to normal sinus complexes and APCs might therefore be missed. It might also be difficult to distinguish between sinus tachycardia and atrial tachycardia on an ambulatory ECG.
Sinus arrhythmia was present intermittently in 60% of the cats with HCM in the present study. Sinus arrhythmia was present in approximately 80% of controls, and other healthy cats when using ambulatory ECG recordings.14,21,22 Escape beats occurred in 3/15 (20%) of the cats with HCM and all of these cats had sinus arrhythmia. The presence of sinus arrhythmia and escape beats in asymptomatic cats with HCM might therefore indicate that the heart is not exposed to high sympathetic tone. In humans with increased vagal tone, respiratory sinus arrhythmia and escape beats occur. 25 A recent study showed that dogs with degenerative mitral valve disease and a regular sinus rhythm at physical examination had a higher risk of cardiac death than dogs that maintained their respiratory sinus arrhythmia during physical examination. 26 Minimum and mean heart rate has been shown to increase significantly with increasing severity of degenerative mitral valve disease in dogs, suggesting that sympathetic tone increases or parasympathetic tone decreases when the heart disease deteriorates. 27
The relatively low number of cats in the study is a limitation. Only cats with asymptomatic HCM were included and cats with more advanced cardiomyopathy might have more VPCs and APCs, and this needs to be evaluated in future studies
Conclusions
Ambulatory ECG in the home environment showed that 80% of cats with asymptomatic HCM had few VPCs and that APCs were scarce. Sinus arrhythmia was observed in 60% and escape beats in 20% of the cats with HCM. The results are similar to those previously described in healthy cats. The present study shows that the heart rate and the number of VPCs and APCs in cats with asymptomatic HCM are similar to that found in healthy cats.
Footnotes
Acknowledgements
The technical staff at Evidensia Djurdoktorn Animal Clinic are acknowledged for their assistance. We thank Anna Nilsson for the ambulatory electrocardiogram recording device garments.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the SKK and Agria Research Fund; the Swedish Women’s Animal Protection Association; and the Rasehorn Cat Research Foundation.
