Abstract
Case series summary
Six cats were diagnosed with renal abscesses. Common clinical findings were lethargy, dehydration, abdominal pain and nephromegaly. Fever was noted in half of the cases. Diagnosis was established by ultrasonography, cytological examination and bacterial culture of abscess aspirates. At least one possible contributing factor could be identified in all cases. Antibiotics were consistently used and in two cats the abscess was surgically drained. The short-term outcome was fair but the long-term outcome was dependent on the underlying condition.
Relevance and novel information
The results of this small case series suggest that renal abscess should be considered when nephromegaly and/or abdominal discomfort are noted. Diagnosis of renal abscess is straightforward when ultrasonography and fine-needle aspirate analysis can be performed. Medical treatment is assumed to be preferable but surgical treatment may be warranted on a case-by-case basis. Given that almost every affected cat was diagnosed with at least one comorbidity, a thorough evaluation is recommended for all cats with renal abscesses.
Introduction
Renal abscess (RA) is a generic term encompassing both intrarenal and perirenal abscesses. Such abscesses seem to be rarely encountered in companion animal medicine. They have only been described in isolated case reports, in association with pyelonephritis in cats and dogs, and with diabetes mellitus, polycystic disease and nephrolithiasis in dogs.1–4 A case of post-traumatic RA with nephrocutaneous fistula has also been reported in a dog. 5 This report describes six cats diagnosed with RA and managed with a combination of medical and surgical treatments.
Case series description
RAs were diagnosed in six cats over a 5 year period at two different institutions (see Table 1 for details).
Information relative to the diagnosis, treatment and follow-up of renal abscesses in six cats

Reference intervals for cases 1–5: WBC: 2870–17,020/µl; PMN: 1480–10,290/µl; Mono: 50–670/µl; PME: 170–1570/µl; creatininaemia: 70.4–211.2 µmol/l; uraemia: 12.1–20.4 mmol/l; calcaemia (total): 1.95–2.83 mmol/l; glycaemia: 4.4–6.7 mmol/l; total proteins: 57–89 g/l; ALP: 14–111 U/l; natraemia: 150–165 mmol/l: chloraemia: 112–129 mmol/l. Reference intervals for case 6: PMN: 1480–10,290/µl; PME: 170–1570/µl; creatininaemia: 80–229 µmol/l; albuminaemia: 27–39 g/l
CBC = complete blood count; F = female; DSH = domestic shorthair; NF = neutered female; NM = neutered male; PUPD = polyuria and polydipsia; UTI = urinary tract infection; temp = temperature; NP = not performed; WBC = white blood cells; PMN = polymorphonuclear neutrophils; Mono = monocytes; PME = polymorphonuclear eosinophils; ALP = alkaline phosphatase; USG = urine specific gravity; prot = protein; RBC = red blood cells; CFU = colony-forming unit; PKD = polycystic kidney disease; NA = not applicable; y = years; glu = glucose
Case 1
A 4.5-year-old intact female Persian cat was presented with a 5 day history of lethargy, anorexia and vomiting. Polyuria and polydipsia (PUPD) were also noted but their duration was not reported. Dehydration, painful abdominal palpation and right nephromegaly were noted during the physical examination. Biochemical analyses revealed mild azotaemia (plasma [P-] creatinine: 266.0 µmol/l; reference interval [RI] 70.4–211.2 µmol/l) and hypercalcaemia (P-total calcium: 3.28 mmol/l; RI: 1.95–2.83 mmol/l). Urine (collected by cystocentesis) specific gravity (USG) was inappropriately low (1.014), proteinuria (2+; ‘normal’ result: negative or trace) and haematuria (4+; ‘normal’ result: negative) were revealed by the urine dipstick test, and leukocyturia (>5 leukocytes per high-power field [HPF]) and bacilluria by microscopic sediment examination. Subsequent urine culture yielded growth of Escherichia coli. The ultrasonographic examination revealed severe polycystic kidney disease (PKD) but one cavity of the right kidney was very large (5 cm) and filled with an echogenic fluid (Figure 1). This lesion was completely drained (65 ml) under ultrasound guidance. Microscopic examination showed that the aspirate contained degenerate neutrophils with intracellular rods. Culture revealed an E coli strain with the same sensitivity profile as the one cultured from urine. The cat received intravenous (IV) fluids (lactated Ringer’s solution) and cephalexin (Rilexine 20 mg/kg IV q12h; Virbac) but was discharged the next day at the owners’ request (owing to financial constraints) with a prescription for oral cephalexin (Rilexine; Virbac) at the same dosage. The patient was subsequently lost to follow-up. The owners were telephoned during the preparation of this article. They reported that the cat had apparently recovered after the administration of cephalexin for only 10 days but had eventually been euthanased 15 months after discharge because of end-stage renal failure.
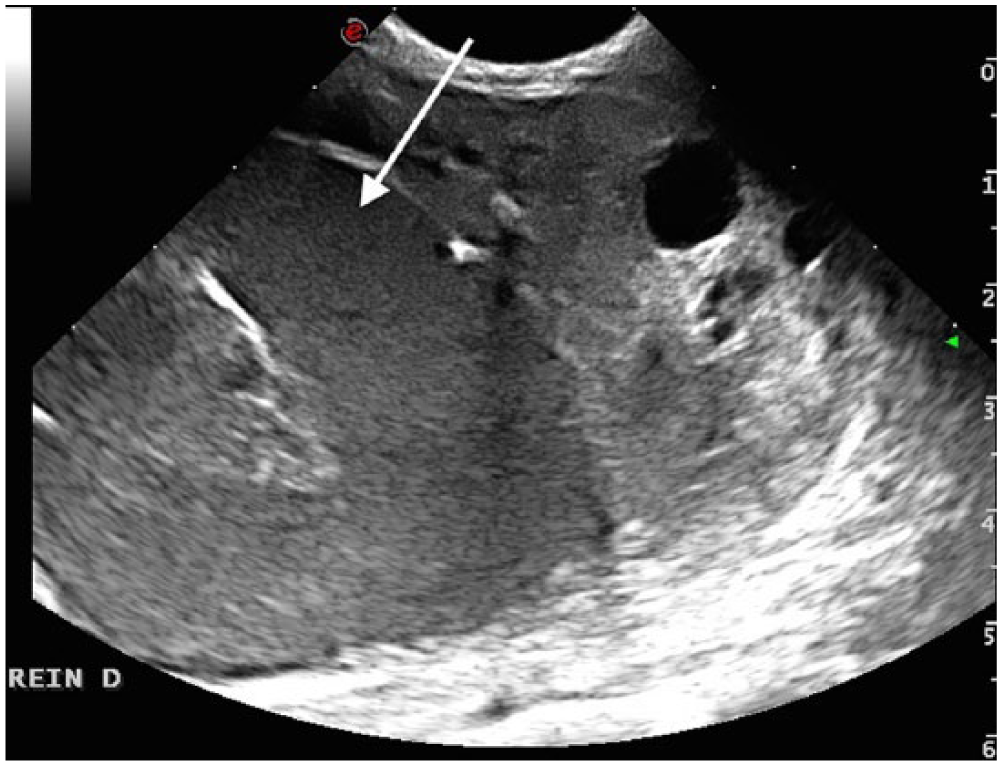
Ultrasonographic view of the right kidney in case 1: the abscess presented as a large, round hypoechoic lesion (arrow). Cysts are identified in the remaining renal cortex
Case 2
A 7-year-old intact female domestic shorthair (DSH) cat was presented with dysorexia and PUPD of 1 month’s duration. This cat had been receiving megestrol acetate (Megecat; Vetoquinol) 1 mg/kg orally every 2 weeks to prevent oestrus. Mild hyperthermia (39.2°C) and several small mammary nodules were apparent during the physical examination. A complete blood count (CBC) revealed leukocytosis (26,810 white blood cells [WBC]/µl; RI: 2870–17,020/µl), with neutrophilia (22,430 neutrophils/µl; RI: 1480–10,290/µl) and the serum biochemical analyses indicated hyperglycaemia (P-glucose: 33.0 mmol/l; RI: 4.4–6.7 mmol/l). The persistence of hyperglycaemia was confirmed by repeated measurements and established the diagnosis of diabetes mellitus, even though serum fructosamine was not assessed at that time. Urine was collected by cystocentesis; USG was 1.034, and glycosuria (4+; ‘normal’ result: negative), proteinuria (3+; ‘normal’ result: negative or trace) and haematuria (2+; ‘normal’ result: negative) were noted on the urine dipstick test. Sediment examination revealed leukocyturia (>5 leukocytes per HPF) and culture was negative. Abdominal ultrasound revealed small kidneys with hyperechoic cortices, poor corticomedullary definition and a 6 × 9 mm hypoechoic cortical lesion in the left kidney. This lesion was aspirated under ultrasound guidance and a purulent fluid was withdrawn. Cytological examination of the aspirate showed degenerate neutrophils and the culture was negative. Treatment consisted of IV fluids (lactated Ringer’s solution), cephalexin (Rilexine; Virbac) 20 mg/kg IV q12h for 6 days then PO for 3 weeks and glargine (Lantus; Sanofi-Aventis) 1 UI SC q12h. Follow-up ultrasound examinations revealed a residual cortical lesion after 10 days of therapy and no abnormality 2 weeks after stopping the antibiotic treatment. Two separate mastectomies were performed, 6 weeks apart. An ovariectomy was also performed during the second procedure. The cat was euthanased 21 months later because of signs consistent with end-stage renal failure.
Case 3
An 8-year-old spayed female DSH cat was presented with a 4 day history of lethargy and anorexia. Haematuria and dysuria were also reported. Physical examination revealed hypothermia (35°C), dehydration, bradycardia (heart rate: 120 beats per minute) and a left nephromegaly. Abnormalities in the biochemistry panel included marked azotaemia (P-creatinine: 798.0 µmol/l; RI: 70.4–211.2 µmol/l), hyperproteinaemia (P-proteins: 100.2 g/l; RI: 57.0–89.0 g/l) and a mild increase in plasma alkaline phosphatase (ALP) activity (P-ALP: 255 U/l; RI: 14–111 U/l). USG (urine collected by cystocentesis) was inappropriately low (1.018), and proteinuria (1+; ‘normal’ result: negative or trace) and haematuria (4+; ‘normal’ result: negative) were present on dipstick urinalysis. Urine sediment examination revealed leukocyturia (>5 leukocytes per HPF) and extracellular rods. The urine culture yielded growth of E coli. Abdominal ultrasonography demonstrated a pyelic non-obstructive urolith in the left kidney and a 7 mm hypoechoic heterogeneous cortical lesion adjacent to subcapsular hypoechoic heterogeneous material (Figure 2). The right kidney was atrophied (13 mm in length) and hyperechoic, with loss of corticomedullary definition.

Ultrasound examination of the left kidney in case 3 showing nephrolithiasis (dashed arrow), a cortical hypoechoic and heterogeneous lesion (solid arrow) and subcapsular effusion mixed with heterogeneous material
The cat was hospitalised and received IV fluids (lactated Ringer’s solution), cephalexin (Rilexine; Virbac) 20 mg/kg IV q12h and ranitidine (Azantac; GlaxoSmithKline) 1 mg/kg IM q8h. The cat’s condition improved over the following days and P-creatinine decreased to 381.9 µmol/l (RI: 70.4–211.2 µmol/l) over 4 days. At that time a surgical procedure was carried out to drain the abscess and obtain samples for culture and histopathology. Recovery was uneventful and the cat was discharged 3 days after surgery on oral cephalexin (Rilexine; Virbac) and a renal diet (Renal RF23; Royal Canin). Subsequent culture of the purulent exudate revealed E coli growth and the renal histopathology analyses were diagnostic of a chronic suppurative, haemorrhagic pseudocyst and chronic tubulointerstitial nephritis.
The cat’s chronic kidney disease (CKD) stabilised in International Renal Interest Society (IRIS) stage 2. Cephalexin was discontinued after 1 month and the subsequent urine culture was negative. Two months after the initial presentation, the cat was again brought in for acute weakness and anorexia associated with a worsening of azotaemia (P-creatinine: 787.6 µmol/l; RI: 70.4–211.2 µmol/l). The owner decided on euthanasia without further assessment.
Case 4
A 15-year-old neutered male DSH cat was presented with sudden onset of lethargy and anorexia. PUPD had been noted for 6 months. Clinical findings included hyperthermia (39.9°C), dehydration, left nephromegaly and painful abdominal palpation. CBC revealed neutrophilic leukocytosis (WBC: 66,240/µl; RI: 2870–17,020/µl with 60,000 neutrophils/µl; RI: 1480–10,290/µl). The serum biochemical analyses showed moderate azotaemia (P-creatinine: 286.9 µmol/l; RI: 70.4–211.2 µmol/l). Urine was collected by cystocentesis. USG was not recorded and urine dipstick analysis indicated proteinuria (2+; ‘normal’ result: negative or trace) and haematuria (1+; ‘normal’ result: negative). Cytological examination of the sediment revealed leukocyturia (>5 leukocytes per HPF) and urine culture was negative. Abdominal ultrasonography showed a 22 mm hypoechoic heterogeneous cortical lesion and a subcapsular effusion of the left kidney (Figures 3 and 4). The left ureter was mildly dilated. The right kidney showed a slight perirenal effusion and poor corticomedullary definition. Cytological examination of the aspirate from the left subcapsular effusion revealed degenerated neutrophils with intracelllular cocci. Culture yielded a group G Streptococcus.

Ultrasonographic view of the left kidney in case 4 showing a large hypoechoic heterogeneous cortical lesion later identified as a renal abscess (arrow)

Ultrasonographic view of the left kidney in case 4: the perirenal abscess presented as a perirenal collection of hypoechoic fluid (arrow)
The cat was hospitalised, received IV fluids (lactated Ringer’s solution) and cephalexin (Rilexine; Virbac) 20 mg/kg IV q12h. Once its condition had stabilised, the abscess was surgically drained and omentalised and renal biopsies were performed. The cat recovered uneventfully and was kept on IV fluids (lactated Ringer’s solution) and cephalexin until discharged, 3 days after surgery. At that time, creatininaemia had decreased to 156.6 µmol/l (RI: 70.4–211.2 µmol/l). Histological examination of the renal biopsies revealed a poorly differentiated renal carcinoma. The cat was still alive 18 months after the initial presentation.
Case 5
A 9-year-old intact female DSH cat was presented with dysorexia and persistent urinary tract infection despite the antibiotic therapy instigated by the referring veterinarian (cefovecin 8 mg/kg SC every 2 weeks, three times). PUPD had been noted for 3 months. Main clinical findings included a left parasternal systolic heart murmur (grade II/VI) and left nephromegaly. The CBC showed neutrophilia (14,470 neutrophils/µl; RI: 1480–10,290 neutrophils/µl) and the biochemical analyses revealed azotaemia (P-creatinine: 306.2 µmol/l; RI:70.4–211.2 µmol/l) and hypernatraemia (P-sodium: 174 mmol/l; RI: 150–165 mmol/l). USG (urine collected by cystocentesis) was inappropriately low (1.016), and the urine dipstick test indicated the presence of pro-teinuria (3+; ‘normal’ result: negative or trace) andhaematuria (4+; ‘normal’ result: negative). Cytological examination of the sediment revealed leukocyturia (>5 leukocytes per HPF) with intracellular rods. Urine culture produced growth of E coli. Abdominal ultrasound examination revealed a small, irregularly shaped left kidney with hyperechoic cortex, poor corticomedullary definition and slight subcapsular hypoechogenicity. The right kidney was large, surrounded by thick hyperechoic tissue and a hypoechoic cortical lesion 5 mm in diameter was apparent. An aspirate from this lesion revealed degenerate neutrophils with extracellular rods. Culture yielded growth of E coli with the same sensitivity pattern as the urinary strain.
The cat was placed on IV fluids, amoxicillin-clavulanic acid (Augmentin; GlaxoSmithKline) 12.5 mg/kg IV q12h and enrofloxacin (Baytril; Bayer HealthCare) 5 mg/kg SC q24h. Once the culture results were available, the cat was kept on subcutaneous enrofloxacin and discharged 3 days later with the same drug (Baytril; Bayer HealthCare) and dosing PO. Creatininaemia at that time was 178.6 µmol/l (RI: 70.4–211.2 µmol/l). Repeated analyses revealed stable values of plasma creatinine (251.7 µmol/l, 248.2 µmol/l and 275.2 µmol/l, 3, 6 and 9 weeks after discharge, respectively; RI: 70.4–211.2 µmol/l) and the persistence of a moderately low USG (1.030). Oral enrofloxacin was continued for 2 months and the follow-up urine cultures were negative. In the recheck ultrasound examinations, the cortical abscess was cyst-like in appearance and the aspirated fluid acellular. However, persistent thickening of the right perirenal tissue with a perirenal effusion was noted.The cat’s general condition worsened progressivelyand the owners elected euthanasia 3.5 months after the initial diagnosis. A partial necropsy was performed. Histopathological analyses of samples from the right kidney and perirenal tissue revealed an infiltrative carcinoma probably of transitional cell origin.
Case 6
A 7-year-old spayed female DSH cat was presented with lethargy, vomiting, anorexia and polydipsia of 3 days’ duration. Main clinical findings included dehydration, tachypnoea and tachycardia with a left parasternal systolic heart murmur, hyperthermia (39.4°C), painful abdominal palpation and asymmetrical kidneys.
The CBC revealed neutrophilia (11,800 neutrophils/µl; RI: 1450–9620/µl) with a left shift and a packed cell volume of 25% (RI: 25–48%). The biochemistry profile indicated mild azotaemia (P-creatinine: 211 µmol/l; RI: 80–229 µmol/l) and hypoalbuminaemia (P-albumin: 22 g/l; RI: 27–39 g/l). USG was 1.030 and the urine dipstick test demonstrated proteinuria (2+; ‘normal’ result: negative or trace) and haematuria (3+; ‘normal’ result: negative). Examination of the sediment revealed leukocyturia (>5 leukocytes per HPF) with intracellular rods and E coli was subsequently cultured. Abdominal ultrasonography revealed multiple rounded hypoechoic subcapsular lesions of variable size in both kidneys (Figure 5). Cytological examination of aspirates from these lesions revealed suppurative inflammation. Culture of the aspirates yielded E coli with the same sensitivity profile as the urinary strain.

Ultrasonography (US) of the right kidney of case 6 showing multiple well-circumscribed hypoechoic subcapsular areas (arrows). US aspect of both kidneys was similar
The cat was hospitalised for IV fluid therapy (lactated Ringer’s solution), and amoxicillin–clavulanic acid (Augmentin; GlaxoSmithKline) 25 mg/kg IV q8h, marbofloxacin (Marbocyl; Vetoquinol) 4 mg/kg IV q24h and buprenorphine (Buprecare; Axience) 10 µg/kg IV q8h, were administered. The cat’s condition improved rapidly and after 6 days it was discharged on marbofloxacin (Marbocyl; Vetoquinol) 2 mg/kg PO q24h with monthly follow-up visits. One month after discharge, P-creatinine was 164 µmol/l (RI: 80–229 µmol/l). The antibiotics were discontinued after 3 months and the urine cultures remained negative throughout the treatment period. Repeated ultrasonographic assessments of the kidneys showed a progressive improvement but persistence of residual lesions. At the time of writing, 2 months after treatment discontinuation, the cat’s CKD has remained stable in IRIS stage 2.
Discussion
RAs have been rarely reported in cats and little information is available about this condition.
The six cats in our small series were middle-aged to elderly and only one was male. The reasons for presentation were often non-specific (lethargy, decreased appetite) but, in most of the cases, PUPD was reported, potentially reflecting an underlying condition (ie, CKD, diabetes mellitus) or an alteration in the renal concentrating capacity secondary to RA (ie, secondary nephrogenic diabetes insipidus associated with an infection by a strain of E coli). Abdominal pain and nephromegaly were frequent clinical signs, with mild fever identified in only half of the cases. Fever and abdominal pain are the commonest clinical complaints in human patients with RA (70–84% and 44–78%, respectively).6–10
Not surprisingly, CBC often revealed leukocytosis and/or neutrophilia. Azotaemia and poorly concentrated urine were frequent and might have resulted from CKD or intrinsic renal damage caused by RA. All cats were diagnosed by ultrasonography and subsequent fine-needle aspiration. RA presented as hypoechoic and occasionally heterogeneous rounded lesions of various sizes. A subcapsular effusion was observed in 3/6 cases. Because of its greater sensitivity, computed tomography is preferred for the diagnosis of RA in humans, particularly in corpulent patients.11–13 A hypodense, distinctly marginated area not enhanced after contrast administration is classically observed within the renal parenchyma. 14
In human medicine, RAs are associated with numerous predisposing factors or comorbidities. In several retrospective case series, a predisposing factor could be identified in 74–92% of patients diagnosed with RA.6,9,10,15 Urinary tract diseases such as nephrolithiasis (5–52%), previous uncomplicated urinary tract infection (19–74%), renal neoplasia (4–16%), urinary obstruction (6–24%) and CKD (4–12%) are the most often cited.6–10,16 The most common general condition associated with RA in humans is diabetes mellitus, reported to be present in up to 62% of RA cases occurring in patients with an anatomically normal urinary tract. 15 Other conditions include immunosuppressive drugs or disease (HIV), liver disease or hypertension.6–10,16
CKD was diagnosed in all the cats in our series, and specific lesions, including perirenal pseudocyst and nephrolithiasis (n = 1), renal carcinoma (n = 2) and PKD (n = 1), were occasionally identified. In addition, one case was receiving progestagens and may have developed diabetes mellitus as a complication of this treatment. These results suggest that when a RA is identified in a cat, a search for a urinary or general predisposing condition should be carried out in every case.
In human medicine, RAs are classified as intrarenal or perirenal. Intrarenal corticomedullary abscesses are considered to result from an ascending urinary tract infection, whereas those located in the cortex are thought to develop secondarily to haematogeneous bacterial spread. 7 Perirenal abscesses would result from a ruptured intrarenal abscess or be of haematogeneous origin. 7 All our cats had intrarenal abscesses and in three of them, these abscesses were associated with perirenal extension of the infection. No cat had a perirenal abscess without also having an intrarenal abscess. In 4/6 cats, the same enteric microorganism (E coli), a very common urinary tract pathogen, was retrieved after urine and abscess culture, indicating that an ascending infection was most likely. Interestingly, all those cats were female and their urine was poorly concentrated, both characteristics being considered as risk factors for urinary tract infection. 17 In one cat, the urine culture was negative and RA culture yielded a group G Streptococcus, which suggested that the RA was of haematogenous origin.
Surgical treatment of RA can consist of open surgical drainage or partial or complete nephrectomy. This approach used to be widely practised in human medicine until improvements were achieved in antibiotic therapy and non-invasive interventional procedures. A small retrospective comparative study of immediate surgical treatment with percutaneous drainage of RA in humans did not demonstrate any difference in cure rates. 16 Surgery remains a treatment option in cases that are refractory to less invasive treatment, in the case of multi-loculated or septated abscesses, or when the abscess concerns a non-functioning kidney. 10 Surgical treatment was advocated for case 3 in our series because a tissue sample was considered pivotal for diagnosis. In case 4, surgery was proposed because the abscess was large and extended into the subcapsular space. Owing to financial constraints, surgical drainage was declined for case 1. Thus, surgical intervention regarding feline RAs needs to be considered on a case-by-case basis. Factors to be taken into consideration include the size of the abscess, its extension into the subcapsular space, the degree of azotaemia, the need for diagnostic samples or the failure of a non-invasive treatment. Whenever possible, nephrectomy should be avoided given the high incidence of CKD in the series described here.
Percutaneous abscess drainage is widely used to treat various abdominal abscesses in humans. 18 Depending on the authors, it is advocated for medium- (3–5 cm) to large-sized (⩾5cm) renal or perirenal abscesses.6–9 Several procedures have been described. They consist of inserting a catheter into the abscess cavity to drain the pus effectively, then flushing it with saline. 18 The catheter is generally left in place in a position that allows effective drainage until the clinical signs disappear and fluid production abates. 18 In our case, no such procedure was attempted. We do not think it advisable to leave such a catheter in place because of the risk of leakage associated with the mobility of the feline kidney. For the same reason, nephrostomy tube placement is not recommended in this species without surgical nephropexy. 19 Each of our cases was diagnosed by fine-needle aspiration of the lesion which, in cases 1, 2 and 5, allowed complete drainage of the abscess. When suspicion of RA is high prior to aspiration, puncture with a needle connected to a three-way stopcock should allow irrigation with saline before removal of the needle if the collected fluid has a purulent aspect.
In humans, medical treatment is advised for lesions <3 cm in stable patients. 8 However, one study reports a good success rate for lesions of 3–5 cm treated medically. 6 Perirenal abscesses are preferentially treated by percutaneous drainage, owing to the lower success rate with medical therapy alone.10,14 Medical treatment was successful in three of the cases in our series. In case 2, cure was assessed by normal renal follow-up ultrasonography. In cases 5 and 6, cure was determined from resolution of the renal lesion as assessed by ultrasonography and negative follow-up urine cultures.
The initial antibiotic treatment was probabilistic with one or two drugs administered intravenously but, because of the small number of observations, it cannot be stated whether a combination of drugs, rather than monotherapy, should be recommended. Subsequent long-term oral antibiotic treatment was based on the sensitivity profiles as the urine and/or purulent exudate cultures were positive in 5/6 cases. Dosage adjustments (ie, decreased dosing or increased intervals) might be relevant in cats with RA and an associated decline in renal function. This would theoretically be required for renally eliminated antibiotics/drugs with a narrow therapeutic index (ie, that could become toxic if accumulation occurred), which was not the case for the drugs used in our case series. It is difficult to draw any conclusions from this series regarding the recommended duration of antibiotic therapy. In human medicine, the antimicrobial treatment is maintained until complete clinical and radiological recovery, which is usually observed after 2–4 weeks. 14 The current recommendations regarding the management of complicated urinary tract infections in small animals advocate a 4 week course of therapy. 20 This recommendation may apply to RA, along with an observed resolution of the renal lesions at follow-up ultrasound examination.
Conclusions
To our knowledge, this is the first case series of RA reported in cats. It suggests that this condition most often presents as an acute episode in a pre-existing CKD and generally appears to develop in cats with at least one predisposing factor, thus warranting a complete medical evaluation. The treatment modality needs to be selected on a case-by-case basis, but the least invasive option should be preferred given the high incidence of CKD in this series. The short-term prognosis in most of the cases was fair but the long-term outcome was more variable and dependent on the underlying condition.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
