Abstract
Summary of cases:
A 6-month-old Burmese kitten developed focal skin lesions following a routine ovariohysterectomy. These were eventually attributed to the patient struggling during catheter placement and induction of anaesthesia. The lesions were caused by fluid extravasation in the subcutis and ischaemic necrosis of the overlying dermis, giving rise to an eschar-like appearance. Such lesions have been seen previously in Burmese cats with cutaneous asthenia and it is thought that they arise due to poor collagenous support for dermal blood vessels. An increased skin extensibility index (>23%) supported a diagnosis of cutaneous asthenia (Ehlers–Danlos-like syndrome), which has been reported as an inherited condition of Burmese cats in Australia, New Zealand and Europe. An additional Burmese cat with cutaneous asthenia is presented in detail, with lifetime follow-up and further salient observations by the owner, a veterinarian. Photographs of three other affected Burmese cats are provided to illustrate the range of presentations encountered with this condition. All five affected cats were presented with eschars, atrophic alopecia and increased skin extensibility, while one cat also had skin ulcers. Routine histopathological examination, including use of special stains such as trichrome, was unhelpful in establishing the diagnosis.
Clinical review:
The clinical features of this genetic disease of Burmese cats are reviewed, especially in relation to the postulated ‘vasculopathy’ that gives rise to characteristic skin lesions. Long term management of this condition is discussed briefly.
Introduction
Ehlers–Danlos syndrome (EDS) (synonym: hereditary collagen dysplasia) refers to a heterogeneous group of inherited connective tissue disorders caused by defective collagen synthesis or assembly. It is characterised by skin extensibility, tissue fragility and joint hypermobility.1 –4 Collagen is the key structural protein of the connective tissue matrix, providing it with strength, shape and an ability to resist deformation. These attributes make collagen integral to the functional integrity of skin, joints, muscles, ligaments, blood vessels, visceral organs and bone. Defective collagen synthesis can render these structures more deformable than normal.1 –4 In relation to the integument, the key feature of EDS is hyperextensibility and decreased tensile strength, manifesting clinically as skin stretching beyond normal limits, with a tendency to sag, because of loose attachment to subjacent tissues. Decreased tensile strength can lead to lacerations after minimal trauma.
The features of EDS were first described by Hippocrates in 400 BC. The syndrome is named after two human physicians, Edvard Ehlers from Denmark and Henri-Alexandre Danlos from France, who described the entity at the turn of the 20th century. This group of diseases has been observed in humans,1 –4 dogs,5,6 horses,7 –9 rabbits, 10 cats,11 –15 cattle, sheep and mink. 16 EDS in humans is caused by mutations in the genes coding for collagen and other fibrous proteins, or the enzymes involved in collagen assembly.1 –4,16 Such mutations cause alterations either in the structure or the post-translational processing of collagen, or in other proteins that interact with collagen. Defects affecting collagen integrity can weaken connective tissue, resulting in the various clinical manifestations that feature in this disorder.
In humans, a number of different types of EDS are recognised dependent on (i) the clinical phenotype and tissue involved (ie, skin, vasculature, joints, etc) and (ii) the molecular basis for abnormal collagen synthesis or assembly, which usually results in only certain forms of collagen being affected. One human classification system recognises six major types of EDS based on clinical, biochemical and molecular genetic features (Table 1).1 –4
The dermatosparaxis (‘torn skin’) form of EDS, characterised by severe skin fragility, is very rare in humans and inherited as an autosomal recessive trait.1 –4 Dermatosparaxis was first described in cattle, more than 20 years before being identified in humans. 16 Cutaneous asthenia (CA) (‘weak skin’) is a term generally restricted to animals and is synonymous with the dermatosparaxis form of EDS, in which clinical and pathological manifestations are largely restricted to skin. 16 CA in animals is rare and thought to be inherited, with autosomal dominant and autosomal recessive forms documented.5 –16 Affected animals have fragile skin that is thin, friable and hyperextensible.5 –16 Skin is therefore prone to injury, tearing easily, with associated cutaneous lesions occurring most commonly on the back and head.5 –16 Unlike EDS in humans, where wound healing is affected, animals with CA appear to have wound healing comparable to non-affected animals.16,17
In cattle and sheep, CA (dermatosparaxis) is inherited as an autosomal recessive trait, with a defect in procollagen peptidase demonstrated in some breeds. 16 In horses, a form of CA referred to as hereditary equine regional dermal asthenia (HERDA) is observed in Quarter Horses and related breeds, with a postulated autosomal recessive inheritance.7,8 Genetic tests are available for this mutation (G to A substitution at codon 115 on equine chromosome 1, in equine cyclophilin B). 18 In canine CA, autosomal dominant inheritance is proposed. 16 Interestingly, despite the same biochemical defect, the clinical picture may vary in nature and severity both between and within species. 16 There is no specific treatment for the disease, although management to prevent trauma to the skin can allow affected animals to live long lives of good quality.
In cats, CA is recognised as a presumed heritable defect in Himalayan 11 and Burmese14,15 breeds and some domestic shorthaired cats.12,13 Both dominant 13 and recessive11,14 autosomal inheritance has been proposed. The entity in Burmese cats is quite distinct from that reported in other cats with EDS in that lesions, consisting of necrotic eschars or atrophic alopecia, generally develop in the absence of skin lacerations.
The Burmese EDS-like phenotype was first described by Burton and colleagues, whose investigations were published in 2000 as a conference abstract. 14 Their study cohort consisted of nine Burmese cats (four male, five female), ranging in age from 5–12 months. Lesions consisted of purpura, necrotic eschars and atrophic alopecia. Tearing of the skin was not reported, in contrast to acquired skin fragility syndromes in cats reported in association with spontaneous or iatrogenic hyperglucocorticism, 19 feline infectious peritonitis and hepatic lipidosis, 20 cholangiohepatitis and hepatic lipidosis, 21 and multicentric follicular lymphoma. 22 Furthermore, acquired skin fragility syndromes generally occur in middle-aged to older cats with no previous history of skin tearing, and the skin is typically thin and fragile without being hyperextensible.
The skin extensibility index (SEI) of affected Burmese cats in Burton’s case series varied from 22–28%, 14 whereas the SEI in normal cats ranges from 11 ± 2% (mean ± SD) 23 to less than 19%. 13 Histological examination of lesional skin showed cutaneous infarction with wedge-shaped areas of epidermal and dermal necrosis. Adjacent skin showed follicular, dermal and epidermal atrophy. Routine histological sections from non-lesional skin were normal on light microscopic examination. Transmission electron microscopy (TEM) revealed marked variation in collagen fibril diameter, irregular collagen arrangement and decreased perivascular collagen deposition when compared with normal cats used as controls. Individual collagen fibrils showed ‘hair pin’ bends not seen in control cats. The clinical and histological appearance of the eschar-like primary skin lesions was consistent with vascular injury. Stretching of the blood vessels as a result of the hyperextensible skin or reduced collagen investment around vessels was proposed as the underlying basis for these lesions. Long-term follow-up of affected cats was not provided, and there was no indication of whether there was involvement of other structures or body systems; for example, whether joint laxity was present, heart valves were abnormal or whether affected cats developed degenerative joint disease prematurely.
The purpose of this report is to complement the conference abstract by Burton and colleagues with the provision of detailed case studies of two affected Burmese cats, photographs of macroscopic disease features in these cats and three additional cats, and discussion of the long term sequelae and aspects of clinical management.
Case summaries
Case 1
History and clinical findings
A 6-month-old female Burmese (2.6 kg) was presented 3 days following a midline ovariohysterectomy. The cat developed a raised swelling over the right scapula (approximately 20 mm diameter). This swelling appeared painful on palpation. The attending veterinarian’s presumptive diagnosis was a local reaction to either a non-steroidal anti-inflammatory injection (meloxicam) administered subcutaneously at this site or to amoxicillin clavulanate administered further dorsally. No treatment was prescribed and the cat was discharged for monitoring at home.
The cat was re-evaluated 7 days later (10 days postovariohysterectomy). The swelling over the right scapula had progressed to a raised, fluid-filled structure (60 mm diameter) covered by hard, dry, leathery skin (Figure 1a,b). The lesion appeared non-painful. The cat was reportedly a little subdued but was otherwise normal and eating well. The cat did not have any other cutaneous lesions and the ovariohysterectomy wound had healed uneventfully.
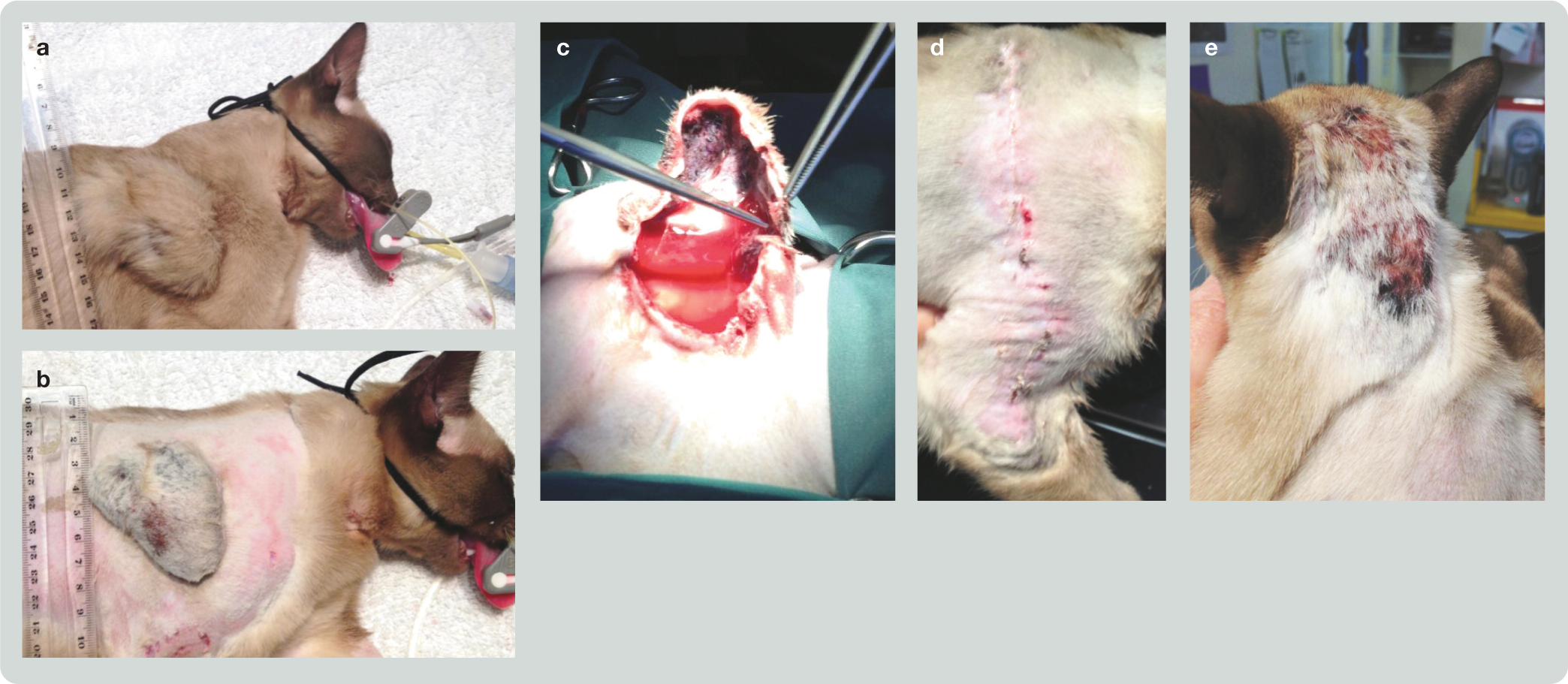
Case 1. (a) Ten days postovariohysterectomy: shoulder lesion prior to surgical debridement. (b) Same as (a), but after clipping and skin preparation. Note the blue discoloration of the eschar-like skin covering the lesion. The skin surrounding the lesion was easily torn during the pre-surgical clip. (c) Surgical excision of necrotic skin overlying the granulation tissue. (d) Twenty-two days postovariohysterectomy: the skin of this cat healed unremarkably by primary intention. (e) Thirty days postovariohysterectomy: the head lesion prior to surgical debridement
Diagnosis
The primary author (NH) recalled that Burmese cats had an increased risk of displaying CA and, at the suggestion of a feline specialist (RM), a qualitative skin extensibility examination was performed. Skin extensibility was obviously increased (see later), and a clinical diagnosis of CA was made. The swelling was attributed, retrospectively, to a traumatic incident that occurred during anaesthetic induction. It is the hospital’s protocol to premedicate surgical patients with a combination of acepromazine and methadone and then induce anaesthesia with alfaxalone via an indwelling cephalic catheter. This cat, however, resisted catheter placement and leapt from the table, falling awkwardly on its right side. Anaesthesia was subsequently induced using isoflurane in 100% oxygen delivered via a Bain system using a tight-fitting face mask.
As Burton et al 14 had proposed that weakness in collagenous support of arterioles causes a vasculopathy in affected cats subjected to minor trauma, this provided the most likely explanation for the lesion developing in this patient, with rupture and/or thrombosis of affected blood vessels causing cutaneous infarction over an accumulation of extravasated blood.
Surgical management and subsequent clinical course
The cat was admitted for excisional biopsy of the lesion. Pre-anaesthetic haematology and biochemistry values were within reference intervals (RIs). The patient was premedicated with acepromazine (0.03 mg/kg) and methadone (0.2 mg/kg) mixed in the same syringe and administered subcutaneously. Anaesthesia was induced with alfaxalone and maintained using isoflurane in 100% oxygen. Perioperative intravenous fluid therapy consisted of Hartmann’s solution (initially 10 ml/kg/h; adjusted based on indirect systolic blood pressure measurements recorded during the procedure).
A cavity containing blood-stained fluid was present under the necrotic epidermis and dermis, with a thin zone of granulation tissue at its base (Figure 1c). Necrotic skin and much of this granulation tissue was excised using a combination of sharp and blunt dissection and submitted for histopathological assessment. The wound was closed using a simple interrupted pattern of a synthetic polyamide suture (3/0 Monosyn).
Postoperatively, 50 mg cephalexin was administered intravenously and the same drug was subsequently given orally (75 mg PO q24h) when the cat was discharged, while tramadol (10 mg PO q12h) was given to provide postoperative analgesia. The surgery site healed well, although the cat developed an area of alopecia over the left scapula, possibly associated with a scratch. Sutures were removed 12 days later (Figure 1d).
Unfortunately, a further lesion developed on the patient’s head. This was managed conservatively with topical silver sulphadiazine (q12h for 5 days) but, by 30 days postovariohysterectomy, it had progressed to an eschar similar to the lesion removed from the shoulder (Figure 1e). The owners consented to surgical excision of the additional lesion. The anaesthetic and surgical procedures were similar to those used for the shoulder lesion. Healing was again unremarkable.
After all wounds had healed, quantitative skin extensibility testing was undertaken. The SEI was greater than 7 cm (vertical height of dorsal skin fold)/30 cm (length of the cat measured from tail base to occipital crest [crown–rump length]); ie, greater than 23% (Figure 2). Note that the endpoint for this test was hard to gauge as there were concerns about causing iatrogenic damage to the dorsal skin if the procedure was performed too forcibly.

(a,b) Skin extensibility examination in case 1. Note the extreme ‘tenting’ of the skin. The extensibility index was greater than 7 cm (height of skin
Histopathology
Microscopic evaluation of the lesion over the scapula revealed coagulative necrosis of the full thickness of the epidermis and much of the dermis. There was a moderate neutrophilic inflammatory infiltrate throughout the necrotic tissue, with myriad gram-positive cocci superficially and in some hair follicles. In the necrotic dermis, collagen fibre thickness, density and arrangement appeared normal. The underlying deep dermis and subcutaneous tissues contained a narrow zone of fibrovascular granulation tissue with moderate collagen deposition and a mild diffuse mixed cell inflammatory infiltrate. Beneath the zone of granulation tissue there was a mild multifocal perivascular infiltrate of mainly mononuclear inflammatory cells. Collagen fibre thickness, density and arrangement appeared normal in the viable deep dermis and subcutis.
Long-term management
To minimise the likelihood of trauma, including self-trauma, the nails on both cats in the household were trimmed and it was recommended that this be done regularly. A suggestion was made to try polyvinyl chloride (PVC) coverings (Catcaps; Narrabeen, NSW, Australia) manufactured to be attached to the cat’s nails using cyanoacrylate. Furthermore, an exclusively indoor lifestyle was strongly recommended to avoid cat fight injuries and other traumas. Finally, strict weight control was recommended, as excessive subcutaneous fat causes the skin of these patients to stretch ventrally, resulting in friction with the floor in extreme cases, or the requirement for cosmetic surgery.
Case 2
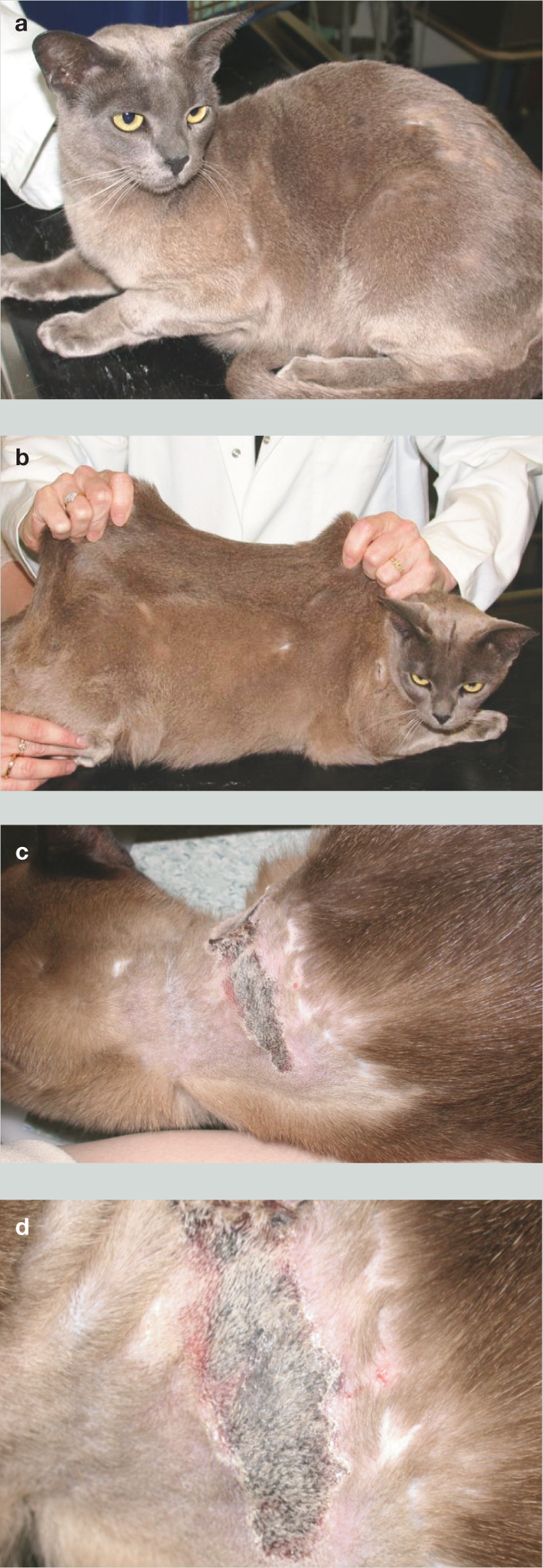
An approximately 12-month-old castrated Burmese was referred for investigation of recurrent ulcerated skin lesions (Figure 3a). Lesions were located on the dorsum and trunk. Biopsy of representative skin from the border of the ulcers failed to demonstrate any definitive aetiology. Previous ulcers had healed as shiny ‘cigarette burn’ eschars with focal alopecia (Figure 3b). An in-house consultation with a specialist veterinary dermatologist (Dr Ralf Mueller) led to a presumptive diagnosis of CA, although the SEI was measured and found to be within the normal RI. There was no overt evidence of ‘stretchy skin’, excessive skin or joint laxity. Due to the recurrent nature of the clinical signs the owners surrendered the cat, and it became a pet of one of the authors (SF).

Case 2. (a) ‘Billy’ at about 1 year of age with ulcerative skin lesions, but otherwise normal skin appearance. (b) An area of focal alopecia resulting from the initial eschar (arrow), just caudal to a pruritic lesion. (c) A small resolving ulcerated skin lesion on the left lateral neck. Later lesions developing in this location were often far more extensive. (d) Billy at approximately 5 years of age, with evidence of excessive skin between the forelimbs and around the head and neck. Note the tendency of the ventral thoracic skin to sag toward the ground. (e) At 11 years of age, the patient had markedly excessive skin folds sagging ventrally (arrow). The ocular lesions were likely attributable to feline herpesvirus type 1 infection
The initial presenting complaint did not persist into older age. Within 6–12 months, the cat no longer developed ulcerated skin lesions. Instead, there were repetitive episodes of self-trauma to the left lateral cervical skin (Figure 3c), with subsequent ulceration. Lesions bled excessively, consistent with a ‘vasculopathy’ triggering the pruritus. A woollen hiking sock was often taped around the patient’s neck to prevent recurrence, permitting the lesions to heal. This measure was well tolerated.
As a 4-year-old, the cat developed a small intestinal obstruction subsequent to ingestion of a foreign body. The surgeon who performed the enterotomy commented that the small bowel was unremarkable in appearance, thickness and estimated tensile strength. Post-surgical healing was uneventful. The patient was examined on a number of occasions by specialist surgeons and joints were judged to be of normal laxity.
The cat was extremely territorial. Although it was kept deliberately as an indoor cat with supervised outdoor access, occasional accidental excursions resulted in cat fights, with extreme tearing of large portions of integument. The skin was noticeably thin on handling but healed routinely with either suturing (single interrupted sutures of 3-0 synthetic polyamide) or stapling (eventually used for speed, ease and convenience). Routine histopathological examination of skin biopsies obtained at the time of suturing continued to show normal collagen morphology using conventional light microscopy and special stains such as trichrome. Sections of skin collected into glutaraldehyde for TEM were unfortunately never processed.
As the cat became middle-aged, the skin appeared excessive and, despite normal body condition, sagged due to the effect of gravity (Figure 3d,e). Interestingly, the SEI was obviously increased at this time (Figure 3d). The cat also ‘howled’ in a most disturbing fashion. It was uncertain whether it was experiencing pain or if this represented a partial seizure. Attempts to ease these episodes were unsuccessful and they would resolve without treatment, usually within 5–10 mins. The cat demonstrated unambiguous partial seizure activity after inadvertently receiving a high dose of enrofloxacin (5 mg/kg PO in the evening, then 10 mg/kg PO the following morning). The cat also developed an adverse behavioural response after receiving buspirone; this resulted in manic eating of cat toys, one of which resulted in the aforementioned small intestinal obstruction. Repeated neurological examinations could not be conducted as the cat’s ‘laissez faire’ attitude meant he would flop on his side if hopped. Similarly he would collapse gently onto his head when wheelbarrowed. The cat was without doubt capable of normal ambulation, could negotiate obstacles and managed to fight other cats.
The cat died suddenly when 11 years of age after a 48 h period of inappetence and malaise. A necropsy failed to reveal any gross lesions apart from some mucoid material in the frontal sinus and slightly flattened cerebral gyri. The patient led a reasonably long life. Substantial owner commitment was required and it was helpful that veterinary attention was available at subsidised rates.
Cases 3–5
Detailed case notes for three additional Burmese cats with CA were not easily retrievable; however, representative photographs are presented (Figures 4–6) to show the range of lesions evident in these affected cats.
Case 3, another Burmese cat with cutaneous asthenia. Note that without provocative testing the patient looks remarkably normal in appearance (a). However, skin extensibility testing (b) demonstrates marked tenting of the skin, unmasking the cutaneous asthenia phenotype. Characteristic ischaemic skin lesions on the dorsal cervical region are shown in (c) and close up in (d)

Case 4, a further Burmese cat with cutaneous asthenia. (a) The skin extensibility is obviously increased. (b) Characteristic ischaemic-type lesion occurring on the shoulder region. (c and d) Close-up views of the lesions, to emphasise their eschar-like surface. Photographs courtesy of Dr Michael Shipstone
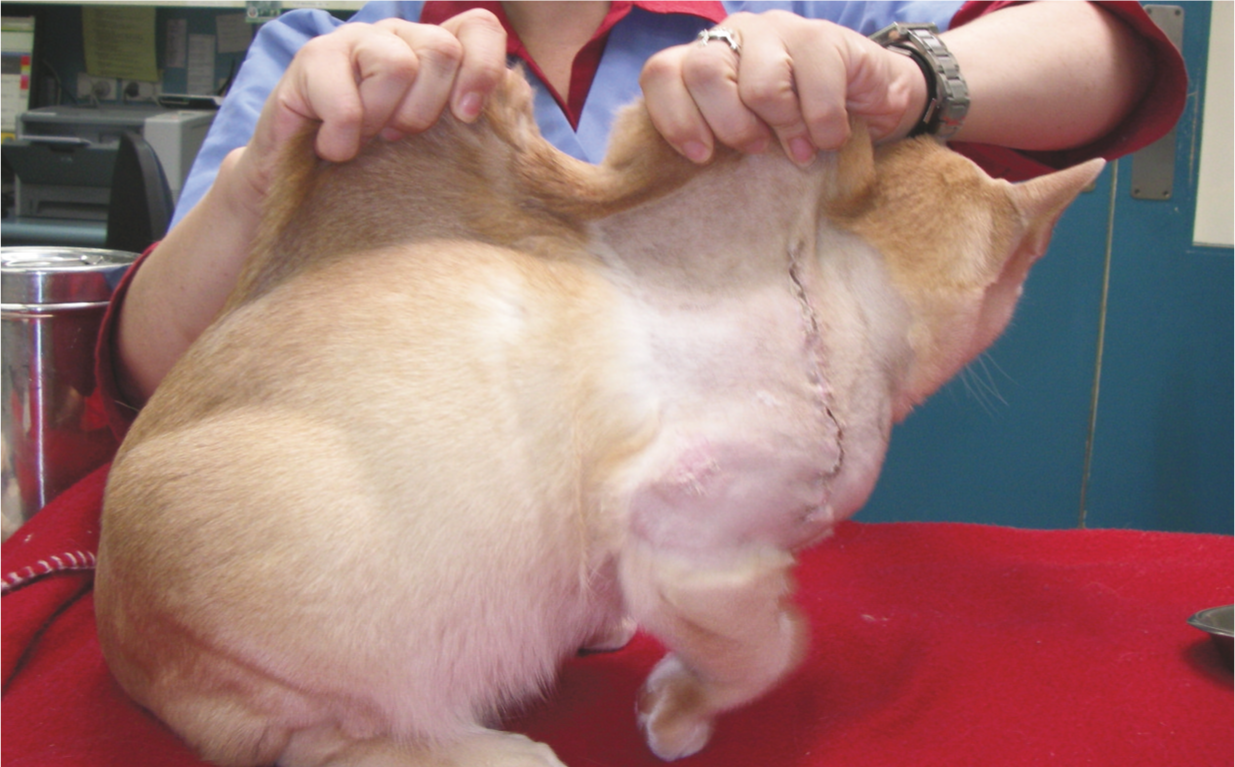
Case 5, yet another Burmese cat with cutaneous asthenia. Note the healing wound (following surgical excision of a large ischaemic lesion) on the shoulder and the obviously increased skin extensibility
Discussion
Feline CA is a rare disorder of collagen production. In Australia, there is a marked preponderance of Burmese cats among reported cases, strongly suggesting it is an inherited condition in this breed. This may be compounded by the fact that Burmese are currently the most commonly kept pedigree cat in Australia. 24 The stud, queen and siblings of case 1 of this report were normal on physical examination (with SEI values in the RI), which would support autosomal recessive inheritance, similar to that previously proposed for Himalayan cats. 11 Apparently, the original affected tom cat in Burton’s series was mated 50 times over 18 months and produced many normal litters, which also is consistent with a recessive mode of inheritance (G Burton, 2010, personal communication).
EDS in humans consists of a spectrum of clinical presentations, which are classified into several distinct types, ultimately explained by the underlying biochemical and genetic defect, in addition to the clinical phenotype.1 –4 In cats, there seems to be most similarity with the dermatosparaxis form of EDS, 1 hence the choice of the term CA. 14
The diagnosis of CA in case 1 was only considered following the development of the first lesion, initially believed erroneously to be an injection-site reaction. Clinical signs were not evident during the pre-neutering clinical examination, although skin extensibility testing was not performed as there was no index of suspicion for the condition at this time. With the benefit of hindsight, we would now recommend that this simple test, as illustrated in Figure 6, be performed on every Burmese cat presented for physical examination.
Case 1 was one member of a two-cat household and had been examined previously on several occasions for vaccination and microchip insertion, with no abnormalities detected. This cat first displayed lesions suggestive of CA ‘vasculopathy’ 14 only after complications arising from routine neutering. These lesions appeared to be grossly similar to those reported in HERDA-affected horses, but differed from the lesions reported in other affected animal species.7 –9,16
Retrospectively, we believe the initial lesion in case 1 was iatrogenic, induced by restraint for induction of general anaesthesia. We suspect that either ‘scruffing’ the cat (to provide firm but gentle restraint) or the subsequent fall to the floor resulted in the first lesion, while pressure applied to the back of the head to secure the face mask during anaesthetic induction may have contributed to the second lesion. The patient’s skin seemed particularly intolerant of pressure or friction, but healed satisfactorily following surgical intervention. Interestingly, both intestines, linea alba and subcutaneous tissues apparently heal unremarkably in Burmese cats with CA (G Hunt, 2001, personal communication).
In some cases of CA in animals, including cats, abnormalities in collagen can be appreciated on light microscopy. More commonly, however, abnormalities are not obvious or differences are subtle and difficult to discern except by comparison with breed- and age-matched controls. 16 In most cases, ultrastructural examination and biochemical analyses are required to confirm the collagen abnormality. Ultrastructural examination requires fixation of appropriate tissue specimens in glutaraldehyde and processing for TEM. Nevertheless, ultrastructural findings can be equivocal or difficult to interpret, and the presumptive diagnosis is more often based on history and clinical features (eg, as in HERDA-affected horses). In other animal species affected with cutaneous asthenia, TEM examination of the dermis may reveal an irregular and disorganised pattern of the fibrils that comprise the collagen fibres. Fibrils tend to be diminished in number, with variable spacing and random orientation within the fibre. Collagen fibres tend to be fewer in number, fragmented and shortened.1 –16 Usually there does not appear to be any alteration to the thickness of the epidermis or dermis.
In case 1, the histological appearance of skin was consistent with vascular injury, with haematoma formation and ischaemia contributing to a circumscribed necrotic swelling covered by an eschar. There was no obvious evidence of granulomatous inflammation, vasculitis, or other possible immune-mediated changes such as vesicle formation that may be seen with a cutaneous drug reaction, although the extent of epidermal and dermal necrosis complicated microscopic assessment. It was difficult to discern the collagen fibres and their arrangement in the superficial/mid-dermis because it was necrotic, though there was no obvious evidence of abnormal collagen. While this cat did not demonstrate histological evidence of abnormal collagen in the viable deep dermis/subcutis, the presumptive diagnosis of CA is almost certain to be correct given the clinical course and the markedly increased SEI value. No further biopsies were taken as the clinical picture strongly supported CA. Blood was collected from the patient, parents and siblings to provide sources of DNA for whole-genome sequencing.
The age at presentation for CA in cats seems to vary from a few months to 2 years, with the history often reporting an apparent ‘onset’ of hyperextensibility from 3 weeks to 4 months prior to presentation. Affected cats were reported to have ‘bleeding’ skin wounds and skin that was easily injured.11 –15 The frequency and severity of skin lesions and their tendency to recur in response to minor trauma (especially scratching) seem to be the critical reason for euthanasia in many cases.
In case 1, hyperextensibility of the skin was not appreciated until after the skin lesions were identified and the clinical team had considered potential aetiologies. Retrospectively, the owners observed many subtle indicators earlier in the cat’s life, such as wrinkling of the forehead and pendulous ventral skin over the belly, that may have facilitated an earlier diagnosis. Case 2 was diagnosed almost instantaneously by a veterinary dermatologist, presumably as the ‘illness script’ 25 for CA in Burmese cats was highly accessible to an elite clinician.
In reported cases of feline CA in Australia, there is an overrepresentation of Burmese cats and one report from Poland described a ‘Burman’ (sic) cat with Australian lineage (likely to be Burmese). 15 In case 1, the primary author (NH) examined both parents and a normal sibling; all were normal on physical examination, with normal SEI values. There is therefore the potential for further genetic investigations using a genome-wide association study approach using the feline single nucleotide polymorphism array 26 or by whole-genome sequencing this trio of closely related cats. CA cannot be treated, but if it is indeed transmitted as an autosomal recessive trait, then it can be prevented using appropriate molecular screening tests to identify both affected and carrier cats. This approach has recently been implemented for Burmese cats with hypokalaemia. 26
It is also important to document the natural history of this disease in cats that are not euthanased. There was no suggestion of joint laxity in either case 1 or case 2 on general physical examination, but specific quantitative orthopaedic testing was not conducted. Likewise there was no suggestion of a murmur due to mitral valve prolapse or pain referable to involvement of musculoskeletal structures such as tendons, ligaments, cartilage or bone. It was with great concern that we became aware after the death of case 2 that intractable migraines can be a feature of EDS in some human patients. 27 It is not known whether the distress and howling displayed by this cat were actually a manifestation of pain or headache. Brain histopathology and/or magnetic resonance imaging could have potentially addressed this question.
It could be argued that chronic administration of pentoxifylline may be useful for patients with CA to prevent the development of vasculopathic lesions, even though there is no evidence-based medicine to support this contention. Anecdotally, pentoxifylline is thought by many experts to be beneficial in the management of dogs with immune-mediated vasculitis. It was trialled in case 2, but not thought to be of any benefit.
Conclusions
The clinical features of CA in Burmese cats are quite characteristic, and as long as the attending clinician is familiar with the ‘illness script’, diagnosis by skin extensibility testing is straightforward. Unfortunately affected cats can have ongoing issues and many are euthanased as the owners become saddened and frustrated by the ongoing problems.
Without any doubt, the best way forward is to harness the power of modern genomics to determine the underlying molecular defect. Whole-genome sequencing of case 1 and its normal sibling and parents was instrumental in the very recent discovery of the genetic defect causing CA in Burmese cats (E Creighton, B Gandolfi and L Lyons, unpublished data). The authors anticipate that within a year a commercial PCR test will be available that will enable breeders and veterinarians to determine whether a cat is affected, a heterozygous carrier or unaffected using DNA obtained from cheek swabs or blood. Hopefully from this point in time onward, we will be able to prevent affected cats being produced by appropriate genetic counselling.
Footnotes
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Date accepted: July 6, 2015

