Abstract
Objectives
The aim of this study was to evaluate the analgesic efficacy of intraperitoneal (IP) bupivacaine in cats undergoing ovariohysterectomy (OVH).
Methods
Forty-five cats were included in a randomized, prospective, blinded study after owners’ written consent was obtained. The anesthetic protocol included acepromazine–buprenorphine–propofol–isoflurane. A ventral midline incision was made and cats (n = 15/group) were administered either IP saline 0.9% (negative and positive control groups; NG and PG, respectively) or IP bupivacaine (2 mg/kg; bupivacaine group; BG). Cats in the PG received meloxicam (0.2 mg/kg SC). An OVH was performed and postoperative pain was evaluated using a dynamic interactive visual analog scale (DIVAS), the UNESP-Botucatu multidimensional composite pain scale (MCPS) and mechanical nociceptive thresholds (MNT) for up to 8 h after the end of surgery. Postoperative sedation was evaluated using DIVAS. Rescue analgesia was provided with buprenorphine and/or meloxicam. Repeated measures linear models and a Cochran–Mantel–Haenszel test were used for statistical analysis (P <0.05).
Results
There was a significant effect of treatment on the number of times rescue analgesia was administered (P = 0.002) (PG, n = 2, 13%; NG, n = 12, 80%; BG, n = 4, 27%) with the number of rescues being higher in the NG group than in the PG (P = 0.0004) and BG (P = 0.02) groups. The DIVAS, MCPS and MNT were significantly different when compared with baseline values at different time points; however, data were not significantly different among groups.
Conclusions and relevance
Treatments PG and BG produced similar analgesia in terms of pain scores, number of times rescue analgesia was administered and MNT. Based on rescue analgesia, IP administration of bupivacaine provides analgesia in cats after OVH.
Introduction
Intraperitoneal (IP) administration of local anesthetics (LA) (ie, IP analgesia) such as bupivacaine reduces early postoperative analgesic requirements, pain scores and time to first intervention analgesia after abdominal surgery in humans.1–6 These findings have been corroborated by systematic reviews and meta-analyses, and IP analgesia is now recommended for laparoscopic surgery in humans as an adjuvant analgesic technique.7,8
In dogs undergoing ovariohysterectomy (OVH), pain scores after IP administration of bupivacaine were lower compared with a control group.9,10 Indeed, a recent study showed that the technique may blunt the surgery-induced stress response in dogs following laparoscopic OVH. 11 A panel of experts has recommended this technique for postoperative pain relief in dogs and cats. 12
To our knowledge, there are no studies reporting the use of IP analgesia in cats. The aim of this study was to evaluate the efficacy of IP bupivacaine in combination with an opioid in client-owned cats undergoing OVH, and compare these effects with a positive control group receiving an opioid and a non-steroidal anti-inflammatory drug (NSAID), and a negative control group receiving the opioid alone. We hypothesized that the prevalence of rescue analgesia and pain scores would be lower, and mechanical nociceptive thresholds (MNT) would be higher in both positive and testing groups when compared with negative controls.
Materials and methods
Animals
The study protocol was approved by the animal care committee of the Faculty of Veterinary Medicine, University of Montreal (14-Rech-1744). Forty-five client-owned mixed-breed healthy female cats scheduled for elective OVH were enrolled in a prospective, blinded, randomized, controlled, clinical trial. Owners of all cats included in the study provided written informed consent. Cats were admitted to the veterinary teaching hospital at the Faculty of Veterinary Medicine, University of Montreal, at least 24 h before surgery. They were housed individually in adjacent cages in a room exclusively for cats. Cats were included if they were considered healthy, based on medical history, complete physical examination and hematology (hematocrit and total protein) testing. Exclusion criteria included aggression, cardiac arrhythmias, pregnancy, lactation, obesity (body condition score >7 on a scale from 1–9), anemia and clinical signs of disease. Abdominal ultrasonography was performed on the day before surgery to ensure that cats were not pregnant.
Anesthetic protocol and surgery
Food, but not water, was withheld for up to 12 h before anesthesia. Cats were premedicated with acepromazine (0.05 mg/kg Atravet Acepromazine maleate, USP; Boehringer Ingelheim) and buprenorphine (0.01 mg/kg Vetergesic; Champion Alstoe Animal Health) injected into the epaxial muscles. Approximately 20 mins later and under aseptic conditions, a 22 G intravenous (IV) catheter (BD Insyte W, BectonDickinson Infusion Therapy System) was inserted into a cephalic vein. Anesthesia was induced with IV propofol (Diprivan 1%; AstraZeneca Canada) to effect. Lidocaine (2 mg) was instilled on the vocal cords, and cats were intubated with an appropriately sized, cuffed endotracheal tube. Anesthesia was maintained with isoflurane (Isoflurane USP; Pharmaceuticals Partners of Canada) administered with 100% oxygen using a non-rebreathing circuit with an oxygen flow rate of 200 ml/kg/min. Cats were positioned in dorsal recumbency over a circulating warm-water blanket. Briefly, monitoring was performed using a multi-parametric monitor (Lifewindow TM 6000V Veterinary Multiparameter Monitor; Digicare Animal Health), which included electrocardiography, capnography, inspired and expired concentrations of isoflurane, pulse oximetry and esophageal temperature. Blood pressure was measured with a Doppler ultrasonic flow detector. Lactated Ringer’s solution was administered at a rate of 10 ml/kg/h IV throughout surgery.
OVH was performed by a single surgeon (BM) and anesthesia was administered by a board-certified veterinary anesthesiologist (PVS). Briefly, a 3–4 cm long ventral midline incision was made through the skin, subcutaneous tissues and aponeurosis of the rectus abdominis muscle, and a modified three clamp technique was used. The abdominal wall and subcutaneous tissues were closed with a simple continuous pattern of absorbable suture material. The skin was closed with an intradermal suture pattern. Surgery time (time elapsed from the first incision until placement of the last suture), anesthesia time (time elapsed from injection of propofol to turning off the vaporizer dial) and time to extubation (time elapsed from turning off the vaporizer dial until extubation) were recorded for each cat. Extubation was performed once the cats’ palpebral reflexes were evident, as is commonly performed at our institution.
Treatment groups
Cats were randomly assigned using an online software program (www.randomization.org) to receive one of the following three treatments (n = 15/group): saline 0.9% IP (0.9% Sodium Chloride USP; Hospira) (negative control group [NG]); saline 0.9% IP and meloxicam SC (0.2 mg/kg Metacam 0.5%; Boehringer Ingelheim) (positive control group [PG]); or bupivacaine IP (2 mg/kg Sensorcaine, Bupivacaine HCl 0.5% USP; AstraZeneca Canada) (bupivacaine group [BG]). In the BG, the solution of bupivacaine was diluted with an equal volume of isotonic saline 0.9%, resulting in a final concentration of 0.25%. The same volume of bupivacaine was administered in the PG and NG using saline 0.9%. For the IP injection, the solution was equally divided in three parts and equivalent volumes were instilled into the peritoneal space, specifically in its right and left ovarian pedicles, and caudal uterus immediately before OVH using a 3 ml syringe attached to a 22 G catheter. Surgery was carried on approximately 2 mins later.
Pain and sedation scores
Assessments were made by two observers who were blinded to the analgesic treatments. These individuals did not interfere with each other’s assessment of pain. The first observer (JB) assessed pain and sedation scores as part of the present study. The second observer (AML) evaluated pain as part of a different study. Pain and sedation were evaluated using a dynamic and interactive visual analog scale (DIVAS).13–15 The DIVAS was derived by using a 100 mm bar where ‘0’ corresponds to ‘no pain’ or ‘no sedation’ and ‘10’ corresponds to the ‘worst pain’ or ‘deepest sedation’ that the observer imagined could ever arise from an OVH. Pain was also evaluated using the UNESP-Botucatu multidimensional composite pain scale (MCPS), which is a valid, reliable and responsive instrument that involves different domains such as ‘pain expression’ and ‘psychomotor change’. 16 For scoring, the cats were initially evaluated inside their cages without being disturbed; then they were gently handled and encouraged to move around while the incision site and abdominal area were palpated. 16 The DIVAS and MCPS evaluations were performed 60 mins prior to the induction of anesthesia (time 0) and at 0.5, 1, 2, 3, 4, 6 and 8 h following the end of surgery.
MNT was measured by applying the tip of a commercial algometer device (Bio-algo-GT; EB instruments). The tip was applied with steadily increasing force within approximately 1 cm of the surgical wound until the cat reacted (aversion to the device, vocalization, opening of the mouth as if to vocalize, attempts to bite or scratch the device or the operator).13,17 The stimulus was randomly applied along the lateral side of the wound by one experienced individual (JB) but not always at the exact same location. It was immediately stopped and the holding force (Newtons [N]) was recorded. This was performed twice with an interval of 30 s between stimuli, and the mean value was used as the MNT for that time point. The MNT was assessed at 0 and 2, 4, 6 and 8 h following surgery, and after DIVAS and MCPS evaluations.
Rescue analgesia was provided if MCPS scores were ⩾6 and consisted of SC administration of meloxicam (0.2 mg/kg) in the NG and BG, and IV administration of buprenorphine (0.02 mg/kg) in all groups. Data collected after the administration of rescue analgesia were not included in the statistical analysis; however, cats continued to be monitored for additional need of analgesics. A second dose of buprenorphine (0.02 mg/kg, IM) was administered to cats at the end of the study period or at any time point if needed. Postoperative treatments were administered by two investigators who were not involved with pain assessment (PVS and BM). The number of cats requiring rescue analgesia and the number of times that rescue analgesia was administered for each individual were recorded.
Statistical analysis
Sample size calculations were performed before the study had begun. Overall, the prevalence of rescue analgesia was treated as the main outcome of this study as data from cats receiving supplemental analgesia were discarded afterwards. The authors assumed that the prevalence of treatment failure would be 70% in the NG and 20% in the treated groups (either PG or BP). 10 A power analysis with Fisher’s exact test indicated that a sample size of 15 would be needed in each group to detect such a difference in 80% of the times at the α level of 5%. From Giordano et al, 15 the prevalence of rescue analgesia in cats ranged from 15–68%, falling in the above range.
Data were tested for normality with a Shapiro–Wilk test. Demographic data for each treatment group were analyzed using one-way ANOVA or χ2 test. The number of cats receiving rescue analgesia was analyzed in two separate ways. Temporal changes within their treatment group (response variable ‘number of rescues’) was analyzed with the Cochran–Mantel–Haenszel test for ordinal scores followed by pairwise comparisons between analgesic treatment groups. A separate subgroup restricted to the cats that received rescue analgesic treatment (response variable ‘rescue time’) was analyzed with a general linear model with treatment as a factor; only the first rescue time was used for analysis.
Mean MCPS, mean DIVAS for pain and sedation before, at 30, 60 and at 120 mins after surgery, and the mean MNT before and at 120 mins after surgery were investigated with repeated measures linear models with treatment class as a between-subject factor and time as a within-subject factor followed by Bonferroni correction for multiple comparisons. These analyses were not performed after the 2 h time point because most of the cats in the NG received rescue analgesia and data were discarded afterwards. Group comparisons would have been inappropriate. Statistical analyses were performed by a statistician (GB) with standard software (SAS, version 9.3; SAS Institute). Values of P <0.05 were considered significant.
Results
Table 1 shows body condition score, body weight, hematocrit, total protein, anesthesia and surgery times, and time to extubation. These variables were not significantly different among treatments. All cats were discharged from hospital at least 24 h after surgery; none of the cats developed postoperative complications.
Body condition score (BCS), body weight, hematocrit, total protein, anesthesia and surgery time, and time to extubation in cats undergoing ovariohysterectomy
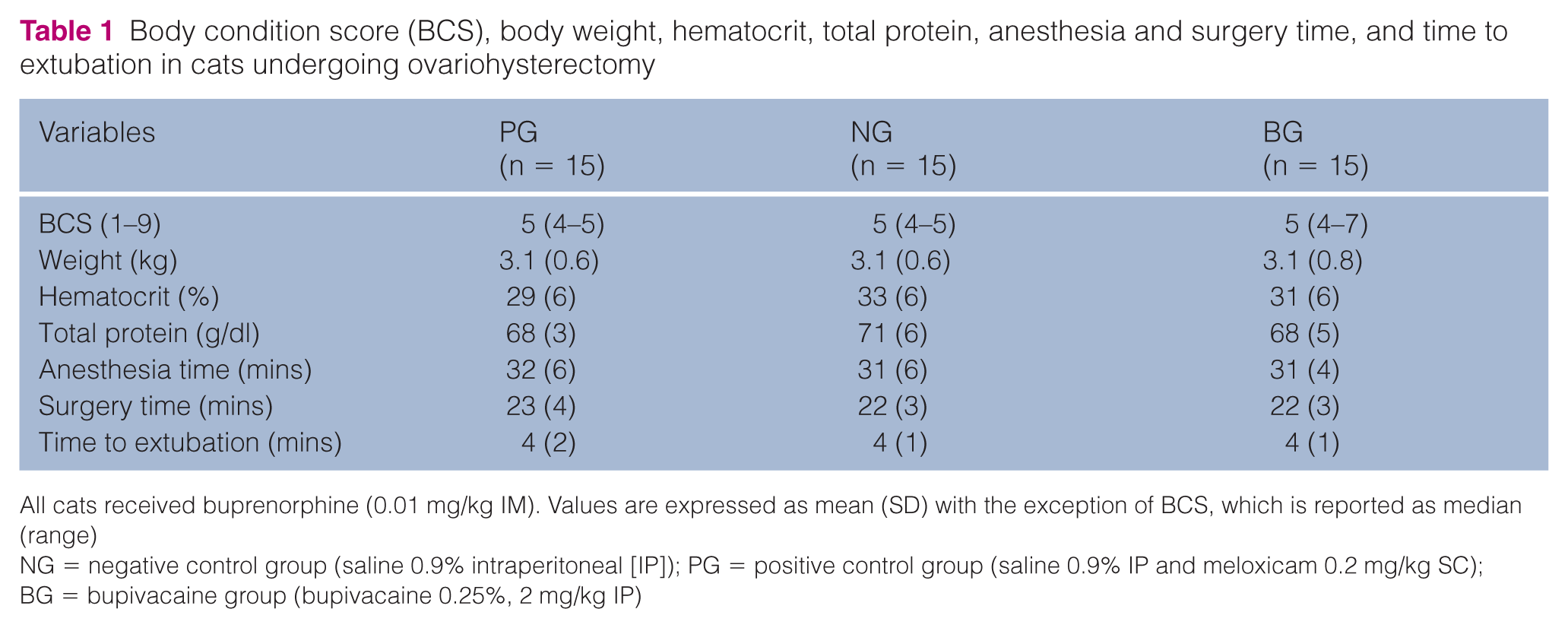
All cats received buprenorphine (0.01 mg/kg IM). Values are expressed as mean (SD) with the exception of BCS, which is reported as median (range)
NG = negative control group (saline 0.9% intraperitoneal [IP]); PG = positive control group (saline 0.9% IP and meloxicam 0.2 mg/kg SC); BG = bupivacaine group (bupivacaine 0.25%, 2 mg/kg IP)
A total of 18 cats required rescue analgesia (PG, n = 2, 13%; NG, n = 12, 80%; BG, n = 4, 27%) (Table 2). There was a significant effect of treatment on the number of times rescue analgesia was administered (P = 0.002). The prevalence of rescue analgesia was significantly greater in the NG vs PG (P = 0.0004) and the BG (P = 0.02), but not between the PG and BG (P >0.05). There were no significant differences among groups for the number of cats requiring a second dose of rescue analgesia (PG, n = 0; NG, n = 1; BG, n = 1). Furthermore, when the analysis was restricted to cases in which rescue analgesia was administered, there were no significant differences among groups regarding timing for first rescue.
Number of cats receiving rescue analgesia over time following ovariohysterectomy. Table includes the first administration of rescue analgesia

NG = negative control group (saline 0.9% intraperitoneal [IP]); PG = positive control group (saline 0.9% IP and meloxicam 0.2 mg/kg SC); BG = bupivacaine group (bupivacaine 0.25%, 2 mg/kg IP)
There were no significant differences among groups for the number of cats requiring a second dose of rescue analgesia (PG, n = 0; NG, n = 1; BG, n = 1). Rescue analgesia was provided with administration of meloxicam (0.2 mg/kg SC) in the NG and BG groups, and buprenorphine (0.02 mg/kg IV) in all the groups, if the UNESP-Botucatu multidimensional composite pain scale scores were ⩾6. All cats received buprenorphine (0.01 mg/kg IM)
There was a significantly higher incidence of treatment failure in the NG group when compared with the PG group (P = 0.0004) and BG group (P = 0.02)
The DIVAS sedation scores were not significantly different among treatments (Figure 1). Sedation scores were significantly increased in the PG at 0.5 h (P <0.0001), in the NG at 0.5 h (P <0.0001) and 1 h (P = 0.0002), and in the BG at 0.5 h (P <0.0001) when compared with baseline values (Figure 1). The DIVAS pain scores were not significantly different among treatments (Figure 2). These pain scores were significantly increased in the PG at 0.5 h (P = 0.002), 1 h (P = 0.0008) and 2 h (P = 0.0006), in the NG at 0.5 h, 1 h and 2 h (P <0.0001 at all time points), and in the BP at 1 h and 2 h (P <0.0001, both time points) (Figure 2). The MCPS scores were not significantly different among treatments (Figure 3). The MCPS scores were significantly increased for all treatments at 0.5 h, 1 h and 2 h (P <0.0001 for all groups and time points) when compared with baseline values (Figure 3).

Mean ± SEM dynamic and interactive visual analog scale (DIVAS) sedation scores in cats following ovariohysterectomy. DIVAS sedation scores were not significantly different among treatments. All cats received buprenorphine (0.01 mg/kg IM). NG = negative control group (saline 0.9% intraperitoneal [IP]); PG = positive control group (saline 0.9% IP and meloxicam 0.2 mg/kg SC); BG = bupivacaine group (bupivacaine 0.25%, 2 mg/kg IP). *Significantly increased in the PG at 0.5 h (P <0.0001), in the NG at 0.5 h (P <0.0001) and 1 h (P = 0.0002), and in the BG at 0.5 h (P <0.0001) when compared with baseline values

Mean ± SEM dynamic and interactive visual analog scale (DIVAS) pain scores in cats following ovariohysterectomy. DIVAS pain scores were not significantly different among treatments. All cats received buprenorphine (0.01 mg/kg IM). NG = negative control group (saline 0.9% intraperitoneal [IP]); PG = positive control group (saline 0.9% IP and meloxicam 0.2 mg/kg SC); BG = bupivacaine group (bupivacaine 0.25%, 2 mg/kg IP). *Significantly increased in the PG at 0.5 h (P = 0.002), 1 h (P = 0.0008) and 2 h (P = 0.0006), in the NG at 0.5 h, 1 h and 2 h (P <0.0001 at all time points), and in the BP at 1 h and 2 h (P <0.0001, both time points) when compared with baseline values

Mean ± SEM multidimensional composite pain scale (UNESP-Botucatu MCPS) pain scores in cats following ovariohysterectomy. MCPS pain scores were not significantly different among treatments. All cats received buprenorphine (0.01 mg/kg IM). NG = negative control group (saline 0.9% intraperitoneal [IP]); PG = positive control group (saline 0.9% IP and meloxicam 0.2 mg/kg SC); BG = bupivacaine group (bupivacaine 0.25%, 2 mg/kg IP). *Significantly increased for all treatments at 0.5 h, 1 h and 2 h (P <0.0001 for all groups and time points) when compared with baseline values
Owing to technical issues, MNT were performed in only 35/45 cats (PG, n = 12; NG, n = 11; BG, n = 12). MNT were not significantly different among treatments at any time point (Table 3). MNT were significantly decreased for all treatments at 2 h (PG and NG, P <0.0001; BP, P = 0.0003) when compared with baseline values.
Mean (SEM) of mechanical nociceptive threshold (MNT) (Newtons [N]) in cats

MNT values were not significantly different among treatments (P ⩾0.05). All cats received buprenorphine (0.01 mg/kg IM).
NG = negative control group (saline 0.9% IP); PG = positive control group (saline 0.9% IP and meloxicam 0.2 mg/kg SC); BG = bupivacaine group (bupivacaine 0.25%, 2 mg/kg IP)
Significantly decreased for all treatments at 2 h (PG and NG, P <0.0001; BP, P = 0.0003) when compared with baseline values
Discussion
This study demonstrated that IP administration of bupivacaine in combination with preoperative buprenorphine provides effective early postoperative analgesia in cats undergoing OVH; the prevalence of rescue analgesia was similar to a positive control group receiving meloxicam and buprenorphine, and lower when compared with a group receiving buprenorphine alone. In addition, DIVAS pain scores in the BG were not significantly increased at 0.5 h when compared with baseline values; the same finding was not observed in the NG and PG, and could potentially show that cats in the BG could be significantly more comfortable in the early (30 mins) postoperative period.
The IP technique showed to be a simple adjunctive analgesic method to reduce pain after OVH in cats for the early postoperative period. Considering that (1) local anesthetics are non-controlled and low-cost drugs that are available worldwide and (2) there are a large number of cats that undergo OVH every year, IP administration of bupivacaine may represent a significant advance in pain management with immediate impact in feline clinical practice. However, it is important to highlight that IP bupivacaine is not considered a ‘stand-alone’ analgesic technique and the study protocol included the administration of buprenorphine by the intramuscular route in all groups. This study also suggests that the administration of IP bupivacaine or an NSAID (meloxicam) in combination with buprenorphine may not provide adequate postoperative analgesia for all cats undergoing OVH. Therefore, postoperative pain should be evaluated on a case-by-case basis; each individual is unique and it might require further administration of analgesics based on pain assessment.
In dogs, IP administration of bupivacaine blunted the postoperative stress response to laparoscopic OVH and reduced postoperative pain. 11 Pain scores after IP bupivacaine were lower than with IP saline in two canine studies.9,10 In agreement with our study, these findings suggest that there is now enough evidence that IP analgesia provides postoperative pain relief after OVH in dogs and cats. As part of a multimodal analgesia protocol, we routinely administer IP bupivacaine in combination with an opioid and an NSAID for OVH in cats.
Pain scores (DIVAS and MCPS) were not significantly different among treatments. However, statistical analysis included data excluded after the administration of rescue analgesia. This approach may overestimate the analgesic effect of a treatment because higher pain scores are possibly omitted and analysis bias is introduced. The loss of data leads to uneven group sizes limiting analysis. In addition, the DIVAS is a subjective evaluation method that has not been validated in cats and may be subject to large inter-observer variation and a high amount of variability in pain scores. 18 However, the DIVAS is thought to have more sensitivity than the numerical rating scale or the simple descriptive scale. 19 The MCPS is a validated pain scale for use in cats in the perioperative period. 16 Analysis bias may have also occurred with the MCPS after data exclusion. The MCPS did not detect significant differences among treatment groups; however, its cut-off for rescue analgesia was sensitive to treatment effect. Overall, there was a trend for lower DIVAS pain scores and MCPS in the PG and BG when compared with the NG but the P values were not significant after the Bonferroni adjustment.
The prevalence of rescue analgesia was significantly greater in cats receiving buprenorphine alone (NG) when compared with the PG and BG. In fact, 80% of cats in this group required additional doses of analgesics showing that, at the dosage regimens used here, a single dose of buprenorphine provides inadequate control of postoperative pain in cats as it has been previously reported even using higher doses. 20 In a previous report, higher doses of buprenorphine (0.02 mg/kg) significantly increased MNT than the dose used in the present study (0.01 mg/kg). 21 It is possible that lower pain scores could have been observed if higher doses of buprenorphine had been used.
The MNT baseline values of approximately 13–15 N were similar to previous studies using the same device. 13 Similarly, thresholds were significantly reduced following surgery herein as in the aforementioned study. However, our study did not detect significant differences in MNT among treatments at 2 h after extubation. The lack of significant changes in MNT may be also explained by analysis bias as previously mentioned. Further studies are warranted to validate this MNT device using a larger number of cats. The employment of such a device in this study was an attempt to add objective measurement tools as part of our clinical pain assessment.
There are some limitations with this study. First, there was a lack of significant differences among treatments using pain scoring systems. This study was designed to evaluate eight postoperative time points. However, there was a high incidence of rescue analgesia in the NG (80%), which precluded further treatment comparisons. However, the prevalence of rescue analgesia was a sensitive outcome method to differentiate treatment efficacy. Second, one may argue that plasma concentrations and pharmacokinetics following IP administration of bupivacaine have not been determined in cats making any clinical recommendations difficult. Nevertheless, before this study had begun, a safety study was performed in eight healthy cats after IP administration of bupivacaine 0.25% (2 mg/kg) (unpublished data). Peak plasma concentration of the drug was approximately half of the concentration that has been reported to cause arrhythmias or convulsive electroencephalography in cats (3.6 ± 0.7 μg/ml), and about one-sixth of that required to produce hypotension.22,23 Third, it has been suggested that bupivacaine concentrations >0.25% should be used as lower concentrations may decrease efficacy.24–26 In the present study, a final concentration of bupivacaine 0.25% was administered and it is not known whether using a higher concentration would increase the magnitude and duration of analgesia. We chose to dilute the drug to achieve a reasonable volume of anesthetic spread. In addition, the doses of bupivacaine used here were based on clinical experience and it is not clear how different doses would have affected the results of our study. The dose of 2 mg/kg is lower than the dose that has been reported for cardiotoxicity.22,23 Fourth, pain was assessed for up to 8 h postoperatively. It is not possible to know what the duration of action of IP bupivacaine is. Finally, it is not clear how sedation affects pain assessment. Therefore, DIVAS sedation scores were included in this study and were not significantly different among treatments. However, DIVAS sedation scores were increased at 1 h in the NG but not in the BG or PG when compared with baseline values. It is not clear how this may have affected pain assessment as pain scores were not different among treatments.
Conclusions
IP administration of 2 mg/kg of bupivacaine (0.25%) in combination with a single dose of preoperative buprenorphine provided early postoperative analgesia. Treatments in the PG and BG produced similar analgesia in terms of pain scores, amount of rescue analgesia and MNT. The technique is a safe, simple and efficacious adjuvant analgesic method to reduce pain after OVH in cats; however, some animals may require additional administration of analgesics (opioid and NSAID) for pain control.
Footnotes
Acknowledgements
‘Centre Hospitalier Universitaire Vétérinaire’ (CHUV), Faculty of Veterinary Medicine, University of Montreal.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by the Morris Animal Foundation. A-ML received a scholarship from the Fonds de Recherche Clinique Zoetis (Canada).
