Abstract
Objectives
The aim of the study was to evaluate, in a controlled, randomised, masked clinical trial, the influence of administration rate of alfaxalone at induction on its relative potency in cats and to report the incidence of cardiorespiratory adverse effects.
Methods
Twelve healthy female domestic cats admitted for ovariohysterectomy were premedicated with buprenorphine 20 µg/kg intramuscularly and alfaxalone 3.0 mg/kg subcutaneously. Sedation scores were established (using a published scale ranging from 1 [no sedation] to 5 [profound sedation]) prior to anaesthesia induction with alfaxalone intravenously at 2 mg/kg/min (group A2; n = 6) or 0.5 mg/kg/min (group A0.5; n = 6) to effect until orotracheal intubation was achieved. Sedation scores and alfaxalone induction doses were compared between the groups, using a Mann–Whitney exact test. Results are reported as median and range. Presence of apnoea (no breathing for more than 30 s) or hypotension (mean arterial blood pressure <60 mmHg) within 5 mins postintubation was also reported.
Results
Although sedation scores (1.5 [range 1.0–3.0] and 2.5 [range 1.0–3.0] for A2 and A0.5, respectively) were not significantly different (P = 0.32), cats in group A2 required significantly more alfaxalone (4.3 mg/kg [range 3.4–7.0 mg/kg]) than group A0.5 (2.1 mg/kg [range 1.5–2.5 mg/kg]) (P = 0.002). Two cats in each group presented postinduction apnoea, and two cats in group A2 and three cats in group A0.5 presented postinduction hypotension.
Conclusions and relevance
The use of a slower induction infusion rate resulted in an increase in the alfaxalone relative potency without obvious cardiorespiratory benefit.
Introduction
Previous studies in cats have found a dose-dependent decrease in heart rate (HR), cardiac output and arterial blood pressure following induction of anaesthesia with alfaxalone.1,2 Alfaxalone also induces dose-dependent decreases in respiratory rate (RR) and tidal volume.3,4 These effects support alfaxalone titration whenever administered intravenously (IV). 5 In humans, using slower infusion rates of propofol allowed induction of anaesthesia with significantly smaller doses than faster rates, causing less cardiovascular and respiratory depression. 6 The aim of the present study was to evaluate the influence of alfaxalone administration rate at induction of anaesthesia on its relative potency in cats and to report the incidence of cardiorespiratory adverse effects. The hypothesis was that a slower induction infusion rate would achieve anaesthesia using a smaller alfaxalone dose.
Materials and methods
The randomised controlled masked clinical trial was approved by the animal ethics committee of the University of Melbourne Faculty of Veterinary and Agricultural Sciences. Between February 2014 and September 2014, domesticated female cats of 16 weeks to 4 years of age that were admitted to the University Veterinary Clinic and Hospital for elective ovariohysterectomy were included in the trial after agreement and signed consent was obtained from the owners. The cats were determined to be healthy by means of thorough physical examination and basic blood analysis (volume of packed red blood cells, total proteins, glucose, urea). Using a commercially available software package (Microsoft Excel for Mac 2011), 12 cats were randomly allocated to group A2 (n = 6) or group A0.5 (n = 6) groups by one of the authors (SHB).
Cats were premedicated with buprenorphine (20 µg/kg IM) (Temgesic; Reckitt Benckiser) and alfaxalone solubilised in 2-hydroxypropyl-β-cyclodextrin (3.0 mg/kg SC) (Alfaxan; Jurox). Twenty minutes later, sedation level was assessed using a published scoring system (Table 1), and a 22 G catheter (Optiva; Smiths Medical) was placed in a cephalic vein. 7 Anaesthesia induction commenced 30 mins after premedication using alfaxalone administered IV at 2 mg/kg/min for group A2 or 0.5 mg/kg/min for group A0.5 until orotracheal intubation was achieved without coughing. Orotracheal intubation was attempted once the cat was not supporting its head, the jaw tong was relaxed and the cat was not retracting its tongue. If coughing occurred, the intubation process was paused for 15 s.
Sedation scores were ascribed following a published scale 7

The time that elapsed from the beginning of intravenous infusion until intubation was defined as induction time. The endotracheal tube was connected to a paediatric rebreathing system and isoflurane in oxygen was administered to effect. Alfaxalone was administered via a calibrated infusion pump (Baxter Colleague 3CX Volumetric Infusion Pump; Baxter Healthcare).
A multi-parametric anaesthesia monitor (Vet Advisor Vital Signs Monitor V9203; SurgiVet, Smiths Medical) was used to monitor electrocardiogram, HR, RR, partial pressure of end-tidal carbon dioxide (PET
HR was monitored continuously starting at induction and MABP was measured immediately postintubation and every 3 mins thereafter.
Postoperative analgesia consisted of meloxicam (Metacam 5 mg/ml; Boehringer Ingelheim) 0.2 mg/kg SC and buprenorphine 0.02 mg/kg IV if required.
All cats were assessed for sedation, monitored and their trachea intubated by the same trained anaesthetist (LNW) who was unaware of the treatment. Sedation scores, alfaxalone anaesthesia induction doses and duration of the inductions were compared between the groups (Mann–Whitney test, P <0.05). The presence of excitement during induction, bradycardia (HR <95 beats per min), hypotension (MABP <60 mmHg) or apnoea (no evidence of breathing for 30 s or more) within the first 5 mins postintubation were recorded. Positive pressure ventilation (PPV) was only implemented if the cat became hypoxaemic (SpO2 <90%). Results are reported as median and range.
Results
For the following parameters there was no statistically significant difference noted between the two groups: median weight was 2.5 kg (range 2.0–4.6 kg) and 2.5 kg (range 1.9–3.7 kg), for groups A2 and A0.5, respectively (P = 0.70). Median age was 9 months (range 5–24 months) and 12 months (6–16 months) for groups A2 and A0.5, respectively (P = 0.60).
Sedation scores, doses of alfaxalone used, induction times and lowest MABP recorded within the first 5 mins post-tracheal intubation are represented in Figure 1. The sedation scores for group A2 (1.5 [range 1.0–3.0]) were not significantly different to those from group A0.5 (2.5 [range 1.0–3.0]) (P = 0.32); however, cats in group A2 required more alfaxalone (4.3 mg/kg (range 3.4–7.1 mg/kg]) than those in group A0.5 (2.1 mg/kg [range 1.5–2.5 mg/kg]) to achieve induction of anaesthesia (P = 0.002). Induction time was shorter in group A2 (129 s [range 103–212 s]) than in group A0.5 (257 s [178–304 s]) (P = 0.01).
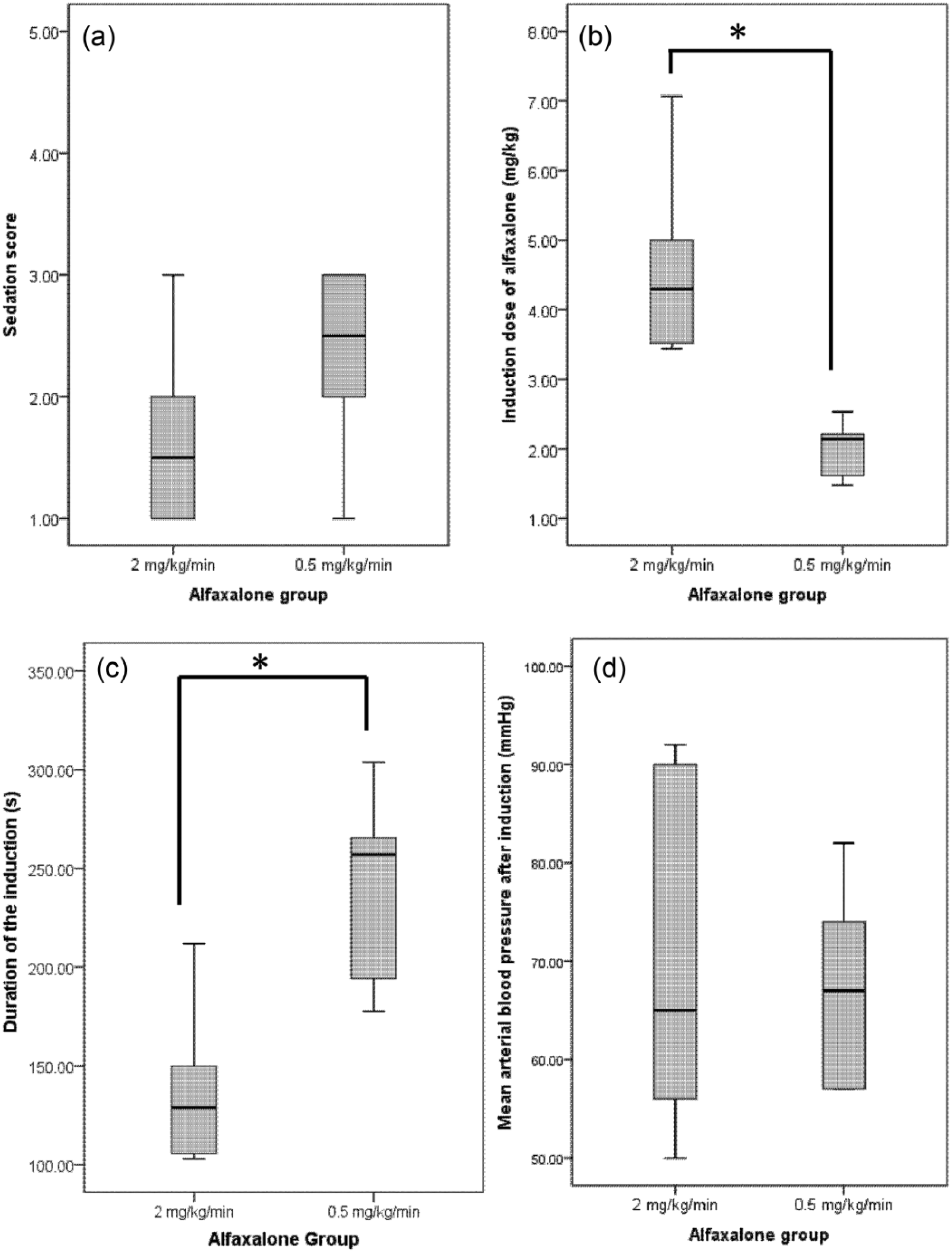
Box and whisker plots of the (a) preinduction sedation scores performed 20 mins after premedication, (b) alfaxalone induction dose performed 30 mins after premedication, (c) duration of the induction of anaesthesia and (d) lowest mean arterial blood pressure recorded within the first 5 mins post-tracheal intubation. Twelve cats undergoing ovariohysterectomy received alfaxalone 2 mg/kg/min IV (group A2, n = 6) or 0.5 mg/kg/min IV (group A0.5, n = 6) following premedication with buprenorphine (0.02 mg/kg IM) concurrently with alfaxalone (3 mg/kg SC). Sedation scores were obtained by use of a published scale ranging from 1 (no sedation) to 5 (profound sedation).The box indicates the interquartile range (25th–75th percentile), the bold black line in the box indicates the median and the whiskers indicate the range. *Statistical difference between the two groups: cats in group A2 required more alfaxalone than those in group A0 to achieve induction of anaesthesia (P = 0.002), and induction time was shorter in group A2 than in group A0.5 (P = 0.01)
Apnoea was present in two cats of each group but no PPV was required. The PETCO2 remained ⩽50 mmHg in all cats. No bradycardia was seen; two cats in group A2 and three in group A0.5 presented mild postinduction hypotension (50 ⩽MABP <60 mmHg).
Discussion
To our knowledge, this is the first study to demonstrate that the use of a slower induction infusion rate resulted in an increase of the alfaxalone relative potency.
Cats involved in this study were representative of the domestic population and there were no outstanding differences in their temperament. The level of sedation was generally poor and could have been due to less effective alfaxalone absorption by the subcutaneous (SC) route and/or to the recording of the sedation score prior to the peak sedative effect (30–45 mins after SC administration). 5 The quality of anaesthesia induction was fair, with no cats exhibiting signs of excitement or dysphoria. However, as the number of animals in each group was small, it cannot be excluded that the lack of significance in the difference of sedation scores observed between the two groups could be due to a lack of power. Thus, the comparison in the amount of alfaxalone necessary to induce anaesthesia could have been biased.
As reported with propofol induction of anaesthesia in humans, the slower administration resulted in a decreased alfaxalone induction dose requirement. 6 Stokes and Hutton described how the slow increase in propofol plasma concentration associated with a slow infusion provides a lower but more sustained gradient for even drug delivery throughout the central nervous system. 6 This is confirmed by the data extracted from a physiological model of induction of anaesthesia with propofol in sheep that also demonstrated that a slower administration rate was associated with significant decrease in propofol peak arterial concentration. 8 This decrease could benefit animals with decreased cardiovascular function by limiting the extent of its depression, and animals with decreased hepatic function when using induction agents that undergo hepatic metabolism (ie, alfaxalone). 5
Although the prediction of the optimal infusion rate for inducing anaesthesia depends on the blood:brain equilibrium half-life of the anaesthetic agent, general pharmacological principles demonstrated using propofol will likely apply to alfaxalone. 9 The lack of evidence for cardiovascular advantages in the present study using a slower infusion rate is probably owing to the nature of the study (pilot study), the inadequate cardiovascular monitoring (non-invasive blood pressure), the small size of the groups and the good cardiovascular functional reserve of the population (young and healthy animals). The durations of apnoea were, unfortunately, not recorded; however, they were not clinically significant as none of the cats required positive pressure ventilation.
Conclusions
For the first time, this pilot study demonstrated that a slower alfaxalone induction infusion rate resulted in a reduction in the alfaxalone dose needed to achieve orotracheal intubation in the cat. However, further studies with larger numbers of animals would need to be performed to confirm this result and to evaluate if any cardio vascular benefit would arise from using a slower induction rate.
Footnotes
Acknowledgements
We would like to thank Drs Donna Scott, Isobel Monotti and Takanori Sugiyama for technical assistance.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
We gratefully acknowledge the partial financial support from Jurox Pty Ltd, Rutherford, NSW, Australia.
