Abstract
Objectives
The aim of this study was to evaluate the feasibility and efficacy of serially administered adipose-derived mesenchymal stem cells (MSCs) in an experimental feline asthma model.
Methods
Allergic asthma was acutely induced with Bermuda grass allergen in six purpose-bred cats. Five intravenous infusions of allogeneic MSCs (n = 4; MSC-treated) or saline (n = 2; placebo-treated) were administered over the first 130 days after asthma induction. Infusions contained 2 × 106, 4 × 106, 4.7 × 106, 1 × 107 and 1 × 107 cryopreserved MSCs/cat. For thoracic imaging additional cats were enrolled as control groups: four untreated, experimentally asthmatic cats (combined with placebo-treated cats), and six healthy, non-asthmatic cats. Outcome measures included airway eosinophilia, pulmonary mechanics, thoracic computed tomography and several immunologic assays.
Results
Cats were assessed for 9 months after treatment. At early points, airway eosinophil percentage was not affected by MSC administration (post-treatment average of days 12, 26, 47, 108 and 133 in MSC-treated cats was 41 ± 15% and in placebo-treated cats it was 34 ± 16%). By month 9, eosinophil percentages in all MSC-treated cats decreased to normal reference intervals (MSC-treated 6%; placebo-treated 20%; normal <17%). Diminished airway hyper-responsiveness was noted in all MSC-treated compared with placebo-treated cats at day 133 (dose of methacholine to double baseline airway resistance: MSC-treated median 22.9 mg/ml [range 6.4–64.0]; individual placebo-treated cats 1.1 and 5.0 mg/ml). Lung attenuation (mean ± SEM MSC-treated −865 ± 12 Hounsfield units [HU]; untreated asthmatics −820 ± 11 HU; P = 0.004) and bronchial wall thickening scores (median [interquartile range] MSC-treated 0 [0–1.5]; untreated asthmatic 11.6 [7.3–27.3]; P = 0.010) were significantly reduced in MSC-treated vs untreated asthmatic cats, consistent with decreased airway remodeling at month 9. No clear immunologic mechanisms by which MSCs act were determined.
Conclusions and relevance
MSCs may have a delayed effect in reducing airway inflammation, airway hyper-responsiveness and remodeling in experimentally induced asthmatic cats. Results warrant additional investigation of MSC therapy for asthma in cats.
Introduction
Feline asthma, characterized by airway eosinophilia, airway hyper-responsiveness (AHR) and remodeling, is driven by a T helper (Th)2 immune response to inhaled aeroallergens. 1 Standard therapy with glucocorticoids and bronchodilators address inflammation and airflow limitation in many cats, but no therapy can consistently prevent or reverse all pathologic aspects of asthma.2–5 Some cats remain resistant to this therapy, with continued clinical signs and airway eosinophilia. 2 Other cats may have concurrent diseases (eg, cardiac disease or diabetes mellitus) that are relative contraindications to glucocorticoid usage; thus, novel therapies are needed. An ideal treatment would attenuate the Th2 response and decrease all characteristic features of asthma.
Mesenchymal stem cell (MSC) therapy has been investigated in murine asthma models. MSCs may be immunomodulatory, restoring the Th1/Th2 balance in asthmatics, as evidenced by decreased Th2-related cytokines after treatment.6–8 MSCs may also promote immune tolerance through proliferation of T regulatory cells (Tregs) and secretion of regulatory cytokines such as interleukin (IL)-10.6,7 MSC therapy has been associated with reduced asthmatic features in murine models, including airway eosinophilia, AHR and remodeling.7–12 The aim of the current study was to evaluate feasibility and efficacy of serially administered adipose-derived MSCs in an experimental feline asthma model.
Materials and methods
Animals
Six cats (four intact males and two intact females; 5–6 months old at time of first infusion) were selected from a high-responder asthmatic research colony (University of Missouri). High-responder cats are bred to display a more robust asthmatic phenotype. Four cats were included in the MSC-treated group and two cats in the placebo-treated group for all analyses except for thoracic imaging. For thoracic computed tomography (CT) scans, an additional six healthy research cats were used as healthy controls; four untreated, age-matched research cats with experimentally induced asthma for a similar period were combined with the placebo-treated group to form an untreated asthmatic group. This study was approved by the University of Missouri Animal Care and Use Committee and performed in accordance with the National Institutes of Health’s Guide for the Care and Use of Laboratory Animals.
Experimental treatment
MSC-treated and untreated/placebo-treated cats were sensitized to Bermuda grass allergen (BGA) to induce an asthmatic phenotype as previously described; 13 >17% bronchoalveolar lavage fluid (BALF) eosinophils were required for study enrolment. 1 Cats received challenges of aerosolized BGA (500 μg/ml) to maintain an asthmatic phenotype (Figure 1).

Schematic overview of the study design. After acute induction of experimental asthma, cats were enrolled in the study and randomized to receive mesenchymal stem cells (MSC; n = 4) or phosphate-buffered saline (n = 2). Aerosol allergen challenges were administered weekly for the first 2 months and bimonthly thereafter. Five infusions of MSC or placebo were administered between day 0 (baseline) and day 130. Blood was collected at baseline and at days 12, 26 and 47 for the evaluation of T-lymphocyte phenotype, total Bermuda grass allergen-specific lymphocyte proliferation, interleukin (IL)-10 production from lipopolysaccharide (LPS)-stimulated whole blood and numbers of IL-10 producing cells. Bronchoalveolar lavage fluid (BALF) was collected at the same time points and at days 108 and 133 for determination of the eosinophil percentage. Evaluation of BALF T regulatory cells (CD5+[CD4+foxP3+]) was performed on days 0, 12, 26 and 47. Pulmonary function testing was completed on day 133. During the remaining study period, no further treatments were administered. At month 9 (day 287), BALF was collected to determine eosinophil percentages. Additionally, tandem thoracic computed tomography (CT) in conjunction with abbreviated pulmonary function testing was performed
Allogeneic MSCs were expanded from subcutaneous adipose tissue harvested from healthy specific pathogen-free donor cats as previously described. 14 Cells were culture-expanded and harvested between passages 2 and 5. 14 Feline adipose-derived MSCs prepared by this technique have been assessed for tri-lineage differentiation and the presence of CD44, CD105 and CD90, and lack of CD4 and major histocompatibility complex class II.14,15
Cats were randomized to receive five infusions (days 0, 14, 28, 98, 130; Figure 1) of MSCs (n = 4) or phosphate-buffered saline (PBS; n = 2). Initial MSC infusions contained 2 × 106 and 4 × 106 cryopreserved MSCs/cat that were shipped frozen and thawed immediately prior to use. While the study was in process, low recovery of cryopreserved cells and data suggesting thawed cryopreserved MSCs may have adverse reactions and decreased function led to a protocol change.14,16 The remaining infusions used 4.7 × 106, 1 × 107 and 1 × 107 MSCs/cat expanded from cryopreserved adipose and shipped overnight in expansion medium. Cells were washed three times, resuspended in 20 ml PBS with 200 U of unfractionated heparin and administered through a cephalic catheter over 1 h. Temperature, pulse and respiratory rate were monitored before and 10 mins after infusions.
Sample collection
Samples were collected as outlined in Figure 1. Six milliliters of blood, collected by jugular venepuncture was placed in EDTA, heparin and clot tubes. Peripheral blood mononuclear cells (PBMCs) were harvested with density gradient centrifugation (Histopaque 1077; Sigma-Aldrich). Serum was harvested after centrifugation at 1720 × g for 20 mins. BALF was collected blindly, 1 and supernatant harvested after centrifugation at 300 × g for 10 mins. Serum and BALF supernatant was banked in aliquots at −20°C.
Immune assays
T-lymphocyte phenotype, BGA-specific lymphocyte proliferation, BALF Tregs, IL-10 production from LPS-stimulated whole blood and IL-10-producing cells were evaluated at baseline and days 12, 26 and 47.
T-lymphocyte phenotype in whole blood was determined using flow cytometry according to a published protocol with minor modifications. 1 Briefly, 100 μl lysed and washed EDTA whole blood was placed in tubes with the following monoclonal antibodies: CD4 (antifeline, clone 3-4F4; Southern Biotech), CD5 (antifeline, clone f43; Southern Biotech) and biotinylated CD8 (antifeline, clone fCD8; Southern Biotech). After incubation and washing, streptavidin APC was added. Cells were washed and resuspended in 1% formalin. Flow cytometry was performed with a CyAn ADP Flow Cytometer (Becton Dickenson). A minimum of 20,000 gated events were collected. Data were expressed as percentage of lymphocytes with each marker. Tregs in BALF were determined using flow cytometry as previously described. 17
BGA-specific lymphocyte proliferation was performed with PBMCs, as previously described, 13 using a commercially available kit (Cell proliferation ELISA, BrdU [colorimetric] #1647229; Roche Diagnostics). Results were reported as a stimulation index (SI) (ie, optical densityBGA/optical densitymedia). At month 9, a more sensitive flow cytometry assay was utilized to evaluate BGA-specific proliferation of CD4+ T lymphocytes. 18
For determination of IL-10 production, whole blood was diluted 1:2 in RPMI media with 1 μg/ml LPS and plated at a final volume of 1 ml. Samples were incubated at 37°C, 5% CO2 for 24 h. Supernatant was assayed in triplicate using a commercial ELISA kit (Feline IL-10 DuoSet DY736; R&D Systems) with limits of detection being 125–8000 pg/ml. Numbers of IL-10-producing cells in blood was determined with an ELISpot assay(Feline IL-10 Development Module: SEL736; R&D Systems) under previously published conditions. 13
BALF eosinophilia
BALF eosinophilia was evaluated on days 0, 12, 26, 47, 108, 133 and 287. A 200-nucleated cell differential count was performed on Wright’s stained cytocentrifuged BALF and expressed as percentage of nucleated cells that were eosinophils.
Lung mechanics and CT imaging
Ventilator-acquired pulmonary mechanics were performed on day 133 as previously described, 19 with minor modifications. Cats were premedicated with ketamine (30 mg IV). Anesthesia was induced and maintained with propofol (6 mg/kg; 0.3 mg/kg/min IV). Cats were mechanically ventilated (Engström Carestation; GE Healthcare) with a positive end expiratory pressure of 2 cmH2O. The tidal volume was set at 10 ml/kg and adjusted at 2.5 ml/kg increments without exceeding 15 ml/kg to ensure an end-tidal CO2 of 36–40 mmHg.
After a 5 mins equilibration, saline was nebulized for 30 s and baseline measurements obtained. Bronchoprovocation was performed by nebulizing methacholine starting at 0.0625 mg/ml and doubling the concentration for each dose up to 32 mg/ml for 30 s. Data were collected for 4 mins after each dose. Bronchoprovocation ended after airway resistance increased to 200% of baseline. Results were expressed as the effective concentration of methacholine increasing baseline airway resistance by 200% (EC200Raw) as determined by linear interpolation of the log-log plot of the dose–response curve. 1
Thoracic CT scans were performed at month 9 (day 287) to evaluate airway remodeling. Images were obtained with multi-slice CT (Aquilion 64; Toshiba America Medical Systems) under resting conditions and in tandem with pulmonary mechanics following nebulization with saline and three concentrations of methacholine (0.0625, 2 and 32 mg/ml). Anesthetized cats were in sternal recumbency to minimize atelectasis. Contiguous transverse images were acquired with 0.5 mm collimation, matrix of 512 × 512, pitch of 1.0, small focal spot size, 10–12 cm field of view, 120 kVp and 250 mAs. During bronchoprovocation, 5 s breath-hold exhalatory and inspiratory CT scans were acquired at minutes 1 and 3 of data collection, respectively. Image acquisition stopped when the EC200Raw was reached. CT parameters at resting conditions (before saline/methacholine nebulization) were assessed on images through the entire thorax, while dynamic conditions (bronchoprovocation) were evaluated on images of the caudal thorax to maximize lung in the field of view. Images were reconstructed with a high-resolution algorithm and 1 mm-thick sections (no interval). Images were transferred to an Empiric PACS system (Encompass; Fujifilm Medical Systems USA), viewed using an OsiriX workstation (v. 3.9.4, 32 bit; see www.osirix-viewer.com), displayed with a constant window width of 1600 Hounsfield units (HU) and level of −550 HU, and blindly evaluated by a board-certified radiologist (IM).
Semiquantitative analysis was performed under resting conditions. Lung attenuation (LA) and linear opacities were evaluated using a scoring system adapted from Warrick et al (Table 1). 20 This scoring system combines the severity and the extent of the disease. To determine severity of disease, the percentage of each lung lobe affected was determined and given a corresponding score. The extent of the disease is determined based on the total number of lung lobes or segment of lobes (for left cranial lung lobe only) involved for each abnormality. The severity and extent scores are combined to calculate a global score. Lesions scored included linear opacities, ground-glass opacity, and complete attenuation or consolidation. Linear opacities included proximal peribronchovascular thickening (extending from the pulmonary hila into the peripheral lung), interlobular or intralobular lines (short irregular lines), or parenchymal band (thin and/or thick non-tapering linear opacity potentially crossing the pulmonary parenchyma). Ground-glass opacity was defined as a hazy increase in lung density with preservation of airway and vessel margins. Complete attenuation or consolidation was defined as an increase in lung density with obscuration of the underlying vessels. Before evaluating bronchial wall thickening (BWT) in treated cats, images from six healthy cats were reviewed and scored. A healthy cat with a mid-range score was chosen to represent a threshold level for BWT, as done by others. 21 MSC-treated and untreated asthmatic cats were compared with the healthy cat and the number of thickened bronchi and severity (mild [1], moderate [2], severe [3]) were determined for each lung lobe and summed for a global BWT score.
Semiquantitative scoring method for lung attenuation (adapted from Warrick et al 20 ). This scoring system combines the severity and the extent of the disease to calculate a global score

For quantitative analysis under resting conditions and in both phases of respiration after bronchoprovocation, Digital Imaging and Communications in Medicine (DICOM) images were imported into three-dimensional Slicer airway inspector software (v. 4.2.; see http://www.slicer.org). Density mask analysis of lung parenchyma was performed on 1-mm-thick sections and automated extraction of LA based on the Otsu Method. 22 Lung density histograms were obtained for HU of −300 to −1000 using the chronic obstructive pulmonary disease–emphysema module as carried out previously. 23 Distribution curves were generated for each phase of respiration, and values for mean and SD of LA and lung volume were recorded.
Statistical methods
Owing to small sample sizes, only descriptive assessment of BALF eosinophil percentage, EC200Raw and immunologic assay results was performed. For CT data, normality was assessed using a Shapiro–Wilk test. A one-way ANOVA was used to assess resting variables – except for BWT score, which was assessed with a Mann–Whitney rank sum test. A two-way ANOVA was used to analyze CT variables after bronchoprovocation with post-hoc multiple comparisons. Commercially available statistical software was used (SigmaPlot 12; Systat Software). A P value <0.05 was considered significant.
Results
Immunologic assays
Published species- and cell type-specific ranges or manufacturer-supplied controls were used to document appropriate assay function. Table 2 shows baseline and post-treatment average values (days 12, 26 and 47) for immunologic assays. There was minimal variation in peripheral blood T-cell phenotype, BALF Treg percentages and whole blood IL-10 restimulation in placebo- and MSC-treated cats. For the IL-10 re-stimulation assay, 33% of samples from all time points were below the assay’s lower limit of detection in placebo-treated vs 8% of samples in MSC-treated cats. There was an approximately two-fold increase in IL-10-producing cells in placebo-treated cats and an approximately four-fold increase in MSC-treated cats from baseline. For the BGA-specific lymphocyte proliferation assay an SI <2 indicates a lack of lymphocyte proliferation. 24 Treatment with MSCs resulted in a decrease in the group average SI <2. The SI was <2 in 88% of all post-treatment samples measured in MSC-treated vs 16% in placebo-treated cats. At month 9, using a more sensitive flow cytometry assay, the subset of CD4+ T cells that proliferated in response to BGA was 3.3 ± 2.6% in MSC-treated and 0.3 ± 0.3 in placebo-treated cats.
Selected immunologic parameters for mesenchymal stem cell (MSC)-treated and placebo-treated asthmatic cats before and after treatment. The immunologic assays were performed early in the course of the study in placebo- (n = 2) and MSC-treated (n = 4) cats

Average of days 12, 26 and 47
SI = stimulation index; LPS = lipopolysaccharide; BALF = bronchoalveolar lavage fluid; Tregs = T regulatory cells; BGA = Bermuda grass allergen
Airway eosinophilia
The average of early time points (days 12, 26, 47, 108 and 133) was calculated and reported as the post-treatment average. The post-treatment average of BALF eosinophil percentages was 41 ± 15% in the MSC-treated and 34 ± 16% in the placebo-treated groups. By month 9, average BALF eosinophil percentage was 6% in the MSC-treated and 20% in the placebo-treated groups.
Lung mechanics and CT structural changes
At day 133, the EC200Raw was higher in all MSC-treated compared with placebo-treated cats (MSC-treated 22.9 mg/ml [range 6.4–64.0]; placebo-treated 1.1 and 5.0 mg/ml; Figure 2).

Airway hyper-responsiveness, as determined by ventilator-acquired pulmonary mechanics. The EC200Raw was determined at day 133 after five infusions in the MSC- (n = 4) and placebo-treated (n = 2) cats. Each point represents the effective concentration of methacholine, which induced a 200% increase in baseline airway resistance
Bronchoprovocation in tandem with CT imaging provided quantitative measures of dynamic changes in LA and lung volumes at month 9 (Table 3). There was significantly lower LA during bronchoprovocation in MSC-treated asthmatic cats vs untreated asthmatic cats on inhalation (P <0.001), with MSC-treated cats being similar to healthy cats (P = 0.461). The lung heterogeneity (ie, variation of LA) on inhalation differed significantly among all three groups (asthmatic vs healthy P <0.001; MSC vs healthy P = 0.008; asthmatic vs MSC P = 0.005). During exhalation, lung heterogeneity was not significantly different between MSC-treated and healthy or untreated asthmatic cats, while untreated asthmatic cats had significantly more lung heterogeneity vs healthy cats (MSC vs healthy P = 0.054; MSC vs asthmatic P = 0.07; asthmatic vs healthy P <0.001).
Quantitative assessment of lung parenchymal changes via thoracic computed tomographic imaging after bronchoprovocation at day 287 in healthy, untreated asthmatic and mesenchymal stem cell-treated asthmatic cats

Vs healthy group for main effect of group
Vs asthmatic group for main effect of group
HU = Hounsfield units; BW = body weight
Representative CT images from an MSC-treated and untreated asthmatic cat under resting conditions are shown in Figure 3. Semiquantitative global LA and BWT scores were significantly lower in MSC-treated vs untreated asthmatic cats (LA, P = 0.004; BWT, P = 0.011; Figures 4 and 5). There was no difference in LA scores between healthy cats and MSC-treated cats (P = 1.000).

Thin-section computed tomography (CT) images obtained in mesenchymal stem cell (MSC)-treated and untreated asthmatic cats. (top) CT scan obtained on (a) inhalation and (b) exhalation in an MSC-treated asthmatic cat. (a) In the MSC-treated cat, lung attenuation was homogeneous with minimal focal hazy areas of increased attenuation ventrally (arrowheads) due to cardiac motion. (b) On exhalation, lung attenuation was slightly increased, consistent with decreased air in the airspaces. (bottom) CT scan obtained on (c) inhalation and (d) exhalation in an untreated asthmatic cat. (c) In the untreated asthmatic cat, increased lung parenchymal density characterized by multifocal ground-glass opacity is seen in the left caudal, right caudal and accessory lung lobes. These lesions contributed to an increase in the mean and variation of lung attenuation (ie, lesions were heterogeneous in distribution). The white arrows delimit an area of ground-glass opacity in the accessory lung lobe. (d) Aforementioned lesions were exacerbated during exhalation. Interestingly, air trapping was not identified in exhalation despite the severity of the lesions. Several bronchi had prominent walls (black arrows) and were classified as thickened. An increased number of smaller caliber bronchi were easily seen

Semiquantitative global lung attenuation score measured on thoracic computed tomographic images. Global lung attenuation scores were determined during inhalation in healthy (n = 6), untreated asthmatic (n = 6) and mesenchymal stem cell (MSC)-treated cats (n = 4) at end of the study (month 9). In the box and whisker plots, the central line represents the median, and the upper and lower hinges represent the 75th and 25th percentiles, respectively. The whiskers represent the maximum and minimum values. *P = 0.004

Semiquantitative global bronchial wall thickness score measured on thoracic computed tomographic images. Mean ± SEM of bronchial wall thickness scores measured during inhalation in untreated asthmatic (n = 6) and mesenchymal stem cell (MSC)-treated cats (n = 4) at month 9 (day 287) after treatment. *P = 0.011
Quantitative analysis of baseline conditions is presented in Table 4. Mean LA on inhalation in MSC-treated cats was significantly lower compared with untreated asthmatic cats (P = 0.013), but was not different from healthy cats (P = 0.815), as illustrated by the overall darker lung parenchyma in an MSC-treated cat (Figure 3a) compared with an untreated asthmatic cat (Figure 3c). Untreated asthmatic cats had significantly higher LA on inhalation compared with healthy cats (P = 0.006). On exhalation, there was no difference between healthy and MSC-treated cats (P = 0.154) or asthmatic and MSC-treated cats (P = 0.101), but the mean LA was significantly higher in untreated asthmatics vs healthy cats (P = 0.004). The tissue heterogeneity was significantly higher in untreated asthmatics vs healthy cats (P = 0.016), but not compared with MSC-treated asthmatic cats during inhalation. In MSC-treated cats, only a few lesions were seen interspersed with normal lung parenchyma; untreated asthmatics had more lesions (Figure 3a,c). Tissue heterogeneity was not significantly different among groups on exhalation.
Quantitative assessment of computed tomographic-derived indices of airway remodeling under resting conditions at day 287 in healthy, untreated asthmatic and mesenchymal stem cell (MSC)-treated asthmatic cats
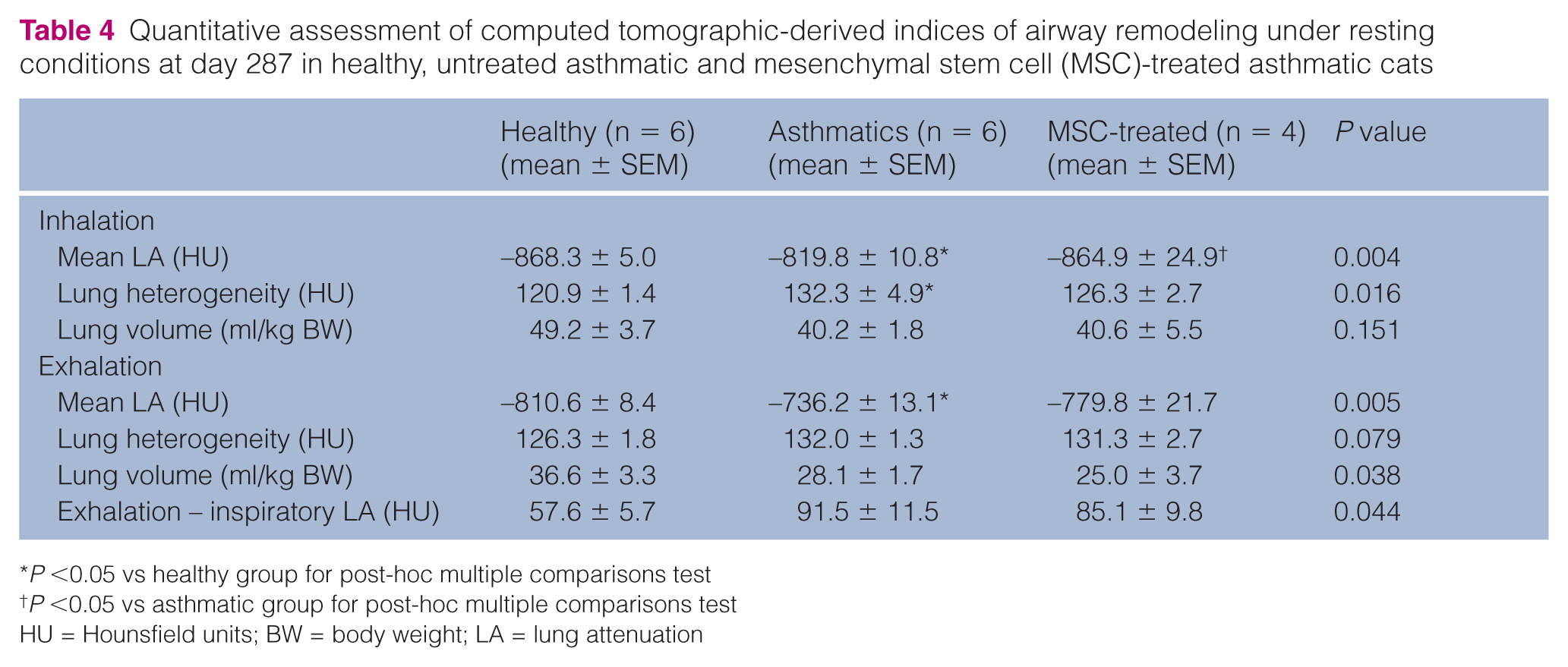
P <0.05 vs healthy group for post-hoc multiple comparisons test
P <0.05 vs asthmatic group for post-hoc multiple comparisons test
HU = Hounsfield units; BW = body weight; LA = lung attenuation
Safety
Cats tolerated five allogeneic MSC infusions generated from multiple donors with no adverse events suggestive of a transfusion type reaction such as marked tachypnea/labored breathing, erythema or collapse.
Discussion
Treatment with MSCs early in the course of asthma development demonstrated a potential delayed beneficial effect on the major hallmark features of asthma in this pilot study. Most strikingly, by 9 months asthmatic cats treated with five allogeneic MSC injections had significant reductions in CT indices of structural changes/remodeling. Serial administration of allogeneic MSCs was well tolerated with no adverse events noted.
In murine models MSCs localize to the lungs after intravenous administration.7,8,12,25 The total time MSCs are present within the lungs has not been definitively determined; however, studies suggest most MSCs fail to remain in the lung for more than a few days.7,26,27 However, MSCs’ immunomodulatory role in lung tissue may be longer lasting.28–30 Murine studies have shown MSCs normalize the Th1/Th2 balance, as supported by decreases in Th2-related cytokines.6,8 Additionally, MSCs promote immune tolerance through Treg proliferation. 7 In the current study, several immunologic assays were performed to determine MSC effects on the immune response in asthmatic cats. In addition, a recently developed flow cytometry assay 18 evaluating CD4+ T cell BGA-specific responses with a higher assay sensitivity was utilized at month 9 in place of the less sensitive BrdU-based ELISA initially used in this study. 13 Despite this, no clear cut mechanisms were elucidated for any of the immunologic assays, perhaps because of the small sample size, imbalanced group sizes and low power of the pilot study, or the presence of different immune mechanisms than were assayed. A larger study would allow further verification of the results obtained in this pilot study and additional investigation into the immunomodulatory functions of MSCs and potential species differences due to the more complex immune systems in cats compared with rodents.
In murine models of asthma, MSC therapy decreases airway eosinophilia.6,8–11,25 In the current study, MSC administration early in asthma development did not appear to immediately blunt airway inflammation. Interestingly, by month 9 all MSC-treated cats had BALF eosinophil percentages within reference intervals for non-asthmatic cats; however, both groups had reduction in airway eosinophilia. The significance of this finding is hampered by the small and imbalanced sample sizes in this pilot study. Additionally, the placebo-treated group also had a reduction in airway eosinophilia, which may reflect decreased frequency of allergen challenges in the chronic study phase. Additional study will help better evaluate the effects of MSC on eosinophilia in cats with asthma.
Studies in rodent models have demonstrated a reduction in AHR following single infusions of MSCs.6,8,11,25 Most studies utilized barometric whole-body plethysmography to measure AHR, which is a non-invasive technique that does not reflect true airway resistance. 31 In the present study, a direct measure of airway resistance using ventilator-acquired pulmonary mechanics was performed to more accurately determine AHR. In this pilot study, the EC200Raw was higher in all MSC-treated cats than in the placebo-treated cats, indicating decreased AHR.
As a less invasive modality than histopathology, thoracic CT can document chronic airway remodeling in inflammatory lung diseases.23,32 In the current study, thoracic CT scans revealed improved BWT and LA in MSC-treated vs untreated asthmatic cats. Without histopathology it is unknown which specific tissue alterations contributed to these CT findings, but could include decreased epithelial hyperplasia, smooth muscle hypertrophy, submucosal gland hyperplasia or inflammatory infiltrates. 32 In resting conditions, lung volume was similar between groups, supporting similar inspiratory capacities and ventilation. Thus, greater scores in asthmatic cats likely can be attributed to lesions causing increased lung density and not lack of air in airspaces or unequal lung expansion.
Interestingly, MSC-treated asthmatic cats had significantly lower inspiratory LA than asthmatic cats while maintaining levels similar to healthy cats during bronchoprovocation, suggesting that MSCs attenuated parenchymal changes associated with AHR. During exhalation, LA with higher doses of methacholine in MSC-treated cats was similar to that of asthmatic cats. Unlike the traditional methacholine challenge where doubling doses are titrated to effect, CT bronchoprovocation in this study used only three doses. As methacholine acts directly on smooth muscle and does not require inflammation or activation of neural networks to work, 33 the highest dose used in this study perhaps represents a ‘sledgehammer’, inducing maximal change and resulting in MSC-treated and untreated asthmatics being similar.
There are no established protocols for MSC infusions in cats. Previous murine models have administered between 0.1 and 2.0 × 106 cells per infusion (average ~ 0.5 × 106).8,11 In this study, it was planned to administer 5 × 106 cells/infusion to account for differences in lung mass of cats compared with mice. Owing to variations in cell numbers that could be expanded prior to each administration, the exact number of cells differed with each infusion. Murine models typically administered between one and three total infusions 1–2 weeks apart.7,8 To investigate the effect of number and timing of infusions, five total infusions were administered with data gathered immediately before, shortly after and at a prolonged period of time after infusions. Certainly, there are other protocols that could be utilized, and this study will be useful in modeling future investigations of MSC therapy in asthma.
In this study, allogeneic adipose-derived MSCs were utilized. Adipose tissue is an excellent, easy-to-obtain source of clinically relevant quantities of MSCs. Allogeneic MSCs are a viable clinical option in many instances as they are considered to be immune privileged owing to their low MHC I expression and lack of MHC II expression. 34 In addition, they lack important co-stimulatory molecules necessary for inducing an immune response.34,35 Inhibition of T-cell proliferation by MSCs may add to their overall immune privilege.26,36 No adverse events suggesting an abnormal immune response to infused MSCs were noted in this study.
Conclusions
This study provides important pilot data supporting evaluation of MSCs as a novel therapeutic for feline asthma. In a small number of asthmatic cats, we demonstrated that repeated administration of allogeneic adipose-derived MSCs decreased remodeling indices and may diminish airway inflammation and AHR. Compared with rodent asthma models, these changes were temporally delayed, suggesting the full benefit of MSC therapy in feline asthma may not be immediate. Further studies are needed to confirm these findings in larger numbers of experimentally induced asthmatic cats and to optimize MSC protocols.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Partial funding was provided by Frankie’s Fund for Feline Stem Cell Research at the Center for Immune and Regenerative Medicine, CSU. This work was also supported, in part, by grant K08AI071724 from the National Institute of Allergy and Infectious Disease. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases or the National Institutes of Health.
