Abstract
Objectives
The aim of this study was to evaluate the effect of topical tropicamide at two concentrations (0.5% and 1.0%) on the Schirmer tear test (STT) results in clinically normal cats.
Methods
Twenty-four adult domestic shorthair cats were randomly assigned to three groups. In all three groups, ophthalmic solutions were instilled in a randomly selected eye and the opposite eye served as the control. In groups 1, 2 and 3 one drop of 0.5% tropicamide, 1.0% tropicamide and distilled water was used, respectively. Tear production in both eyes was tested 30 and 60 mins after instillation in all three groups.
Results
Baseline mean ± SEM STT values for the treated eyes in groups 1, 2 and 3 were 13.37 ± 2.91 mm/min, 10.87 ± 1.39 mm/min and 11.37 ± 1.65 mm/min, respectively. Thirty minutes after the drug instillation in the treated eye, mean ± SEM STT values in groups 1, 2 and 3 were 4.87 ± 3.05 mm/min, 2.00 ± 0.84 mm/min and 11.25 ± 1.81 mm/min, respectively. The mean ± SEM STT levels of the treated eye after 60 mins were 3.75 ± 1.87 mm/min, 0.5 ± 0.37 mm/min and 11.42 ± 1.78 mm/min in groups 1, 2 and 3, respectively.
Conclusions and relevance
Use of 0.5% topical tropicamide, instead of 1.0% tropicamide, as a mydriatic agent, can be recommended in cats. Tear production measurement should be performed prior to the administration of tropicamide, regardless of the concentration of this drug.
Introduction
Pharmacologically induced mydriasis is a common diagnostic and therapeutic procedure in both veterinary and human ophthalmology.1,2 Mydriatics are commonly used to facilitate the visualisation of lens, vitreous and ocular fundus during the ophthalmic examination, or to provide exposure for cataract or vitreo-retinal surgeries. 3
Tropicamide is a non-selective muscarinic antagonist (parasympatholytic), which produces a short-lived mydriasis. 4 Inhibition of the parasympathetic constrictor muscle in the iris results in pupil dilation because the action of sympathetic dilator muscles is unopposed. 5 Tropicamide is bound by pigments; therefore, in heavily pigmented eyes, tropicamide takes longer to work and the pupil stays dilated longer. 5
When applied topically, tropicamide is rapidly absorbed within a few minutes from the eye into the systemic circulation. Despite being considered a very safe drug when used topically, it has been associated with an acute transient sialoadenomegaly, sialorrhoea, conjunctival hyperaemia, chemosis, blepharospasm and nictitating membrane protrusion in cats. 6
In veterinary medicine, the most commonly used quantitative tear film assessment method is the Schirmer tear test (STT). This test is performed by placing a 5 mm wide, 35 mm long strip of filter paper in the ventral fornix of the eye at the lateral canthus or a location approximately one- or two-thirds of the eyelid length (ie, medial from the lateral canthus) for 1 min. This test (called STT I) provides measurements of the residual tear volume in the lacrimal lake and of basal and reflexive tearing. Following the administration of a topical ophthalmic anaesthetic and wicking away the tears in the lacrimal lake before performing the test, basal tearing alone can be measured. The latter test is termed STT II and is not widely used in veterinary species because low values (including 0 mm/min) in clinically normal animals confound the diagnosis of low tear production. 7
Tropicamide is used at two concentrations (0.5% and 1.0%) to dilate the pupil for diagnostic purposes. Effects of the administration of topical tropicamide 1.0% have been reported for dogs, cats and horses.8,9 However, no published information is available about the effect of topical tropicamide at a concentration of 0.5% on tear production in dogs and cats.
In the present study, the effect of topical tropicamide at two concentrations (0.5% and 1.0%) was evaluated on the STT results in clinically normal cats.
Materials and methods
This study was approved by the Iran Society for Prevention of Cruelty to Animals in accordance with Iranian ethical codes for studies on laboratory animals. All parts of this study were performed in double-blind manner.
Twenty-four adult domestic shorthair cats of both sexes (12 males, 12 females; age range 2–4 years) were used for this study. The cats were individually housed in an animal room under standard controlled environmental conditions for 5 days before the beginning of the study. A commercial dry food was offered twice daily throughout the study. Water was given ad libitum for 24 h. Simple random sampling was used in the study.
For all the cats, results of complete blood counts and serum biochemical profile were within the reference intervals prior to enrolment in the study. A full physical examination was performed and the cats were selected on the basis of a normal physical and ophthalmic examination, which included direct and indirect ophthalmoscopy, fluorescein staining, STT and slit lamp biomicroscopy.
A baseline STT-I was performed in all the cats. Twenty-four cats were randomly assigned to three groups. In group 1, all eight cats received one drop 0.5% tropicamide (Mydrax 0.5%; Pharma Sina Darou); in group 2, they received one drop 1.0% tropicamide ophthalmic solution (Mydrax 1%; Pharma Sina Darou). In group 3, they received one drop distilled water. In all three groups ophthalmic solutions were instilled in a randomly selected eye and the opposite eye served as the control. A 35 × 5 mm sterile commercial tear test strip (lot STR090201, Opstrip; Ophtechnics Unlimited) from one manufacturer along with a batch with a single lot number was used to record tear production in all three groups. Gently, the cats were physically restrained; the lower eyelid of each cat was everted and the STT was placed in the ventral conjunctival fornix at one-third the distance from the temporal to nasal canthus for 60 s. Then, the wetted portion of the strips was measured as mm/min. In both groups, tear production in both eyes was tested 30 and 60 mins after tropicamide administration. One examiner (MSG) performed all the ocular tests, examinations and measurements. Three days after end of the study no signs of conjunctivitis, keratitis, blepharitis, corneal ulceration or intraocular disease were detected in any of the cats.
Statistical analysis was performed using SPSS for Microsoft Windows (SPSS 17.0; IBM). Normality was tested by one-sample Kolmogorov–Smirnov test and a paired sample t-test was used for the statistical analysis of the effect of drugs within the groups. Differences in mean values between three groups of eyes were analysed by ANOVA. A P value <0.05 was considered statistically significant.
Results
All the numerical data obtained in the population used in this investigation were normally distributed according to one-sample Kolmogorov–Smirnov test (P ⩾0.1). All data were expressed based on mean ± SEM in mm wetting/min. Significant differences were not found between the treated and control eyes in the baseline STT in each group (group 1, P = 0.6; group 2, P = 0.3; group 3, P = 0.4).
Group 1 (tropicamide 0.5%)
The baseline mean ± SEM STT values for the treated and control eyes were 13.37 ± 2.91 mm/min and 13.14 ± 2.52 mm/min, respectively. After the unilateral administration of tropicamide, the mean ± SEM STT values at 30 and 60 mins in the treated eye were 4.87 ± 3.05 mm/min and 3.75 ± 1.87 mm/min, respectively. In the non-treated eye, mean ± SEM STT values 30 and 60 mins after the drug adminstration were 2.87 ± 1.39 mm/min and 2.62 ± 1.64 mm/min, respectively. Significant differences were detected in both treated and non-treated eyes between the baseline STT and STT values 30 mins (treated eye, P = 0.009; non-treated eye, P = 0.009) and 60 mins (treated eye, P = 0.001, non-treated eye, P = 0.004) after drug administration.
Group 2 (tropicamide 1.0%)
The baselines mean ± SEM STT values for the treated and control eyes were 10.87 ± 1.39 mm/min and 11.10 ± 1.54 mm/min, respectively. In this group, the mean ± SEM STT values 30 and 60 mins after drug administration to the treated eye were 2.00 ± 0.84 mm/min and 0.5 ± 0.37 mm/min, respectively, while in the control eye mean ± SEM STT values after 30 and 60 mins were 1.12 ± 0.44 mm/min and 0.62 ± 0.49 mm/min, respectively. Significant differences were detected in both treated and control eyes between the baseline STT and STT values after 30 mins (treated eye, P = 0.00; non-treated eye, P = 0.002) and 60 mins (treated eye, P = 0.00; non-treated eye, P = 0.001) of drug administration (Figure 1).
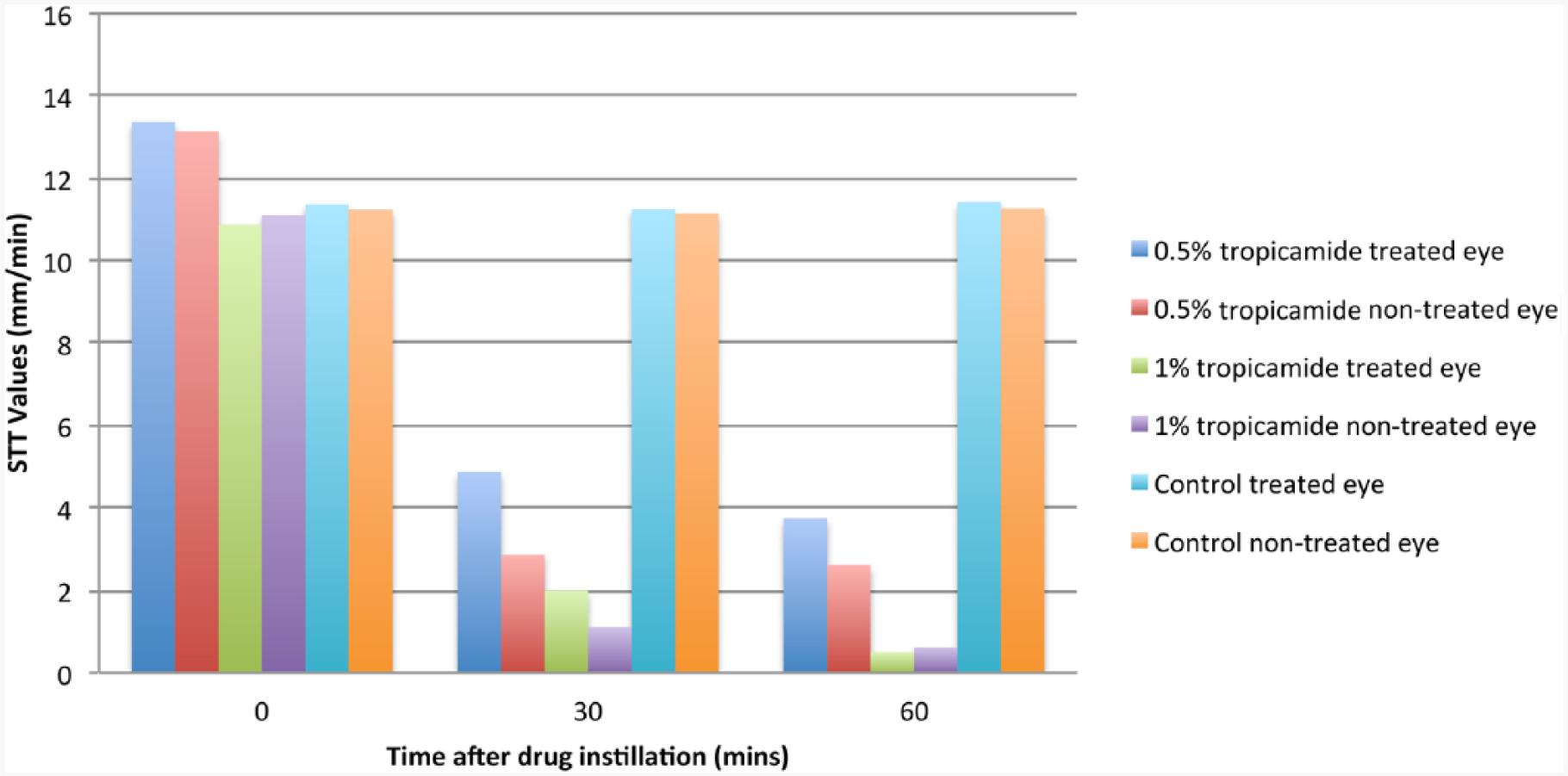
Schirmer tear test (STT) results for 0.5% tropicamide, 1.0% tropicamide and control groups at baseline, 30 and 60 mins after drug instillation
Group 3 (control)
The baselines mean ± SEM STT values for the treated and control eyes were 11.37 ± 1.65 and 11.24 ± 1.28 mm/min, respectively. In this group, the mean ± SEM STT values 30 and 60 mins after distilled water administration to the treated eye were 11.25 ± 1.81 mm/min and 11.42 ± 1.78 mm/min, while in the control eye, mean ± SEM STT values after 30 and 60 mins were 11.15 ± 1.18 mm/min and 11.17 ± 1.28 mm/min, respectively. Significant differences were not detected in both treated and control eyes between the baseline STT and STT values after 30 mins (treated eye, P = 0.2; non-treated eye,P = 0.3) and 60 mins (treated eye, P = 0.1; non-treated eye, P = 0.3) of drug administration.
Discussion
In this investigation, the decrease in mean ± SEM STT levels with tropicamide 0.5% was less than that of tropicamide 1.0% after 30 mins (4.87 ± 3.05 vs. 2.00 ± 0.84 mm/min) and 60 mins (3.75 ± 1.87 vs. 0.5 ± 0.37 mm/min) of drug administration. A more pronounced reduction in tear production was observed after topical administration of both 0.5% tropicamide and 1.0% tropicamide in the treated eyes compared with non-treated eyes of the studied population. Parasympatholytic effects of tropicamide were responsible for these marked reductions in tear production in both eyes. However, the exact cause of the more pronounced reduction in tear production in the non-treated eye is still unknown.
Keratoconjunctivitis sicca (KCS) is a deficiency of the aqueous portion of the tear film. Causes of KCS, other than immune-mediated diseases, include trauma, congenital causes, radiation therapy, neurological deficit and uncorrected prolapse of the nictitans gland. Several systemic diseases (including diabetes mellitus, Cushing’s disease and hypothyroidism) and systemic/topical administration of certain pharmaceutical agents (eg, sulfonamides, etodolac, general anaesthetics, topical and systemic atropine, sedatives and opioids) have been reported to cause dry eye. 10 Diagnosis of KCS in dogs is achieved by measuring both basal and reflex tear production using the STT. 8
Atropine is one of the most commonly used mydriatics that is potent and long-acting, but has a risk of a reduction in tear production and an increased risk of corneal ulceration. Substitution of topical 1% atropine by 1% tropicamide has been suggested as a method to eliminate the adverse effects of these anticholinergics on tear production. 8 Tropicamide, like atropine, causes mydriasis and cycloplegia, but has more mydriatic than cycloplegic activities. Tropicamide causes maximum mydriasis at 15–30 mins of action. 11 This drug has also been indicated following cataract removal to reduce the risk of synechiae formation that is associated with postcataract atropine administration. As the half-life of tropicamide is shorter than that of atropine, iris contraction can prevent synechial adhesions. 11 In humans, 0.5% solution of tropicamide is administered 15–20 mins before the eye examination. To produce cycloplegia, a 1% solution is used; after 5 min, a further administration may be necessary to prolong the effect after 20–30 mins. 12
By reviewing the veterinary literature, it seems that only one study has discovered the effect of 1% tropicamide on tear secretion in dogs and cats. 8 Margadant et al suggested that single doses of topical 1% tropicamide in dogs did not significantly decrease tear production, measured by STT. 8 In contrast, in cats, a single dose of 1% tropicamide in one eye caused a significant reduction in tear production in both eyes after 1 h. 8
The mean ± SEM STT value in the study by Margadant et al after 1 h of 1% tropicamide instillation was 5.3 ± 0.8 mm/min; 8 however, in the present study, 1 h after administration of one drop of tropicamide, the mean ± SEM was 0.5 ± 0.37 mm/min. The cause of this significant difference between these two studies is unknown; however, the small sample size in the present study, animal breed differences and geographical conditions could be responsible for the difference in findings: the baseline mean ± SEM STT levels of the present work in the eye treated with 1% tropicamide was 7.50 ± 0.84 mm/min compared with 14.7 ± 0.8 mm/min in the study by Margadant et al. 8
In the present study, STT values showed a significant decrease in tear secretion after the administration of both 0.5 and 1.0% concentrations of tropicamide in both treated and contralateral control eyes of all the cats. Consistent with the study by Margadant et al, 8 in the present study there are two explanations for the significant decrease in tear production: (1) anticholinergic effects of topical tropicamide probably through absorption to the systemic vasculature; and (2) adrenergic stimulation affecting tear formation rates in cats, probably from changes in blood flow to the tear glands. 8
Systemic absorption of topical ophthalmic applied tropicamide has not yet been studied in cats; however, it seems legitimate to assume that the substance is absorbed via conjunctival vessels, mucous membranes of the mouth and nose, and corneal diffusion with further transport into aqueous humor circulation and outflow. 13 In humans, tropicamide has been detected in the serum after topical ophthalmic application; however, the serum levels decline very rapidly to an undetectable concentration within a few hours. 4
Hassler-Hurst et al compared the effects of two concentrations of tropicamide (0.5 and 1.0%) on pupil dilation and ocular discomfort in humans, and stated that administration of 0.5% tropicamide reduced discomfort during mydriasis in adolescents. 14 The study by Hassler-Hurst et al demonstrated that a lower concentration of tropicamide could produce sufficient pupil dilation at 30 min post instillation. 14 They were unable to determine whether the duration of mydriasis was any shorter when 0.5% tropicamide was used or not. 14 According to this human study, 14 a lower concentration of tropicamide could have sufficient mydriatic effects in terms of improving the ease of examination of the posterior segment of the eye.
Regarding pupil dilation, Schmidt et al used lingual application of 0.5% tropicamide and stated that the lingual application of tropicamide could result in systemic absorption and cause significant papillary dilation. 15 Stadtbäumer et al demonstrated the appropriate mydriatic effects of 0.5% tropicamide after the unilateral application of this drug in the feline eye. 16 In this study, pupillary diameter was not evaluated as a separate parameter. Infrared evaluation of pupil size would have been valuable but, unfortunately, this technology was not available to us. However, clinically, sufficient mydriasis was achieved and the clinician was able to do appropriate examination of posterior segment of eyes in all cats.
There were some limitations in the present research. The first limitation was the small population of the experimental groups. Indeed, larger studies to monitor STT levels sequentially in cats could help veterinarians identify the time at which STT values recover to baseline values. This issue was the second limitation of the present study.
Conclusions
Despite these limitations, results of this study could suggest use of 0.5% topical tropicamide, instead of 1.0% tropicamide, as a mydriatic agent in cats that are to undergo funduscopic examination. Also, veterinarians must be aware of the fact that assessment of tear secretion using a quantitative method, like the STT, must be performed prior to the administration of tropicamide, regardless of the concentration of this drug.
Footnotes
Conflicts of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
