Abstract
Objectives
This study was designed to evaluate the efficacy and safety of reducing ciclosporin (CsA) dosing frequency from daily to every other day (EOD) or twice a week (TW) according to clinical response in cats with hypersensitivity dermatitis (HD) and treated with CsA.
Methods
One hundred and ninety-one cats with HD were given 7 mg/kg CsA daily for at least 4 weeks. Depending on clinical response, the dosing frequency was tapered from daily to EOD over the next 4 weeks and further to TW for an additional 4 weeks. Safety was evaluated through physical examinations, clinical pathology and the monitoring of adverse events (AEs).
Results
The majority of cats were able to have their dose of CsA tapered to either EOD (15.5%) or TW (62.9%) according to the clinical response. Observed AEs were most frequently mild and self-limiting vomiting and diarrhea. A higher percentage of AEs occurred with daily administration (73%) compared with other dosing regimens (27%).
Conclusions and relevance
Following 4 weeks of daily dosing at 7 mg/kg, CsA may be tapered to EOD or TW while maintaining the desired therapeutic response in cats with HD. Additionally, CsA appears to be well tolerated with fewer AEs at EOD or TW dosing. Establishing the lowest effective dosing frequency of CsA improves the drug’s safety profile.
Introduction
Ciclosporin (CsA), a selective immunosuppressor, is increasingly used in veterinary medicine to treat a variety of conditions. 1 An oral solution of CsA (Atopica for Cats [ciclosporin oral solution, USP] modified; Elanco Animal Health) has been approved for the control of feline allergic dermatitis. Allergic dermatitis has also been referred to as ‘hypersensitivity dermatitis’ (HD), a term used to describe a wide variety of allergic diseases such as flea bite hypersensitivity dermatitis, cutaneous adverse food reactions, urticaria, angioedema and atopic dermatitis. 2 The International Committee on Allergic Diseases of Animals recently adopted the term ‘feline atopic syndrome’, which specifically excludes parasitic causes but includes environmental allergen causes, and at least some manifestations of asthma, and food reactions; as such, manifestations may occur concurrently in a single cat. 3 As feline allergic dermatitis is a chronic condition, prolonged or continuous administration of CsA is required to control clinical signs. An effective and well-tolerated dose of CsA for the treatment of HD in cats at 7 mg/kg was identified by King et al. 4 In laboratory studies, even at multiples of the recommended dose of 7 mg/kg and administered daily over a 6 month period, CsA was reported to be well tolerated. 5 Studies suggest that after a daily dosing induction period of 4–10 weeks, decreasing the dosing frequency may reduce the incidence of adverse events while the clinical signs of feline HD can still be well controlled.6–9 This study was designed to confirm that tapering the dosing frequency of CsA following a 4 week induction period would maintain an adequate clinical response in cats with HD.
Materials and methods
Study design
This study was conducted in two phases. Phase 1 demonstrated the field efficacy of CsA for the control of HD, administered at a target dose of 7 mg/kg body weight, while the outcome of phase 2, in which a dose-tapering regimen was examined, is presented here.
Briefly, the first phase (phase 1) was a blinded, randomized, parallel group, placebo-controlled study in which CsA, dosed orally at 7 mg/kg administered once daily for up to 6 weeks, was evaluated for the treatment of HD. Clinical response was assessed 6 weeks after the start of treatment.
The second phase (phase 2), a non-randomized, open-label study carried out immediately after phase 1, explored the effect of a dose-tapering regimen on the clinical response to CsA. Clinical observations were made by veterinarians at the inclusion visit (V1; [day 0 of phase 2 and the same day as the final visit for phase 1] and after a further 4 weeks [28 ± 5 days; V2], 8 weeks [56 ± 5 days; V3] and 12 weeks [84 ± 5 days; final visit (FV)])of treatment. Both phases were multi-center, with sites located in the USA and Canada, and were conducted in accordance with the procedures and principles of Good Clinical Practice. All procedures were reviewed and in compliance with Novartis Animal Health (NAH) Animal Welfare Guidelines and the US Department of Agriculture Animal Welfare Act (2009). In addition, cat owners signed an informed consent for their cat to be included in the study and were free to withdraw their animals at any point.
Inclusion/exclusion criteria
Cats were included in phase 1 after a diagnosis of HD was made by ruling out flea bite hypersensitivity, bacterial and fungal infections and, if possible, food allergies. Cats were also required to have non-seasonal localized or generalized pruritus. Cats with positive serological status for feline leukemia virus or feline immunodeficiency virus, or clinical signs of active Toxoplasma species were excluded. Cats withdrawn because of a lack of efficacy following a minimum of 10 days treatment, cats completing phase 1 without experiencing a serious adverse event, and cats that experienced good owner treatment compliance were eligible to participate in phase 2.
In addition, to be included in either phase, cats had to be at least 6 months of age, ⩾1.4 kg body weight and currently on a flea adulticide (a flea adulticide was not administered in two sites, one in Colorado and one in Nevada, as they were not considered to be flea-endemic areas). Cats were excluded for the following reasons: breeding, pregnant or lactating cats; presence of an active systemic infection; evidence of or history of any type of malignancy; uncontrolled ectoparasite infestation; cats fed uncooked home-prepared diets; cats that were unavailable for the duration of the study; and cats that received drugs which may interfere with the evaluation of CsA (see ‘Authorized and prohibited treatments’ section).
Assessment of the clinical response
Clinical effect was assessed to determine if dose tapering was appropriate by comparing the initial clinical examination at inclusion in phase 1 (V1) with the overall clinical improvement seen at the phase 2 visits (visits 2 [V2] and 3 [V3]). Clinical improvement was assessed as: 0 = excellent (clinical signs observed during the first examination have completely disappeared); 1 = good (clinical improvement compared with the initial examination); 2 = fair (clinical improvement compared with the initial examination but cat has responded only slightly to treatment); 3 = poor (clinical signs of the cat compared with the initial examination have not changed); or 4 = worse (clinical signs of the cat compared with the initial examination have worsened).
The same veterinarian evaluated the severity of the lesions and pruritus at every visit. The lesion severity was measured using SCORFAD (SCORing Feline Allergic Dermatitis), a scoring system previously described and partially validated. 9 A visual analog scale (VAS) was used to assess an owner pruritus score based on the owner’s observations of their cat’s behavior during the previous 3 days. Owners were asked to draw a vertical line on the horizontal scale with endpoints represented by ‘comfortable with normal grooming behavior’ (far left) and ‘uncomfortable, grooming all of the time’ (far right).
In addition, a five-point numerical rating scale from 0 to 4 was used to determine a veterinarian’s pruritus score, as previously described. 4 An overall assessment of clinical response (scored from 0 to 4) was made by the investigator and the owner at each visit of the phase 2 study beginning at V2.
Dosage and administration
CsA was provided by NAH (Basel, Switzerland) as a 100 mg/ml micro-emulsion liquid formulation contained in a 50 ml glass bottle. The dose of CsA was administered using a graduated syringe calibrated to deliver the required volume of treatment according to body weight. CsA was administered to the cat either in food or directly into the mouth. The owner was instructed to withdraw food for a period of time, where possible, to encourage the cat to eat the medicated food. If the cat refused the treatment for two consecutive dose attempts when offered with food, the owner was instructed to administer the treatment directly into the cat’s mouth after feeding. If the cat vomited or regurgitated within 5 mins of the treatment administration the cat was re-dosed, otherwise owners were instructed not to re-dose the cat until the next scheduled treatment.
From inclusion to V2 (day 28 ± 5), all cats were administered a once-daily oral dose of CsA at 7 mg/kg, based on the body weight obtained at the FV of phase 1 (day 0, phase 2), and irrespective of previous treatment used in phase 1. From V2, dose tapering was determined according to the veterinarian’s overall assessment of clinical response in comparison with the first day of inclusion into phase 1. All cats received CsA at a target dose of 7 mg/kg, but the frequency of dosing could be altered to every other day (EOD), then to twice weekly (TW), as shown in Figure 1. The investigator could adjust the dose frequency between the visits back to the previous dosing regimen at any time if deemed necessary. If a cat was returned to the previous dose regimen between visits, the cat was evaluated at the next scheduled visit and the dose regimen maintained or adjusted according to the clinical response as described above. If a cat failed to respond (rated poor or worse) to CsA treatment on daily dosing, the case was withdrawn.

Decision procedure for the dose-tapering regimen. From inclusion to day 28, all cats were administered a once-daily oral dose of ciclosporin (CsA) at 7 mg/kg, based on the body weight obtained at the final visit of phase 1 (day 0, phase 2), and irrespective of previous treatment used in phase 1. From day 28, dose tapering was determined according to the veterinarian’s overall assessment of clinical response in comparison with the first day of inclusion into phase 1. All cats received CsA at a target dose of 7 mg/kg, but the frequency of dosing could be altered to every other day (EOD) then to twice weekly (TW). ‘No response’ was defined as an overall score of 3 or 4, an ‘insufficient’ response was defined as an overall score of 2 and a ‘good’ response was defined as an overall score of 0 or 1
Treatment acceptance was evaluated by the owner during dose administration.
Authorized and prohibited treatments
Throughout the duration of the phase 2 study, cats received flea adulticide treatment at regular intervals according to the manufacturer’s recommendations, except at two sites which were located in geographical areas considered to be flea free (Colorado and Nevada). Flea collars were not permitted throughout the study. Medications or interventions likely to have a significant effect on efficacy parameters, such as topical or systemic glucocorticoids (including ophthalmic preparations), megoestrol acetate, antihistamines, clomipramine, amitriptyline and fluoxetine, topical calcineurin inhibitors and allergen-specific immunotherapy, were prohibited during the study. Essential fatty acids and shampoos were authorized only if used before study initiation (day 0 of phase 1) and maintained throughout the study. Drugs interfering with CsA, such as ketoconazole, itraconazole, erythromycin and phenobarbital, were also prohibited if given continuously for ⩾7 days. Any vaccinations were to be given prior to the study start in phase 1. Other concomitant medications or treatments such as parasiticides and medications having therapeutic effects unrelated to the treated dermatological disease were allowed as long as strictly documented. In cases of severe facial/neck pruritus, at the discretion of the investigator, an Elizabethan collar could be used at the beginning of the study and for up to a maximum of 10 days thereafter.
Housing and feeding
Cats remained with their owners in their usual home environment for the duration of the study. Owners were instructed not to change the home management or diet of their cats during the study and to allow for the cat(s) to be fed separately from other animals in the household to ensure that non-study animals did not receive the CsA when administered in food. Owners were also directed to restrict their cats from hunting and scavenging, and to offer only a commercial or cooked diet for the duration of the study.
Assessment of safety
In addition to a full physical examination and body weight determination at each visit, blood samples were collected for hematology and biochemistry (Antech Diagnostic) at inclusion into phase 1 and at study end, and in the case of premature withdrawal. Toxoplasmosis screening (IgM and IgG titers) was performed at the FV and for any cat in which clinical signs consistent with toxoplasmosis were observed. CsA blood concentrations (monoclonal whole-blood test TDx FLx; Abbott Diagnostics) were measured at V2 or FV. The potential for adverse events (AEs) was monitored by the owner and the investigator throughout the study. An AE was considered to be any observation that was unfavorable or unintended and occurred after the administration of CsA, whether or not considered to be related to treatment. A serious AE was defined as an adverse reaction that resulted in death, was life threatening, resulted in significant disability or incapacity, or which resulted in permanent or prolonged signs. A significant AE included those events that required active medical intervention and were considered by the investigator to be clinically significant.
All AEs, regardless of causality, were recorded during the study.
Statistical analysis
Descriptive statistics were computed for all variables. Dose tapering was measured between visits in phase 2. The dose-tapering regimen for each cat was calculated based on the animal’s dosing schedule as determined by the owner’s diary. Dosing schedules were determined by the dosing scheme used most often during the period in question.
Change in body weight from V1 to FV was calculated and analyzed using a Student’s t-test (SAS, version 9.1.3, PROC TTEST; SAS Institute); body weight change was also analyzed using an ANOVA model (SAS) with dose schedule (daily vs other) as a fixed effect, and random effects of site and schedule-by-site. Hematology and clinical chemistry parameters were compared between subgroups using an analysis of covariance model with the pretreatment value at V1 used as the covariate, the fixed effect of dose schedule (daily vs other), and random effects of site and schedule-by-site.
Results
Study population and demographics
A total of 191 cats (109 female, 82 male) out of 217 cats that had participated in phase 1 were enrolled into phase 2. At the time of enrollment, cats were approximately 11 months to 16 years of age and weighed between 2.3 and 9.8 kg. Fourteen breeds were represented, where approximately 71% of the cats were domestic shorthairs. One hundred and fifty-seven cats were included in the efficacy analyses (see Figure 2); of the 34 cases excluded, cats were primarily excluded because of dose tapering compliance (44%), dosing compliance (18%), visit compliance (15%) and administration of a prohibited concomitant medication (15%). Loss to follow-up and sponsor-requested withdrawal (one cat was removed from the study as it had increased thyroxine levels and exited for evaluation for hyperthyroidism) constituted the remaining excluded cases (8%). All cats treated with CsA were included in the safety analyses.

Flow chart of study population. Cats were included in phase 1 after a diagnosis of hypersensitivity dermatitis was made by ruling out flea bite hypersensitivity, bacterial and fungal infections and, if possible, food allergies. Cats with a positive serological status for feline leukemia virus or feline immunodeficiency virus, or clinical signs of active Toxoplasma species infection, were excluded. Cats withdrawn because of a lack of efficacy following a minimum of 10 days of treatment, cats completing phase 1 without experiencing a serious adverse event and cats that experienced good owner treatment compliance were eligible to participate in phase 2
Treatment administration and dose tapering regimen
All cats received a dose of 7 mg/kg CsA for the first 4 weeks of phase 2. Of the 157 animals included in the efficacy calculations, the dose could be tapered to EOD for the next 4 weeks in 79% of cats. During the final 4 weeks, 63%, 22% and 15% of cats remaining in the study could be treated at TW, EOD or daily, respectively (Figure 3). Of the 115 owner-compliant cats (those cats that had all visits included in efficacy calculations) the majority (71%) were able to have their dose tapered to either EOD (18%) or TW (53%) by FV based on their clinical response (Table 1). The remaining cats were either maintained on daily dosing (13%, including eight cats that had attempted EOD dosing but required an increase back to daily dosing) or were withdrawn.

Percentage of cats for each dosing frequency and overall clinical score. The daily ciclosporin dosing regimen could be tapered to every other day (EOD) or twice weekly (TW) in >70% of all cats enrolled into the study as soon as 4 weeks of beginning treatment without deterioration of clinical response. Dosing period: V1–V2 = between visit 1 and visit 2; V2–V3 = between visit 2 and visit 3; V3–V4= between visit 3 and visit 4
Study and dosing outcome for 115 per-protocol cats*

Data are n (%)
These calculations do not include cats that were completely excluded from the study, or those that had at least one visit excluded from the efficacy calculations
V2 = visit 2; V3 = visit 3; FV = final visit; EOD = every other day; TW = twice weekly
Efficacy results
Primary efficacy variables
The veterinarian’s assessment of the overall clinical improvement score at each visit was used to determine if dose tapering was appropriate. The majority of cats were evaluated as ‘excellent’ or ‘good’ (score 0 or 1) in overall clinical improvement by the veterinarian at all visits (82.0%, 78.7% and 75.7% for V2, V3 and FV, respectively).
Secondary efficacy variables
Owner assessment of overall clinical improvement score agreed with that of the veterinarian, with ‘excellent’ or ‘good’ scores recorded in 87.3%, 85.8% and 80.0% of cats at V2, V3 and FV, respectively.
At all visits, the majority of cats had no lesions or only one lesion, of which self-induced alopecia was the most frequently observed. The mean ± SD total lesion score (TLS) decreased from 3.41 ± 3.36 at V1 to 1.87 ± 2.41 at V2 and 1.87 ± 2.46 at V3, representing an overall reduction of 45%. The TLS increased slightly to 2.25 ± 2.83 on the FV, representing a 34% reduction compared with day 0 of phase 1. A single lesion type or no lesions were observed in 72.2% of cats at the FV. The number of cats having body regions containing at least one lesion decreased from 72.6% at V1 to 56.0%, 62.2% and 62.6% at visits V2, V3 and FV, respectively.
Pruritus severity, as assessed by the veterinarian, improved in a similar manner to TLS. Baseline pruritus values were 1.39 ± 1.36 and decreased at V2 to 0.71 ± 0.90, with a slight increase at V3 and FV to 0.83 ± 1.04 and 1.00 ± 1.15, respectively. The percentage of cats with a pruritus score of 0 or 1 (indicating cats were comfortable or tolerable and calm) was 80.0%, 76.4% and 73.9% at V2, V3 and FV, respectively. Owner assessment of pruritus (VAS) was similar to that observed by the veterinarian, with a score of <30% at FV compared with a score of 38% at baseline.
At study conclusion, the investigator assessed the overall clinical improvement of the cat in owner-compliant cases as excellent in 46 (40.0%) cats; good in 41 (35.7%) cats; acceptable in 18 (15.7%) cats; poor in four (3.5%) cats; and bad in six (5.2%) cats. A similar result was obtained from the owners (data not shown).
Tolerability
AEs were observed in 130/191 cats included in phase 2. The most frequently reported AE was vomiting. In most cases, the vomiting spontaneously resolved with continued dosing. A higher percentage of AEs occurred with daily administration of CsA (73%) compared with other dosing regimens (27%). Twenty-five AE reports were considered clinically serious (17 reports for 15 animals treated daily and eight reports in six cats treated either EOD or TW). Those clinically serious reports in cats with a possible relationship to treatment with CsA included such clinical signs as vomiting, lethargy, weight loss, infection, hepatic lipidosis and neoplasia. No deaths were reported during the study. Two deaths were reported following study exit. One cat was diagnosed with feline infectious peritonitis (FIP) and died approximately 2 weeks following a normal study exit. In addition, one cat was noted to have a pre-existing anemia (prior to phase 1). Eight days following exit from phase 2 the patient was presented to an emergency clinic with a severe aplastic anemia (haematocrit of 20.5% at V1, 15.4% at FV and 10.3% at the emergency clinic [normal reference interval 29.0–48.0%]). The cat was euthanized (15 days after the study) owing to the grave prognosis.
Twenty-nine cats were reported to have positive Toxoplasma gondii titers (IgM, IgG or both) during the study. All positive titers, with the exception of one cat, occurred in the absence of clinical signs of disease. One cat had signs of respiratory disease and was subsequently diagnosed with toxoplasmosis. The cat recovered following study withdrawal and treatment with antibiotics.
Evaluation of clinical pathology indices showed no overall clinically relevant abnormalities associated with CsA treatment. In addition, there was no statistically significant change in body weight when measured from V1 to FV (Table 2), nor was there a statistically significant difference in body weight change (P = 0.1950) between animals dosed daily with CsA vs animals dosed with a schedule other than daily. However, persistent progressive weight loss (weight loss >20% of initial body weight) that resulted in hepatic lipidosis occurred in two cats treated with CsA daily.
Summary statistics and Student’s t-test analysis results for body weight change
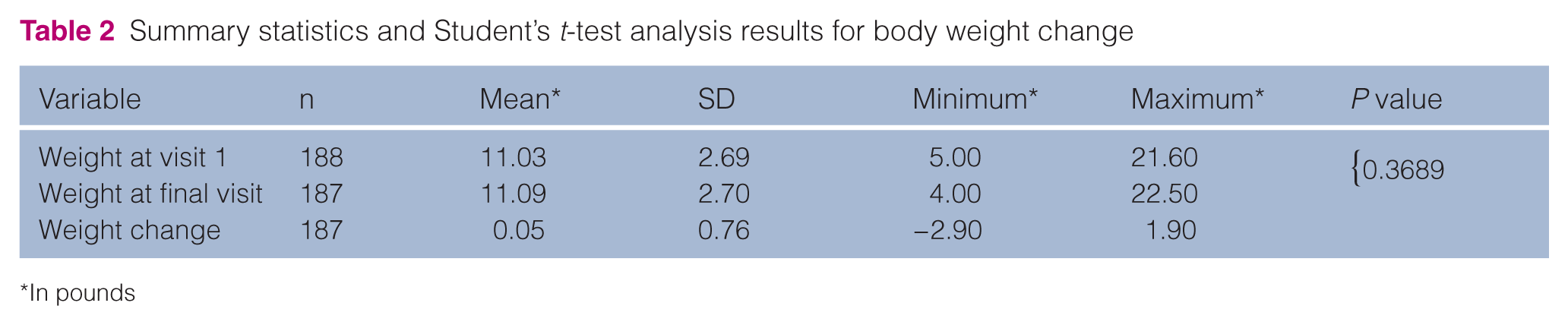
In pounds
The average dose for cats enrolled in phase 2 that received CsA was 7.09 ± 0.36 mg/kg. The average CsA dose for cats reporting an AE was 7.11 ± 0.38 mg/kg vs 7.05 ± 0.31 mg/kg for those not reporting an AE. The average dose for cats on daily dosing and experiencing an AE was 7.05 ± 0.45 mg/kg and for those dosed by any other regimen was 7.13 ± 0.34 mg/kg. CsA levels were obtained from 184 cats at V2 (or FV if exited before V2). Mean CsA was 1113.64 ± 865.72 ng/ml. CsA was used in conjunction with various medications, including parasiticides, antimicrobials and nutritional supplements.
Discussion
Although CsA has been used in cats for some time, an oral solution of CsA has only recently been approved for the control of feline allergic dermatitis in cats of at least 6 months of age and 1.4 kg in body weight. Treatment at a dose of 7 mg/kg for up to 6 weeks was shown to be efficacious and well tolerated. 4 The current study, including a large population of cats, shows that following 7 mg/kg per day of CsA for a minimum of 4 weeks, the frequency of CsA administration could be reduced to EOD while maintaining control of HD in >70% of treated cats. The dosing frequency could be further reduced to TW in 53% of treated cats. This agrees with previous reports from a similar study in European cats with HD, where up to 70% of cats had their CsA dosing frequency reduced to at least EOD, and 57% were maintained on TW dosing. 7 A reduction in dosing frequency has also been reported in a retrospective study of cats treated with CsA for idiopathic pruritus, eosinophilic plaque, indolent ulcer and/or linear granuloma. 8 Although a range of dosages was used in the study by Vercelli et al, 8 after an 8 week induction period of daily dosing, all cats with these conditions could be clinically maintained on a reduced dosing frequency of EOD or less.
Clinical response assessments were used to determine if a reduction in dosing frequency was appropriate based on an overall clinical improvement score provided by the veterinarian. A reduction in frequency of CsA administration from daily to EOD to TW was achieved without relevant loss of response to treatment as reflected in the owner and veterinarian overall clinical assessment, severity of pruritus, total lesion scores and extent of lesions. Only 7% of cats that attempted dose tapering required a return to daily dosing based on a deterioration of their condition. This indicates that most cats maintain stable disease when tapering the CsA dose according to clinical response, and confirms the observations of Steffan et al, who reported that dose adjustment to compensate for deteriorating disease was rarely needed. It would have been interesting to assess the 7% of cats that required a dose increase for a longer period of time and determine if it was possible to re-attempt dose tapering based on clinical response. However, Steffan et al suggested that extending the duration of treatment with the inducing dose was unlikely to result in a better or more stable clinical response. 7 In this study, dose tapering was attempted following up to either a 4 week (phase 2) or 10 week (beginning treatment in phase 1 and continuing treatment into phase 2) induction period. While this study did not specifically evaluate the effect of induction period, the ability to dose taper in most cats suggests that tapering based on clinical response is appropriate irrespective of induction period of 4 weeks or greater. Further, the clinical response should be closely monitored following a reduction of dosing frequency as some cats may need to return to their original dosing regimen.
Approximately 68% of all cats treated experienced at least one AE. The majority (73%) of reported AEs occurred during daily CsA administration (compared with 27% of AEs occurring with EOD and TW dosing). This agrees with previous reports of 80% of AEs occurring during daily administration and confirms that dose tapering reduces the undesirable effects of CsA therapy. 7 The AEs observed were also consistent with previous reports in cats.1,5,7,8,10,11 Digestive tract disorders, in particular vomiting, were the most frequently reported AE. Most vomiting reports were single occurrences and resolved despite continued administration of CsA. Two cats demonstrated severe anorexia that resulted in significant weight loss and hepatic lipidosis during CsA treatment. Hepatic lipidosis in cats is associated with overt liver dysfunction where excess lipid is mobilized to the liver because of starvation. 12 Monitoring the body weight of cats treated with CsA is recommended. In the event of persistent, progressive weight loss a complete clinical examination should be performed. Hepatic lipidosis has not been reported in dogs treated with CsA for atopy. 13
Toxoplasmosis has been previously reported in association with CsA therapy.14–17 A total of 29 cats were reported to have positive Toxoplasma titres (either IgM or IgG or both) during the study. Follow-up titers showed that some animals returned to a negative status, while some titers persisted. All positive titers, with the exception of those for one cat, occurred in the absence of clinical signs of disease. This cat was diagnosed with toxoplasmosis and responded well to stopping treatment with CsA and subsequent antibiotic therapy. Cats that are seronegative for T gondii are considered to be at risk of developing clinical toxoplasmosis if they become infected while undergoing treatment with CsA. 18
As CsA is a systemic immunosuppressant, it may increase the susceptibility to infection and the development of neoplasia. 14 One cat died of the effusive form of FIP 2 weeks after completing the study. In vitro studies have shown that CsA inhibits FIP virus replication. 19 Therefore, while it is possible that cessation of CsA therapy at the end of the study could have precipitated the disease, the immunosuppressive effects of CsA rendering the cat susceptible cannot be ruled out.
The average CsA dose for cats in the study was 7.09 ± 0.36 mg/kg. The average dose for cats with a reported AE was 7.11 ± 0.38 mg/kg. The mean CsA level was 1113.64 ± 865.72. While CsA blood samples were taken at V2 (or FV), the sample was collected at variable times in relation to the last treatment administered such that the mean CsA concentration reflects only an overall exposure. Among cats with a similar clinical response and with or without the report of an AE, a correlation between blood CsA levels and clinical response or prevalence of AEs was not observed. Though plasma concentrations did not appear to be a predictor of effectiveness or safety, the variable sampling time in relation to treatment confounds this assessment.
Conclusions
These results suggest that the inducing dose of 7 mg/kg CsA can be tapered to EOD or TW as soon as 4 weeks without deterioration of clinical response in cats with HD. Although CsA treatment generally appears to be well tolerated, these data also suggest that establishing the lowest effective dose regimen of CsA treatment will improve the safety profile of the drug.
Footnotes
Acknowledgements
We wish to thank all the study investigators who contributed cases: Drs Beale, Bloom, Burkett, Byrne, Cannon, Daigle, DeManuelle, Friberg, Grieshaber, Griffin, McKeever, Messinger, Rosales, Schick, Schick, Spiegel, Stokking, Tapp, Tater, Thomas, Trimmer, Vitale, Waisglass, Wellington, White and Williamson. We also thank the cats and cat owners, and the team at Novartis Animal Health.
Conflict of interest
ESR, LR and SK are full-time employees of Novartis Animal Health. TT and AT have received related and unrelated consultancy fees, lecture fees or investigator fees from Novartis Animal Health.
Funding
This study was funded by Novartis Animal Health US, Inc.
