Abstract
Objectives
Body temperature is commonly used for assessing health and identifying infectious diseases in cats. Rectal thermometry, the most commonly used method, is stressful, invasive and time consuming. Non-contact infrared thermometry (NIRT) has been used with mixed success to measure temperature in humans and other species. The purpose of this study was to determine if NIRT measurements were comparable to rectal temperature measurements or, if not highly correlated, could at least identify cats in the hypothermic or hyperthermic range in need of further evaluation.
Methods
From a total of six NIRT devices and 15 anatomic sites, three devices and three sites (pinna, gingiva and perineum) with the highest correlation to rectal temperature were selected for further study. Measurements were made in 188 adult cats housed indoors at animal shelters, veterinary clinics and private homes across a wide range of body temperatures and compared with rectal temperatures.
Results
Bland–Altman analysis revealed poor agreement between NIRT and rectal thermometry. The mean NIRT measurements ranged from 0.7–1.3°C below the mean rectal measurements, but the effect was not consistent; NIRT measurements tended to exceed rectal measurements in hypothermic cats and fall below rectal measurements in normothermic and hyperthermic cats.
Conclusions and relevance
The accuracy of temperature measurements using NIRT devices is not reliable for clinical use in cats.
Introduction
Rectal thermometry (RT) is the most common method used for measuring body temperature in the clinical assessment of cats. Although recognized as a generally sound reflection of core body temperature, rectal temperature can lag behind changes in core temperature and is affected by depth of measurement, presence of feces and local blood flow. 1 When ambient temperature changes or animals undergo heat loss from the skin, respiratory tract and exposed tissues during anesthesia and surgery, the rectal temperature may remain stable as core temperature and skin temperature change.
Collecting individual RT measurements from a large number of cats, such as at a spay/neuter clinic or an animal shelter, can be time-consuming. When RT is used to monitor infectious disease outbreaks, handling and shared thermometers can contribute to cross-contamination among cats. Rectal disease and pelvic, hindlimb and tail injuries can make RT a painful procedure. As a prey species, cats are particularly prone to fear and stress during veterinary visits. Handling for RT measurement can exacerbate fear, particularly if frequent monitoring is required. This can lead to staff injuries from bites and scratches. 2 RT in fractious or feral cats may not be possible at all. A less invasive method of temperature measurement may result in a less stressful veterinary assessment.
Infrared thermometry (IRT) arose as an alternative to RT in the 1980s when devices were developed to be inserted into the ear canal of humans to measure the temperature of the tympanic membrane. 3 It has been reported that cats tolerated this method better than rectal thermography, especially if frequent monitoring was required. 3 Reports differ in their assessment of the accuracy of auricular thermometry in cats, with evidence to support both high and low correlation with RT.3–5 This technique, while possibly less objectionable to cats than RT, still requires handling and restraint. Another alternative is the use of microchip transponder thermography. In this method, an implantable temperature-sensing microchip, which doubles as a cat identification device, is inserted subcutaneously. Although two people are required to implant the microchip, a single person passing a reader over the site without restraining or contacting the cat can then read the temperature of the chip. Subcutaneous thermography was found to correlate reasonably well with RT in cats and was tolerated better than RT. 2 A disadvantage of microchip thermometry is the invasive implantation procedure and cost associated with each microchip.
Non-contact IRT (NIRT) has gained favor as a non-invasive tool for temperature assessment in human medicine, particularly for pediatric practice, in which young patients may not cooperate with rectal or oral devices.6–9 NIRT devices (forehead thermometers) are marketed directly to the public for use in families and their pets. NIRT is performed by aiming an electronic measuring device at a body area from a distance of 2.5–10.1 cm. In humans, the forehead is the most commonly used site because the carotid artery supplies blood at high flow rates to the temporal artery in the forehead region. 6 Recently, IRT has been used for mass screening of travelers for fevers in an attempt to reduce the global spread of infectious diseases such as Ebola and severe acute respiratory syndrome, but with dubious efficacy.10,11 Inconsistent correlation of NIRT with core or rectal temperatures in adult and pediatric practice has led to suggestions that NIRT should be used for fever screening instead of close monitoring, or not at all.6–9 This technology has also been used in rabbits, 12 monkeys, 13 guinea pigs, 14 horses 15 and mice, 16 with mixed success.
Skin temperature varies widely in cats, even within the same individual over a short time interval. This is demonstrated in Figure 1, which shows the marked surface temperature variation in an adult cat imaged in its’ home over a period of 5 mins using a forward-looking infrared camera. Nevertheless, if NIRT is accurate across a wide range of temperatures in cats, it could be used to decrease stress during a veterinary examination, improve the efficiency of measuring temperatures in large numbers of cats, minimize handling of fractious cats, enhance monitoring for infectious diseases and reduce the risk of fomite transmission among diseased cats. The aim of this study was to determine the correlation of NIRT with RT across a range of body temperatures in cats.

Forward-looking infrared image demonstrating marked surface temperature variation in an adult indoor cat. The coolest area is indicated in dark blue (the nose, 22°C) and the warmest area is indicated in white (the eyes, 33°C). A temperature gradient color scale correlates with temperatures recorded in the image
Materials and methods
Phase 1: selection of infrared thermometers and anatomic sites
The objective of this screening phase was to identify the most accurate NIRT devices and the most reliable anatomic sites for further evaluation. Fourteen well-socialized healthy adult shorthair cats housed in an indoor animal shelter were selected for temperature measurements. Six different NIRT devices designed for humans or pets were tested and used according to the manufacturers’ instructions (FeverWatchers model RC802i [OctiveTech]; Santa Medical model RY230 [Gurin]; Mini Pocket IR DT8220 [Luckystone]; Data-Therm IR model JXB-182 [Jinxinbao Electronic]; ProExotics PE2 [TempGun]; TempIR model RC003 [Raycom Electron Technology]). While it would be ideal to measure sites that would not require handling, such as the torso, pinna, feet and face, other sites were also screened because it was considered that an instantaneous measurement with light restraint would still be less stressful for some cats than restraint and rectal manipulation for RT. Measurements were taken at 15 different anatomic sites on each cat: the pinna, preauricular area, nasal planum, gingival mucosa, dorsal neck, lateral neck, ventral neck, thorax, axilla, metacarpal pad, abdomen, medial thigh, lateral thigh, tail and perineum (Figure 2). A plastic ruler was affixed to the front of each device to ensure that all measurements were made at a distance of 2.5 cm. Three temperature measurements in rapid succession were made with each device at each site to assess for repeatability. In total, 270 NIRT measurements were made for each cat. A single rectal temperature was recorded with a digital thermometer (ReliOn; MABIS Healthcare) inserted 3–4 cm into the rectum after all of the IRT measurements were made. The accuracy of the digital rectal thermometer was confirmed using a laboratory organic column thermometer (Fisherbrand; Fisher Scientific) certified to Scientific Apparatus Makers Association and American Society for Testing and Materials tolerances. The thermometers were immersed in a water bath at temperatures from 31.7°C to 43.3°C. The room temperature where cats were housed and temperatures were measured was determined with an ambient scanning thermometer (Minitemp MT6; Raytek) to evaluate any association of ambient temperature with body surface temperature.

Anatomic sites tested by infrared thermometers. Solid circles mark sites with the strongest correlations with rectal temperatures (pinna, gingiva, perineum). Open circles mark sites with the weakest correlations with rectal temperatures (preauricular area, nasal planum, dorsal neck, lateral neck, ventral neck, metacarpal pad, thorax, axilla, abdomen, inner thigh, outer thigh, tail)
Phase 2: correlation of NIRT with rectal temperature
The three NIRT devices and the three anatomic sites found to correlate best with rectal temperature in phase 1 were selected for further evaluation. Temperatures were measured in 188 adult shorthair cats from animal shelters (n = 113), veterinary clinics (n = 57) and private homes (n = 18) across a wide range of body temperatures. In order to assess NIRT function in the hypothermic range, temperature measurements were made in anesthetized cats following spay/neuter surgery. NIRT temperatures were measured in duplicate at a distance of 2.5 cm with three devices at three anatomic sites for a total of 18 measurements per cat. The ambient temperature, cat signalment and cat phenotype was recorded as for phase 1. A total of 188 cats were classified into temperature categories based on RT, including (1) hypothermia (<36.7°C; n = 43); (2) normothermia (36.7–38.9°C; n = 134); (3) hyperthermia (>38.9°C; n = 11). 17 This study was approved by the University of Florida Institutional Animal Care and Use Committee.
Statistical analysis
The correlation of the first reported NIRT measurement and the RT measurements for each device site pair was determined by Pearson’s correlation coefficient. Using the rectal temperature as the gold standard, the accuracy of each NIRT device at each site was assessed using the Bland–Altman agreement statistic. The precision of duplicate infrared measurements was assessed by calculating a Pearson’s correlation coefficient (ρ) for the two NIRT readouts for each measurement. The correlation between ambient temperature and RT and between ambient temperature and NIRT in all 136 cats that were not undergoing anesthesia was also calculated by Pearson’s correlation coefficient. All analyses were performed with statistical software (MedCalc Statistical Software version 13.3.1; MedCalc Software).
Results
Phase 1
The three NIRT devices that correlated best with rectal temperature were the FeverWatchers model RC802i (OctiveTech), Data-Therm IR model JXB-182 (Jinxinbao Electronic) and TempIR model RC003 (Raycom Electron Technology) (Figure 3). The three anatomic sites that correlated best with rectal temperature were the pinna, gingiva and perineum, all of which are sparsely haired or hairless.
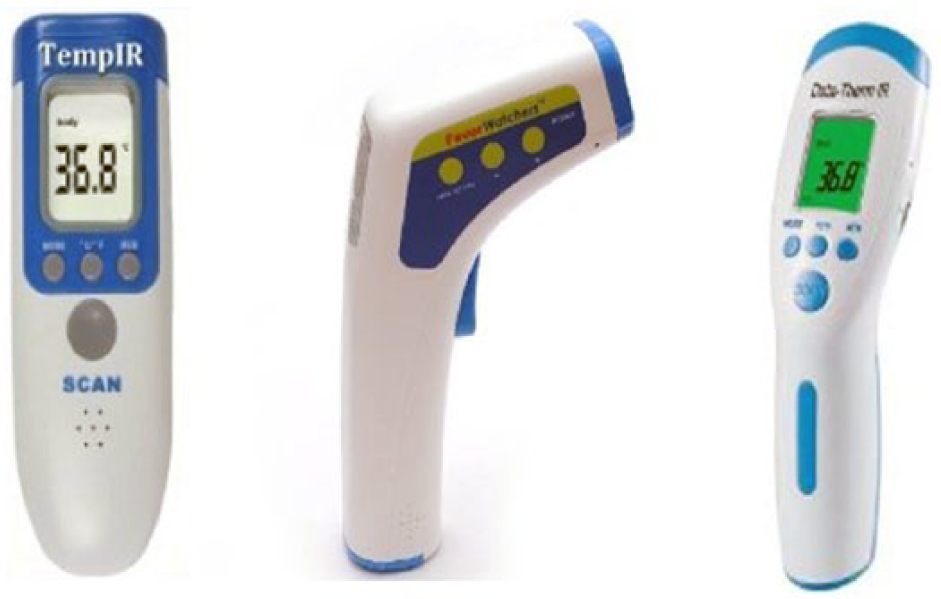
The three non-contact infrared thermometry devices that correlated best with rectal temperature were, from left to right, TempIR model RC003 (Raycom Electron Technology), FeverWatchers model RC802i (OctiveTech) and Data-Therm IR model JXB-182 (Jinxinbao Electronic)
Phase 2
None of the NIRT devices provided a measurement below 36.0°C and instead displayed ‘lo’ readings, even though their instruction manuals reported the ability to measure temperatures as low as 32.2°C (FeverWatchers) and 32.0°C (Data-therm and TempIR). In one cat, three successive ‘hi’ measurements were reported by the TempIR device when aimed at the cat’s gingiva. The device’s instruction manual states that the thermometer will read ‘hi’ when temperatures exceed 42.9°C, but this cat was healthy and had a rectal temperature of 37.6°C. All values of ‘lo’ and ‘hi’ were excluded from the correlation and Bland–Altman analyses because the analysis requires quantitative values, resulting in sample sizes of 112–185, depending on how many ‘lo’ and ‘hi’ readings were omitted.
NIRT measurements with all three devices at all three anatomical sites were only weakly correlated with rectal temperature measurements (r = 0.10–0.42) (Figure 4). Bland–Altman analysis revealed poor agreement between NIRT and RT (Figure 5). The mean NIRT measurements ranged from 0.7 to 1.3°C below the mean rectal measurements, but the effect was not consistent; NIRT measurements tended to exceed rectal measurements in hypothermic cats and to fall below rectal measurements in normothermic and hyperthermic cats. Precision of duplicate NIRT measurements in rapid succession was moderate to high (r = 0.76–0.90). Ambient temperature ranged from 20.3–29.2°C. RT and NIRT were not significantly correlated with ambient temperature.

Correlation of rectal thermometry with three different non-contact infrared thermometry (NIRT) devices used on the (a) gingiva, (b) pinna and (c) perineum. NIRT measurements with all three devices at all three anatomical sites were only weakly correlated with rectal temperature measurements (r = 0.10–0.42)

Bland–Altman comparisons of rectal thermometry with three different non-contact infrared thermometry (NIRT) devices used on the (a) gingiva, (b) pinna and (c) perineum. The dotted line represents the line of equality, the solid line represents the mean difference of the NIRT device measurements from the rectal temperature measurements, and the dashed line represents 1.96 SD from the mean. Bland–Altman analysis revealed poor agreement between NIRT and rectal thermometry. NIRT measurements tended to exceed rectal measurements in hypothermic cats and to fall below rectal measurements in normothermic and hyperthermic cats
Discussion
The large discrepancies between NIRT compared with rectal temperature of cats preclude the use of NIRT in clinical practice. Not only did NIRT devices have poor agreement with RT, but low accuracy also prevented allocation of cats into hypothermic, normothermic or hyperthermic categories. In addition, lack of any consistent pattern of variation from RT prevented the development of a standardized correction equation to improve accuracy of NIRT measurements in cats.
The reason for poor correlation of NIRT with RT was not determined. It is likely that the haircoat impeded infrared emission detection at some sites, which would explain why hairless or sparsely haired sites (pinna, gingiva, perineum) were more accurate than other sites. However, two hairless sites (nasal planum, metacarpal pad) yielded a substantially lower temperature by NIRT, and were not further evaluated. Other factors may also play a role, such as potential differences in arterial blood flow, skin emissivity and skin pigmentation of cats compared with that of humans, for whom the devices were developed.
Conclusions
Currently available NIRT devices are not suitable for measuring body temperature in cats.
Footnotes
Acknowledgements
We thank Laura Adkins, Rose Worobec, Patricia Diskant and Yoonju Cho for their technical assistance.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
This study was made possible by grants from Maddie’s Fund, the Humane Society Veterinary Medical Association and the Department of Small Animal Clinical Sciences at the University of Florida College of Veterinary Medicine.
