Abstract
Objectives
This study describes the sonographic and computed tomographic (CT) characteristics of primary and recurrent feline injection-site sarcomas (FISSs).
Methods
Between 2005 and 2013, 32 cats were selected for prospective and retrospective studies. Tumor shape and margins, presence of thickening of the adipose tissue, muscular and bone involvement, pre- and postcontrast attenuation, blurring of fat planes, calcification and liquefactive necrosis, intratumoral areas and skip metastasis were analyzed in CT scans. Echogenicity, echotexture, tumor margins and peritumoral tissue characteristics were analyzed by ultrasound (US).
Results
Irregular shape (62.5%) with digitiform projections (100.0%), mixed (peripheral and intratumoral) contrast enhancement (67.7%), blurring of fat planes (68.8%) and signs of liquefactive intratumoral necrosis (68.8%) were the prevailing CT findings. Ultrasonography revealed irregular tumor margins, peripheral hyperechoic capsule-like rim, heterogeneous echotexture, and hyperechoic tissue contiguous with the formations and thickening of adjacent subcutaneous tissues in all cases. Mixed echogenicity with areas suggestive of tumor liquefactive necrosis was documented in 83.3% of cases. Skip metastases were highly correlated with tumor recurrence (P = 0.001). The incidence of muscular involvement tended to be higher (P = 0.003) in tumors presenting with thickening of adjacent adipose tissue.
Conclusions and relevance
CT and US features common to FISS lesions were highlighted in this study. The imaging modalities employed allowed assessment of peritumoral inflammation, particularly adipose tissue inflammation. Imaging data may contribute to FISS diagnosis, therapeutic planning and patient follow-up.
Introduction
Feline injection-site sarcomas (FISSs) are mesenchymal aggressive and locally invasive neoplasms.1,2 The condition affects the subcutaneous and muscular tissues, particularly following local injections or trauma,3–7 and was first described in the 1990s. 3 Despite low metastatic potential, FISS tend to project into surrounding tissues; 8 high recurrence rates have been reported. 1 After more than two decades of scientific debate, the etiology of FISS remains undetermined.2,5,6 FISS is thought to be related to chronic inflammation. 9 Individual factors, such as genetic changes or obesity,10,11 may also be involved.
The need for cross-sectional imaging modalities such as computed tomography (CT) and magnetic resonance imaging for accurate recognition of neoplastic margins and appropriate surgical planning in cases of FISS is an area of consensus among clinicians.1,5,9,12,13 However, FISS imaging studies are scarce as most publications to date have not focused primarily on diagnostic imaging.2,8,11,14–16
This study was designed to describe the CT and ultrasound (US) features of FISS and to compare imaging findings between recurrent and non-recurrent neoplasms.
Methods
The information contained in this article was extracted from part of a Master’s thesis by the first author (KMZ). 17
This project was approved by the animal ethics committee of the School of Veterinary Medicine and Animal Science, University of São Paulo (protocol number 2705/2013). The study comprised a prospective (CT and US characterization of FISS) and a retrospective phase (CT characterization of FISS). Cats with a history of subcutaneous tumors at injection sites in accordance with the 3–2–1 rule (masses present at injection sites for >3 months and/or masses >2 cm in diameter and/or increase in mass size 1 month after injection), 5 and with cytological and/or histological diagnosis of FISS were included, regardless of sex, body weight and age. The retrospective study was based on CT records of cats diagnosed with FISS scanned at the veterinary hospital between 2005 and 2012.
Data on clinical history, physical examination and cytologic/histologic results were collected. The cytological criteria adopted were consistent with those employed in diagnosis of mesenchymal neoplasms (sarcomas), that is, the presence of low-to-moderate amounts of fusiform cells with oval nuclei, distinct nucleoli and anisokaryosis. Histologic criteria corresponded with a combination of : (1) cells with large irregular nuclei, and high pleomorphism and a high mitotic index; (2) central area of necrosis; (3) lymphoid aggregates around tumors and irregular macrophage aggregates.
Neoplasms were characterized according to anatomic location, progression (days), recurrence, type of surface (smooth/irregular), insertion base (sessile/pedunculated), consistency (firm/soft/mixed), and presence of ulceration, alopecia, erythema and sensitivity. For non-recurrent lesions, progression time was defined as the time (days) elapsed between detection of a lump by the owner and the day of CT scanning. For recurrent cases, progression time was defined as the time (days) elapsed between surgical resection and the day of CT scanning.
For the purposes of this study, cats were classified into three groups based on a body condition score (BCS) of 1–9 (1 = underfed, 2 = properly fed, 3 = overfed; BCS of 1–4, 5 and 6–9, respectively). 18 Physical examination of neoplasms and assignment of BCS to cats in the retrospective study had been performed by a single examiner (CSC) as part of a previous research project. 11 Corresponding data were collected by a single examiner (KMZ) in the prospective study.
All CT images in the prospective study (6/6) and part of the CT images in the retrospective study (4/26) were acquired using a single-slice helical CT scanner (Xpress, GX Spiral; Toshiba America Medical Systems). A third-generation CT scanner (CT-Max 640; GE Healthcare) was used for the remaining CTs (22/26). CT images employed in the prospective study were in Digital Imaging and Communication in Medicine (DICOM) format and were stored and analyzed in Synapse PACS (Synapse 4.0; Fujifilm Medical System). CT images employed in the retrospective study were either in DICOM or JPG format (four and 14 images, respectively), or printed on film (eight images).
Cats were submitted to general inhalation anesthesia according to the standard protocol employed by the anesthesia care team of veterinary hospital. The anesthetic protocol was adjusted to patient’s individual needs (American Society of Anesthesiologists’ classification system). Anesthetized cats were positioned in sternal recumbency on the CT table and constantly monitored for signs of anesthetic complications or adverse reactions to iodinated contrast medium injection that might require discontinuation of the image acquisition procedure.
In the prospective study, helical CT images were acquired with a slice thickness of 3 mm at reconstruction intervals of 1.5 mm, before and after intravenous (IV) injection of contrast medium; images were reconstructed using soft tissue and bone filters. In the retrospective study, slice thickness and increments or reconstruction intervals between 2.5 and 10 mm were used according to tumor size. All scans were performed using 120 kVp and 150 mA; slices were planned to encompass the entire neoplasm.
Low osmolality non-ionic iodinated contrast medium (Iohexol, Omnipaque 300; GE Healthcare) was employed in prospective and some (2/26) of the retrospective contrast-enhanced CT scans. Remaining contrast-enhanced CT scans (retrospective group; 24/26) were performed using ionic water-soluble iodinated contrast medium (Meglumine amidotrizoate and sodium amidotrizoate, Urografina 292; Berlimed SA). Owing to chronic renal failure, two cats in the retrospective group were not eligible for contrast-enhanced CT. Contrast agents were administered by IV bolus injections at doses of 1.5 ml/kg.
All CT images were evaluated by a single examiner (KMZ). The following characteristics were analyzed and classified: tumor shape (amorphous, spindle-shaped, oval or multilobated); presence of flat component in tumor shape (flat soft tissue structure connecting two or more neoplastic nodules); presence of thickening of the adipose tissue adjacent to the neoplasm compared with contralateral adipose tissue; secondary muscular and bone involvement; tumor margins (poorly or well defined); precontrast attenuation (homogeneous or heterogeneous); contrast enhancement (homogeneous, heterogeneous, peripheral or mixed: heterogeneous as well as peripheral); presence of digitiform projections; presence of blurring of fat planes (variable heterogeneous enhancement of peritumoral adipose tissue); signs of intratumoral calcification; presence of hypodense non-enhancing areas suggestive of liquefactive intratumoral necrosis. Imaging findings suggestive of skip metastasis (small discontinuous subcutaneous nodules adjacent to the primary neoplasm 8 ) were also analyzed in CT scans.
All cats included in the prospective study (6/6) were submitted to B-mode ultrasonography (MyLab 40 scanner; Esaote Healthcare) using a multifrequency (3–11 MHz) linear transducer. Frequency, focal zone and gain/depth settings were adjusted according to tumor size. US assessment was performed by a single examiner (KMZ) with cats in lateral recumbency. Longitudinal and transverse scans were performed for comprehensive tumor assessment following clipping of the target area. The following US parameters were considered and classified: echogenicity (hypo-, iso- or hyperechoic compared with surrounding tissues, or mixed echogenicity); tumor margins (regular or irregular); echotexture (homogeneous or heterogeneous); presence of thickened hypoechoic peritumoral subcutaneous tissue; presence of hyperechoic rim; presence of intratumoral calcification; presence of hypo-/anechoic areas suggestive of liquefactive intratumoral necrosis.
Categorical variables were described using relative and absolute frequencies, and continuous variables using measures of position and scale. Categorical variables were compared using the Fisher’s exact test. Continuous variables were compared using the non-parametric Mann–Whitney test (two groups) or the non-parametric Kruskal–Wallis test (three or more groups). Statistical analyses were performed using R software (R Development Core Team). The level of significance was set at 5%.
Results
Thirty-two cats were selected for the study (n = 26, retrospective study; n = 6 cats, prospective study). All cats had a diagnosis of mesenchymal neoplasm (sarcoma) based on cytology. Histologic diagnosis was available in 20 cases and revealed fibrosarcoma, mixosarcoma, neurofibrosarcoma or poorly differentiated sarcoma (80%, 5%, 5% and 10% of cases, respectively). Not all medical data regarding the physical examination of one of the cats in the retrospective study were complete. A total of 32 CT images were analyzed (13 recurrent cases and 19 non-recurrent cases). US assessment was performed in all six cases of FISS included in the prospective study.
The distribution of categorical and continuous clinical variables is given in Tables 1 and 2, respectively. In cases with two CT scans (primary and recurrent tumor respectively; 3/13), only CT images of recurrent lesions were considered in order to avoid the introduction of biases in the analysis. Whenever variables had two characteristics, only the most representative characteristic was presented (Table 1). In this study, clinical variables were not correlated (P >0.05) with tumor recurrence. Thoracoabdominal/abdominal wall (55%) and paralumbar area (19%) were more commonly affected by FISS in the 32 cats of the sample, followed by limb (13%), cervical/interscapular area (8%) and thoracic wall (5%).
Comparisons of categorical clinical variables between recurrent and non-recurrent neoplasms
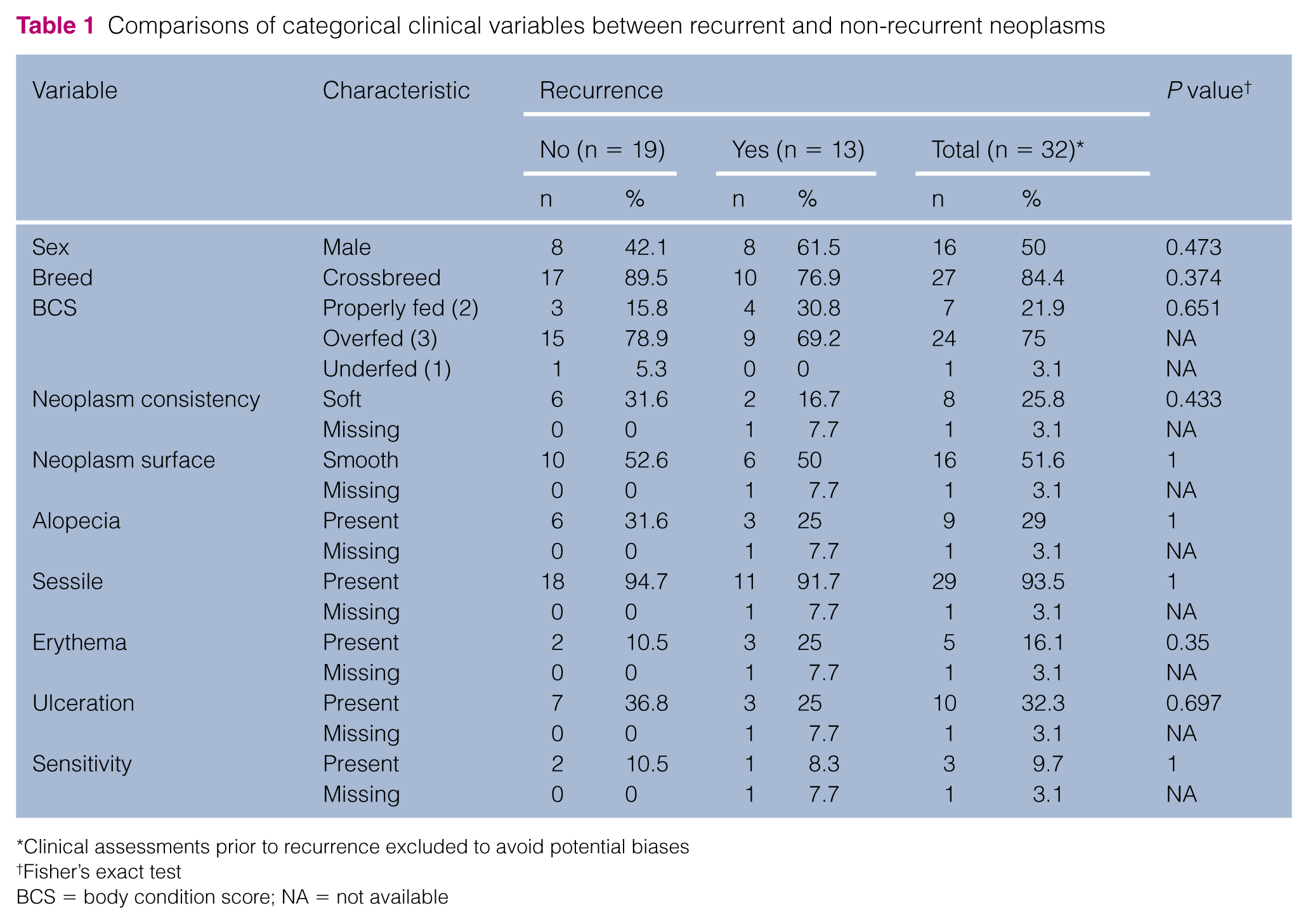
Clinical assessments prior to recurrence excluded to avoid potential biases
Fisher’s exact test
BCS = body condition score; NA = not available
Comparisons of continuous clinical variables between recurrent and non-recurrent neoplasms

Clinical assessment prior to recurrence excluded to avoid potential biases
Mann–Whitney test
CT findings
CT findings did not differ significantly (P >0.05) between recurrent and non-recurrent neoplasms (Table 3).
Comparisons of categorical computed tomographic (CT) variables between recurrent and non-recurrent neoplasms

CT prior to follow-up CT excluded to avoid potential biases
Fisher’s exact test
Most FISSs studied (62.5%) had poorly defined margins, which could be more clearly demarcated in postcontrast CT images. Irregularly shaped neoplasms (amorphous [31.3%] and multilobated [31.2%]) were more commonly observed, followed by spindle- (25.0%) and oval-shaped (12.5%) formations (Figure 1). Flat components (Figure 2), and thickening of adjacent adipose tissue (Figure 3), were documented in 37.5% and 31.2% of cases, respectively (Table 3), and were present in recurrent and non-recurrent neoplasms, with no significant differences between groups (P >0.05).

Transverse tomographic images displaying different neoplastic shapes (arrows) and digitiform projections (arrowheads) in cats affected by feline injection-site sarcoma. (a) Amorphous; (b) multilobated, (c) spindle-shaped and (d) oval-shaped neoplasms

Transverse tomographic images displaying different presentations of flat components (arrows) in the shape of feline injection-site sarcomas. (a) Recurrent neoplasm; (b) non-recurrent neoplasm
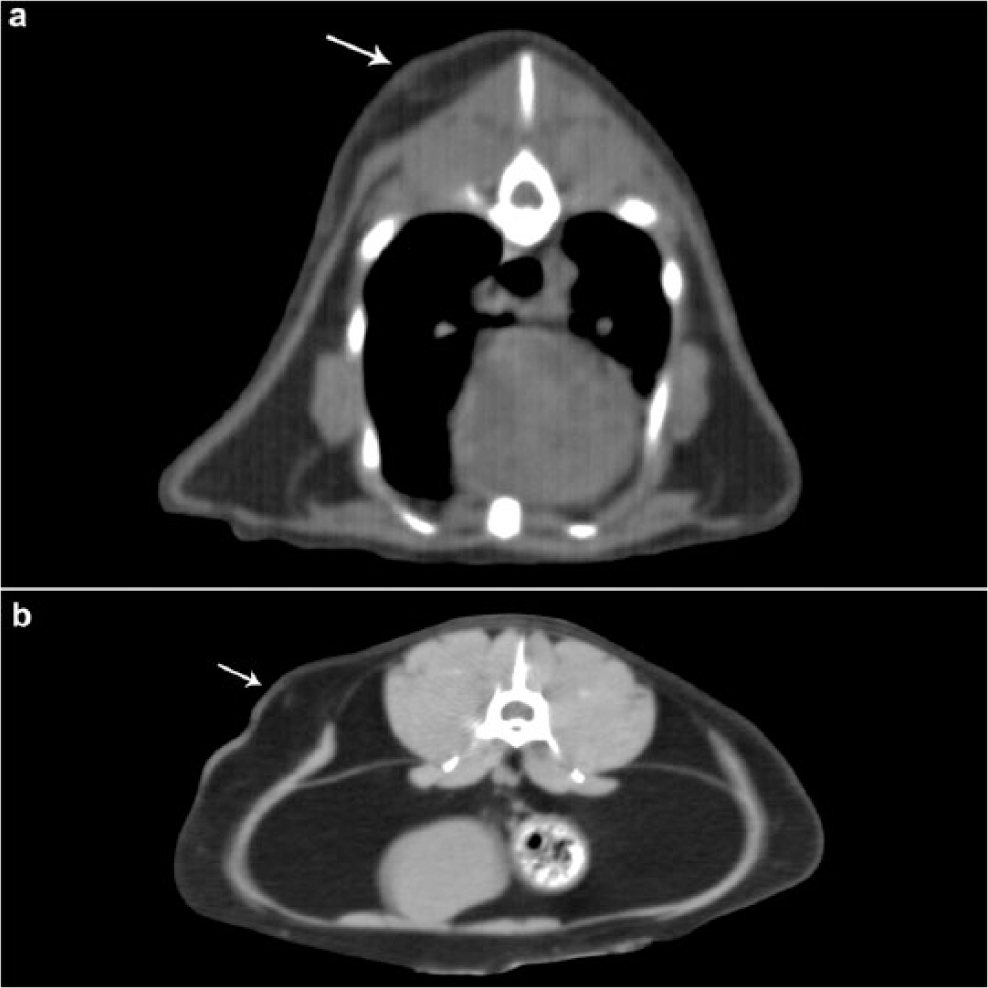
Transverse postcontrast tomographic images of feline injection-site sarcomas displaying thickening of adjacent subcutaneous adipose tissue to the tumor. Thickening of subcutaneous adipose tissue (arrows) in slices in close proximity to the primary neoplasm (not shown in these images). (a) Dorsal thoracic and (b) lumbar subcutaneous adipose tissue
Heterogeneous precontrast attenuation (59.4%) with mixed contrast enhancement (67.7%) prevailed in the neoplasms studied (Figure 4). Contrast enhancement was purely heterogeneous, homogeneous or peripheral in 16.1%, 9.7% and 3.2% of cases, respectively. Blurring of fat planes (Figure 5), and images suggestive of intratumoral liquefactive necrosis, were also frequently observed (Figure 4), and did not differ significantly between groups (68.8% of cases, respectively; P = 1). Areas suggestive of mild intratumoral calcification (attenuation between 120 HU and 180 HU) were observed in 12.5% of non-recurrent FISS; however, recurrent and non-recurrent neoplasms did not differ significantly with respect to intratumoral calcification (P >0.128; Table 3).

Transverse tomographic images of feline injection-site sarcomas displaying mixed neoplastic enhancement. (a) Precontrast heterogeneous attenuation; (b) mixed postcontrast enhancement (peripheral and heterogeneous) (arrows). N = area suggestive of intratumoral liquefactive necrosis

Transverse tomographic images of feline injection site sarcomas displaying blurring of peritumoral fat planes. (a) Precontrast attenuation of peritumoral fat; (b) mild postcontrast enhancement typical of blurring of fat planes (arrows). *Homogeneous abdominal fat tissue can be seen in both phases
Digitiform projections were recognized in all FISSs to a higher or lesser degree (Figure 1). In this study, evidence of secondary bone involvement was not identified on CT images.
In each tumor, the number of secondarily affected muscles ranged from one to 10 (mean 4.28 ± 2.47), with no significant differences between groups (P = 0.506, Mann–Whitney test). Twenty-nine different muscles were secondarily affected by FISSs (Figure 6), as shown by changes in shape and/or attenuation, or absence of a defined cleavage plane between muscular and tumoral tissues and unclear margin demarcation. Tumor shape was not correlated with the number of secondarily affected muscles (P = 0.228, Kruskal–Wallis test); however, incidence of secondary muscular involvement tended to be significantly higher in cases associated with thickening of adjacent adipose tissue (P = 0.003, Mann–Whitney test). Seven of 11 patients (63.6%) presenting with thickening of adjacent adipose tissue were overfed (BCS 3).

Prevalence of muscular involvement in 32 cases of feline injection-site sarcoma (FISS). The cutaneous trunk and oblique abdominal muscles were more frequently affected by FISS in the 32 cats studied
Skip metastases were recognized in eight cats (21.6%) and were more common in recurrent than non-recurrent neoplasms (53.8% and 4.2%, respectively; P = 0.001, Fisher’s exact test) (Figure 7). Fat blurring was not significantly correlated with thickening of adjacent adipose tissue, evidence of skip metastases on CT or BCS (P = 0.279, P = 1 and P = 0.177, respectively, Fisher’s exact test).
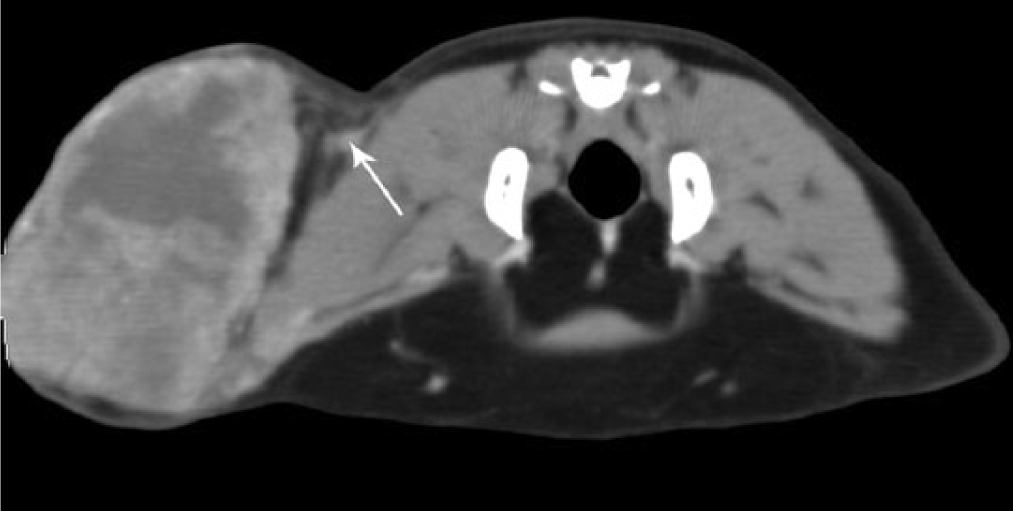
Transverse postcontrast tomographic image of feline injection-site sarcoma. Nodular image consistent with skip metastasis (thin arrow) adjacent to the neoplasm
US findings
Mixed echogenicity prevailed in the neoplasms studied. All FISSs scanned had a heterogeneous echotexture, irregular margins and projected into surrounding tissues to a higher or lesser degree (Figure 8). Peripheral hyperechoic capsule-like rim and variable thickening (0.06–0.13 mm) of adjacent subcutaneous tissues were also observed. Signs of liquefactive necrosis were recognized in 83.3% of cases and were consistent with changes documented in corresponding CT images. Intratumoral calcifications were not observed.
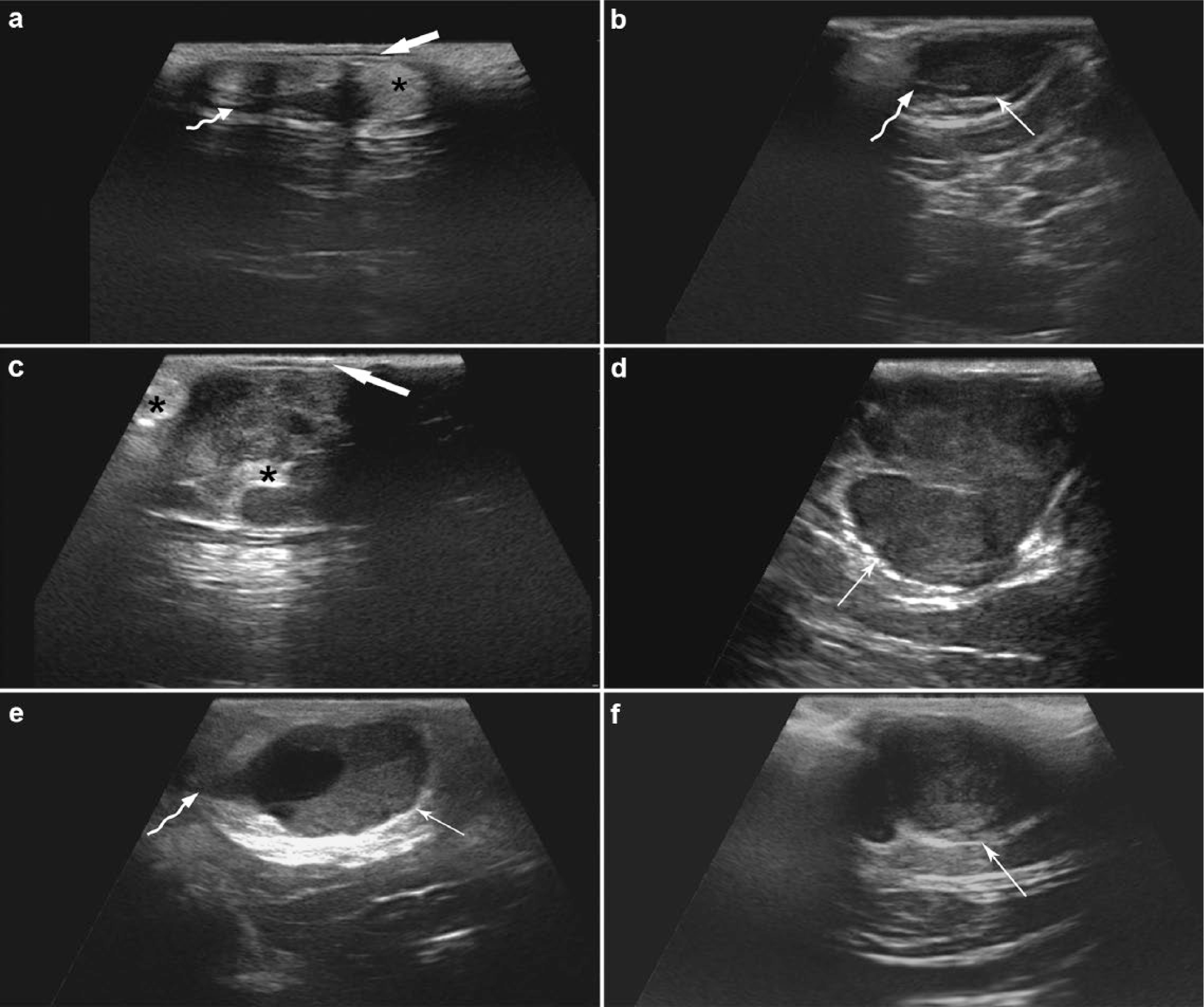
Main sonographic features of the six feline injection-site sarcomas scanned in the prospective study (B mode ultrasound). (a,b,e) Prevailing mixed echogenicity with heterogeneous echotexture, irregular margins and digitiform projections (irregular arrows); (b,d,e,f) capsule-like hyperechoic rim (thin arrows); (a,c) hyperechoic tissue contiguous with the neoplasm (*); (a,c) thickening of the surrounding subcutaneous tissue that corresponds to the fine, linear, hypoechoic superficial image (wide arrows)
Discussion
FISS has been widely investigated; however, diagnostic imaging has received limited attention in FISS studies to date. 8 Only one publication describing the absence of significant correlations between CT findings (appearance, attenuation, enhancement, necrosis, blurring of fat planes, invasiveness and volume) and the primary or recurrent nature of FISS was found in the literature. 11
Fibrosarcomas accounted for 80% of all neoplasms with histologic diagnosis in this study. The predominance of fibrosarcoma in cats affected by FISS has been reported elsewhere. 19 The remaining cases in the sample (37.5%) had history and cytologic findings suggestive of FISSs. The choice of fine-needle aspiration cytology over biopsy collection in these cases was owing to risk-related and economical concerns; presumptive diagnosis of FISS was based on cytologic evidence, anatomic location (injection site), and well-established clinical criteria (3–2–1 rule). 5
Compared with other types of sarcoma, FISS is thought to have no sex or breed predisposition,2,11 and to affect younger cats.4,11,14 In the present study, male and female cats of different breeds (mean age 10 years) were equally affected by FISSs. The predominance of overfed cats (75.0%) supports the hypothesis that fat cats are prone to the development of FISS. 11
Firm, sessile and deeply rooted neoplasms with smooth or irregular surfaces prevailed in the cases studied. Similar characteristics have been described eslsewhere. 11 Differences in preferential sites of FISS development in this and previous studies (thoracoabdominal/abdominal wall or paralumbar vs interscapular area,3,8 respectively) may have reflected typical injection sites at different geographical locations. 8
CT recognition of highly inflamed and perfused peritumoral tissues, often unrecognized on physical examination, plays an important role in surgical planning.8,11 Contrast enhancement improved tumor margin demarcation in this study (including the digitiform projections), showed a prevalence of mixed tumor enhancement and should be employed whenever possible. Clearer demarcation of tumor margins and enhancement of tissue vascular patterns by IV iodinated contrast media has been reported elsewhere.8,11,20 Heterogeneous enhancement has been associated with tissue necrosis in cases of FISS.6,8,11,14 Digitiform projections seem to be strongly associated with the infiltrative nature of FISS.6,8,11,14,20,21 Hence, in an effort to ensure comprehensive tumor assessment, CT scans should be planned to include a wide area. 8 Further details could be highlighted, such as the prevalence of amorphous and multilobated shaped neoplasms in recurrent and non-recurrent cases. Flat components in tumor shape were cited in a preliminary study by the authors of this article, 20 and thickening of adjacent adipose tissue has not been described in FISS studies to date.
Blurring of peritumoral fat planes is thought to reflect fat tissue inflammation, neovascularization or neoplastic infiltration of the panniculus, and was a common CT finding in this and previous studies.8,11,14, 20 In obese animals, blood flow impairment by hypertrophied adipose cells result in tissue hypoxia, inflammation and macrophage infiltration. 22 Yet, obesity-related inflammation tends to be subclinical (low grade) 23 and would not be expected to appear as blurring of fat planes on CT. Thickening of adipose tissue adjacent to FISS in this study may have reflected adipocytic hyperplasia/hypertrophy in response to the higher energetic demands of adipocytes in obesity and inflammatory states, 22 or of neoplastic cells undergoing continuing mitosis. In humans, adipose tissue deposition in mammary glands has been shown to have a vital role in breast cancer progression and aggressiveness. 24 Further studies are warranted to clarify the significance of this process in FISS. The higher incidence of muscle involvement in FISS cases showing thickening of the adjacent adipose tissue may indicate a relevant role of adipose tissue in FISS aggressiveness.
Variable secondary muscular involvement in cases of FISS has been confirmed in this study.8,11,14,20 In contrast to earlier findings, 8 the cutaneous trunk muscle was more frequently affected than the interscapular muscles, possibly owing to the superficial location and large extension of this muscle in cats.
Further studies, with a larger sample size, focused on the correlation between blurring of fat planes and thickening of adjacent adipose tissue or skip metastases are recommended. Skip metastases have been associated with higher recurrence and lower survival rates in cases of human osteosarcoma. 25 In the present study, skip metastases were more common in recurrent than non-recurrent FISS (P = 0.001). However, the prevalence of these metastases prior to surgical resection and the potential effects of surgical intervention on neoplastic dissemination could not be determined owing to the lack of primary tumor CT data in most recurrent cases.
Mixed echogenicity, heterogeneous echotexture, irregular margins, digitiform projections associated with hyperechoic tissue contiguous with the primary tumor (possibly corresponding to inflamed adipose tissue) and capsule-like hyperechoic rims were common to all FISSs scanned. Irregularly shaped intratumoral anechoic areas suggestive of liquefactive necrosis were recognized in most neoplasms studied. In other species, injection-site sarcomas have been described as heterogeneous cavitating masses with peripheral hyperechogenicity and a highly cellular anechoic core, suggesting a necrotic area. 26 The irregular shape and highly infiltrating nature seem to be the main sonographic differences between FISSs and soft tissue tumors described in dog.21,27
Variable hypoechoic thickening of subcutaneous tissues adjacent to the neoplasms studied suggest superficial inflammatory reaction and may have reflected the extension of the severe inflammatory process to the subcutaneous tissue.
The sonographic appearance of tumor margins reflects differences in acoustic impedance between different tissues. Therefore, recognition of neoplastic capsules on US scans may not reflect a true physical plane between tissues but rather peripheral inflammatory infiltration (inflammatory pseudocapsule).9,27 Further studies are required to investigate this hypothesis.
Irregular tumor surface and large tumor size precluded complete inclusion of the neoplasm in the US field of view in some cases. Similar difficulties have been reported in canine lipoma studies. 28 The subjective nature of ultrasonography may also have influenced the results presented. This study described general ultrasonographic features of FISS. Given the small number of tumors scanned, further studies with larger samples and including inter-observer comparisons are warranted.
Future studies comparing imaging (CT and US) and histopathologic findings should also be conducted to investigate potential correlations between the presence of areas of necrosis, infiltration, inflammation and calcification of FISS.
Conclusions
The imaging modalities studied allowed the characterization of the recurrent and non-recurrent FISS lesions and the recognition of peritumoral tissue involvement. This study confirmed the value of CT in establishing tumor features, and provided important contributions to diagnosis, therapeutic planning and patient follow-up. US proved to be an ancillary tool in clinical investigation of FISS lesions.
Footnotes
Acknowledgements
The authors thank Carolina Scarpa Carneiro, PhD, School of Veterinary Medicine and Animal Science, University of São Paulo, for the medical records used in the retrospective phase of this study.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
