Abstract
Overview:
Cardiopulmonary nematodes are emerging parasites of cats in Europe. A number of helminth parasites may be involved. The most prevalent lungworm in domestic cats is Aelurostrongylus abstrusus. Oslerus rostratus and Troglostrongylus species are found mainly in wild cats. The trichurid Capillaria aerophila has a low host specificity and is not uncommon in cats. Additionally the lung flukes Paragonimus species are reported in many species outside of Europe, including cats.
Clinical signs:
Lungworm infections may be asymptomatic, or cause mild to severe respiratory signs, dependent on the worm species and burden; mixed infections are observed. Kittens can be vertically infected and may develop a more severe disease. Affected cats show a productive cough, mucopurulent nasal discharge, tachypnoea, dyspnoea and, in severe cases, respiratory failure and death.
Management:
Early diagnosis and treatment greatly improves the prognosis. First-stage larvae can be easily detected in fresh faecal samples; the Baermann migration method is the enrichment technique of choice, but takes 24 h. Lungworm larvae can be found in tracheal swabs and bronchoalveolar lavage fluid, but with less sensitivity than in faeces. Molecular methods have been developed that exhibit high specificity and sensitivity, and allow diagnosis in the prepatent phase. Treatment options include fenbendazole paste, milbemycin oxime/praziquantel and various spot-on formulations. Severe cases should receive prompt medical care in an intensive care unit.
Prevention:
Avoiding predation is at present the only preventive measure for pulmonary worms with indirect life cycles.
Zoonotic risk:
C aerophila has zoonotic potential, causing severe pulmonary disease in humans. Some Paragonimus species are also of zoonotic concern.
Introduction
Cardiopulmonary nematodes are emerging parasites of cats and dogs in Europe and have received growing attention from researchers in recent years.1 –4 Significant progress has been made, mainly in the diagnosis and treatment of infection.
Disease agents
Infection of the lower respiratory tract can be caused by a number of parasitic nematodes. Certain metastrongyloid worms are commonly defined as lungworms because the adult stage is located in the lungs of their hosts, but actually some trichurids and flukes also live in the respiratory system.1 –3,5 Aelurostrongylus abstrusus (Strongylida, Angiostrongylidae) is the most well known feline lungworm and is regarded as the most prevalent in domestic cats. It is small (5–10 mm) and very narrow (less than 100 μm) and capable of colonising the respiratory bronchioles and alveolar ducts of domestic cats and other felids worldwide.2,6 Other respiratory mollusc-borne metastrongylids are commonly reported at necropsy in wild felids but are considered very rare in domestic cats. Oslerus rostratus (Strongylida, Filaroididae) exceeds 30–40 mm in length and infects the bronchial submucosa mainly in wild cats such as bobcats or in feral cats.5,7 –10 Troglostrongylus species (Strongylida, Crenosomatidae) is reported in a wide variety of wild cats and occasionally in domestic cats;4,9,11 –14 these worms vary in length, according to the species, from about 10–25 mm and are up to 0.5 mm in width. They are located in the trachea and bronchi or even the bronchioles for the smallest species (T brevior).12,15
The trichurid Capillaria aerophila (syn Eucoleus aerophilus) has a low host specificity and is not uncommon in cats and dogs as well as wild carnivores. 16 It is also a zoonotic parasite, causing a potentially severe pulmonary disease in humans. 17 C aerophila is found in the submucosa of the trachea, bronchi and bronchioles.2,16
Mixed infections by respiratory nematodes are sometimes reported7,9,14,18 –20 and both Troglostrongylus species and O rostratus may be more prevalent than presumed in domestic cats since there is a risk that these infections are being misdiagnosed as A abstrusus because of morphometric similarities of their first-stage larvae (L1) in faeces.3,4
Paragonimus species are lung flukes reported in many animals, including cats and humans, and some species are of zoonotic concern. Many species are found in cats, including P kellicotti, and between one and 10 adults measuring 8–18 mm x 4–8 mm live in subpleural cysts or bullae. 1
Life cycle and transmission
A abstrusus, O rostratus and Troglostrongylus species all have an indirect life cycle involving terrestrial molluscs. Eggs of A abstrusus laid by female worms hatch in the respiratory tract and L1 larvae are coughed up, swallowed and eliminated in the environment with the faeces. They can actively enter slugs or snails where they moult into the infectious L3 stage.21 –24 The biological cycle in the intermediate host is influenced by environmental temperature: a higher rate of larval development is observed at warmer temperatures. 23 The L3 larvae are also found in a wide range of paratenic hosts (rat, mouse, lizard, frog, bird) commonly predated by cats.1,5,22 The ingestion of L3 larvae by the cat is the best recognised means of transmission of lungworms, but vertical transmission via the placenta or milk cannot be excluded, as adult egg-laying worms have been found in kittens as young as 8 weeks of age. 14 Experimentally, it has been demonstrated that egg production starts 4–6 weeks after infection and may last for months, although it can be irregular.6,25 –28
Vertical transmission of T brevior was recently observed in a queen and patent infection was detected in 1-month-old kittens.13,14,29 T brevior and A abstrusus larvae may develop simultaneously in the same mollusc host (Helix aspersa) and overwinter for at least 120 days. 24 Very recently environmental contamination has been suggested as an alternative means of transmission for both A abstrusus and T brevior L1 on the basis of an experimental study; 30 live larvae were found in the pedal mucus excreted by H aspersa and in the water where the snails died.
C aerophila has a direct life cycle and eggs laid by female worms in the respiratory tract are swallowed and reach the environment in the faeces. After 30–45 days, embryonated eggs become infective when ingested by cats. Earthworms are facultative paratenic hosts. 16 When cats ingest infective eggs or earthworms carrying larvae, the larvae migrate to the lung and develop into the adult stage in 3–6 weeks. 31
The life cycle of Paragonimus species is associated with freshwater environments and is complex as it involves two intermediate hosts. Motile miracidia are released from eggs when swallowed and then passed in faeces from infected cats and penetrate aquatic snails; cercarial stages developed in snails will move from them, actively entering the second intermediate host (crab or crayfish). Cats are infected after eating the second intermediate host where metacercariae finally develop. Young flukes develop from metacercariae in the cat intestine, and cross the intestinal wall and the diaphragm to the pleural cavity where they penetrate the lung parenchyma and become reproducing adults in about 6 weeks. 1

Epidemiology
Feline lungworm infection is receiving increasing attention.2,6 A abstrusus is a well recognised agent of lower respiratory tract disease in cats.1,2 Epidemiological studies and case reports have confirmed the presence of the parasite in the Americas, Europe, Asia and Australia.1,14,32 –40 Prevalence rates vary and endemicity is linked to climatic and ecological factors that may influence: (a) the vitality and developmental capacity of L1; (b) the presence of suitable intermediate hosts in the environment; and (c) the number of days needed for development of the infective stage (L3). The diagnostic method used in epidemiological studies and the characteristics of the population investigated heavily influence the results.2,37,41,42 Feral and free-roaming cats are at higher risk because of their predator activity, as are cats with respiratory signs and young cats.43,44 In Tirana (Albania), post-mortem examination of the lungs of 18 feral cats revealed that nine (50%) were positive for A abstrusus. 45 Use of a low-sensitivity diagnostic method, such as the standard faecal flotation technique, showed a prevalence rate of 1–25% in a general cat population (see Table 1).14,46 –49
CR = case report
O rostratus is considered an uncommon parasite in domestic cats, but the prevalence in feral cats was found to be 24% in Majorca (Spain). It was also reported in a cat in northern Spain.7,8 Very recently, the incidental occurrence of a few adult O rostratus worms was reported in Sicily (Italy) at the necropsy of an adult cat that had died following a road traffic accident. 10
C aerophila has a sporadic occurrence in cats, dogs and humans in Europe. In central Italy, a prevalence of 3–14% was found in the feline population.2,16,36
Single cases of Troglostrongylus species infection were recently reported in cats from Ibiza (Spain), central and southern Italy and Crete (Greece).9,12,13,15,19,20,29,50 The first epidemiological data on T brevior in domestic cats were recently provided in Sardinia (Italy) where 6.5% of a sample of 107 cats tested positive compared with 25.2% that tested positive for A abstrusus, 14 confirming that Troglostrongylus is not a negligible lungworm of domestic cats.
The recent development of molecular assays specific for mollusc-borne feline lungworms sharing the same ecological niches, as well as for C aerophila, is likely to be of great value for epidemiological investigations, overcoming the difficulties of copromicroscopy for differentiating the metastrongylid L1 larvae.3,9,50 –52
Paragonimus species infections are reported in cats from the Americas, Africa and Asia.1,53,54 Paragonimiasis is most prevalent in cats and dogs in some parts of Asia. 55
Prevalence rates and/or documented cases of A abstrusus, C aerophila, O rostratus and Troglostrongylus species in some European countries are shown in Table 1.
Pathogenesis
The severity of lesions depends on the worm species and burden. Kittens also seem to develop a more severe disease.18,26,27,67 This may be explained by the smaller lung volume and small diameter of the trachea and bronchi, which are more easily blocked by worms, in particular by the larger Troglostrongylus species. The immature immune system also seems to facilitate infection: experimental reinfection of kittens with A abstrusus L3 larvae about 1 year after the initial symptomatic infection failed to induce respiratory signs or lung lesions. 25 In cats with natural aelurostrongylosis, the more severe radiological abnormalities and the higher larval burdens were found in younger animals (Figure 1). 68

Right lateral thoracic radiograph of a kitten with severe aelurostrongylosis, showing a diffuse focal alveolar pattern. Courtesy of Maria Grazia Pennisi, Department of Veterinary Sciences, University of Messina, Italy
An infective dose of <100 L3 A abstrusus larvae does not induce clinical signs but infective doses of 800–3200 larvae severely affect the lung and may even be lethal.69,70 However, at normal infective doses, the individual immune response significantly disrupts the parasite life cycle. 28 Cats repeatedly infected with a low number of larvae do not develop clinical disease when challenged with a high dose. 71
The role of immunity is confirmed also by the protective effect of passive immunisation in experimentally infected kittens. In some cases it can halt the parasite life cycle and the patent phase of infection does not occur.28,72
It has been recognised for a long time that eosinophilia is evident 2–6 weeks after the ingestion of L3 larvae of A abstrusus and that immune-mediated reactions of types I, III and IV are associated with alveolar, interstitial, peribronchial and vascular lesions and may lead to the death of parasites several months later.1,73 A more recent experimental study provides more detailed information on the clinical signs, haematology, biochemistry, coagulation analysis, computed tomography, coprology and post-mortem findings in young adult cats.28,70 Infected cats had moderate, non-specific clinical signs (fever, lethargy, weight loss, lymph node enlargement) and respiratory signs (dyspnoea, respiratory sounds, cough). Leucocytosis, massive and persistent eosinophilia and, in some cases, severe lymphocytosis were the most frequently observed abnormalities but no changes were detected on serum biochemistry. Various coagulation abnormalities were found, with a frequent occurrence of low fibrinogen values suggesting an increased consumption of coagulation factors. Imaging changes in the thorax were related to the dose and consisted of pulmonary nodules, bronchial pattern and lymphadenomegaly and were found even in a cat that did not develop a patent infection. 70
A abstrusus eggs accumulate in alveoli and bronchioles, inducing an inflammatory reaction in the lung (Figure 2). Multiple subpleural nodules (Figure 3) are caused by the granulomatous reaction surrounding clusters of eggs and adult worms, and emphysema is due to parasitic accumulation in the alveolar spaces. Bronchitis is severe and diffuse, usually manifested by bronchial and peribronchial lymphoid hyperplasia, hypertrophy of the smooth muscle layer and mucosal hyperplasia with increased mucous cell secretion in the bronchi. Vascular and perivascular changes are also seen, with hypertrophy and hyperplasia of pulmonary arteriolar smooth muscle, subendothelial fibrosis associated with eosinophilic infiltrates, endothelial and perivascular hyperplasia. Pulmonary hypertension may be the consequence of lung disease and arteriolar and bronchial changes may persist after the parasite dies, mimicking the changes found in feline asthma.27,73 –75 Bacterial complication is frequent and can be associated with pleural effusion. 26 Salmonella typhimurium, Pseudomonas species and Escherichia coli have been isolated in some cases and infection with enteric bacteria probably results from larvae migrating from the intestine.54,76

Alveolitis with larval accumulation, bronchiolitis and bronchiectasis in the lung of a cat with aelurostrongylosis (haematoxylin and eosin stain). Courtesy of Maria Grazia Pennisi, Department of Veterinary Sciences, University of Messina, Italy

Multifocal subpleural nodules and haemorrhages in a severe case of aelurostrongylosis. Courtesy of Maria Grazia Pennisi, Department of Veterinary Sciences, University of Messina, Italy
In a kitten with severe pulmonary aelurostrongylosis, enteritis and mild diarrhoea were associated with the presence of a high number of L1 larvae invading the small intestinal mucosa. 40
Lethal T brevior infection was associated in three kittens with catarrhal bronchitis occluding the lumen together with the adult worms, and multifocal pulmonary haemorrhages, consolidation and emphysematous foci.12,15
O rostratus does not seem to be associated with severe pathological changes in domestic cats, as few adult worms are found embedded in bronchial or peribronchial tissues inside pseudocysts.7,10 C aerophila usually induces chronic bronchitis.16,77
The penetration of Paragonimus species in the lung is associated with haemorrhagic foci, usually in the diaphragmatic lobe. Fluke cysts enter the bronchi and may evolve into bullae, with a consequent risk of pneumothorax.
Clinical signs
Although the majority of the publications in the literature concern A abstrusus, it has been suggested that many cases of infection or co-infection by other metastrongylids may have been erroneously attributed to the better-known A abstrusus because of difficulties with the morphometric differentiation of L1 larvae.3,4,9 Genetic characterisation of larvae now offers new insights and is likely to permit more accurate diagnosis.
Lungworm infections may be asymptomatic, or cause mild to severe respiratory signs due to bronchopneumonia, sometimes complicated by pleural effusion or pneumothorax.26,67,78,79 A productive cough is, therefore, the main clinical sign, together with mucopurulent nasal discharge, tachypnoea, dyspnoea with laboured, abdominal breathing and end-inspiratory crackles upon auscultation. In more severe cases, respiratory failure causes cyanotic mucosae and respiratory acidosis.9,18,27,42,80
Diagnostic imaging (eg, thoracic radiography or computed tomography) reveals bronchial thickening and poorly defined, small nodules during the patent phase. These findings may persist after clearing the infection and should be differentiated from other chronic bronchial disease such as asthma.59,81 Imaging changes may be evident even before the patent phase of disease.28,70
Right-sided cardiomegaly associated with eccentric hypertrophy and secondary to pulmonary hypertension has been described in two kittens affected by a severe bronchopneumonia caused by A abstrusus. 27 Both kittens presented with heart murmurs with maximum intensity on the right hemithorax due to tricuspid and pulmonary regurgitation. One of the kittens died but, in the surviving kitten, the heart murmur disappeared several months after parasitological and clinical cure. Echo-Doppler examination confirmed the resolution of pulmonary hypertension. 27 It is, therefore, advisable to investigate for the presence of lungworm infection in cases of right heart disease associated with signs of pulmonary hypertension in outdoor cats. In a study of 54 cats that died during anaesthesia in spay–neutering programs in the USA, 9% of post-mortem investigations revealed the presence of A abstrusus. 82 Stray outdoor cats, such as those included in trap–neuter–release programs, are at higher risk of lungworm infection.
Eosinophilia is a frequent abnormality but is not found consistently in cell blood counts or in bronchoalveolar lavage (BAL) cytology.18,67,76,83
Troglostrongylus species was considered the cause of death of parasitised kittens presenting with a cough and severe respiratory failure at diagnosis, but cases of asymptomatic infection have also been reported.12,13,15,19,84
Capillaria infection may induce coughing (mostly dry cough), sneezing and wheezing in cats but asymptomatic carriers have also been reported.16,85
Mixed infections are increasingly reported but they do not necessarily have a more severe clinical picture or poorer outcome.19,84,85

Diagnosis
L1 larvae are very active in the faeces and are readily detected in fresh faecal samples. Care should be taken to prevent soil contamination of samples, as the presence of free-living nematodes may lead to misdiagnosis. L1 larvae can be observed in direct faecal smears or by the flotation technique. Note that, in the latter method, high specific gravity concentrated salt or sugar solutions may induce osmotic damage to the larvae, making identification difficult. 1 The Baermann migration method is considered the enrichment technique of choice for metastrongyloid lungworms, and is based on the positive hydrotropism observed in live nematode larvae (see box and Figure 4, page 630).41,42,86 It can provide quantitative information on the number of larvae found in each gram of faeces, which correlates well with the severity of disease.47,68 Unfortunately, 24 h are necessary to obtain the result and the test should be repeated three times in the event of negative results, for optimum sensitivity.
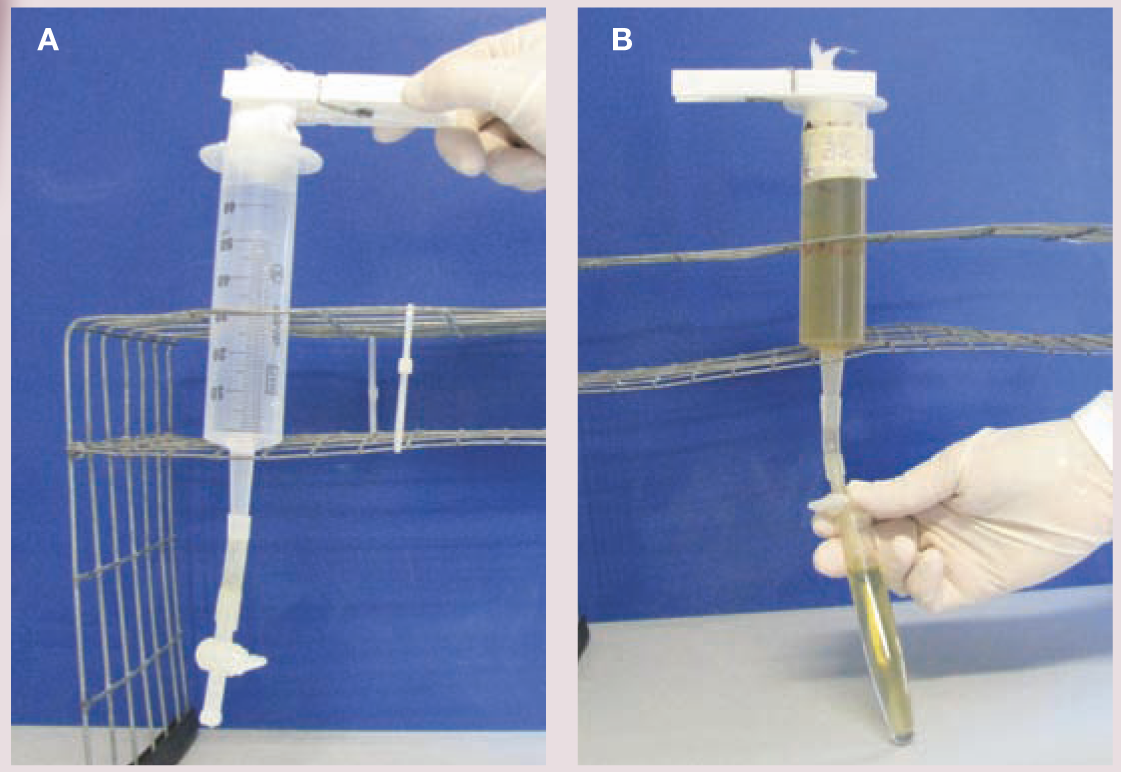
(A,B) Baermann apparatus. Courtesy of Emanuele Brianti, Department of Veterinary Sciences, University of Messina, Italy
A newer parasitological device for multivalent quantitative estimation of eggs, larvae and oocysts, named FLOTAC, was evaluated for suitability in the diagnosis of A abstrusus infection. The authors reported that it was more sensitive than the Baermann method. 87 However, the major limitation of copromicroscopy in general is the impossibility of making a diagnosis in the prepatent period, which lasts about 1–2 months, or when egg shedding has stopped but parasites persist and clinical signs are manifest. A well-trained observer is required to distinguish between the different strongylid L1 forms on the basis of their morphometric and morphological characteristics (Figures 5 and 6).3,12
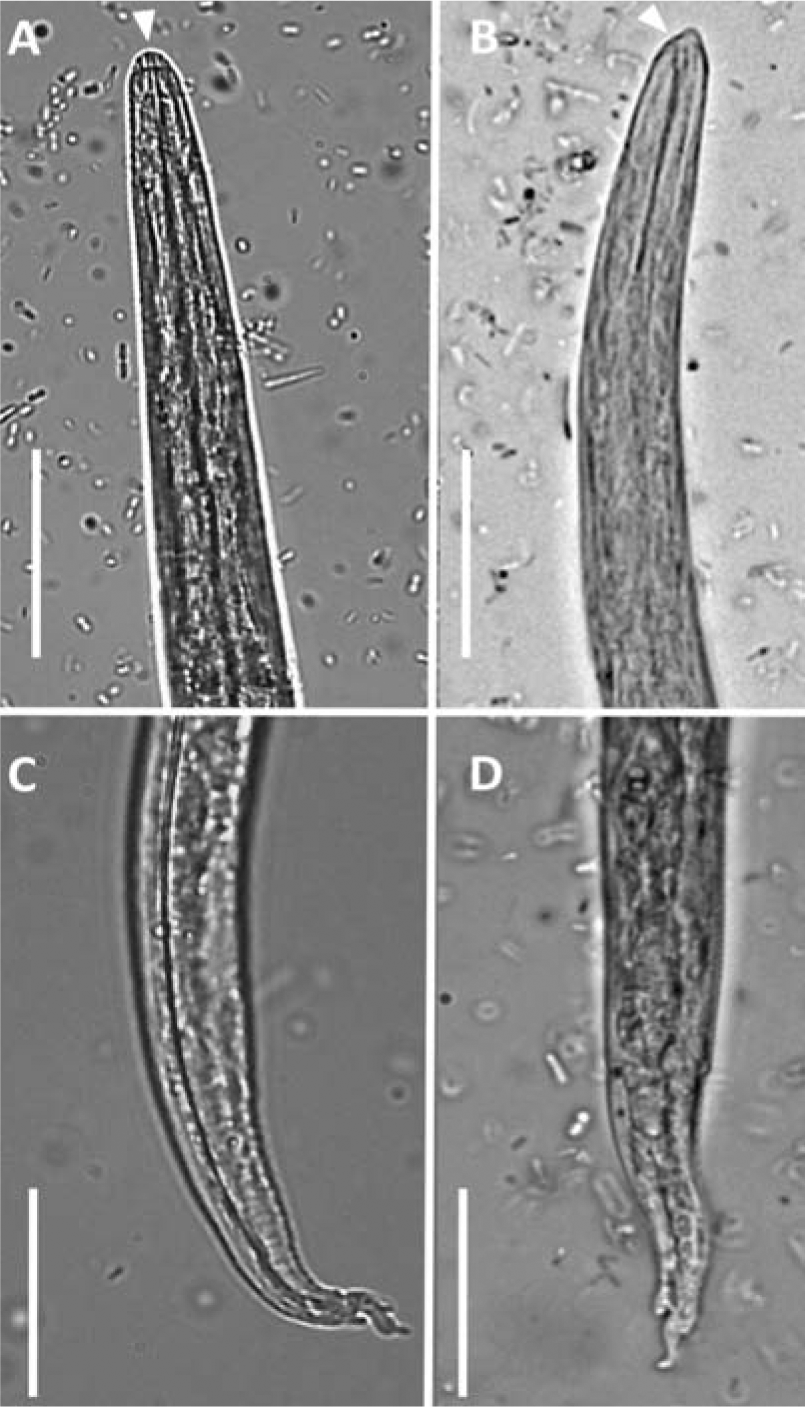
First-stage (L1) larvae of Aelurostrongylus abstrusus (A and C) and Troglostrongylus brevior (B and D) viewed by light microscopy (scale bars = 25 mm). (A) Anterior extremity of A abstrusus, lateral view. Note the terminal oral opening (arrowhead). (B) Anterior extremity of T brevior, lateral view. Note the pointed head and the subterminal oral opening (arrowhead). Morphology of the tail of A abstrusus (C) and T brevior (D) showing a dorsal spine at the end of the tail. Courtesy of Emanuele Brianti, Department of Veterinary Sciences, University of Messina, Italy

First-stage (L1) larvae of Oslerus rostratus viewed by light microscopy (scale bars = 100 μm). Note the morphology of the cephalic (A) and caudal (B) regions. Courtesy of Emanuele Brianti, Department of Veterinary Sciences, University of Messina, Italy
Lungworm larvae can be found in tracheal swabs or wash and BAL cytology but with less sensitivity than in faeces, so there is no benefit in using these more invasive procedures that risk severe respiratory disease. 41 Antibodies to A abstrusus can be detected as early as 3 weeks postinfection using an immunofluorescence antibody test, but past and currently active infections cannot be differentiated by serology. 88
Significant progress has been made diagnostically with the advent of molecular methods. A nested-PCR assay specific for A abstrusus has been validated on different biological samples (faeces, flotation supernatant, Baermann sediment and pharyngeal swabs) collected from cats with natural infections. A specificity of 100% and a sensitivity of up to 96.6% were recorded and the best results were obtained using pharyngeal swabs. 51 This method allows early diagnosis in the prepatent phase, with a potential positive impact on prognosis. Molecular techniques are expected to significantly improve the understanding of lungworm infections. A new multiplex PCR has also been developed for the simultaneous detection of A abstrusus and T brevior. 50
Capillariosis is diagnosed by standard faecal flotation but molecular techniques are also available for screening and for human cases.2,52
Paragonimiasis is diagnosed using a formalin-ether sedimentation technique. 53 Molecular methods are available for epidemiological purposes in cats and are used for human cases.89,90

Treatment
Information on the efficacy of various drugs in the treatment of feline lungworm infection is available from controlled studies and clinical case reports (Table 2). Oral administration of fenbendazole has been suggested, with different doses and durations of therapy (from 20 mg/kg for 5 days to 50 mg/kg for 15 days), but an oral paste is licensed in the UK for treating aelurostrongylosis in cats at 50 mg/kg q24h for 3 days [
Drugs used for the treatment of pulmonary nematode infections in cats

CS = controlled study, CR = case report, EI = experimental infection
Off-label use of ivermectin has been reported, with inconclusive results, and should not be considered because of the risk of toxicity, principally in kittens [
Two spot-on formulations administered at the recommended dosage were compared with a 3 day course of fenbendazole therapy and were found to be effective and safe in the treatment of 12 naturally infected cats each: one formulation contained imidacloprid 10% and moxidectin 1% (Advocate; Bayer), the other emodepside 2.1% and praziquantel 8.6% (Profender; Bayer) [
In a case series study, cats with natural infection treated with the combination of imidacloprid 10% and moxidectin 1% were rechecked at day 14, and those still found positive (4/7) were retreated and checked 1 week later. At that stage, one cat remained positive and was treated for a third time. At the end of the study (day 50), two negative faecal tests had been obtained for all treated cats, confirming the efficacy of the treatment with this combination [
A combination of milbemycin oxime (4 mg) and praziquantel (10 mg) (Milbemax; Novartis) was administered as a single oral dose (half a tablet per kg) three times, 15 days apart, to a kitten with A abstrusus bronchopneumonia and pulmonary hypertension, achieving parasitological and clinical cure [
Information on the treatment of Troglostrongylus, as well as on mixed infections, is derived from case reports only. Cases of severe respiratory disease associated with Troglostrongylus infection were not cured by imidacloprid 10% and moxidectin 1% or febendazole treatment [
Bacterial secondary infections may contribute to the severity of lungworm disease and broad-spectrum antibiotics should always be given together with corticosteroids at anti-inflammatory doses in cases with signs of bronchopneumonia. Pleural effusion and pneumothorax require immediate resolution by thoracocentesis, and medical care in an intensive care unit (oxygen administration) is required for all cats with respiratory failure.
Prognosis
In cases of A abstrusus infection, a delay in diagnosis and treatment may lead to fatal cardiopulmonary lesions, while early diagnosis and treatment greatly improves the prognosis. The level of larval burden determined by the Baermann test is usually related to the severity of the disease but the prognosis should be based mainly on physical examination (severity of dyspnoea and occurrence of cyanosis) and radiographic findings (severity of diffuse bronchial, alveolar and interstitial disease).
Prevention
Stray and free-roaming cats have a higher risk of becoming infected with lungworms in endemic areas.
43
Avoiding predation is at present the only preventive measure for metastrongyloid or trematode pulmonary worms with indirect life cycles. The prophylactic activity of some molecules used to treat nematode respiratory infections – as for A vasorum infection in dogs – is currently unknown; but the spot-on combination of fipronil 8.3%, (S)-methoprene 10%, eprinomectin 0.4% and praziquantel 8.3% (Broadline) was found effective as a preventative treatment for aelurostrongylosis in an experimental setting [

Key Points
Aelurostrongylus abstrusus (Strongylida, Angiostrongylidae) is the most well known feline lungworm and is regarded as the most prevalent worldwide in domestic cats.
Other lungworms in the cat include Oslerus rostratus, Troglostrongylus species, Capillaria aerophila and Paragonimus species.
A abstrusus, O rostratus and Troglostrongylus species may cause mixed infections as they share the same intermediate and paratenic hosts.
Lungworm infections may be asymptomatic, or cause mild to severe respiratory signs due to bronchopneumonia, sometimes complicated by pleural effusion or pneumothorax (nasal discharge, tachypnoea, dyspnoea, coughing). The disease can be fatal.
Kittens may be vertically infected and develop a more severe disease at an early stage, due to the smaller diameter of the respiratory tract and their immature immune system.
It is advisable to investigate for the presence of lungworm infection in outdoor cats with right-sided heart disease associated with signs of pulmonary hypertension.
Stray outdoor cats are at higher risk of lungworm infection.
The Baermann migration method is considered the enrichment technique of choice, but takes 24 h to produce results and false negatives may occur.
The major limitation of copromicroscopy is that it is not diagnostic in the prepatent period, which lasts about 1–2 months.
A nested-PCR assay specific for A abstrusus has been validated.
Treatment options include fenbendazole paste, milbemycin oxime/praziquantel and various spot-on formulations (imidacloprid 10 %/moxidectin 1%; emodepside 2.1%/praziquantel 8.6%; fipronil 8.3%/(S)-methoprene eprinomectin 0.4%/praziquantel 8.3%; or selamectin).
In severe cases, broad-spectrum antibiotics should be given, together with corticosteroids.
C aerophila has zoonotic potential and sporadic cases of human capillariosis, manifesting with a productive cough, haemoptysis and lung lesions, have been described .
Footnotes
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this article. The ABCD is supported by Merial, but is a scientifically independent body and its members receive no stipends from Merial.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.

